Abstract
In vivo assessment of ventricular function in rodents has largely been restricted to transthoracic echocardiography (TTE). However 1.5 T cardiac magnetic resonance (CMR) and transoesophageal echocardiography (TOE) have emerged as possible alternatives. Yet, to date, no study has systematically assessed these three imaging modalities in determining ejection fraction (EF) in rats. Twenty rats underwent imaging four weeks after surgically-induced myocardial infarction. CMR was performed on a 1.5 T scanner, TTE was conducted using a 9.2 MHz transducer and TOE was performed with a 10 MHz intracardiac echo catheter. Correlation between the three techniques for EF determination and analysis reproducibility was assessed. Moderate-strong correlation was observed between the three modalities; the greatest between CMR and TOE (intraclass correlation coefficient (ICC) = 0.89), followed by TOE and TTE (ICC = 0.70) and CMR and TTE (ICC = 0.63). Intra- and inter-observer variations were excellent with CMR (ICC = 0.99 and 0.98 respectively), very good with TTE (0.90 and 0.89) and TOE (0.87 and 0.84). Each modality is a viable option for evaluating ventricular function in rats, however the high image quality and excellent reproducibility of CMR offers distinct advantages even at 1.5 T with conventional coils and software.
Keywords
Small animal models are frequently used to evaluate cardiovascular disorders and response to therapeutic interventions. The primary endpoint in many of these studies is the evaluation of ventricular function, with in vivo methods now largely superseding ex vivo assessment. There are specific challenges to imaging rodent hearts, namely their minute size (≤10 mm left ventricles) and extreme heart rates (∼300–600 beats/min). 1 Accordingly, adequate spatial and temporal resolution of candidate imaging modalities is essential.
Transthoracic echocardiography (TTE) has been the principal imaging modality employed owing to its widespread availability and excellent temporal, yet modest spatial resolution. In conjunction with high frequency echo transducers and skilled operators, a variety of images can be readily obtained. In practice, most TTE assessments of ventricular function rely on either 1-dimensional M-mode evaluation, or 2-D left ventricular (LV) short axis views at one or more levels. 2 M-mode only evaluates two opposing myocardial walls, e.g. the anteroseptum and the posterior wall obtained from parasternal views, thus providing no information regarding the remaining myocardium. Calculation of LV ejection fraction (EF) from short axis views can provide a more representative evaluation of function, as a 360° view of myocardium is obtained. However even this ignores important changes in a geometric shape in diseased states, such as after myocardial infarction (MI). 3 Additionally TTE can suffer from reduced reproducibility, as it is more operator-dependent and ensuring that the views obtained are truly orthogonal to the LV long axis can be challenging.
Transoesophageal echocardiography (TOE) might offer an alternative echocardiographic strategy. Suitable transducer probes, of a sufficiently small calibre and cost-effective nature, have recently become available in the form of intracardiac echocardiogram (ICE) catheters. 4 These devices are routinely used in many interventional cardiology centres for assistance with some structural or electrophysiology procedures. A typical ICE catheter is 8 Fr (2.7 mm) in diameter with a high frequency (10 MHz) transducer and is amenable to repeated use. However there is a paucity of published evidence demonstrating the utility of rodent TOE.
Cardiac magnetic resonance (CMR), however, is the gold standard method of assessing LV function in humans and large animals. The high spatial and moderate temporal resolution of magnetic resonance imaging (MRI) provides highly reproducible imaging with minimal inter- and intra-observer variations.5,6 Until recently, CMR of rodents has been most often performed using very expensive high-field MR scanners which are available only in a minority of research institutions. Accordingly investigators have explored the utility of standard low-field clinical MR scanners for this purpose with promising results. 7 However, whether CMR retains any advantage over TTE or TOE when performed on 1.5 T scanners is unclear. To the best of our knowledge, no evaluation of 1.5 T CMR, TTE and TOE has been undertaken in small animals. Therefore we sought to determine the utility of these three modalities, all directly relevant and available to many research groups, in a rat model of MI.
Animals, materials and methods
Study protocol
All procedures were performed in accordance with local animal research and ethics committee approval. Male Sprague–Dawley rats (n = 20, weight 350–370 g) participating in a separate study evaluating novel therapy after surgically-induced MI were used. All animals underwent imaging by all three modalities (CMR, followed by TTE and TOE) at day 28 post-MI. Anaesthesia was maintained using 1% isoflurane, which provided effective sedation but no significant effects on cardiac function. 8 Cine CMR sequences were acquired after 5–10 min of isoflurane and both echocardiography investigations were also performed after 5–10 min of a second period of anaesthesia (≥2 h after CMR), ensuring comparable depth and duration of anaesthesia was provided in all animals.
CMR protocol
MRI was performed on a 1.5 T MR system (Magnetom Sonata, Siemens, Erlangen, Germany), equipped with a dedicated cardiac software package. The thorax and right paw were shaved, cleaned with alcohol and adhesive electrocardiogram (ECG) electrodes attached thereby generating a vector ECG. Animals were placed in a supine recumbent position in the isocentre of the magnet, with a 4-channel phased array carotid radiofrequency coil placed over the thorax, with 1% isoflurane administered via a nose cone to maintain anaesthesia. Accordingly, all CMR images were free breathing, ECG-gated, acquisitions. Transverse and coronal localizer images were acquired followed by long (4-chamber and 2-chamber) and short axis pilot images, from which a true short axis stack was prescribed. Balanced steady-state free-precession (SSFP) cine images (gated to alternate R waves) were acquired. The stack comprised three contiguous LV slices (each 3 mm thick, with no intersection gap) providing full coverage of the LV (Figure 1). The image matrix was 384 × 384, field of view 185 mm, repetition time 14.72 ms, echo time 1.55 ms, flip angle 90°, image resolution 0.5 × 0.5 mm, and eight phases per cardiac cycle were acquired. Three cardiac cycles were acquired at each level, with an imaging time of 52 s per short axis slice.
Cardiac magnetic resonance. Short axis stack comprising three short slices, each 3 mm thick covering the base (a), mid-left ventricle (d) and apex (g) of the heart. Representative examples of images at end-diastole (b, e, h) and end-systole (c, f, i) at each of the above levels, with endocardial and epicardial contours annotated.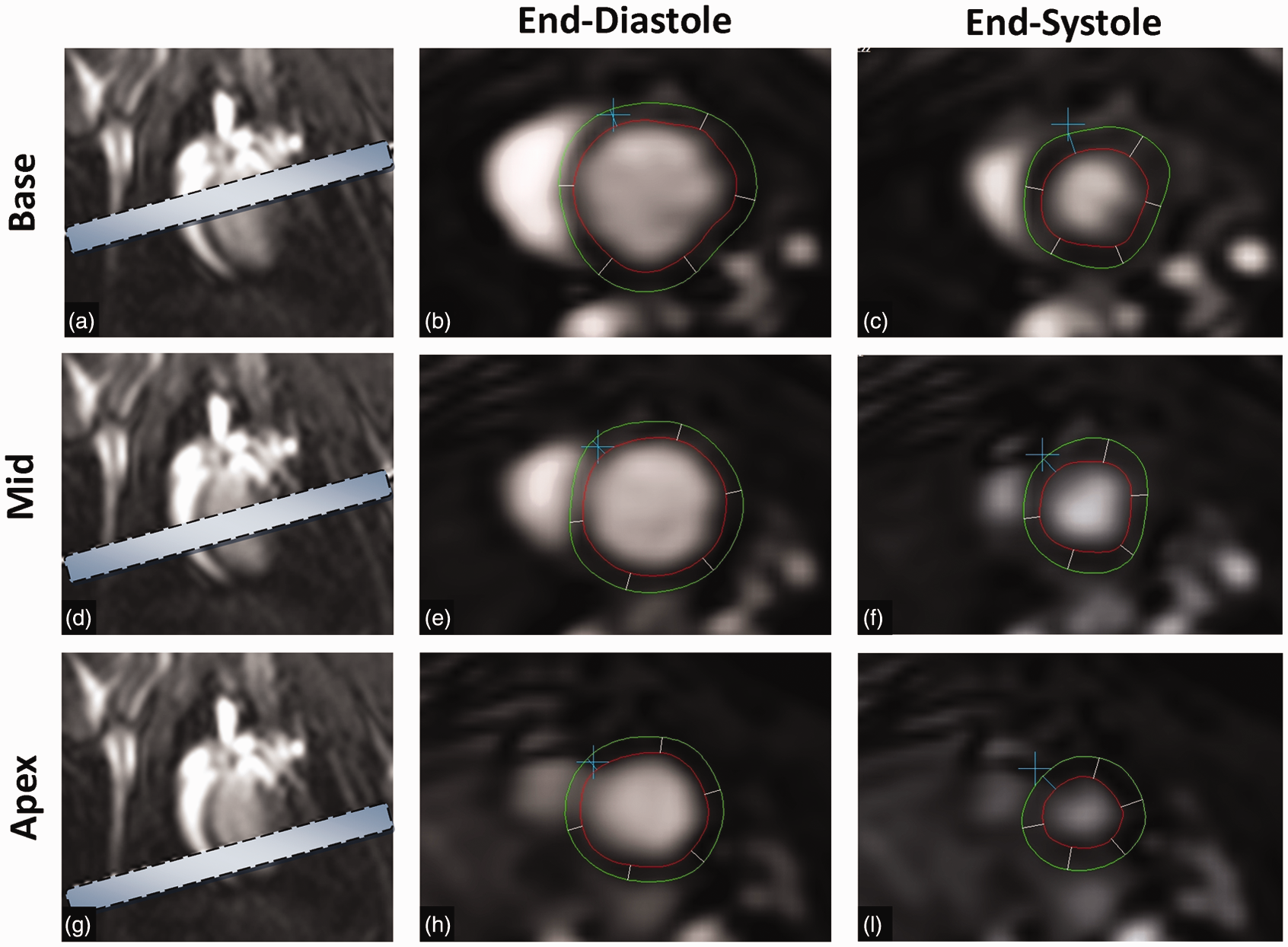
CMR image analysis
LV volumes and derived EF were measured off-line from cine images using commercially available software (QMass v7.2, Medis, Leiden, The Netherlands). Of the three cardiac cycles acquired at each level, the largest and smallest silhouettes corresponding to the end-diastole and end-systole were identified and the endocardial border was manually traced. Papillary muscles were excluded from calculations. The end-diastolic (EDV) and end-systolic (ESV) volumes were then calculated using the true disk summation technique (i.e. sum of cavity volumes across all continuous slices), as previously described. 9
TTE protocol
TTE was performed on a clinical Siemens Acuson X300 ultrasound system (Siemens) in conjunction with a Siemens P9-4 (9.2 MHz) transducer. Animals were placed in a supine recumbent position while maintained on 1% isoflurane. An LV short axis slice at the level of the papillary muscles was obtained and used to calculate EF. Additionally, an M-mode image was acquired by prescribing a cursor beam in the antero-inferior orientation of the above short axis slice (Figure 2). A minimum of three cardiac cycles were acquired and the largest and smallest silhouettes identified from these were analysed. Endocardial and epicardial borders were manually traced off-line (ProSolv Cardiovascular V4.0, FujiFilm, USA), with EF and fractional shortening (FS) calculated. We utilized the widely-used Teichholz formula (Volume = 7D3/2.4 + D, where D = dimension) to calculate EF.
Transthoracic echocardiography (TTE). Photograph illustrating TTE set-up with 9.4 MHz transducer over the shaved thorax (a). Representative 2-D short axis image (b) and M-mode acquisitions (through the anterior and inferior walls) of stem cell recipients (c) and controls (d) respectively.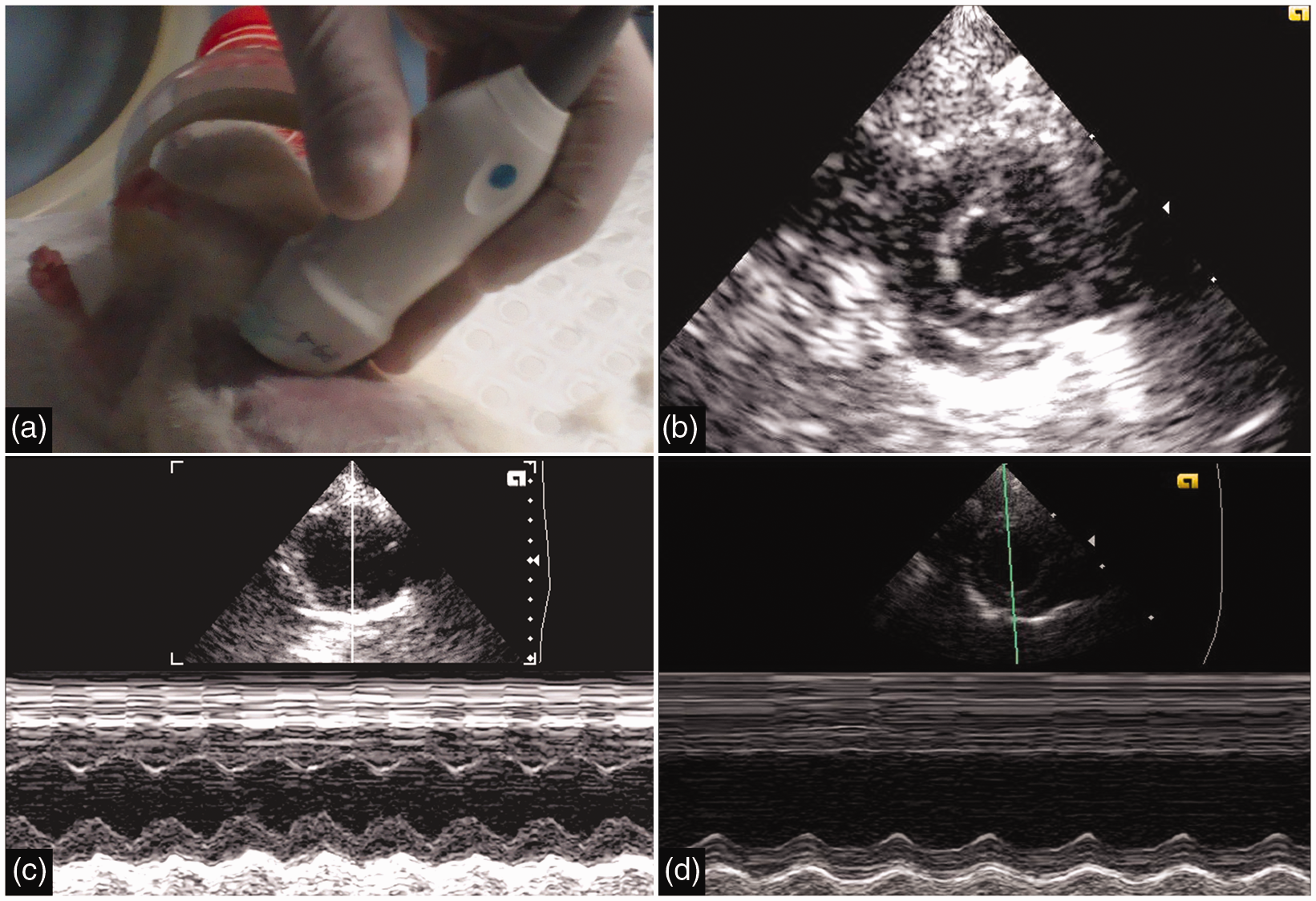
TOE protocol
Following TTE, rats were placed on an intubation rack by their front teeth, thereby lying in a vertical orientation (Figure 3) and 1% isoflurane was delivered via a nose cone. An 8 Fr (2.7 mm diameter) Biosense Webster AcuNav (10 MHz) ICE catheter was used in combination with the Siemens Acuson ultrasound system used for TTE imaging. The ICE catheter was introduced into the oesophagus by directing the catheter posteriorly using minimal pressure. The catheter, steerable in the antero-posterior and right–left directions, readily identified a 2-chamber view which illustrated the anterior and inferior walls from base to apex. As above, a minimum of three cardiac cycles were acquired and the largest and smallest silhouettes were analysed. EF was calculated as for TTE using the same analysis software. In addition, FS was calculated using end-diastolic and end-systolic diameters, derived from the calliper measurement of the mid-LV just below the papillary muscle, using the same 2-chamber cine.
Transoesophageal echocardiography (TOE). Photograph of an 8 Fr (2.7 mm) intracardiac echocardiogram (ICE) catheter, with an enlarged view of the transducer tip (a). Picture of a rat undergoing TOE, while suspended from an intubation rack (isoflurane nose cone temporarily removed) (b) and representative TOE image (c).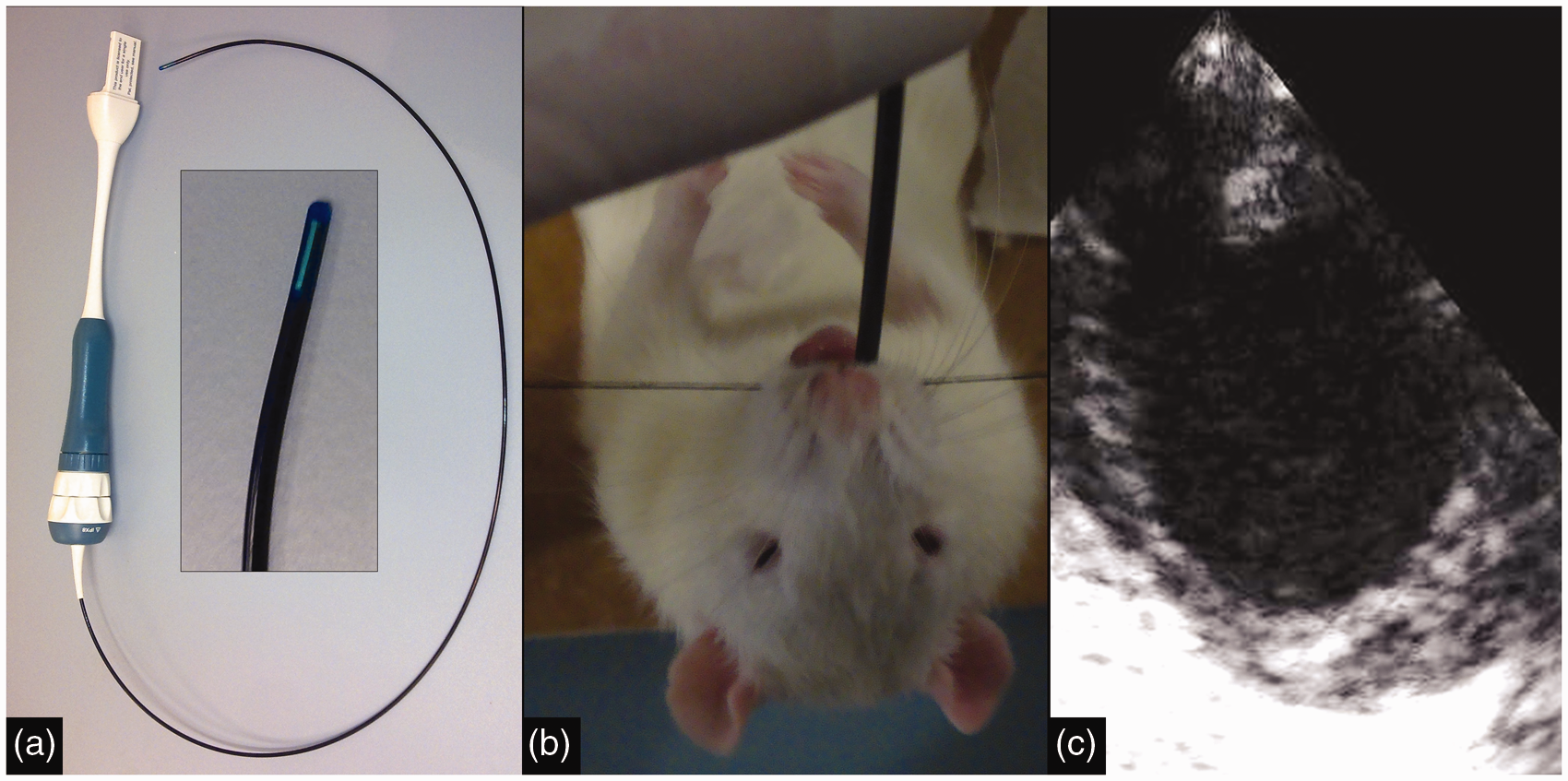
Data analysis
CMR studies were independently analysed by two CMR cardiologists (AGB, JDR). For the purpose of intra-observer assessment, the entire data-set was analysed on two separate occasions, with an interval of at least one month. An analogous format was followed for both TTE and TOE, with analysis performed by two experienced ECG readers (BK, JDR). Image quality was quantitatively graded by two observers as good (=1), adequate (=2) or poor (=3) and mean scores for each 2-D technique were determined (40 scores per imaging modality). Criteria included image clarity, blood–myocardium border definition and absence of artefact.
Statistical analysis
Values are expressed as mean ± standard deviation. Groups were compared using paired t-tests (if normally distributed) or using Wilcoxon rank test for non-parametric data. Correlation between modalities, intra- and inter-observer agreements were assessed using Pearson or Spearman correlation (if normally distributed or not, respectively) and intraclass correlation coefficients (ICC). Bland–Altman analysis was conducted, with mean bias, standard deviation and 95% limits of agreement calculated. A P value <0.05 was considered statistically significant. Statistical analysis was performed with SPSS 17.0 (SPSS Inc, Chicago, IL, USA).
Results
Imaging with the three technologies was completed in all 20 animals without complication and with the full set of views obtained. The typical duration of the CMR investigation was 10 min, 5–7 min for TTE and 3–4 min for TOE. Mean heart rate was 300 beats/min for each imaging assessment. Superior image quality was noted with CMR, which overall was graded as good (mean 1.16 ± 0.41, P < 0.0001), whereas TTE and TOE were scored as adequate (2.16 ± 0.55 and 2.09 ± 0.53) respectively.
Correlation between modalities
The mean EF values determined by CMR and TOE were very similar (40.1 ± 17.4% and 39.9 ± 14.6% respectively, P = 0.96), whereas there was a strong trend towards higher EF values when assessed by TTE (45.7 ± 14.7%, P = 0.06). FS results, derived from TTE M-mode and TOE 2-chamber images, were also highly comparable (31.8 ± 2.9% and 30.7 ± 2.3% respectively, P = 0.90). Conformity of EF measurements between each imaging modality was evaluated (Figure 4). Excellent correlation was noted between CMR and TOE (r = 0.89, ICC = 0.89, P < 0.0001), whereas moderate–good agreement was noted between TTE and TOE (r = 0.75, ICC = 0.70, P < 0.0001) and CMR and TTE (r = 0.67, ICC = 0.63, P < 0.001). Bland–Altman plots demonstrated a bias towards lower EF for both CMR (bias –5.86) and TOE (bias –5.78) when compared with TTE across the range of EF measurements (Figure 4). No significant bias was observed between CMR and TOE, with all measurements falling within 95% limits of agreement.
Correlation between imaging modalities. Linear correlation chart with regression line for each comparison pair (a, b, c), with correlation r value and intraclass correlation coefficient (ICC). Corresponding Bland–Altman plots (d, e, f) with mean bias, standard deviation and 95% limits of agreement displayed. CMR: cardiac magnetic resonance, TTE: transthoracic echocardiography, TOE: transoesophageal echocardiography, EF: ejection fraction.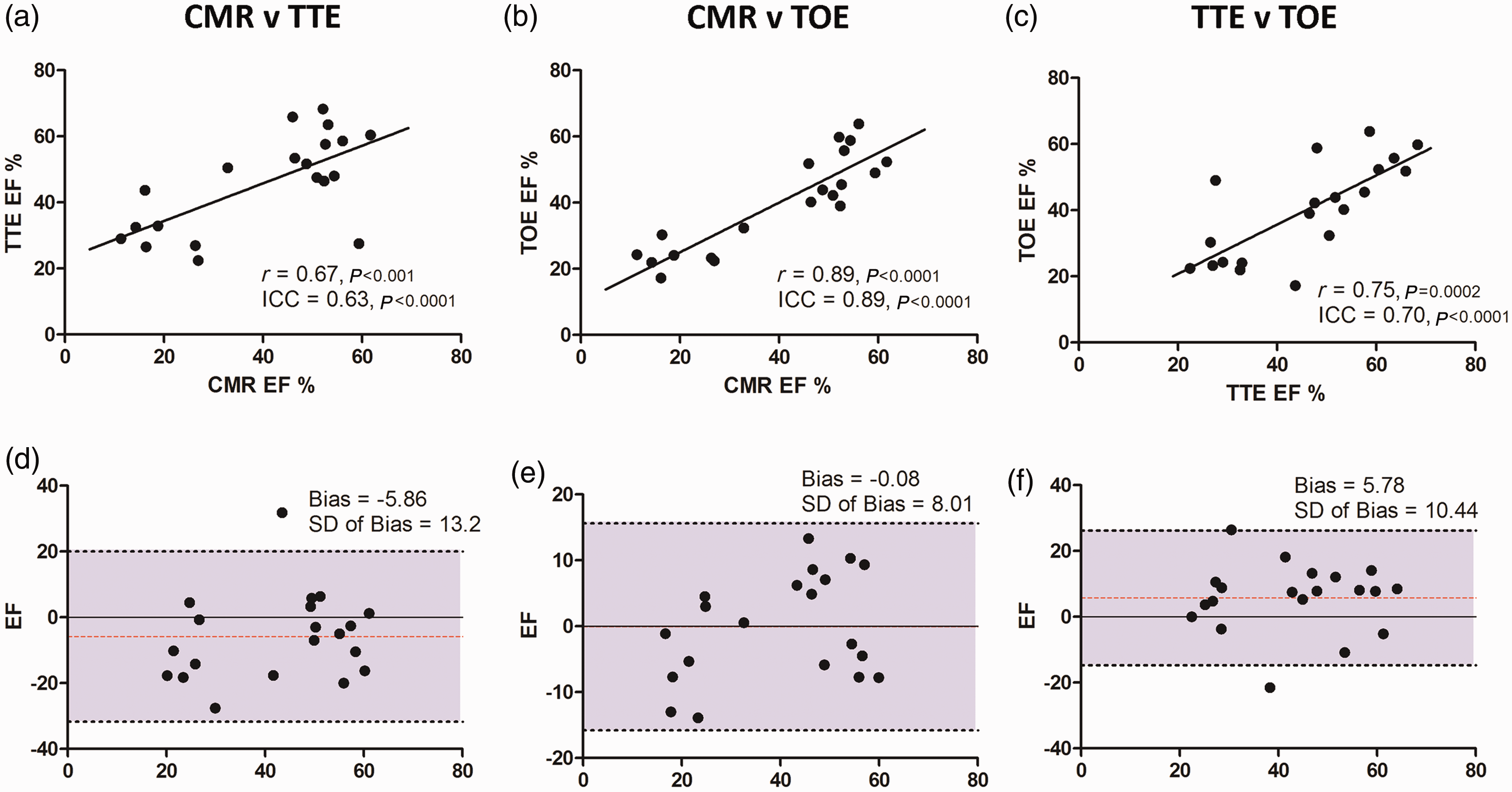
Intra-observer agreement
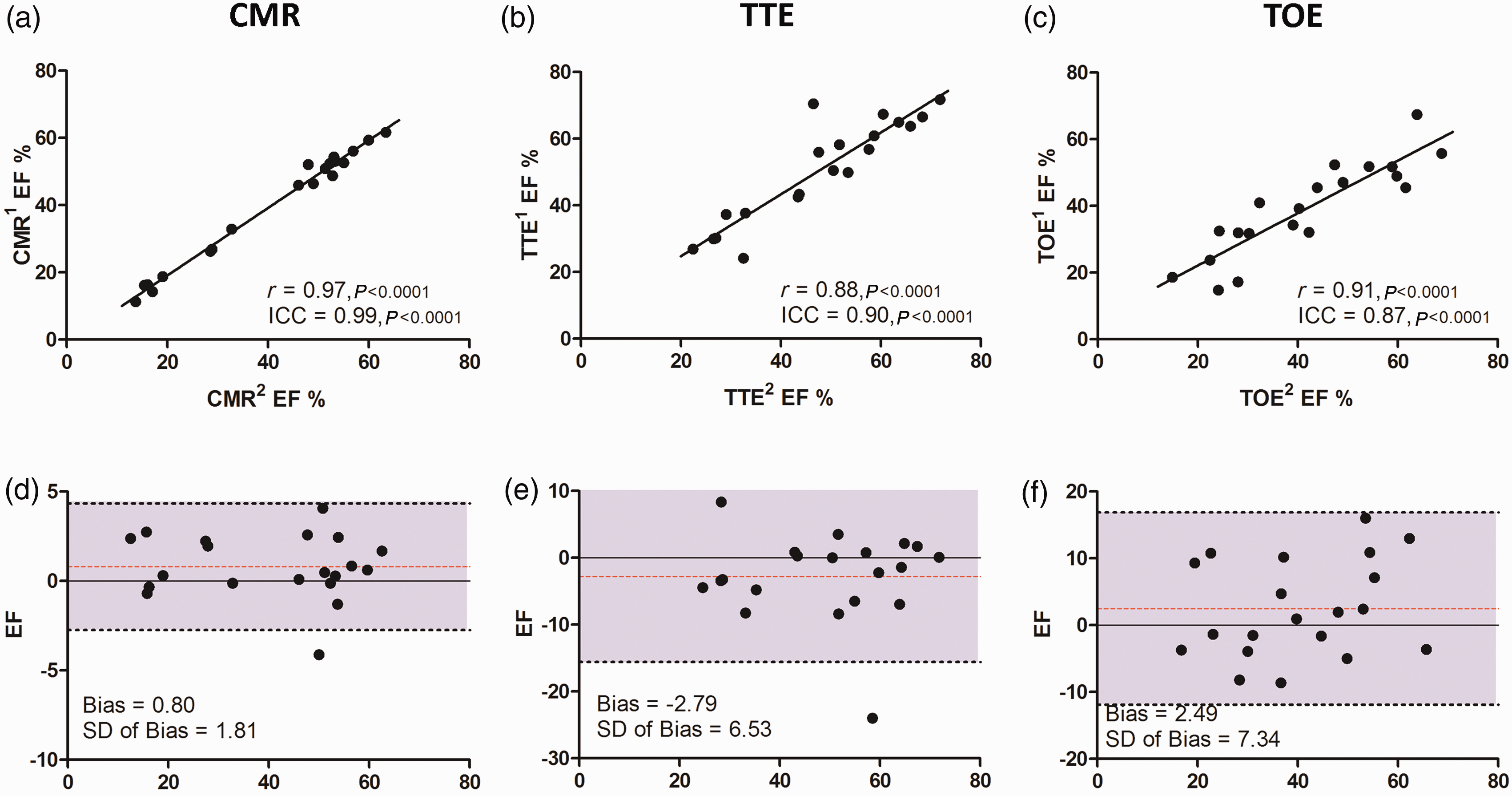
The absolute differences in EF (ΔEF) for each animal study between the first and second analyses were evaluated for each modality (Figure 5). For CMR the mean difference was 1.47 ± 1.29%, but this was appreciably higher for TTE (ΔEF = 4.85 ± 3.78%, P < 0.05) and TOE (ΔEF = 5.84 ± 4.41%, P < 0.0001). Intra-observer agreement was excellent for CMR (r = 0.97, ICC = 0.99) and very good with TTE (r = 0.88, ICC = 0.90) and TOE (r = 0.91, ICC = 0.87). Reproducibility for FS was excellent for TTE-derived M-mode images (ICC = 0.97) and very good for TOE (ICC = 0.87). Bland–Altman analysis reiterated the high level of intra-observer agreement for all three modalities, particular CMR (Figure 6).
Mean absolute differences in paired ejection fraction (EF) measurements. Bar chart illustrating intra-observer data for each modality (white) and inter-observer data (grey). Statistical significance of the differences between modalities displayed. *P < 0.05, ****P < 0.0001, ns = non-significant, CMR: cardiac magnetic resonance, TTE: transthoracic echocardiography, TOE: transoesophageal echocardiography, EF: ejection fraction. Intra-observer agreement. Linear correlation chart with regression line for each comparison pair (a, b, c), with correlation r value and intraclass correlation coefficient (ICC). Corresponding Bland–Altman plots (d, e, f) with mean bias, standard deviation and 95% limits of agreement displayed. CMR: cardiac magnetic resonance, TTE: transthoracic echocardiography, TOE: transoesophageal echocardiography, EF: ejection fraction.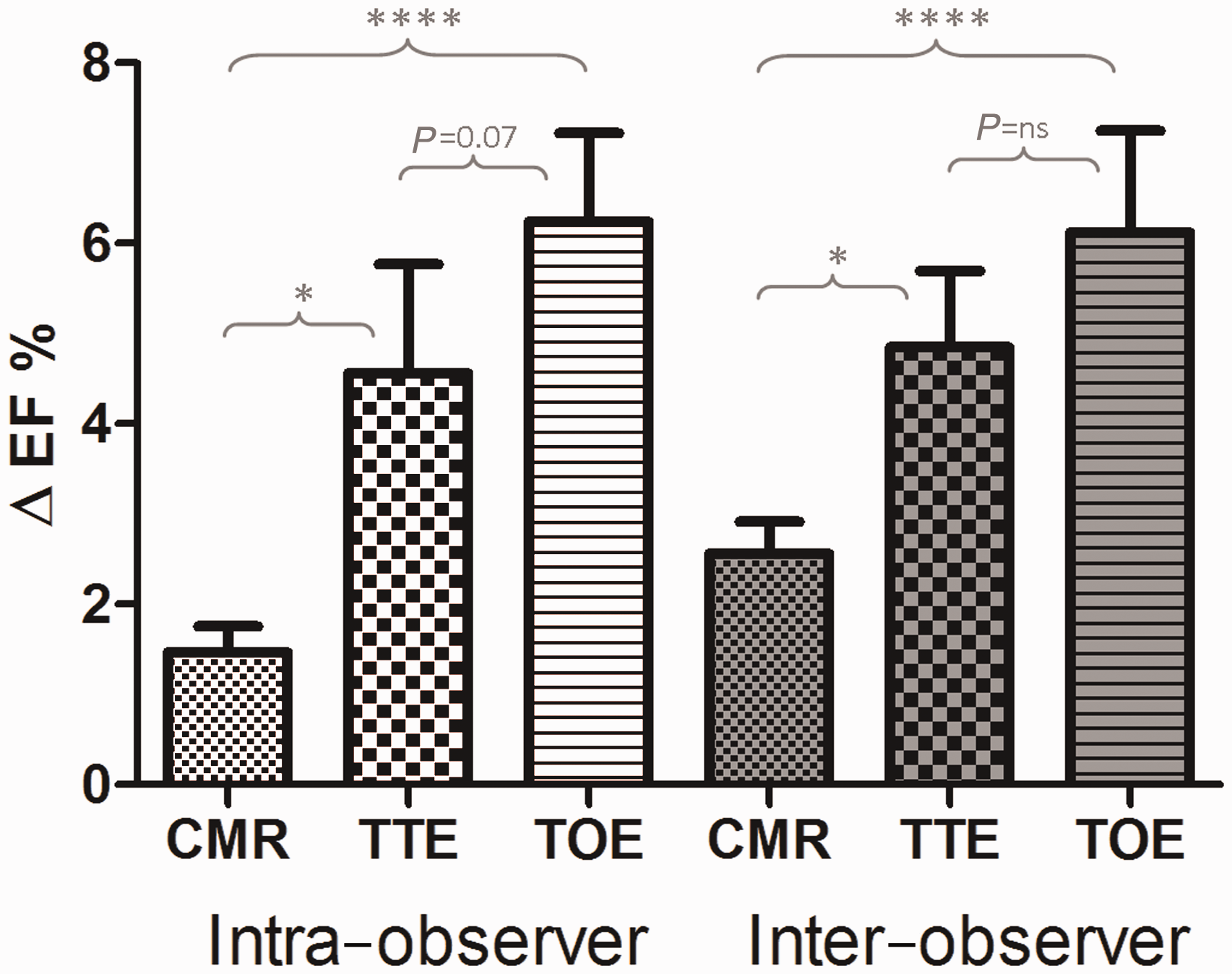
Inter-observer agreement
Mean differences in EF between two independent observers were lowest for CMR (ΔEF = 2.6 ± 1.5%) compared with TTE (ΔEF = 4.6 ± 5.4%, P < 0.05) and TOE (ΔEF = 6.3 ± 5.7%, P < 0.01) (Figure 5). Inter-observer agreement was excellent for CMR (r = 0.97, ICC = 0.98) and very good for both TTE (r = 0.87, ICC = 0.89) and TOE (r = 0.87, ICC = 0.84). Regarding FS, TTE-derived M-mode demonstrated excellent reproducibility (ICC = 0.93), whereas TOE-derived FS was only moderate in agreement (ICC = 0.71). Bland–Altman analysis (Figure 7) revealed a trend for one observer to systematically measure CMR EFs as marginally higher (bias 1.71). For TTE, there was no overall significant bias, but differences were found between observers at either end of the EF spectrum. As with CMR, TOE analysis was systematically rated higher by one reader, but with a greater variation (bias 4.32 ± 6.74).
Inter-observer variation. Linear correlation chart with regression line for each comparison pair (a, b, c), with correlation r value and intraclass correlation coefficient (ICC). Corresponding Bland–Altman plots (d, e, f) with mean bias, standard deviation and 95% limits of agreement displayed. CMR: cardiac magnetic resonance, TTE: transthoracic echocardiography, TOE: transoesophageal echocardiography, EF: ejection fraction.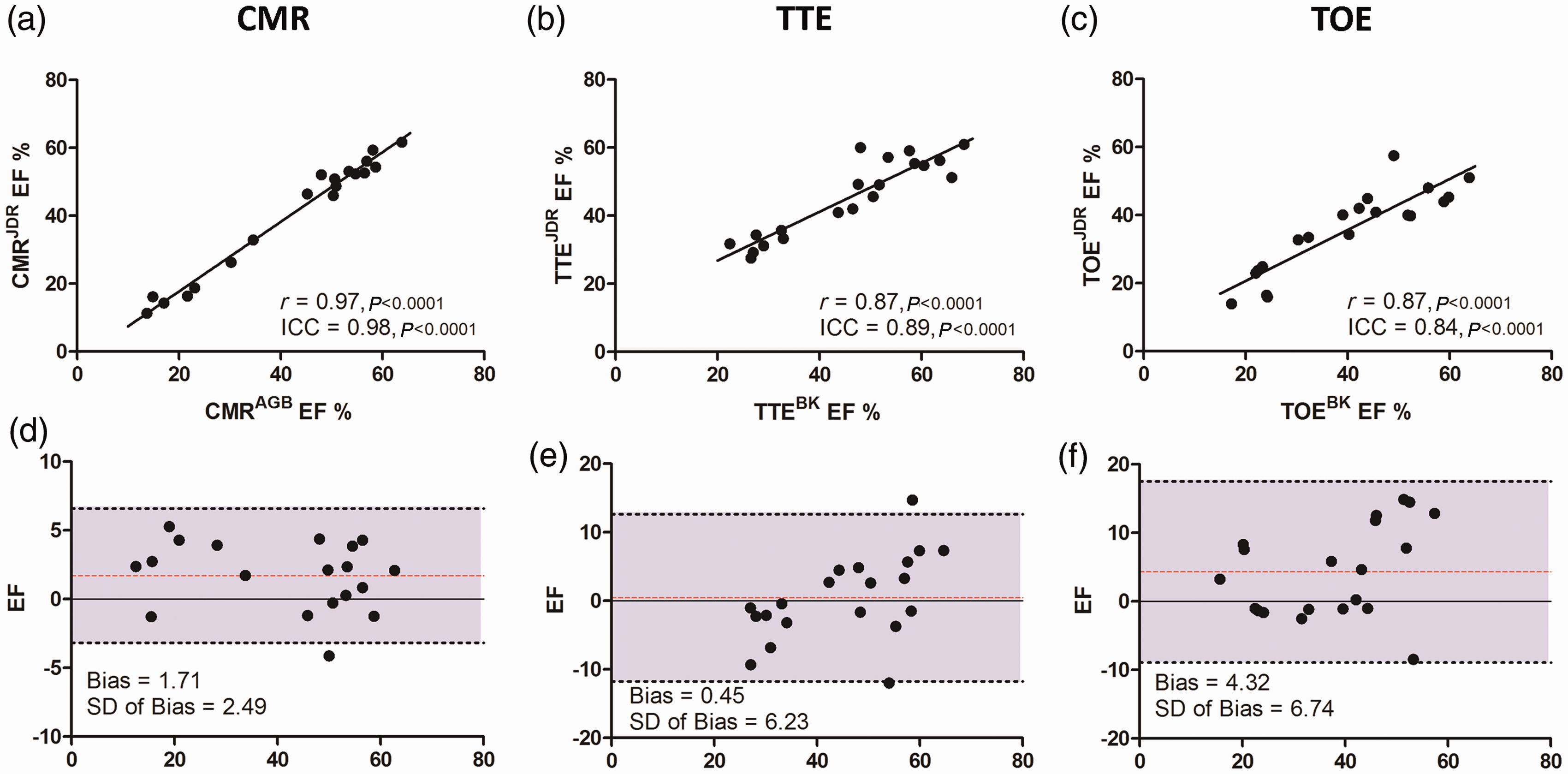
Discussion
This study demonstrates: (1) The first systematic comparison of 1.5 T CMR, TTE and TOE in rodents and suggests that 1.5 T CMR is a feasible and highly reproducible method of evaluating LV EF in rats; (2) Support for TOE as a feasible and reproducible method to evaluate LV function; (3) CMR has greater reproducibility than the alternative modalities studied, with excellent intra- and inter-observer agreements.
Utility of 1.5 T CMR
We believe this is only the second description of balanced SSFP cine imaging of rodents using 1.5 T CMR. 10 This cine sequence has been widely adopted in clinical practice given the high signal-to-noise ratio (SNR) and excellent subendocardial contrast that it provides. By contrast, in the few other published studies using 1.5 T CMR, investigators have used older generation spoiled gradient echo sequences, (e.g. FLASH).5,11–13
CMR imaging of small animals had until recently been the preserve of research-dedicated high-field (≥4.7 T) magnets in conjunction with customized surface coils. In order to accurately image such small anatomy, spatial resolution of up to 10-fold higher than that used for humans is required, inevitably causing significant SNR losses. 14 Higher magnetic fields and small/customized coils can help boost signal to compensate for this obstacle, hence the use of this hardware in the few institutions where it is available. However, 1.5 T CMR scanners are the principal MR technology used in clinical practice, consequently this equipment is more widely available to researchers. Accordingly, the utility of small animal imaging on 1.5 T scanners is of significant interest to potential investigators. At present, reports have documented the utility of 1.5 T CMR in evaluating cardiac structure/function for MI,7,12,15 myocarditis, 16 transplant rejection,11,13 stem cell tracking 10 and myocardial mass. 17 In only one study was an alternative imaging modality used to provide a comparison, 7 which demonstrated a high correlation between single slice CMR and transthoracic M-mode FS (r = 0.86). We have demonstrated that it is feasible to conduct rat CMR on a 1.5 T scanner using clinical software in combination with a conventional carotid coil. This provided image quality qualitatively rated as superior to both TTE and TOE, thereby enhancing the potential for accurate and reproducible data to be collected.
This study shows a moderate–strong correlation of CMR to both echocardiographic-based techniques. Differences in EF measurements are expected, given the inherent differences in how the LV is interrogated by the different modalities. TTE usually depends upon a single measurement – either 1-D M-mode evaluating two opposing myocardial walls, or a short axis slice at a solitary LV level (usually mid-LV/papillary muscle level). In comparison, CMR allows a complete assessment of the entire LV by means of contiguous LV slices with no gaps, as utilized in this study (Figure 1). Accordingly, as the entire LV is evaluated it is more likely to be a representative assessment of ventricular function. It was notable that in rodents with extremely poor LV function (EF < 20%) as determined by CMR, TTE tended to provide higher EFs with none recorded as being below 20%. Reliance on a single echocardiography measure can therefore cause tighter grouping of measurements, so making it more difficult to discern smaller differences in function.
Reproducibility
For 1.5 T CMR to be a feasible alternative, analysis of the images obtained must be highly reproducible. The findings from this study show this to be conclusively the case, with excellent intra- and inter-observer agreements observed for CMR. Rates of intra- and inter-observer agreements were also high with both TTE and TOE, albeit lower than with CMR. The very low variability observed between repeated EF measurements by CMR has important implications for study sample sizes used in research projects. For experiments using EF as an endpoint, the sample size will be determined by the expected effect of the therapy, but importantly, also the variability of EF measurement in a population. The implication of the improved reproducibility with CMR is that it enables the systematic detection of smaller changes in ventricular function and therefore potentially allows a substantial reduction in sample size. This is of considerable scientific, ethical and economic values.
Reproducibility is crucial when serial imaging is performed to monitor function over time and CMR provides a number of advantages over echocardiographic-based techniques in this respect. In addition to the breadth of LV coverage discussed above, CMR also allows LV short axis slices to be prescribed at clearly identifiable anatomical sites in a truly perpendicular manner. When the short axis stack of three slices (3 mm slice thickness, no intersection gap) is prescribed the alignment is checked in two orthogonal (the 2 - and 4-chamber) views, thereby ensuring truly perpendicular images are obtained. This reduces the impact of partial volume effects or undue influence of an obliquely-bisected myocardial wall.
FS data provided by TTE M-mode had excellent intra- and inter-observer reproducibility. One-dimensional data, however, are inherently limited with the provision of information about only two opposing walls, thereby underestimating the potential impact of the remaining myocardium on ventricular function. This is particularly important with disease states associated with regional myocardial dysfunction such as ischaemia/infarction. By contrast, CMR provides almost complete LV coverage in conjunction with excellent reproducibility.
Utility of TOE
Very few studies have described the use of TOE in small animals.18–20 These have used intravascular ultrasound18,20 which has high frequency/resolution, yet is not readily steerable, thereby limiting the potential views that can be obtained. However, one study has utilized ICE to conduct TOE examinations and demonstrated that a complete study, including Doppler acquisitions, is possible in rats. 19 The vertical orientation of the long axis of the rat heart lends itself to TOE imaging and the ICE catheter readily identifies the 2-chamber view, with minimal radial manipulation of the probe. By directing the ICE probe into the stomach, a transgastric short axis view can be obtained with more assertive manipulation. In our study, the first comparative study of ICE-derived TOE measures of EF, we used the 2-chamber image as our default view given the ease and reproducible nature of obtaining this view. This provided a 2-D assessment of the anterior and inferior walls, allowing the calculation of EF, and also FS by manually measuring mid-LV cavity dimension. We observed TOE to have a greater correlation with CMR-derived EF measurements than with TTE. Accordingly, it is possible that the long axis view was more representative of the more extensive assessment provided by CMR, than the single mid-LV slice with TTE. Notably image quality was rated very similar to TTE but not as highly as CMR.
There are a number of scenarios where TOE (or CMR) may be advantageous to TTE imaging. Following thoracic surgery, wound margins may be raised, large intrathoracic collections or residual air may be present, 21 therefore limiting optimal image views with TTE, which can be circumvented by transoesophageal imaging. It is feasible to perform TOE intraoperatively to guide procedures, and for researchers investigating posterior cardiac structures (e.g. atria and pulmonary veins), TOE would also be particularly suitable. We used the same ICE catheter, itself previously used once clinically, for all 20 rat examinations. As ICE catheters are increasingly used in many interventional/electrophysiological cardiology centres, availability should not be limiting. Accordingly, for institutions which do not have easy access to expensive high frequency TTE transducers, ICE TOE is an economically and scientifically sound alternative.
Limitations
Cardiac MRI of rodents takes 1.5 T MRI to the edge of its technological capabilities. Optimal spatial and temporal resolutions, both readily achievable with high-field scanners, is not possible on clinical scanners and compromises in these factors are inevitable. The spatial resolution of the CMR sequence used in this study is marginally lower than other investigators have used, but we considered this to be a better balance with higher temporal resolution, which allowed a minimum of eight phases per cardiac cycle to be acquired. We found that by optimizing other aspects, particularly ECG-trace quality, use of 3 mm thick cine slices, signal averaging and contemporary balanced SSFP software (the latter three enhancing SNR), that high-quality images could be reproducibly obtained. This study evaluated 3-D CMR against TTE and TOE, which will in part explain the favourable results for CMR. Bi-dimensional techniques, such as the area–length method, can provide increased accuracy in EF determination compared with the techniques used in this study. We also acknowledge that relatively low frequency echo transducers were utilized (9.2 MHz), largely as this was the highest frequency transducer available to our unit, which is likely to be the case for many research groups. However, for those facilities with access to high frequency research-dedicated transducers, superior echo image quality should be obtained which might enhance the accuracy and reproducibility of the echo data.
Conclusion
This is the first study to systematically evaluate 1.5 T CMR, TTE and TOE in rodents. It shows 1.5 T CMR to be feasible, productive of high-quality images with extremely high reproducibility. Moderate–strong correlation was observed between all three modalities, identifying all technologies as potential options for researchers evaluating ventricular function in rats. The availability, associated costs and institutional expertise with these imaging modalities will determine the equipment used, but these data suggest that CMR offers distinct advantages even at 1.5 T with conventional coils and software.
Footnotes
Declaration of conflicting interests
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
