Abstract
This study was undertaken to assess the suitability of fentanyl/fluanisone (‘Hypnorm’, VetaPharma; 0.315 mg/mL of fentanyl citrate and 10 mg/mL of fluanisone) alone or combined with midazolam in rhesus macaques. Fifteen rhesus macaques requiring sedation for veterinary procedures received an intramuscular (IM) dose range of Hypnorm from 0.01 mL/kg to 0.3 mL/kg either alone or combined with 0.5 mg/kg of midazolam. To reverse the sedation, flumazenil in combination with either naloxone, buprenorphine or butorphanol was administered intravenously (IV) or IM. Rhesus macaques were successfully sedated with 0.1 mL/kg of Hypnorm and 0.5 mg/kg of midazolam, and sedation was partially reversed by the administration of flumazenil and either naloxone or buprenorphine. However the primates remained slightly sedated and were only released into their home cage several hours post recovery. Butorphanol failed to induce recovery and caused marked respiratory depression. The neuroleptanalgesic combination, Hypnorm and midazolam, effectively immobilized rhesus macaques and was reversible with a combination of flumazenil and either naloxone or buprenorphine.
Fentanyl combined with fluanisone is a powerful neuroleptanalgesic combination that produces heavy sedation and analgesia in rabbits and rodents. In these species the addition of a benzodiazepine produces muscle relaxation and surgical anaesthesia. In most species, this combination produces only moderate respiratory and cardiovascular depression and it can easily be partially reversed by the administration of μ-antagonist agents such as naloxone and butorphanol, and also by buprenorphine.1–3 Given the successful widespread use of this combination in rodents and lagomorphs, it could be of value as an option for providing chemical restraint in non-human primates (NHPs).
Neuroleptanalgesia with a combination of fentanyl and droperidol (‘Innovar Vet’) in NHPs has been reported, 4 however there appear to be no reports of the use of fentanyl/fluanisone either alone or in combination with a benzodiazepine. This study was undertaken to assess the suitability of fentanyl/fluanisone (‘Hypnorm’, VetaPharma) alone or combined with midazolam in rhesus macaques (Macaca mulatta).
Animals
The facility was accredited by the Home Office (the relevant UK Authority) and complied with the Animal Scientific Procedure Act 1986 and the European Directive 2010/63/EU. The use of these primates for research was authorized by the UK Home Office (PPL60/4560; PPL70/7976; PPL60/4041; PPl60/4095), and the Newcastle Animal Welfare and Ethical Review body. The anaesthetic procedures did not require specific ethical approval as they were undertaken under the Veterinary Surgeons Act 1966. This study was performed at the Comparative Biology Centre, Newcastle University, UK.
The animals were housed in indoor pens with a solid floor and windows, allowing a view of the other pens and the corridors. A minimum floor area of 4.40 m2 was provided for each animal or pair of animals. A smaller pen with a squeeze-back system was located between each housing pen. Enrichment devices and substrate for foraging were provided. Animals were maintained on a light:dark cycle of 12 h:12 h at a temperature of 22℃, with 15 air changes per hour and a relative humidity of 24%. Primates were fed with an adapted old world primate diet (Special Diets Service, Witham, UK), and received tap water ad libitum at the time of the study. Forage mix was provided daily to all the animals (LBS Biotechnology, Horley, UK). All the primates were paired-housed except for two males. The single housing was due to exceptional circumstances unrelated to research protocols. The primates had not been sedated or anaesthetized during the two weeks prior to the start of the study.
Materials and methods
Sedation procedure
Compounds used in the study.
Initial use
A pilot study using one dose of 0.3 mL/kg of Hypnorm intramuscularly (IM) to sedate an animal for a veterinary procedure (intensive headpiece margins care) did not produce a degree of sedation that was sufficient to allow safe handling of the primate. The addition of 0.5 mg/kg IM of midazolam increased the degree of sedation and enabled the procedure to be completed safely. Following this initial use of the agents, two other primates, also requiring veterinary procedures, received 0.1 ml/kg and 0.03 mL/kg of Hypnorm IM and 0.5 mg/kg of midazolam IM, respectively. Both animals were sufficiently deeply sedated. The combination was partially reversed by the administration of naloxone (0.01 mg/kg IM). Although the primates were closely monitored, no details of the effects of these agents were recorded during this initial use of the combination.
Main study
Study design.
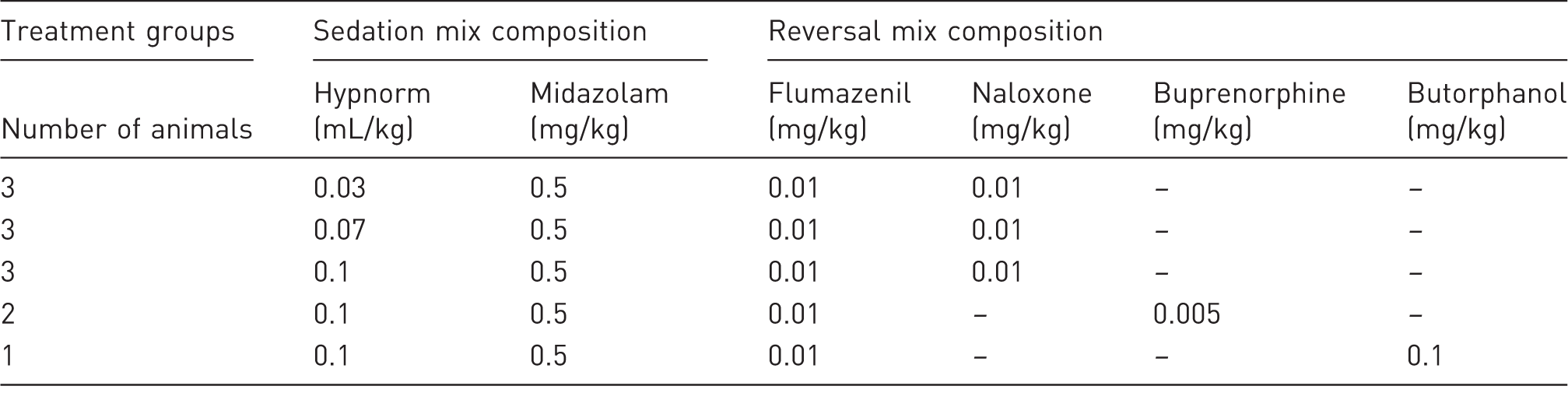
Each millilitre of Hypnorm (VetaPharma, Leeds, UK) contained the equivalent of 0.315 mg of fentanyl citrate and 10 mg of fluanisone. – indicates that the drugs were not administered to the animals.
Score sedation.

Statistical analysis
The physiological parameters, induction time and sedation score, were not normally distributed so a Kruskal–Wallis test was used to compare them between the treatment groups. The recovery times were compared using the Mann–Whitney U-test. The differences were considered significant at P < 0.05. Statistical analysis was performed with SPSS statistics software version 22 (IBM, Armonk, NY, USA).
Results
The rhesus macaques were successfully sedated with 0.1 mL/kg of Hypnorm and 0.5 mg/kg of midazolam, and the sedation was partially reversed by the administration of flumazenil with either naloxone or buprenorphine. In this group the level of sedation was considered to be equivalent to light general anaesthesia, as the palpebral reflex was still present and increases in heart rate and respiration rate were observed while the intradermal tuberculin injection and venous blood sampling were performed.
Physiological parameters results.
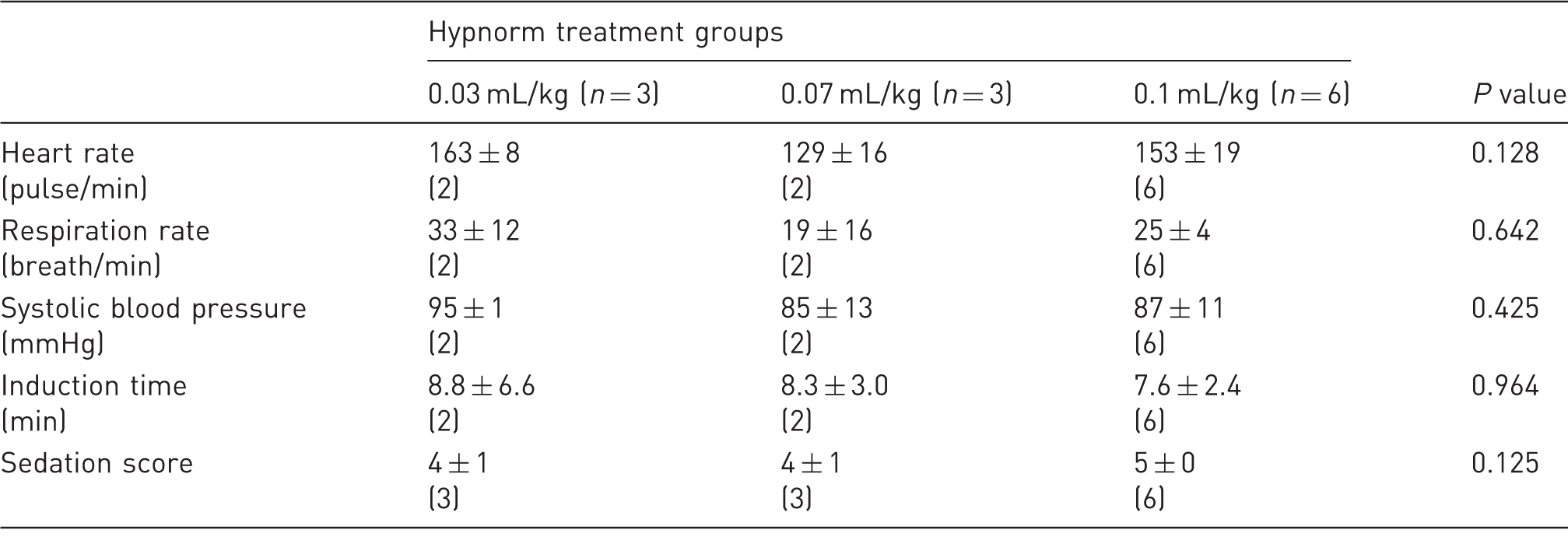
Data are shown as mean ± 1SD. n represents the number of animals per treatment group. The numbers within brackets represent the number of animals that provided data. In the two groups 0.03 mL/kg and 0.07 mL/kg, one primate did not reach a suitable level of sedation that could allow recording of vital parameters. The parameters were compared with the non-parametric Kruskal–Wallis test and a significant threshold at 5%.
The righting reflex return times were not significantly different between the groups that received naloxone or buprenorphine (naloxone: 26 ± 10 s, buprenorphine: 26 ± 18 s; P = 1). Butorphanol failed to induce a rapid recovery and the primate remained deeply sedated. A marked respiratory depression developed and the animal was unable to maintain an oxygen saturation of > 85% without oxygen supplement. Additional administration of a dose of 0.01 mg/kg of flumazenil and two doses of 0.01 mg/kg of naloxone were required to reverse sedation and respiratory depression.
After the return of the righting reflex, all the animals were slightly sedated and were only released into their home cage several hours post recovery. Clinical assessment by the animal care staff indicated that full recovery was not complete until 4 to 6 h after the return of the righting reflex.
Discussion
Veterinary or experimental procedures can necessitate the chemical immobilization of rhesus macaques, which is often performed using ketamine. This agent has a wide safety margin but it is also associated with several side-effects including pain on injection, tissue necrosis, seizures,7–9 and impairment in cognitive development in young primates. 10 Ketamine may also be contraindicated in animals considered to be at risk of having raised intracranial pressure.11,12 Neuroleptanalgesic combinations such as fentanyl/droperidol in humans produce sedation and analgesia with minimal effects on cardiovascular function, but some respiratory depression can occur.13,14 These combinations can be partially reversed with μ-antagonists or partial agonists such as naloxone, buprenorphine or butorphanol, 15 and this can reduce the recovery times. 16
This report describes the first assessment of the use of a combination of Hypnorm and midazolam in rhesus macaques. Although a dose of 0.3 mL/kg of Hypnorm IM has previously been recommended, 15 , 17 this dose recommendation is based only on a single report of the use of fentanyl/droperidol. 4 Fentanyl in combination with fluanisone (Hypnorm) at this dose did not immobilize rhesus macaques sufficiently to allow safe handling, and the addition of 0.5 mg/kg of midazolam IM to 0.1 mL/kg of Hypnorm (fentanyl citrate: 0.031 mg/kg; fluanisone: 1 mg/kg) was necessary to produce a complete loss of the righting reflex. Fentanyl has potent analgesic effects but, at the doses used in this study, the depth of anaesthesia was only sufficient to allow minimally invasive procedures, such as superficial vein catheterization, to be undertaken. Higher dose rates of Hypnorm might provide deeper planes of anaesthesia, sufficient to perform more invasive surgical procedures, but higher doses might also increase the degree of respiratory depression.
In rodents and rabbits, Hypnorm, in combination with benzodiazepines, produces a surgical plane of anaesthesia.1,18,19 This combination has minimal effect on cardiovascular function and tissue perfusion in these species.3,18 Although the use of these agents produces moderate respiratory depression, no severe hypoxaemia associated with cyanosis or major changes in blood gas values have been reported in rabbits and rodents.1,19 The effects of the combination in rhesus macaques appear to be more variable. Fentanyl is a potent full μ-agonist opioid that produces depression of the respiratory centre and this effect is potentiated if benzodiazepines are administered, 20 although macaques appear to be less sensitive to this effect. 21 However one of the primates involved in this study developed a very low respiratory rate (8 breaths/min). This observation and the variable level of sedation in the low (0.03 mL/kg) and middle (0.07 mL/kg) dose groups of Hypnorm indicate a marked individual variability in response to the combination. The variation in expression of μ-opioid receptors observed in macaques might explain this observation. 22 Because of the possible respiratory depression severity, particular attention should be given to providing oxygen and monitoring tissue oxygenation (SpO2).
The use of a μ-antagonist (naloxone) or partial agonist (buprenorphine) was effective in reversing the effects of fentanyl and speeded recovery, as has been shown in other species.2,23,24 In the present study three agents, naloxone, buprenorphine and butorphanol, were selected to partially reverse the neuroleptanalgesia based on previous reports in rodents and rabbits.1,2,19 In contrast to rodents and rabbits, butorphanol did not decrease sedation, and because of the marked respiratory depression that developed in the first animal to receive this agent, no further animals received butorphanol. Butorphanol has not been reported to cause significant sedation in rhesus macaques but respiratory depression can occur at dose rates as low as 0.001 mg/kg. 25 The deep sedation was probably due to the combined effects of butorphanol, fluanisone and midazolam. In other animal species, combinations of either phenothiazines or butyrophenones with butorphanol produced marked sedation.26–29 Naloxone is an antagonist at μ, κ and δ opioid receptors and reverses both full μ-agonist or agonist/antagonist opioids such as fentanyl and butorphanol. 23 In the present study naloxone administration reversed the neuroleptanalgesia and also the depressant effects of butorphanol. Buprenorphine is a partial μ-receptor agonist that produces analgesia but also reverses respiratory depression and narcotic effects of full μ-agonists such as fentanyl.23,30,31 However, respiratory depression has been reported in one paediatric human patient with a therapeutic dose of buprenorphine administered following intraoperative use of fentanyl. 32 In the present study buprenorphine, like naloxone, was effective in reversing the sedation and did not cause respiratory depression.
The prolonged light sedation observed after the return of the righting reflex was considered to be due to the persistent effects of fluanisone and midazolam. Fluanisone is a butyrophenone, a class of drugs with central nervous system depressant and sedative proprieties.29,33 However no antagonist was available at the time of the study so the effects of fluanisone could not be reversed. The benzodiazepine antagonist currently available (flumazenil) has a considerably shorter half-life than that of midazolam, so that re-sedation can occur. 34 The prolonged sedation observed in the present study is also very likely due to the remaining fluanisone. Although the prolonged recovery time with Hypnorm and midazolam in combination compares poorly with recovery times from ketamine,16,35 NHPs were only lightly sedated and were not ataxic.
In conclusion, this preliminary study has demonstrated that the neuroleptanalgesic combination Hypnorm (fentanyl/fluanisone) and midazolam at dose rates of 0.1 mL/kg (fentanyl citrate: 0.031 mg/kg; fluanisone: 1 mg/kg) and 0.5 mg/kg IM respectively, immobilized rhesus macaques, and the effects were partially reversed by the administration of either naloxone or buprenorphine combined with flumazenil. The sedation and the analgesia provided by this combination enabled routine minimally invasive veterinary procedures (intradermal tuberculin injection and blood sampling) to be undertaken. However we recommend that oxygen should be provided and respiratory function monitored because of the risks of respiratory depression. The primates remained lightly sedated for several hours following recovery of their righting reflex. Although Hypnorm could provide a useful alternative to ketamine in particular research protocols, this agent is only available in Europe, and this will limit its use. However it might be of value to assess the use of other combinations of opioids and tranquillizers in this species. Further studies should be conducted to determinate any advantages (e.g. stability of physiological parameters, emergence quality) of this combination in comparison to other protocols such as ketamine combinations.
Footnotes
Acknowledgements
The authors would like to thank Mrs Denise Reed, Ms Caroline Fox, Mr Ashley Waddle, Ms Stevie O’Keefe and Ms Carrie Todd for their technical support during this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
