Abstract
Refinement of scientific procedures carried out on protected animals is an iterative process, which begins with a critical evaluation of practice. The process continues with objective assessment of the impact of the procedures, identification of areas for improvement, selection and implementation of an improvement strategy and evaluation of the results to determine whether there has been the desired effect, completing the refinement loop and resulting in the perpetuation of good practice. Refinements may be science-driven (those which facilitate getting high-quality results) or welfare-driven or may encompass both groups, but whatever the driver, refinements almost always result in benefits to both welfare and science. Refinements can be implemented in all aspects of animal use: improved methodology in invasive techniques, housing and husbandry, and even statistical analyses can all benefit animal welfare and scientific quality. If refinement is not actively sought, outdated and unnecessarily invasive techniques may not be replaced by better methods as they become available, and thus outdated information is passed down to the next generation, causing perpetuation of old-fashioned methods. This leads to a spiral of ignorance, leading ultimately to poor practice, poor animal welfare and poor-quality scientific data. Refinement is a legal and ethical requirement, yet refinements may not always be implemented. There are numerous obstacles to the implementation of refinement, which may be real or perceived. Either way, in order to take refinement forward, it is important to coordinate the approach to refinement, validate the science behind refinement, ensure there is adequate education and training in new techniques, improve liaison between users and make sure there is feedback on suitability of refinements for use. Overall, refinement requires a coordinated ongoing process of critical appraisal of practice and active scrutiny of resources for likely improvements. In the busy world of biomedical research, this process needs help. In order to develop these themes further, a workshop was held at the LASA Winter Meeting 2006, UK, to assist in identifying potential obstacles to refinement, and then to explore and develop strategies for overcoming these obstacles in key areas. A range of strategies appropriate to different circumstances was identified, which should facilitate the implementation of refinements.
Refinement of scientific procedures carried out on animals to minimize adverse effects and to maximize the scientific benefit gained is a legal and ethical requirement, under the Animals (Scientific Procedures) Act 1986, the Animal Welfare Act 2006, and the Animal Health and Welfare Strategy for Great Britain, and also in the wider European and worldwide context (
In attempting to define or describe animal welfare, there is broad consensus that the ‘five freedoms’, as laid down by the British Farm Animal Welfare Council in 1979, can be taken as general indicators of animal wellbeing (FAWC 1979). These are freedom from hunger and thirst, freedom from discomfort, freedom from pain, injury and disease, freedom to express normal behaviours and freedom from fear and distress. Scientific research has the ability to impact on all these. For example, food and water restriction paradigms are commonly used in behaviour studies, metal cages and grid floors can produce discomfort, experimental surgery can cause pain, single housing and restricted space can impact on behaviour and handling methods or procedures can cause fear or distress. There is inevitably tension caused by the potential of science to impact on welfare and the need to maintain the welfare of animals used (Smith 2006). Researchers have an obligation to minimize the impact of the procedures they do on these five freedoms through the active implementation of refinements.
Fundamentally, the drivers for refinement are always the same, improving scientific quality or improving animal welfare, but on a practical level, cultural and organizational factors will impact on how change (including refinement) occurs in different settings.
Culture
The concept of academic freedom is highly prized, sometimes at the expense of more effective management. Within an academic environment the inadequacy of implementation of refinements may be recognized, but there is often a failure to take any action to address this because of the lack of management criteria such as key performance indicators and use of standard operating procedures (SOPs). Conversely, there is much within the culture of pharmaceutical research and development that encourages innovation including refinement. Within the drug hunting process, highly organized and disciplined teams work towards common goals. There are good communication channels and a focus on quality assurance. Investment in facilities, people and equipment is strong, with an inherent focus on technology and innovation, but there is not an ‘open cheque-book’ and a business case must be made for all expenditure to be effectively prioritized.
Drivers for refinement
Academic research tends to be very focused into narrow areas. Scientists are strongly driven to do high-quality research in their field, but may lack similar drive when evaluating the influence of methodology on the scientific quality of their results or on the welfare of animals used. The implementation of the 3Rs is seen almost as a separate subject area, whereas it would be better if seen as a normal part of the research package and integrated into it. Within the pharmaceutical sector, strong corporate drivers for consistency and harmonization within global organizations can be used to bring about refinement (e.g. housing improvements) and in an increasingly tough external environment, a strong focus on corporate reputation can also bring useful leverage to welfare initiatives. The project focus in drug discovery can provide impetus to refinement, such that refinements driven by project need happen ‘automatically’ but conversely refinement cannot be allowed to interrupt project progression. It can be more difficult to influence practice (e.g. change an anaesthetic regimen or animal model) with an established screening cascade: timing and opportunity are critical.
Scale
Academic research groups tend to be small and work independently, so refinements implemented at the individual project level have little impact in a wider institutional setting, whereas in industry, change can take time but when it happens it can typically be on a large scale across a whole department or site.
The refinement loop
Refinement is an iterative process, which begins with a critical evaluation of practice, leading to recognition and assessment of poor or suboptimal welfare, identification of the causes of this, selection of improvement strategies and implementation of these strategies. Any changes then have to be evaluated for their efficacy, thus the process begins again, forming the refinement loop.
Critical evaluation of practice
Researchers have to look critically at what they do and identify if there is a better way of conducting the research that will minimize welfare implications and maximize the scientific output, thus improving the harm: benefit balance and the justification for doing the work. It is particularly important that this is seen as an ongoing requirement: new developments in refinement may turn what was yesterday's best practice into today's outdated methodology.
Objective assessment of animal welfare and scientific quality
This requires first and foremost an understanding of wellbeing and what particular animals need. Only then is it possible to identify appropriate parameters to assess wellbeing and scientific quality in a meaningful manner. Sufficient resources have to be provided to perform the assessments effectively. This requires the researcher to schedule observations appropriately to maximize the likely detection of problems, to recognize where there is deviation from normal, taking into account species, strain and inter-animal differences and to be able to identify when there is room for improvement.
Selection of improvement strategies
Having identified that there is a problem and a likely cause for this, the researcher has to be able to select an appropriate improvement strategy. This requires active and effective utilization of an ever-increasing variety of sources of information and advice. This may include the veterinarian and animal care technicians, other researchers, journals or internet-based resources.
Implementation of improvement strategies
The implementation of any new methodology is accompanied by a period of inexperience, during which time it may not be possible to discern any improvement, and indeed may lead to a period of increased losses while researchers develop the necessary skills for the new technique. This may cause researchers to question the new method. Systems should be in place at institutions to deal appropriately with such a reaction, and make sure that the refinements are implemented so that the long-term benefits are realized. Researchers must also recognize that this will be the case and ensure that they have help, advice and support available while they are learning the new technique.
Evaluation of improvement strategies
Having implemented an improvement strategy, this must be evaluated to determine whether there has been an improvement. It may be that the new method is found to be better, or that the current method is confirmed as being the best available method: either way the information is valuable to others in the field. This information should then be disseminated to others working in the same area, ideally by publication in mainstream journals, but as a minimum it should be used for internal reference.
This process forms the refinement loop (Hartley et al. 2004) (Figure 1).
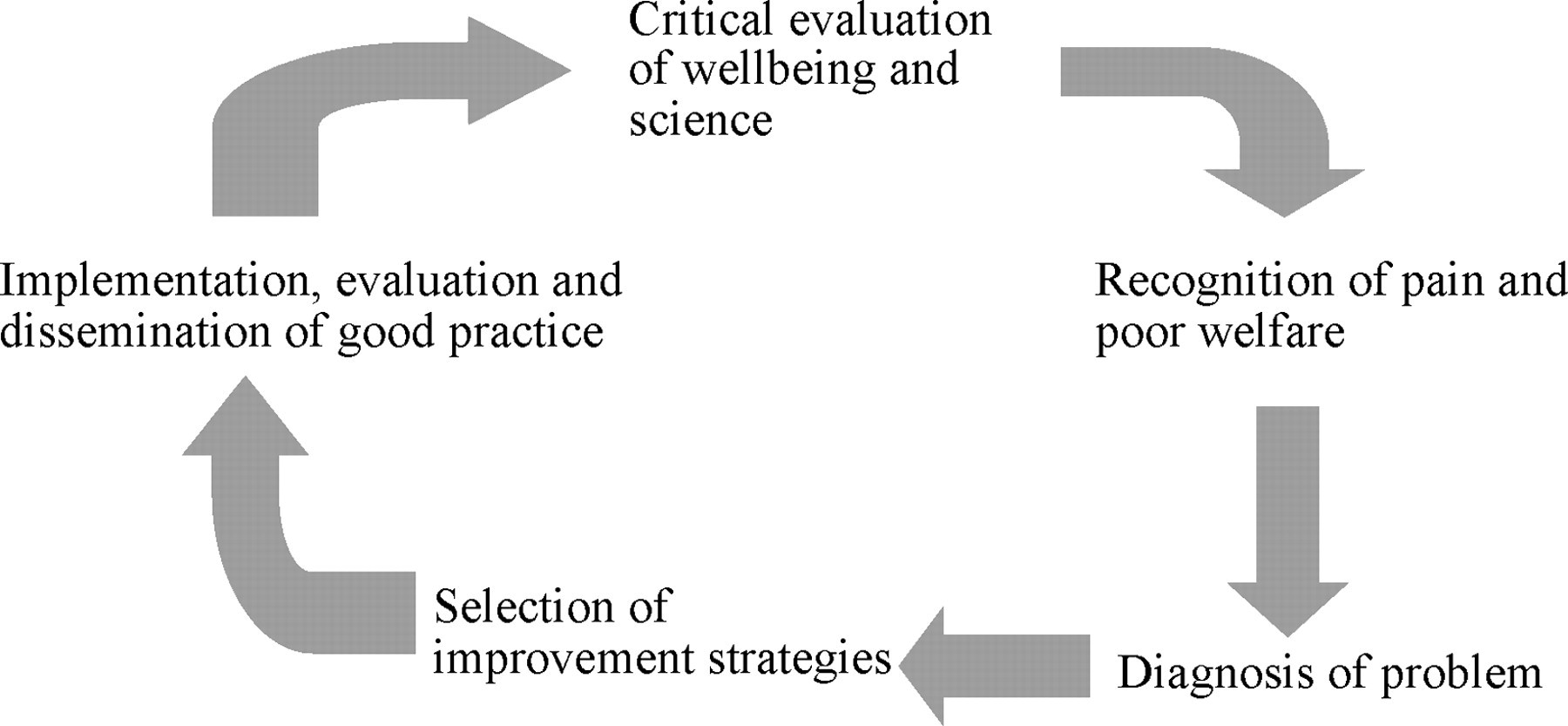
The refinement loop
Obstacles to refinement
Obstacles to refinement may be real or perceived, each requiring different strategies to overcome them. Real obstacles such as lack of money, facilities, personnel or equipment will require a patient, tactical approach using all organizational levers available to overcome them. Perceived obstacles arise from misunderstanding or misinformation and need a different approach: providing evidence-based strategies to tackle key issues, educating and informing to allay unfounded fears and influencing and persuading to change attitudes.
Numerous factors have been identified, which can impede the progress of refinement (Karas 2006). Particular issues include:
Failure to actively seek refinement leads to a situation where techniques are handed down unchanged through generations of practitioners. This is particularly the case in biomedical research since although new researchers undergo a very basic training course the majority of their learning is ‘on the job’. This leads to a spiral of ignorance, in which techniques used become further and further removed from ‘best practice’ (Figure 2).

The spiral of ignorance
Making refinement work
Experience from both industry and academia has shown that it is possible for refinement to be taken forward. Important factors for success include determining what type of obstacle is involved, then determining the appropriate strategy to tackle it, including which people are the best ones to help. A lack of resources requires a different approach to a problem of attitude or knowledge. Strategies for success include capitalizing on opportunities, influencing and persuading, engaging with stakeholders to tackle concerns and good team working. Using a tactical team approach and all organizational levers is critical for success.
In a workshop held at the LASA Winter Meeting 2006, UK, potential obstacles were explored in more depth. Participants were divided into four groups of between eight and 10 participants, and each group was tasked with identifying strategies for overcoming obstacles to refinement in an area where the main issues were attitudinal.
The groups considered four key areas:
(1)
The key issues identified were inexperience and lack of confidence. They simply may not have the knowledge or confidence to try new techniques or challenge their supervisors. Also, new researchers are at the point of unconscious incompetence (Chapman 2007) (Table 1) – they may not recognize that they have a problem. This may also apply to more senior researchers, who have always used a particular technique and do not recognize that times have moved on.
Conscious competence-learning matrix (adapted from Chapman 2007)
These issues can be addressed by considering training, mentoring and providing a culture of support, which is the responsibility of the
New researchers could benefit from having a person who is not their supervisor or manager appointed to provide support and advice. This person could act as a mentor, being available to listen to problems and offer help in a confidential way. Supervision is the responsibility of the
The culture at an establishment very much depends on the attitude of its leader. It is necessary to promote a culture of advice-seeking, so that new researchers feel confident to ask for help in areas where they are uncertain. New researchers must recognize that they are not competent just because they have completed training. The veterinarian and animal care technicians need to be available to give advice, and their expertise should be respected. A culture of support, not a culture of blame, needs to be fostered by the
(2)
The main issue identified was that it is difficult to get people to accept that what they are doing and have always done may no longer be best practice, and that things have progressed. If challenged, the usual reaction is to defend the position held, ‘I've always done it this way and never had any problems’. This is a difficult issue to tackle and may take time to change. Change may be incremental and it may be necessary to go backwards before moving forwards while scientists are learning a new up-to-date technique. Different strategies may be needed for different cases. The terminology used is important, and this may need plenty of gentle persuasion and diplomacy. It is necessary to get researchers on side, so they will come back and ask for help again.
Here, it is necessary to explore why there is reluctance to change, explain why change is beneficial, and demonstrate that this occurs in practice.
Exploring why there is reluctance to change involves psychology – asking ‘Why do you do it this way?’ comes across as a challenge, and is likely to result in the person defending their position. Rather than attacking the methodology being used, it is more effective to use open, non-threatening questioning, such as ‘Have you considered x?’ Change requires time and effort, additional resources, and acceptance that what has always been done may no longer be appropriate.
Legal and ethical requirements encourage and require refinement, and there may be some leverage in explaining the potential benefits of the proposals. Both the local ERP and funders may look more favourably on proposals using more refined methods. It may also be possible to take advantage of academic competitiveness by citing academic competitors using new techniques or the possibility of attracting new talent (and possibly funding) to the laboratory.
It may be possible to demonstrate that refined methods work better or are more likely to attract funding. This may need some kind of proof, such as good-quality papers or the opinion of other experts to back it up.
Many people can be involved in this process, including the
In the event that there is still reluctance to change and there may be implications for animal welfare, it may not be acceptable to wait to implement refinements. Sometimes the establishment has to be up front and say no. There is an ongoing legal and ethical requirement for refinement to be implemented, and the
(3)
Some scientists may perceive research into refinements as less important or peripheral, and such research is rarely published in mainstream journals where scientists get to see it. Welfare research is perceived as poor-quality science and is not considered relevant to many scientists.
A stock of ‘best-practice’ papers to be used as sources of information can be useful. These should be research evidence based, relevant and multidisciplinary with good statistical analyses and must be critically evaluated before being given to scientists. In particular websites such as the National Centre for the 3Rs (NC3Rs) and Centre for Alternatives to Animal Testing act as useful portals to up-to-date 3Rs information and ‘best practice’. It must be noted that often experiments are condition-dependent, and the conditions at the establishment may be different from those used in the study, which can render the results less useful. The
At present, scientists tend to require proof that refinements will not adversely affect the research that they are doing before they will consider implementing them. However, ideally it should be the other way round: scientists should find proof that the refinement does impact on their research negatively before rejecting a refinement. Also, refinements often improve the scientific quality of the results (Ritskes-Hoitinga et al. 2006): scientists should make it clear that they have evaluated the possible positive effects of any refinement on their research before rejecting it. The animals should always get the benefit of the doubt.
(4)
Information about refinements is often passed on by the
A strategy is needed for finding relevant information on refinements simply. This may be achieved by encouraging the involvement of 3Rs experts at the study design stage so that all opportunities for refinement are recognized, encouraging interaction between scientists using intranets or newsletters and setting up fora for the sharing of ideas. Relevant information can come from new staff, laboratory animal science meetings, journals or discussions with experienced colleagues. Most mainstream science groups have 3Rs groups and run workshops, but to maximize the use of appropriate sources of information it may be necessary to employ the services of an
Key points and key personnel for implementation of refinement
ERP = ethical review process
Conclusions
European legislation (the Animals (Scientific Procedures) Act 1986 in the UK) requires implementation of the three Rs.
Refinement is an iterative process starting with critical evaluation of practice.
Failure to actively seek refinement leads to a spiral of ignorance and perpetuation of poor practice.
Numerous obstacles to refinement have been identified, which can prevent refinements from being implemented.
To overcome these, it is essential to coordinate the approach to refinement within an institution or organization. It may be beneficial to identify someone with overall responsibility for this: a ‘refinement champion’ who is responsible for ensuring that refinements are actively implemented at the establishment.
The institution needs to ensure that support is available for key people in developing a culture of care, and that the professional integrity of the experts in welfare assessment is recognized. The animal management team needs to communicate effectively with senior management, to make sure that adequate resources are provided to implement refinements appropriately. There should be a clear process for the implementation and reporting of problems, and the development of a culture of advice-seeking and support, not blame and isolation.
Scientists need to recognize that there is a learning curve to all new methods, and accept that while inexperience may lead to new or increased problems in the short term, there will be longer-term benefits from perseverance.
There is a need to enhance information dissemination, to support and guide scientists through the overabundance of advice and regulations to help them identify what is best practice.
Refinements may be developed to enhance animal welfare, to improve scientific quality or sometimes simply as a defensive reaction to prevent criticism of old-fashioned methodologies. The underlying reason of why a procedure is done matters less than what is done, when, how and by whom, and the validity of the results obtained. For the animal it is not important what we think, what we monitor or how we score it, but what actions are taken that directly affect it (Wolfensohn & Honess 2007): for the animal it is actual quality of life that matters.
