Abstract
Survival of sepsis is related to loss of muscle mass. Therefore, it is imperative to further define and understand the basic alterations in nutrient metabolism in order to improve targeted sepsis nutritional therapies. We developed and evaluated a controlled hyperdynamic severe sepsis pig model that can be used for in vivo multi-organ metabolic studies in a conscious state. In this catheterized pig model, bacteremia was induced intravenously with 109 CFU/h Pseudomonas aeruginosa (PA) in 13 pigs for 18 h. Both the PA and control (nine) animals received fluid resuscitation and were continuously monitored. We examined in detail their hemodynamics, blood gases, clinical chemistry, inflammation, histopathology and organ plasma flows. The systemic inflammatory response (SIRS) diagnostic scoring system was used to determine the clinical septic state. Within 6 h from the start of PA infusion, a septic state developed, as was reflected by hyperthermia and cardiovascular changes. After 12 h of PA infusion, severe sepsis was diagnosed. Disturbed cardiovascular function, decreased portal drained viscera plasma flow (control: 37.6 ± 4.6 mL/kg body weight (bw)/min; PA 20.3 ± 2.6 mL/kg bw/min, P < 0.001), as well as moderate villous injury in the small intestines were observed. No lung, kidney or liver failure was observed. Acute phase C-reactive protein (CRP) and interleukin-6 (IL-6) levels did not change in the PA group. However, significant metabolic changes such as enhanced protein breakdown, hypocalcemia and hypocholesterolemia were found. In conclusion, PA-induced bacteremia in a catheterized pig is a clinically relevant model for acute severe sepsis and enables the study of complex multi-organ metabolisms.
Sepsis is a complex bacteremia-induced pathophysiological state characterized by both cardiovascular and metabolic responses.1–4 Its dynamic character, variability of response, and complicated clinical logistics make it difficult to study the metabolic interaction between different organs in septic patients in a controlled setting.5,6
One characteristic metabolic alteration resulting from sepsis is loss of muscle mass. Loss of more than 25% of muscle mass is incompatible with survival.7,8 Less severe muscle loss contributes to morbidity and mortality, 9 and hinders rehabilitation to normal health and function after recovery. Moreover, intestinal mucosal atrophy, mainly due to reduced protein synthesis, is often present with sepsis, and contributes to increased translocation of bacteria from the gut into the bloodstream.10,11 Increased translocation of gut bacteria perpetuates the septic state. Also, a severe decrease of plasma albumin is associated with higher mortality. 12 Albumin is the principal oncotic protein in plasma, and a decrease in concentration leads to loss of fluid from the vascular space. Finally, the physiological significance of the acute phase response has been debated, 13 but clearly represents a metabolic priority in sepsis. More knowledge about the metabolic changes in sepsis should contribute to more effective nutritional support.9,14
Although the debate over the relevance and validity of various animal models of sepsis is still ongoing,3,15,16 the high morbidity and mortality resulting from sepsis continues to illustrate the scientific and clinical need to further define and understand the basic nutrient metabolism alterations and to improve existing targeted sepsis nutritional therapies. For nutritional therapies in sepsis, an optimal animal model to study metabolic responses in the hyperdynamic septic state should be clinically relevant, controlled, and performed in the conscious state. It should also have a potential therapeutic window, with the possibility for studying multi-organ metabolisms. For this model, the first consideration is that the septic state in the animal model should not exceed severe sepsis. Therefore, septic shock models without potential therapeutic windows would not be valid. Secondly, induced sepsis should start with invasion of the blood stream by microorganisms, usually bacteria. It has been indicated that animal models using live bacteria are the most clinically relevant models.15,17 Thirdly, pig models are preferable as they are omnivores like humans, and the pig has a similar renal, cardiovascular, and gastrointestinal anatomy, physiology and metabolism. Therefore, studying therapeutic nutritional support in a pig model is viewed as a highly translational research model.18,19 Fourthly, the metabolic measurements should preferably be done in a conscious state, because it is more clinically relevant and translational. In addition anesthetic management in sepsis presents challenges, 20 and interferes with metabolism, especially splanchnic metabolism (e.g. down regulation of gut movements, detoxification activity of anesthetics by the liver). Sepsis is associated with clinical symptoms of systemic (body wide) illness such as fever, chills, malaise and a general feeling of sickness. However, as we know from clinical situations, pain is not a primary symptom. Therefore, it is acceptable to measure metabolism in a conscious state as long as the animal is not in shock, which is characterized by multiple-organ failures and the necessity for ventilation with sedation.21,22 Finally, the animal should be instrumented to accommodate painless blood sampling and infusions during any multi-organ metabolism study. 23
Currently, there are no animal models available with these characteristics for relevant metabolic and future nutritional therapies in sepsis. Previously, we have studied the multi-organ metabolism in a 24 h endotoxin-induced catheterized sepsis pig model.24–26 Although endotoxin is the major virulent factor with a Gram-negative bacterium in sepsis, it is possible that important components of the metabolic dynamics are missing in this which are present when sepsis is induced by a live bacterium.15,17
The aim of the present study was to characterize in detail the response in hemodynamics, blood gases, clinical chemistry, inflammation and organ plasma flows in a new Pseudomonas aeruginosa (PA)-induced septic instrumented porcine model. The model was to be used for studying clinically relevant, multi-organ metabolism in sepsis. We modified a widely used, high dose bacteremia porcine septic shock model16,27–30 to obtain a controlled hyperdynamic conscious sepsis model by administrating the bacteria intravenously at a low dose via continuous infusion, while simultaneously preventing low blood pressure by fluid resuscitation. PA was used as it is one of the most frequently isolated bacteria in sepsis in humans in hospital intensive care units, apart from lipopolysaccharides and other virulence factors such as flagella and pili, which inject a set of toxins directly into host cells. 31 The pigs were surgically catheterized and subsequently acclimatized 10 days before the induction of sepsis. This was done to better control the sepsis induction and the subsequent hyperdynamic state, and to perform multi-organ metabolic flux measurements over time in the conscious state.23,24,32
Materials and methods
Animals
Twenty-seven female Yorkshire crossbred pigs (from multiple litters; Metz Farms, Russellville, AR, USA) with an average body weight (bw) of 20–25 kg were used in the present study. General health status was determined by an assigned veterinarian of the animal research facility (Arkansas Children's Hospital Research Institute, Little Rock, AR, USA, AAALAC-certified). The pigs were individually housed in steel pens (2 × 3 m) in a controlled housing, biosafety level 2 facility: large animal cubicle, plastic-coated mesh-style grid floor, room temperature 22–24℃, 12 h light–dark cycle, and standardized food intake (1 kg/day) (Harlan Teklad vegetarian pig/sow grower; Harlan Laboratories, Indianapolis, IN, USA). The pens were enriched with toys to stimulate natural pig rooting behavior and to prevent stereotypic behavior. A heating pad was added to the pen to ensure natural body temperature control post-surgery. Water was provided ad libitum. Clinical examination was carried out on arrival, before surgery (7–10 days after arrival) and frequently (twice daily on the first three/four days post-surgery and then every two/three days until the sepsis study) post-surgery. The present animal study, including its justification for number used, breed and single sex, was approved by the animal experiment ethics committee (IACUC) of the University of Arkansas Medical Sciences (Little Rock, AR, USA).
Surgical procedure
In order to perform multi-organ metabolism studies in the septic pig, catheters were implanted in blood vessels using a surgical procedure, as described in detail previously.23,32 After an acclimatization period of 7–10 days, a pre-surgery clinical exam and anesthesia were performed by an assigned veterinarian. Anesthesia was induced with an intramuscular injection of a mixture of telazol, ketamine and xylazine (PigMix®, dose 0.4 mL/kg bw). With the subsequent intubation of the animal, general anesthesia was started and maintained with isoflurane (2% mixture with oxygen). Via an ear vein cannula, flunixin meglumine (Flunixamine®, 2 mg/kg bw) was administered as an analgeticum and as an antibioticum, and a lincomycin–spectinomycin mixture (Linco-Spectin®, lincomycin 5 mg/kg bw, spectinomycin 10 mg/kg bw) was administered as a prophylaxis. The animal received 500–1000 mL Ringer's lactate during the surgery to maintain its blood pressure and fluid balance. Electrocardiography (ECG), carbon dioxide and oxygen saturation were monitored continuously.
In brief, during midline laparotomy, a catheter was inserted into the abdominal aorta for blood sampling over time, and to monitor mean arterial pressure (MAP). An inferior caval vein catheter was inserted, enabling administration of post-surgery medication and experiment-related infusions. A second arterial catheter was inserted just before the bifurcation, and a splenic vein catheter was inserted to allow infusion of para-aminohippuric acid (PAH) for plasma flow measurements. A Swan Ganz catheter (5 Fr, #132F5; Edwards Life Sciences, Irvine, CA, USA) was inserted via the right jugular vein to monitor mean pulmonary arterial pressure (MPAP). The position of the tip of the Swan Ganz catheter was determined following the pressure readings when the catheter was pushed through the right atrium and ventricle. Additionally, an extra caval vein, a portal vein, and a hepatic vein catheter were inserted for multi-organ blood sampling.
The duration of the surgery was approximately 5 h. Postoperative care was standardized, as has been described in detail previously,23,32 and this was monitored by an assigned veterinarian. After surgery, the animal was kept hooked up to the ECG, pulse oxymeter, and ventilator until it responded with multiple swallow reflexes and eye blinking, and by fighting the ventilator and trying to rise. A second dose of lincomycin–spectinomycin was given intramuscularly. The animal was observed (breathing and heart rates) further until it sat sternally. During the first four days post-surgery, the animals were clinically examined twice daily (for body temperature, appearance and behavior). Buprenorphine (0.03 mg/kg bw) and lincomycin–spectinomycin were administered via the implanted central vein catheter. The animals were fed half of the normal ration of food during the first two days. Catheters were kept patent by daily checks during the first three days, and by use of antimicrobial and anticoagulant catheter filling (20 mg/mL gentamycin and 225 U/mL alfa-chymotrypsin), as has been described previously. 23 Subsequently, every two/three days, catheters were checked for patency and the animals were clinically examined. Before and after surgery, and during the recovery period (7–10 days) the animals were acclimatized to a small movable cage (0.9 × 0.5 × 0.3 m) to ensure that the PA experiments could be performed in this cage in conscious animals in free-standing, sitting or lying positions. Until the day of the sepsis experiment, the pigs were placed at least 10 times in these movable cages.
Experimental setup of the sepsis model
The design for characterizing the septic response of the PA bacteremia is shown in Figure 1. The experiment started after a recovery period of 7–10 days post-surgery. These animals were then allocated to the PA group or the control group in a randomized fashion. Basal blood pressures, plasma flow, blood gas, clinical chemistry and inflammatory data (pre-septic period; t = −2 h to 0 h) measurements were started 4–6 h after the last food intake (half of the daily amount: 0.5 kg). No food was administered during the rest of the experimental period (18 h). At t = 0 h bacteremia was induced by starting the continuous intravenous infusion of PA (IRS 12-4-4, Shriners Burns Institute, University of Texas Medical Branch, Galveston, USA); originally from a burn patient at Brook Army Medical Center in San Antonio, Texas, USA (109 CFU/h in 1 mL of 0.9% NaCl solution). The PA dose initially obtained from the method by Rimmele et al.
16
was tested for virulence and adjusted in a pilot (using three animals). The control group received 0.9% NaCl solution at the same volume. Fluid resuscitation (30 mL/kg bw/h 0.9% NaCl, IV) was started at the same time as the PA infusion. General appearance (alertness, breathing) was monitored and recorded continuously. Multiple blood samples were taken over time (see details in the following section). At t = 18 h the animals were euthanized with 125 mg/kg pentobarbital sodium and 16 mg/kg phenytoin sodium (Euthanasol®), administered via the central vein catheter.
Design induction of sepsis with Pseudomonas aeruginosa bacteremia. MAP: mean arterial pressure; MPAP: mean pulmonary arterial pressure; PAH: para-aminohippuric acid infusion for plasma flow measurements; IV: intravenous; bw: body weight.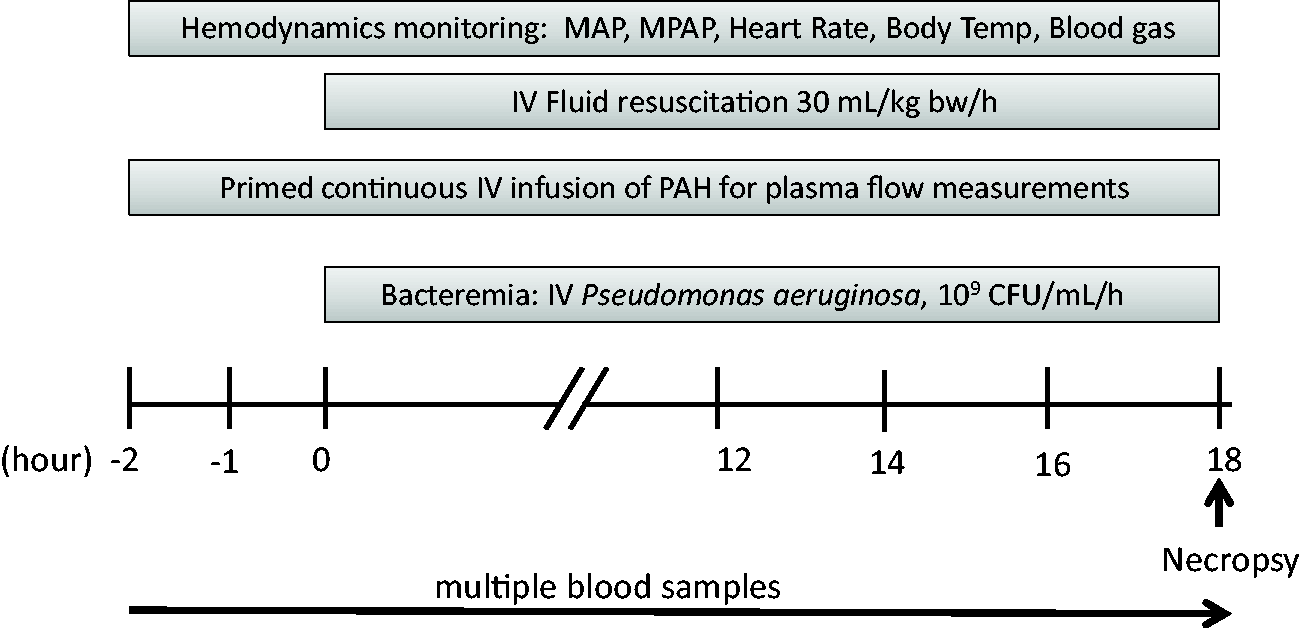
Hemodynamics
Hemodynamics were monitored continuously to ensure that the hyperdynamic state was kept within the expected ranges for severe sepsis (not shock): maximal body temperature (normal range: 37–39.6℃ 33 ) increase or decrease by 2–3℃, maximal MPAP (normal range: 11–24 mmHg 33 ) of 35 mmHg, heart rate (normal range: 90–107 BPM 33 ) of 200 beats per minute (bpm), and MAP (normal range: 86–123 mmHg 33 ) higher than 60 mmHg, using the arterial and Swan Ganz catheters for invasive blood pressure measurement (monitor: Propaq Encore, Welch Allyn, Skaneateles Falls, NY, USA; transducer: TruWave, Edwards Life Sciences). If the heart rate exceeded 200 bpm and/or the MPAP exceeded 35 mmHG, the PA infusion was stopped temporarily to limit the increase of the right ventricular afterload 16 until the heart rate and MPAP fell to below 200 bpm and 35 mmHg, respectively.
Clinical chemistry
At t = 0, 12 and 18 h, 500 µL of serum was collected and stored at −80℃ for a standard clinical chemistry diagnostic panel (see Supplemental Table 2, Veterinary Diagnostic Laboratory, Little Rock, AR, USA; all supplementary material in this article can be found online at http://lan.sagepub.com).
Blood gas
Every 6 h arterial and central venous blood samples were collected for blood gas levels using an on-site blood gas analyzer (i-STAT, cartridge CD4+; Abbott Laboratories Inc, Abbott Park, IL, USA). Blood gas data using the alpha-stat scientific model with no temperature correction were compared and interpreted. 34 Fractional oxygen extraction rates from the capillary bed were calculated: SaO2–SvO2/SaO2 (normal range: 22–32%, where SaO2 = arterial oxygen saturation and SvO2 = venous oxygen saturation). Pulmonary oxygen exchange efficiency was calculated by dividing arterial oxygen pressure (PaO2) with the fraction of inspired oxygen (FiO2 = 0.21; normal range: > 300).
Plasma flow measurements
Organ plasma flow was measured using the PAH dilution model, as has been described previously.
35
In brief: plasma flow was measured with a primed-continuous infusion of PAH (25 mmol/L) via the splenic and one of the arterial catheters (75 mL/h). Arterial, portal, hepatic and caval vein lithium–heparinized blood samples (0.5 mL/port) were collected at t = −1, −0.5, 0, 12, 13, 14, 15, 16, 17 and 18 h for portal drained viscera (PDV), splanchnic (SPL) and hindquarter (HQ) plasma flow determination. These samples were directly cooled (4℃) and centrifuged (8000 ×
Histology
At t = 18 h, immediately after euthanasia, small fragments (approximately 200 mg) of muscle (musculus gastrocnemius, right hind leg, middle part), jejunum, ileum, liver, and lung were obtained from seven control and six PA animals. Tissues were rinsed with saline and fixed in 10% buffered formalin for 24 h. The tissues were then placed in 70% alcohol and stored at room temperature until further routine processing and staining with hematoxylin and eosin (Texas A&M University, Department of Veterinary Pathobiology, TX, USA). The tissues were examined blinded for sepsis-related non-specific inflammation and/or organ-specific histopathological lesions: 37 throughout for the influx of inflammatory cells; and for initial signs of atrophy in muscle; 38 for jejunum/ileum mucosal villous injury using a previously described scoring system; 39 and for subepithelial spacing in the crypt regions. Signs of fatty change were in the liver, and finally signs of diffuse alveolar damage (DAD) in the lung were examined.
Inflammation parameters
Acute phase cytokine interleukin-6 (IL-6) and C-reactive protein (CRP) in arterial plasma were measured at time points –2,–1, 0, 12, 14, 16, 17, 17.5, and 18 h, and analyzed using commercially available enzyme-linked immunosorbent assay (ELISA) according to the manufacturer's instruction (Porcine IL-6 DuoSet, Porcine CRP Duoset ELISA; R&D Systems, Minneapolis, MN, USA). Plasma IL-6 and CRP amounts were expressed in terms of per liter of plasma and milligrams of plasma protein. 30 The dynamics of IL-6 and CRP plasma concentrations over time (Supplemental Figure 1) showed non-physiological relevant changes between 12 and 18 h in a pilot number of animals (IL-6: control n = 2; PA n = 4; CRP: control n = 2; PA n = 2). Therefore, only the pre (t = –2 h) and post PA (t = 17 h) data were presented.
Statistical analyses
Data are expressed as mean ± SE. GraphPad Prism version 6 (GraphPad Software Inc, La Jolla, CA, USA) was used for statistical analysis. Levels of significance were set at P < 0.05. Hemodynamics, organ plasma flows, histological villous scoring, blood gas data, clinical chemistry data and acute phase protein were compared using two-way analysis of variance (ANOVA), where appropriate with repeated measures. Also when appropriate a post hoc Bonferroni, Tukey or uncorrected Fisher's least significant difference (LSD) multiple comparison test was performed, as indicated in the figures and table legend.
Results
Of the 27 animals, five animals were lost due to intestinal complications (ileus, ascites or peritonitis) during the recovery phase from the catheter implantation surgery. However, no animals were lost during the surgery and the PA experiment. General appearance was monitored: PA animals started shivering between 1 and 2 h after the start of PA infusion, deeper breathing (less frequent) started between 4 and 6 h, animals were generally calm but still alert. When needed, PA infusion was stopped between 7 and 18 h (average 14.5 h) after the start of the infusion to prevent any septic response from developing further into an acute septic shock. Thirteen animals with PA (PA group) and nine animals without PA (control group) completed the study.
Hemodynamics and body temperature
The initial increased peak in heart rate was parallel to that of the MPAP up to 6 h (Figure 2). The average temporary stop of PA to limit the increase of the right ventricular afterload
16
infusion lasted 18.5 min. MPAP decreased slowly between 2 and 3 h after the start of PA infusion. The heart rates showed a biphasic pattern: an initial peak in the first 5 h and subsequent increase that showed a stable tachycardia after 10 h. MAP data revealed an initial hypertension phase in the first few hours, followed by a decrease after 5 h. MAP data was kept stable at between 75 and 82 mmHg (hypotension) after 10 h, compared with between 102 and 113 mmHg in the controls. Body temperatures increased within a few hours after the start of PA infusion and reached a maximum after 4 h, followed by a gradual decline. No hypothermia was observed throughout the study.
Hemodynamics and body temperature. Hemodynamic response on the Pseudomonas aeruginosa (PA) induced bacteremia in the pig. A: MAP is mean arterial pressure. B: MPAP is mean pulmonary arterial pressure. C: Heart rate (HR). D: Body temperature. Control n = 9; PA n = 13. Values are mean ± SEM. Statistics: two-way analysis of variance (ANOVA) comparison; when appropriate a post hoc Bonferroni multiple comparison test was performed; hook or *: significance P < 0.05.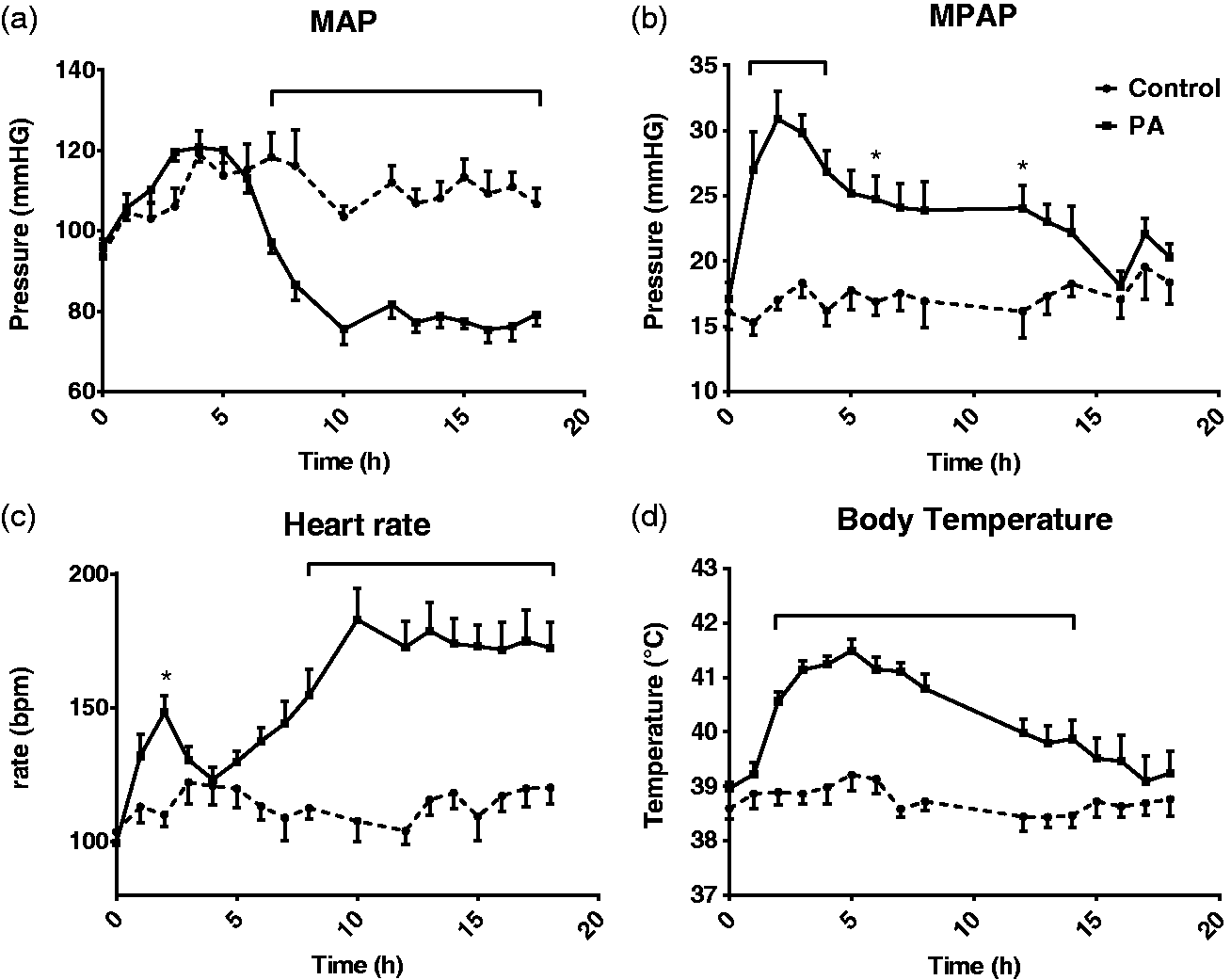
Blood gas characteristics
Arterial and venous oxygen saturations were kept within normal ranges (Supplemental Table 1, all supplementary material can be found online with this article http://journals.sagepub.com/doi/full/10.1177/0023677217718003). The fractional uptake of oxygen from the capillary bed did not change (O2ER). The efficiency of pulmonary gas exchange (average PaO2/FiO2 > 300) was comparable between the groups. The tissue perfusion variable lactate increased over time (>1 mmol/L); however, it did not exceed the upper limit of the normal range. The venous pCO2 and HCO3 were lower than normal in the sepsis group, the resulting pH was expectedly higher; however, both were still within the normal range at t = 18 h.
Blood flow characteristics
Baseline plasma flow was not different between the control and PA groups (Figure 3). PA infusion induced a decrease in plasma flow in the PDV area (P < 0.001), but not in the whole splanchnic area. An increase of plasma flow was observed in the HQ in both the PA and control groups.
: Organ plasma flow. Organ plasma flow changes before and after Pseudomonas aeruginosa (PA) induced bacteremia in the pig. ‘Pre’ is average plasma flow one hour before induction bacteremia. ‘Post’ is average plasma flow between 14 and 18 h after the start of bacteremia induction. Control n = 9; sepsis n = 12. Values are mean ± SEM. Statistics: two-way analysis of variance (ANOVA) repeated measures for time, when appropriate a post hoc Tukey multiple comparison test was performed; hook: P < 0.05.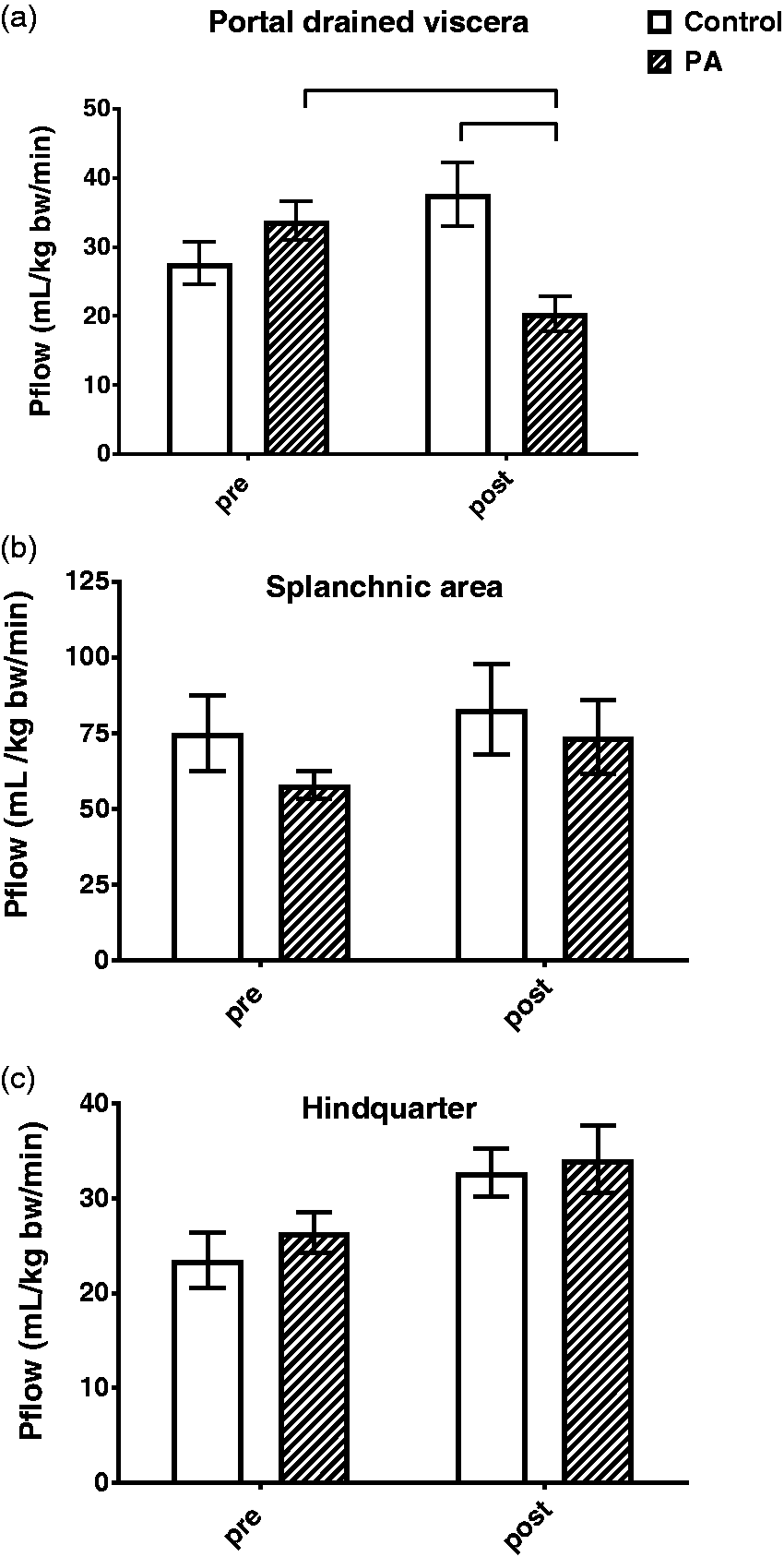
Clinical chemistry
Metabolic parameters, i.e. total protein, albumin, and globulin concentrations, cholesterol, as well as calcium (important for ion homeostasis) decreased in the PA group (Supplemental Table 2). Creatinine phosphokinase (CPK) levels, an indirect marker of muscle degradation, and lactate concentrations (Supplemental Table 1) were increased. The energy parameter glucose reached its lowest value at t = 12 h. Urea (marker of protein metabolism and kidney function) and creatinine levels as well as urea/creatinine ratio were elevated. The liver degradation parameters alanine aminotransferase (ALT) and gamma-glutamyltransferase (GGT) were not altered, but aspartate aminotransferase (AST) and alkaline phosphatase (ALP) were increased.
Histology
The muscle (m. gastrocnemius) showed a mild influx of inflammatory cells between the muscle fibers in all the septic animals, and also in two of the control animals. No initial signs of muscle atrophy were observed. No increases in inflammatory cells were found in the jejunum and ileum, although in the jejunum villous damage was found in both the control and the PA animals. The vacuolization and irregular/blunted villi in the jejunum were more commonly present and severe in the PA animals (Figure 4). Subepithelial space was observed in the jejunal mucosa crypts in five PA and two control animals. Moderate overall flattening of ileum villi occurred in the PA animals. In the liver, fatty change and mild influx of inflammatory cells around the central vein were observed in the PA animals. The lungs showed no signs of DAD.
: Villous injury score. Histological scoring of villous damage in the jejunum and ileum after Pseudomonas aeruginosa (PA) induced bacteremia in the pig. Control n = 7, sepsis n = 6. Values are mean ± SEM. Statistics: two-way analysis of variance (ANOVA) comparison; significance P < 0.05; PA effect: P = 0.022; tissue effect: P = 0.032; interaction: P = 0.572. When appropriate a post hoc uncorrected Fisher's least significant difference (LSD) was used to test PA effect per tissue; hook: significance P < 0.05.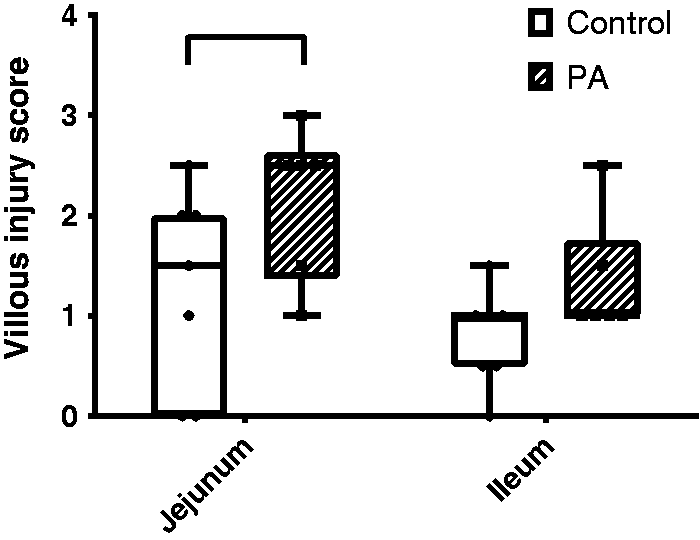
Cytokines and acute phase proteins
Interleukin-6 (IL-6) and C-reactive protein (CRP) plasma concentrations.
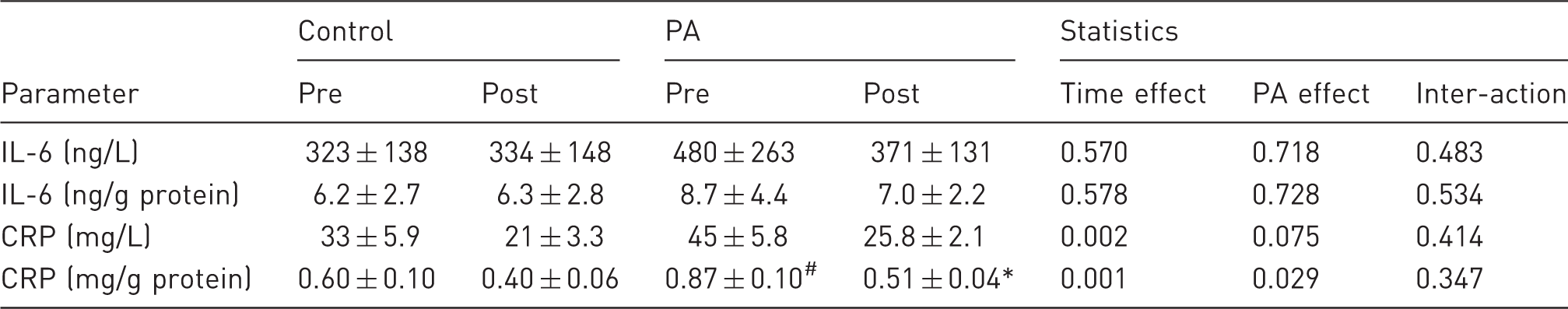
IL-6 and CRP changes before and after Pseudomonas aeruginosa (PA) induced bacteremia in the pig. ‘Pre’ is one hour before induction bacteremia. ‘Post’ is 17 h after start of induction of bacteremia. Values are means ± SEM; control: n = 9; PA: n = 13; statistics: two-way analysis of variance (ANOVA), data of effects are P values; significance P < 0.05. When appropriate post hoc uncorrected Fisher's LSD is done: *significance between pre and post; #significance between control and PA.
Discussion
We developed a controlled hyperdynamic porcine model of sepsis with cardiovascular, inflammatory and metabolic characteristics as those observed in a human septic state. This model enabled us to conduct studies unraveling the disturbances in organ metabolic pathophysiology in the early hyperdynamic septic state.
Diagnosis of the septic state
Sepsis is a clinical syndrome of systemic inflammatory response (SIRS) that complicates severe infection. Diagnostic criteria include a documented pathogen plus two or more SIRS abnormalities.1,3,4 Absolute values of clinical parameters can be slightly different in pigs compared with humans. Therefore in the present study, we compared the changes of these parameters with normal pig values. Physiological and pathophysiological responses of pigs are highly comparable with humans,18,19 therefore described responses (equating to changes in absolute values) of human SIRS criteria can also be used for pigs. With these main diagnostic criteria of SIRS (body temperature: increase of more than 2℃; and heart rate: > 90 bpm; PaCO2: < 32 mmHg), we concluded that the present infection successfully induced sepsis in pigs (Figure 2, Supplemental Table 1). This means that the acute septic response of the chosen rate of PA infusion could be controlled by monitoring the hemodynamics and general appearance (alertness, breathing) of the animals during PA infusion. Sepsis was induced within 6 h after the start of PA infusion. During this initial phase of sepsis, we observed that it was necessary to carefully monitor MPAP and heart rate in the first 2 h to prevent deep, unrecoverable septic shock. We found that stopping the PA infusion for a few minutes was sufficient to stabilize the animals. The increase in the right ventricular afterload is well described in the medical literature, and was likely related to the release of proinflammatory mediators such as tumor necrosis factor alpha (TNF-α) and immunoreactive endothelin-1, which are strong vasoconstrictors in pulmonary vessels. 16
Organ dysfunction
A severe sepsis state is characterized by one or more organ dysfunctions.1,3,4 We found that, despite fluid resuscitation, pronounced hypotension, tachycardia and a decreased PDV plasma flow were present, indicating a disturbed cardiovascular function (Figures 1 and 2). The decreased PDV flow could be responsible for the injured jejunal and ileum mucosa tissues. 40 In particular a potentially compromised PDV microcirculation 41 can result in a decreased absorption capacity/gut metabolism due to mild/moderate damaged villi (Figure 4). In the jejunum of the PA group, it is unclear why subepithelial spacing was not only observed in the villi but also in the crypts. It is likely that the combination of the abnormal translocation of principal oncotic protein in plasma albumin to the intestines, which leads to loss of fluid from the vascular space and the fluid resuscitation, could play a role.12,42 The respiratory system showed no arterial hypoxemia (Supplemental Table 1); therefore, no mechanical ventilation was needed, as was confirmed by the absence of DAD in the lung tissue. The tissue perfusion parameter lactate revealed hyperlactatemia (>1 mmol/L) according to the human criteria, 1 but for a pig it is still within the normal physiological range. 33 Therefore, we can conclude that tissue perfusion was changing in the PA group, however the change was not physiologically meaningful.
Acute kidney failure, as reflected by a substantial increase in the creatinine systemic plasma level, was not observed in the present model (Supplemental Table 2).1,4 Doi et al. 43 have indicated that creatinine levels can be inconclusive due to a potential decrease in creatinine production with sepsis. The fact that we did not find any change in the arterial PAH concentrations, used for organ plasma flow calculations, confirms that renal function was not compromised in the present sepsis model. Although ALT and AST are both biomarkers for liver injury, ALT is more liver-specific. Increased AST alone indicates that there is extrahepatic injury (for instance in skeletal muscle) and fits with the increase of CPK (muscle degradation marker). We concluded that there were no indications of acute hepatic dysfunction (Supplemental Table 2), although liver histology showed mild infection related abnormalities. 37 Therefore, we can conclude that we have developed a sepsis model characterized by a severely compromised function of the cardiovascular system, and a mildly compromised function of the PDV by infusing PA. Severe sepsis was established between 12 and 18 h after starting the PA infusion. Furthermore, we observed several changes in clinical parameters independent of the presence of sepsis, which were potentially induced by fluid resuscitation, 37 indicating that the control group was crucial in discriminating between sepsis and non-sepsis-related changes.
Systemic inflammation symptoms
The steep increase in body temperature indicated the onset of a PA-induced inflammatory response (Figure 2). Additionally, in a follow-up study we conducted a white blood cell (WBC) count (total WBC, lymphocytes, granulocytes and platelets) in a pilot group of animals (four) in the first 6 h after the start of PA infusion (Supplemental Figure 2). The observed disappearance of lymphocytes from the systemic blood to the peripheral tissues was expected as an early response to trapped PA bacteria in the tissue.44,45 This resulted in an initial decrease of total WBC, but was quickly attenuated by a stimulated increase in availability of granulocytes. 46 Therefore, we concluded that a clinically expected systemic WBC response to PA was observed. However, this was not accompanied by a clear cytokine (IL-6) and acute phase protein (CRP) response between 12 and 18 h (Table 1).
Although, a general cytokine response was expected, many factors could be involved, which may explain why we did not see a response. IL-6 and CRP responses to PA in the pig over time are a result of the amount/strain/route of PA administration and the subsequent complex interaction of multiple host and microbial factors. Therefore, this can be highly variable between different studies47,48 and between animals. 49
The following explanations need to be considered. First, the uncontrolled microbial status of the farm pigs may likely have been the cause of the high variability in IL-6 and CRP values before the bacteria were infused. 50 However, Supplemental Figure 1 illustrates that the initial IL-6 or CRP variability was not related to the non-response in the period between 12 and 18 h. Therefore, we can conclude that it is highly unlikely that the differences in initial cytokine status have an influence on PA cytokine responses. Secondly, serum amyloid A (SAA) could potentially be a more sensitive alternative for CRP, but this was not measured in the present study. 51 However acute phase protein response is considered to be initiated by interleukins such as IL-6, also SAA. Therefore, the absence of the CRP response is potentially caused by an absence of an IL-6 response. Thirdly, we have to consider that the absence of a clear IL-6 and CRP response is a reflection of an inefficient or delayed inflammatory response. This phenomenon in humans is associated with higher mortality from sepsis.50,52 Finally, we need to consider time of measurement. Rimmele et al. have presented a response to PA infusion in the pig which was very early (in the first 6 h after administration of PA) but mild. 16 This is in line with observations in humans with experimental endotoxemia. 46 We did not measure IL-6 in this time window in the present study. However, in a follow-up pilot study using this model (four animals), a small IL-6 response was confirmed in the first 6 h (Supplemental Figure 3). Therefore, we can conclude that the cytokine response of IL-6 in the current model occurred at a very early stage, but did not continue to the severe septic stage. On the other hand, measurement was perhaps too early. For instance, Benes et al. have found that an IL-6 response to comparable PA infusion started after 18 h in a ventilated pig model. 30 However, the IL-6 response was still mild in comparison with a peritonitis-induced sepsis model in the pig. Overall, we can conclude that the model described in the present study showed a clinically relevant inflammatory response in the first 6 h.
Potential value for nutritional research in septic state
Nutritional intervention can be beneficial and be of high clinical value in severe sepsis, 9 in contrast to those in a septic shock state. 53 Sepsis-related metabolic changes such as protein breakdown,5,24 hypocalcemia 54 and hypocholesterolemia 55 were observed in the current model (Supplemental Table 2). Therefore, we concluded that with these metabolic characteristics, the presented PA model, with the implanted catheters to ensure the possibility of measuring metabolism across organs,23,24,26 could be very useful for preclinical nutritional research in severe sepsis. Though other PA-infused pig models are available, they are septic shock models in an unconscious ventilated pig,16,30 and are therefore not suitable for nutritional research.
Limitation of the model
First, though PA is a clinically relevant bacterium in sepsis, it is not the only bacterium that can cause sepsis. When comparing other live bacteria used to induce severe sepsis in pigs, the pathophysiology of a single dose of Gram-positive Staphylococcus aureus 22 in a conscious pig model showed slightly different patterns in its hyperdynamics, and a late (after 18 h) cytokine response. A similar late cytokine response was observed in a mixed bacterium model (peritonitis created by intraperitoneal autologous fecal inoculum). 19 Therefore, we can conclude that although metabolic data are lacking from these models, it is highly likely that the metabolic results from the present PA severe sepsis model can be translated to general severe sepsis, independent of the bacteria that caused the septic state. Secondly, we need to consider that the relative young age of the studied pigs cannot represent metabolic implications seen in adult pigs. We made these concessions for practical reasons (handling, catheter protection with the used harness), but with the knowledge that the gastrointestinal tracts in these animals are already mature. 56 Therefore, we can still make the translation of metabolic findings to a broader age range. Finally, only the initial phase of severe sepsis has been represented. This means that results cannot be easily translated to a recovery phase in sepsis (for instance after antibiotic treatment). Therefore further development of the sepsis model is needed for studying metabolism in the recovery phase.
Conclusion
The present acute sepsis model has the characteristics of being hyperdynamic and biphasic with a severe septic phase present between 12 and 18 h after PA infusion. The present PA-induced porcine sepsis model is a clinically relevant model for acute severe sepsis. Implanted catheters can accommodate an in-depth study of multi-organ metabolism and nutrient metabolism in sepsis.
Footnotes
Acknowledgements
We thank the assigned veterinarians and animal caretakers of the Arkansas Children’s Hospital Research Institute for their dedication in providing optimal care for the pigs in our experiment. We also thank Joshua Spore, Bea Zoer, Eva M C Oosterlaken and Sonya R Mehta for their skilled technical assistance in carrying out the animal experiments and/or sample processing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of General Medical Sciences or National Center for Research Resources or the National Institutes of Health.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by Award Number R01GM084447 from the National Institute of General Medical Sciences and by Award Number S10RR027047 from the National Center for Research Resources.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
