Abstract
The liver receives dual blood supply from the hepatic artery and portal vein. The pig is often used as an animal model in positron emission tomography (PET) and pharmacokinetic studies because of the possibility for extensive and direct blood sampling. In this study, we compared measurements of hepatic blood flow in 10 female adult Göttingen minipigs and 10 female pre-pubertal Danish Landrace x Yorkshire (DLY) pigs. Ultrasound transit time flow meter probes were placed around the hepatic artery and portal vein through open surgery, hepatic blood flow measurements were performed, and the liver was weighed. Total hepatic blood flow was on average 363 ± 131 mL blood/min in Göttingen minipigs and 988 ± 180 mL blood/min in DLY pigs (p < 0.001). The mean hepatic blood perfusion was 623 mL blood/min/mL liver tissue and 950 mL blood/min/mL liver tissue (p = 0.005), and the liver weight was 0.58 kg and 1.04 kg, respectively. The mean arterial flow fraction in Göttingen minipigs was 12 ± 7% and lower than in DLY pigs, where it was 24 ± 7% (p = 0.001). Using the gold standard for blood flow measurements, we found that both total hepatic blood flow and blood perfusion were significantly lower in Göttingen minipigs than in DLY pigs. The hepatic blood perfusion and arterial flow fraction in DLY pigs were comparable to normative values from humans. Differences in hepatic blood flow between adult Göttingen minipigs and humans should be considered when performing physiological liver studies in this model.
Introduction
The liver is supplied by oxygenated blood from the hepatic artery and venous blood from the mesenteric circulation through the portal vein. The arterial blood input to the liver is generally believed to account for 25% of total hepatic blood flow in humans,1,2 however, the haemodynamics vary significantly depending on prandial state and pathophysiological changes as observed in chronic liver disease.3–6 The pig is often used as an animal model for preclinical testing of new positron emission tomography (PET) tracers and pharmacokinetic studies because of its size and anatomical resemblance to humans.7–9 Moreover, the large blood volume of pigs allows for invasive and extensive blood sampling as well as accurate flow measurements.
Domestic pig types known from conventional farming and pig production such as the Danish Landrace x Yorkshire (DLY) pig are cheap to produce but weigh up to 300 kg in adulthood, and young pigs of 40 or 60 kg are therefore often used for practical reasons. However, the rapid growth makes the model unsuitable for interventional follow-up studies, since normal physiological changes cannot be excluded. 10 For this purpose, the Göttingen minipig has been introduced and is increasingly used. 11 The Göttingen minipig is bred for research purposes on a strict genetic background and weighs 35–40 kg when fully grown.12,13 In practice, researchers are therefore often faced with using adult minipigs or young domestic pigs, especially in imaging and surgical research.
It is generally assumed that the hepatic blood perfusion and arterial flow fraction in pigs are comparable to values in humans. Having experience with functional PET studies of the liver in both DLY and Göttingen minipigs, we have observed notable differences in hepatic blood supply values between the two, and while crucial for in vivo studies of hepatic kinetics, these are seldom examined thoroughly. In this study, we compared hepatic blood flow measurements from Göttingen minipigs and DLY pigs as we sought to investigate the translational value for human studies.
Methods
Study Design
In this study, we compared invasive hepatic blood flow measurements from two previous PET studies performed in 10 adult female Göttingen minipigs (Ellegaard Minipigs, Dalmose, Denmark) 14 and 10 pre-pubertal female DLY pigs (local commercial pig breeder), 15 giving a total of 20 animals. Both studies comprised dynamic PET recordings of the liver with simultaneous measurements of hepatic blood flow rates and sampling of blood from a femoral artery and the portal vein. Half of the Göttingen minipigs had been exposed to whole-liver radiation, but this did not affect hepatic blood flow or liver weight when compared to control pigs. 14 This was a retrospective analysis, and thus no randomization or blinding was used.
Ethics statement
The protocols from the two studies were approved by The Danish Animal Experiments Inspectorate, and all experiments were performed according to the current law on animal experimentation and ethics.
Animal housing and health status
All pigs were free of scabies (Sarcoptes scabie), lice (Haemotopinus suis), swine dysentery (Brachyspira hyodysenteriae), atrophic rhinitis (toxin-producing strains of Pasteurella multocida), pleuropneumonia (Actinobacillus pleuropneumoniae serotypes 1–10, 12), enzootic pneumonia (Mycoplasma hyopneumoniae), and porcine reproductive and respiratory syndrome (PRRS virus) according to clinical testing and blood samples. The animals were acclimatized for at least one week prior to experiments and housed under environmental conditions of 20°C (50–55% relative humidity), 12:12 hours of light and darkness, and change of air eight times per hour. The pigs were group housed and fed a restricted pellet diet (Göttingen minipigs, SDS Diet, UK; DLY pigs, IA plus FI, DLG, Copenhagen, Denmark) with free access to tap water, as previously described.14,15 The pigs were clinically healthy during this period, and their well-being was monitored twice per day.
Animal handling and flow measurements
All pigs fasted overnight (16 h) with free access to water. The pigs were sedated with an intramuscular injection of midazolam (1.25 mg/kg) and S-ketamine (6.25 mg/kg), and after a few minutes, a catheter (Venflon, 22G, 0.9 mm × 25 mm, Medshop, Denmark) was placed in an ear vein for injection (induction) and subsequent infusion (maintaining) of anaesthesia mixture (midazolam 1 mg/kg/h, S-ketamine 5 mg/kg/h and propofol 6 mg/kg/h). Prior to surgery, the pigs were treated with an intramuscular injection of 0.3–0.6 mg buprenorphine and 10 mL lidocaine applied topically. The pigs were intubated, mechanically ventilated (8–9 mL/kg tidal volume; Model 3000, Veterinary Ventilator, Matrx, Pittsfield, MA) with a mixture of oxygen and medical air (1:2.2), and kept physiologically stable under anaesthesia (placed on a thermostatically controlled heating blanket, keeping the rectal temperature between 38.5 and 39.5 °C; pulse and O2-saturation were measured continuously). Eye and interdigital reflexes were tested at least twice per hour. No humane endpoints were used, since this was a non-survival design.
The liver was accessed through a surgical midline incision followed by dissection of the hepatoduodenal ligament, and transit time ultrasound perivascular flow probes (VeriQ; Medistim, Denmark) were placed around the hepatic artery (FHA, mL blood/min) and portal vein (FPV, mL blood/min). In both pig types, there was variation in the arterial anatomy, and in cases where the hepatic artery branched before entering the liver, flow probes were placed around each branch and the flow measurements were summated. Total hepatic blood flow (F, mL blood/min) was calculated as the sum of FHA and FPV. Hepatic blood perfusion, Q (mL blood/min/mL liver tissue), was calculated as Q = F/V, where V (mL liver tissue) is the liver volume calculated as the liver weight corrected for a tissue density of 1.07 g/mL liver tissue. 16 Flow measurements were performed at least 60 min after surgery to allow for physiological stabilization, and the measurements used in this study were obtained at baseline, that is before administration of radiotracers and blood sampling (reduction of circulatory blood volume).
Termination and weighing
After the experiments, the pigs were euthanized with an overdose of pentobarbital. Immediately after, the liver was removed, drained for excessive blood, and weighed. The body weight was measured ≤ 3 days before studies. The fraction of liver weight compared to total body weight was determined as LW% = liver weight/body weight × 100%.
Statistical analysis
Normality was assessed using QQ plots; all data were normally distributed. Group values are expressed as mean ± SD and were compared using Student’s t test. Correlation analysis was performed using the Pearson’s Product Moment. A p value below 0.05 was considered as statistically significant. Since this was a retrospective analysis of previous PET studies, the sample size was defined on beforehand and no power calculations were performed. Statistical analysis was performed using STATA (Version 16.0, StataCorp, College Station, TX).
Results
The Göttingen minipigs and DLY pigs were examined at mean age 401 (c. 1.1 years) and 99 days (c. 14 weeks), and they weighed on average 29.6 and 39.0 kg (t(18) = –8.94, p < 0.001), respectively (Table 1). The liver volume was lower in Göttingen minipigs than in DLY pigs (mean 0.58 kg v. 1.04 kg; t(17) = –20.68, p < 0.001), and so was the fraction of liver weight compared with total body weight (LW%: mean 1.99% v. 2.67%; t(17) = –12.15, p < 0.001) (Table 1).
Animal characteristics and liver weight. Values are given as group means ± SD. All group differences were statistically significant (p < 0.001). Liver weight was not available for one Danish Landrace x Yorkshire (DLY) pig (n = 9).

LW%: fraction of liver weight compared with total body weight.
Total hepatic blood flow was on average 363 ± 131 mL blood/min in Göttingen minipigs and 988 ± 180 mL blood/min in DLY pigs (t(18) = –8.87, p < 0.001). The difference in hepatic blood flow could not be ascribed to the difference in liver volume alone, as the mean hepatic blood perfusion in Göttingen minipigs was also significantly lower than in DLY pigs (mean 623 mL blood/min/mL liver tissue v. 950 mL blood/min/mL liver tissue; t(17) = –3.18, p = 0.005) (Figure 1). Figure 2 shows that hepatic blood flow and perfusion were not systematically correlated to body weight of the pigs, nor were they correlated with liver weight as proportion of body weight. In Göttingen minipigs, the hepatic blood flow supplied by the hepatic artery and portal vein was 40 ± 18 mL blood/min and 323 ± 132 mL blood/min (mean ± SD). In DLY pigs, it was 249 ± 100 mL/min and 764 ± 111 mL/min (Figure 3). The fraction of total hepatic blood flow supplied by the hepatic artery was on average 12 ± 7% in Göttingen minipigs and 24 ± 7% in DLY pigs (t(18) = –3.85, p = 0.001), yielding a ratio between blood flow supplied by the hepatic artery and portal vein of approximately 1:8 in Göttingen minipigs and 1:3 in DLY pigs.
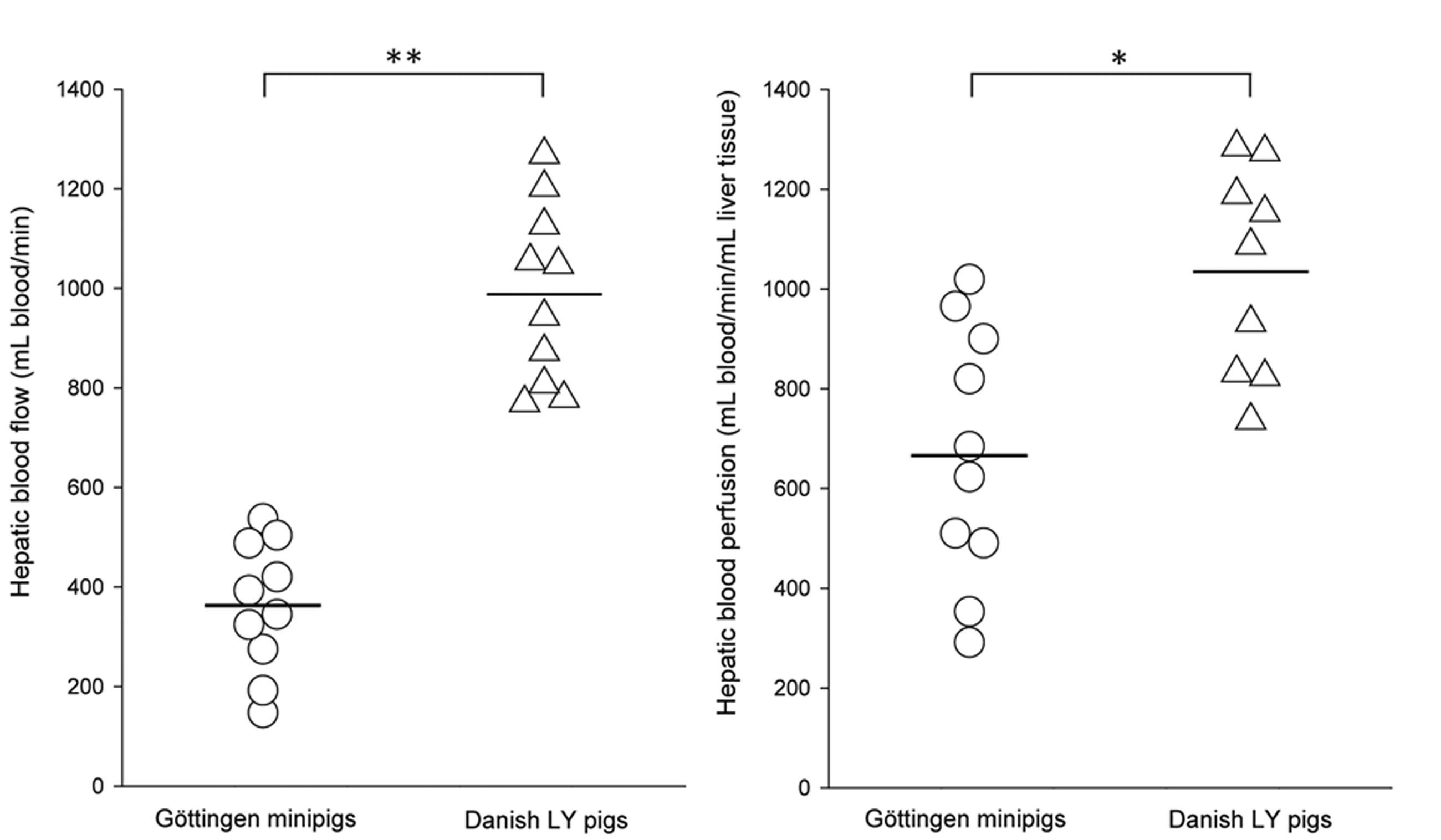
Hepatic blood flow and perfusion measurements in Göttingen minipigs (circles) and Danish Landrace x Yorkshire (DLY) pigs (triangles). Shown are individual measurements with bars indicating group means. Liver weight, and consequentially hepatic blood perfusion, was not available for one DLY pig (n = 9). *p < 0.01; **p < 0.001
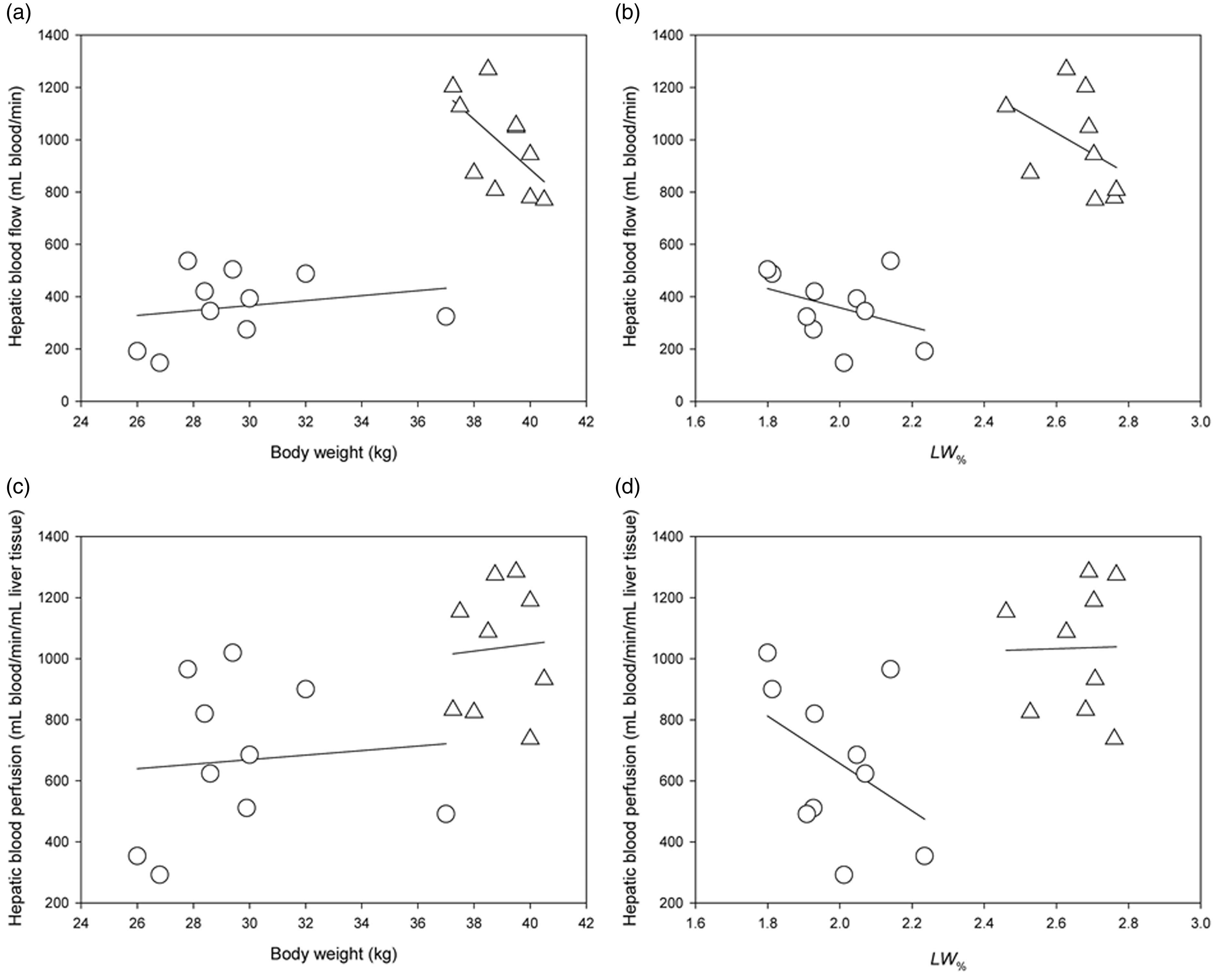
Correlation analyses of hepatic blood flow and hepatic blood perfusion versus body weight and fraction of liver weight to body weight (LW%). Shown are the individual measurements from Göttingen minipigs (circles) and Danish Landrace x Yorkshire (DLY) pigs (triangle). (a) and (b) Scatter plots with linear regressions of hepatic blood flow versus body weight (a), and LW% (b). (c) and (d) Scatter plots with linear regressions of hepatic blood perfusion versus body weight (c), and LW% (d). None of the parameters were significantly correlated.
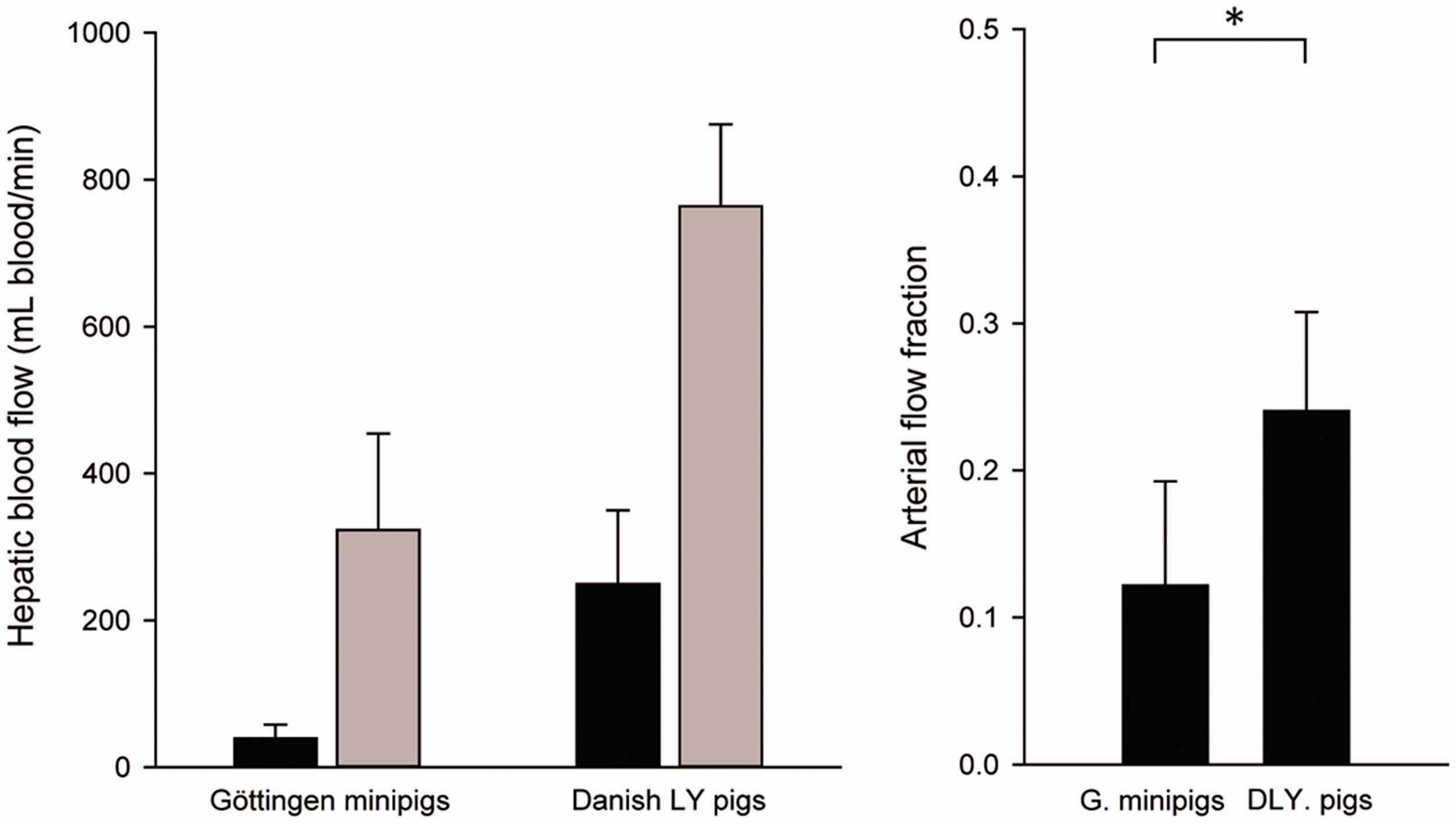
Distribution of hepatic blood flow from the hepatic artery and portal vein in Göttingen minipigs and Danish Landrace x Yorkshire (DLY) pigs. (a) Mean blood flow in the hepatic artery (black bars) and the portal vein (grey bars) with SD (statistical analysis of mean difference not shown). (b) The arterial fraction of hepatic blood flow as compared with total hepatic blood flow. *p < 0.01
Discussion
In this paper, we present detailed hepatic blood flow measurements from two commonly used pig breeds, the adult Göttingen minipig and the pre-pubertal DLY pig. We found that the hepatic blood flow in Göttingen minipigs was approximately one third of that in DLY pigs. Interestingly, this difference could not be ascribed to lower liver volume or body weight in the Göttingen minipigs alone, as the hepatic blood perfusion was only two thirds of that in DLY pigs. Finally, the contribution to total hepatic blood flow supplied by the hepatic artery was only 12% in the Göttingen minipigs as compared with 24% in DLY pigs.
We used direct flow measurements to determine hepatic blood flow, which is considered the most reliant method for estimation of dual-input blood flow from both the hepatic artery and portal vein. 17 Other more non-invasive methods are available, but these cannot distinguish between arterial and portal input or do not measure blood flow with sufficient precision. 18 The most significant limitation to direct flow measurement is the need for invasive surgical placement of ultrasound probes, which in most cases precludes survival designs. The prolonged anaesthesia and trauma from surgery may also affect the mesenteric blood flow. 19 However, this was corrected for by exposing the pigs to one hour of physiological stabilization before flow measurements. Since we used the exact same experimental procedures in both studies,14,15 the differences in hepatic blood perfusion are unlikely to be caused by methodological uncertainty. Moreover, all surgical preparations and placement of ultrasound flow probes were performed by the same surgeon, which also eliminates inter-individual variability.
DLY pigs have been well characterized in the past, 20 but studies on the normal physiology of Göttingen minipigs are more scarce; to our knowledge, hepatic blood flow in Göttingen minipigs have only been described briefly in one other study. 21 The present hepatic blood flow measurements in DLY pigs are in line with previous observations in other domestic pig types.22,23 However, our measurements of hepatic blood perfusion in Göttingen minipigs are markedly lower than those reported by Suenderhauf et al. in 6-month old, 14-kg male Göttingen minipigs, as these were more similar to the DLY pigs. 21 The most probable explanation for this difference seems to be the age and size of the pigs, and it could be hypothesized that the developing liver in pre-pubertal pigs has a higher perfusion than in adulthood. Other than age, health status of the animals can also influence physiological parameters. Hence, although health status was comprehensively monitored, the high Specific Pathogen Free (SPF) standards for rearing of Göttingen minipigs constitutes another difference between the two pig types. It should also be noted that we report hepatic blood flow measurements from exclusively female pigs, which are most commonly used, but potential sex differences should be taken into consideration. We used female pigs because of the possibility for urinary catheterization under anaesthesia, which is not possible in male pigs. Moreover, male pigs have a strong odour, which is unpractical in a clinical research environment.
There was no significant correlation between hepatic blood flow measurements and body weight in the two pig types. Furthermore, there was notable variation in hepatic blood perfusion in both pig types, in particular for Göttingen minipigs. These observations underline the importance of directly measuring individual hepatic blood flow values and that if using estimates from the literature, measurements from pigs of the same age and strain should be applied. Knowing the ratio between arterial and portal flow input is necessary for the analysis of non-steady state kinetics in the liver, 24 and based on our results, standardized values could be unreliable for this purpose. Accordingly, the observed differences in arterial flow fraction further supports direct measurement of dual input blood flow in pigs when performing studies on non-steady state kinetics in the liver, while such estimates must be presumed in human studies. 25
Although body surface area is the most accurate predictor of liver weight, 26 liver weight in humans is commonly reported as being approximately 4% of body weight in the infant, decreasing to around 2% in adulthood.27,28 In this context, the adult Göttingen minipigs in the present study were more comparable to the adult human, whereas in pre-pubertal DLY pigs, the liver accounted for approximately 2.7% of total body weight. Hepatic blood perfusion in healthy humans is approximately 1000 mL blood/min/mL liver tissue, with some variation,29–31 being comparable to our observations in DLY pigs, while the hepatic blood perfusion in Göttingen minipigs was approximately two thirds of this. Hepatic blood flow in humans peaks in early adulthood, decreasing almost linearly with age, 31 and with the likelihood that the same is applicable to pigs, this emphasizes age as a key factor for hepatic blood flow. Also, the arterial fraction of total hepatic blood flow in DLY pigs was practically equal to the average of 25% in healthy humans,1,2 where it was half of that in Göttingen minipigs. It is unclear whether the reduced arterial flow input in adult Göttingen minipigs is an age-dependent physiological phenomenon, since no other studies report such data in humans or pigs. Regardless, our observations suggest that for measurements of hepatic blood flow, the normal physiology of the pre-pubertal DLY pig is more readily translated to adult humans than it is for the adult Göttingen minipig. It should also be mentioned that from a surgical perspective, the Göttingen minipigs had more abdominal fat and larger ventricles as well as a smaller, more fragile portal vein and common biliary duct, thereby complicating liver surgery. Thus, this study shows the importance of using well-characterized and validated pig models and emphasizes that the pig cannot be considered as one entity when studying the liver, but varies with pig type, age and body weight.
Conclusion
Thorough characterization and validation of normal physiological parameters is pivotal for the use of animal models in preclinical research, and accurate measurements of hepatic blood flow dynamics are necessary for the assessment of hepatic kinetics in vivo. We show in this study that hepatic blood flow and perfusion is significantly lower in adult Göttingen minipigs when compared with pre-pubertal DLY pigs and humans. Moreover, the relative flow input from the hepatic artery was markedly lower in Göttingen minipigs than what is generally assumed to be 25% of total hepatic blood flow in humans. The Göttingen minipig is an increasingly used animal model in studies of hepatic kinetics, however, our results suggest some disparity between normal hepatic blood flow in adult Göttingen minipigs and humans, whereas the pre-pubertal DLY pig is more similar.
Footnotes
Acknowledgements
The authors would like to thank Mie Dollerup and Stine Methmann for assistance with surgical procedures and monitoring of the pigs during experiments.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The studies used to generate the data presented in this paper were supported by the Aarhus University Research Foundation, The Independent Research Fund Denmark (Medical Sciences; 8020-00427B, 12-125512), Novo Nordisk Foundation (NNF20OC0059717, NNF17OC0029702), Aase and Ejnar Danielsen’s Foundation, the Foundation of 17-12-1981, the National Institute of Diabetes and Digestive Diseases (R01DK074419), and Helga and Peter Korning’s Foundation.
