Abstract
A large craniotomy survival porcine model is useful for scientific research. The surgical approaches and complications of craniotomies in pigs have not been published before. This study describes how large craniotomies were performed in 46 pigs and how the risk of complications was minimized. The major complications were direct postoperative epidural hematomas (n = 3) and sagittal sinus rupture (n = 4). The measures taken to prevent postoperative epidural hematomas consisted of optimizing anesthesia, using bone wax to stop trabecular bleeding, increasing blood pressure before bone flap replacement, tranexamic acid administration, and postoperative recovery of the pigs in the prone position in a dedicated hammock. After these measures, no pig died from a postoperative epidural hematoma. Iatrogenic sagittal sinus rupture occurred in cases where the dura shifted into the craniotome during craniotomy. The dura was detached from the skull through drill holes with custom elevators before craniotomy to minimize the risk of a sagittal sinus rupture. In conclusion, pigs can undergo craniotomy and survive if the right measures are put in place.
Introduction
Craniotomy has been performed in pigs to provide training for basic neurosurgical techniques such as bone drilling, bleeding management, and brain dissection.1,2
However, a large craniotomy survival pig model for research purposes has not been published before. Craniotomy survival pig models are useful for scientific research, as the porcine brain resembles the human brain in anatomy, growth, and development more than the brains of commonly used laboratory animals. 3 Although craniotomy in pigs is not a complex procedure, it is important to be conversant with the surgical techniques and perioperative care to avoid complications leading to animal loss and/or distortion of study results. This will result in the use of fewer animals (reduction) and the minimization of the risk of pain and distress (refinement). This study aimed to describe how to perform a large craniotomy in a pig model and ensure survival and minimize the risk of intra- and postoperative complications.
Animals, materials, and methods
Craniotomies were performed in 46 pigs at our center between 14 November 2016 and 21 April 2017. The craniotomies described were performed to study medical devices. Therefore, the number of animals used in this study was based on the minimal sample size required to evaluate the safety of a new medical device.
The study was approved by the local ethical review board and the Central Animal Experiments Committee affiliated with the Dutch National Institute for Public Health and the Environment (Approval No. AVD115002016457). The Central Lab Animal Research Facility (CLARF), where the study was conducted, is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC). All procedures were performed by a fully trained neurosurgeon (T.v.D.), an experienced resident in the surgical team (A.K.), a veterinarian anesthesiologist (K.V.), and an experienced biotechnician in the anesthesiology team.
Animals
SPF Topigs Norsvin (TN) genetic female pigs were selected for this study. Male pigs were excluded following the recommendation of our local ethical review committee based on the policy of our research facilities because they have a more aggressive natural behavior. The cranial anatomical variance between male and female pigs is minimal and unlikely to influence the outcome of the study. The pigs were delivered from a specific pathogen-free breeding farm (van Beek SPF varkens BV, Lelystad, The Netherlands) under the supervision of The Netherlands Food and Consumer Product Safety Authority. The microbiological status is provided in Supplementary File S1.
The mean weight of the pigs was 66 ± 5.7 kg and the mean age was 4.3 ± 1.2 months on the day of surgery. The animals were acclimatized for at least 1 week. All the animals were examined and adjudged healthy by a veterinarian (K.V.). The pigs were sacrificed at different times (3, 7, and 14 days, 1, 3, 6, and 12 months) to assess the local effect and the resorption of the medical device over time.
Housing
The pigs were kept in groups of three in neighboring pens on straw. The conditions of the room were strictly controlled at a temperature of 20°C, humidity of 45–80%, and lighting for 12 h daily (between 7:00 and 19:00). The animals were fed twice a day with 750 g cereal (Altromin 9053®) low-calorie diet; depending on the weight gained, this was increased to a maximum of 1500 g twice daily. The pigs received a low-calorie diet to minimize weight gain; otherwise, the growth of the pigs would cause housing issues. They had unrestricted access to drinking water. The environmental enrichment consisted of biting sticks and playing balls. The floors of the pens were covered with straw. The pigs were kept solitary in pens after surgery until the jugular cannula was removed 2 weeks postoperatively due to the risk of biting each other’s cannula. Thereafter, the pigs were kept in pairs.
Anesthesia
For the first three pilot pigs, anesthesia was performed as follows. One day before surgery, a transdermal buprenorphine patch 5 µg/h (Butrans® 5 mg) was applied to the abdominal skin for 7 days. The pigs were food-deprived 12 h before surgery without water restriction. General anesthesia was induced by intramuscular injection of a mixture containing midazolam 0.7 mg/kg (Midazolam Actavis® 5 mg/ml), ketamine 13 mg/kg (Narketan® 10 mg/ml), and atropine 0.05 mg/kg (Atropinesulfaat CF® 1 mg/ml). The larynx was sprayed with lidocaine (100 mg/ml) and the pigs were intubated orotracheally (Covidien Shiley HI-Contour oral/nasal tracheal tube cuffed, 7.7–8.5 mm inner diameter, 30 cm length). Anesthesia was maintained with midazolam (1.0 mg/kg/h) and sufentanil (0.01 mg/kg/h) via an auricular vein. Blood pressure, heart rate, respiratory rate, saturation, electrocardiogram (ECG), and capnography were monitored continuously during surgery. Amoxicillin/Clavulanic acid® (500/50 mg powder for solution, Sandoz) and a bolus meloxicam® 0.4 mg/kg (20 mg/ml, Metacam) was administered intravenously before incision. The basal fluid requirement during surgery was maintained with intravenous NaCl infusion at a rate of 1.5 ml/kg/h. The incision area was infiltrated with 10 ml of lidocaine® HCl (20 mg/ml, B. Braun) subcutaneously and periosteally. After the closure of the galea aponeurotica, the wounds were dripped with neomycin and procaine benzylpenicillin (Neopen® 100 mg/ml). The postoperative analgesia consisted of a transdermal buprenorphine patch and 0.4 mg/kg meloxicam administered intravenously via the jugular cannula for 5 days. Buprenorphine (0.05 mg/kg) was administered intramuscularly twice daily as escape medication until no signs of pain were present; if allowed, the dosage was gradually decreased. Depending on the survival period, the pigs were euthanized under anesthesia with 100 mg/kg intravenous pentobarbital (Euthanimal®, 200 mg/ml). A cannula was placed in the jugular vein perioperatively for frequent blood sampling. The technique for the placement of an external jugular vein cannula has been described previously. 4
Craniotomy
The pig was placed in sternal recumbency with the front and back limbs forward. The head was placed on a pillow block and the ears were taped behind the head to fully expose the epicranium (Figure 1a). The epicranium was shaved and disinfected with chlorhexidine (Hibiscrub®) and povidone-iodine (Betadine®). The incision and the craniotomy line were marked by drawing lines from the opening of the external acoustic meatus to the contralateral pupil on both sides. The intersection point was the center of the linear midline craniotomy incision (measuring 8–9 cm) and the mid-point of the bone flap (measuring 4 × 4 cm) (Figure 1b). After the initial skin incision and skull exposition using a large wound spreader, a mould of 4 × 4 cm was used to mark the placement of four burr holes at the corners of the craniotomy (Figure 1c). The burr holes were made with a disposable 9-mm perforator (Codman®, Johnson & Johnson) (Figure 1d). The perforator was placed perpendicularly to the bone for drilling. During bone drilling, the surgical field was irrigated extensively with sterile saline to reduce thermal damage. The holes were expanded using Kerrison bone punches. The dura was freed from the skull with a dissector. For the craniotomy, a long craniotome (EG1® High-Speed Electric System with a 30.3 mm Fluted Spiral Router, Anspach®, Johnson & Johnson) was used. We angulated the craniotome slightly backward. Subsequently, the bone flap was removed and the dura was exposed (Figure 1e). The bone flap was placed in a solution of amoxicillin/clavulanic acid (Sandoz® 500/50 mg in 100 ml saline). The sharp edges of the skull were removed using a Kerrison punch. Lyostypt® (B. Braun) was placed above the sagittal sinus to stop microbleeding from the sinus. Bleeding was stopped with bipolar coagulation. After hemostasis, the dura mater was incised. Small dural bleedings were stopped by squeezing the dura with a micro-tweezer or by bipolar coagulation. The dura was closed with interrupted sutures using 5/0 absorbable sutures (Vicryl®; Ethicon, Inc.). Finally, the bone flap was replaced and fixed to the skull using one 2/0 absorbable suture (Vicryl® Ethicon, Inc.) on each side (Figure 1f) through one drilled hole (EG1® High-Speed Electric System with a Fluted Router (1.4 × 12.8 mm) Anspach®, Johnson & Johnson). The subcutis was closed with a Vicryl® 2-0 and 2 mm 1/2c needle continuous suture. The cutis was sutured intracutaneously with a 3/0 absorbable monofilament suture (Monocryl®; Ethicon, Inc.). The complications during surgery that were attributable to humane endpoints were, but not limited to, orbital bone damage with the risk of eye damage, untreatable sagittal sinus bleeding, and major brain damage. Major brain damage was assessed by the neurosurgeon or veterinarian based on their discretion.
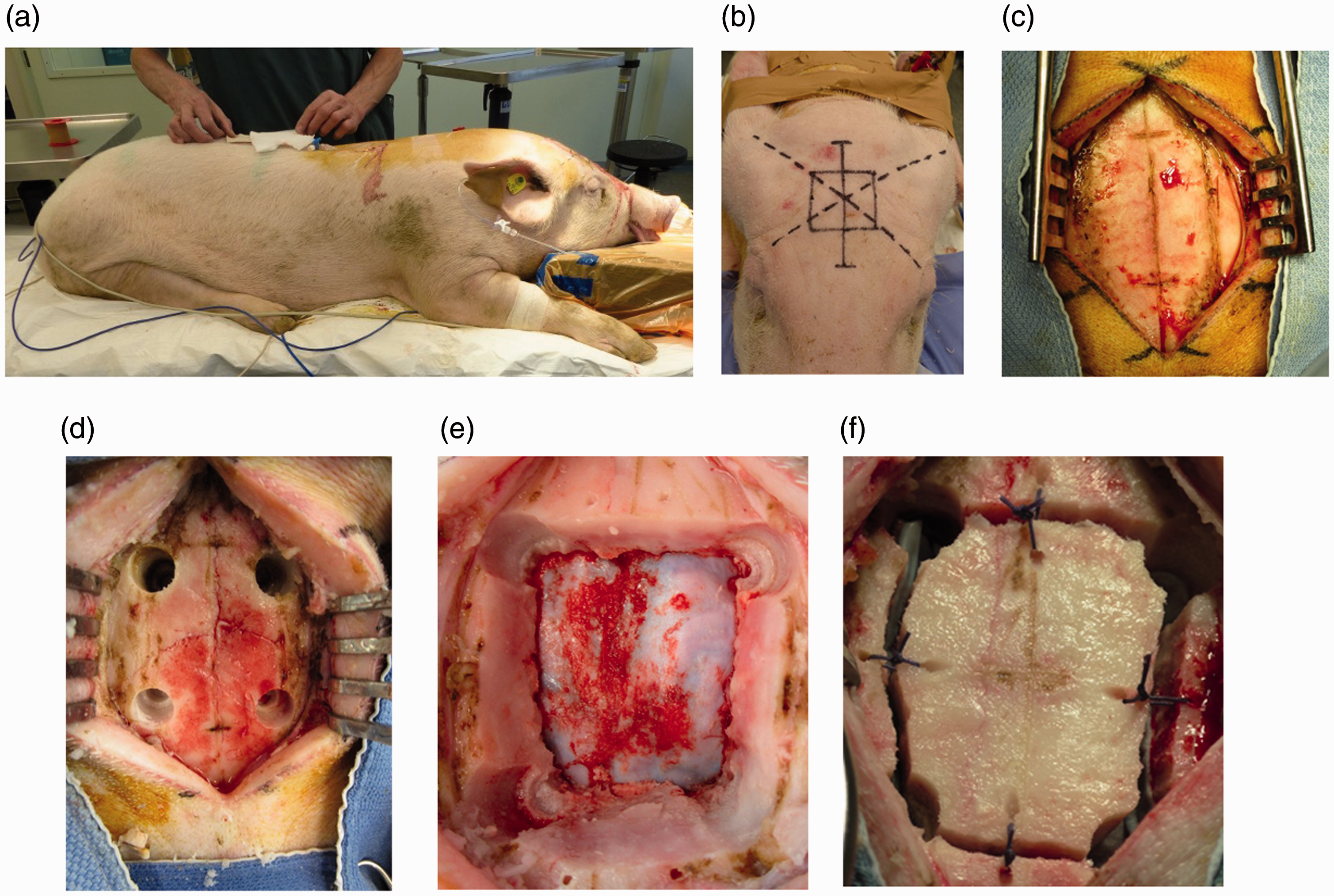
(a) Positioning of the pig. (b) Marking the skin incision. (c) Exposure of the craniotomy side of the skull. (d) Four burr holes forming the edges of the craniotomy. (e) The dura mater is exposed after the removal of the bone flap. Lyostypt® and gauze were placed above the sagittal sinus. (f) Bone flap fixed to the skull using one suture per side.
Postoperative course
The pigs were kept in the operating room after they were extubated until respiration and vital signs were normalized. They were observed by the same animal caretakers who cared for them before surgery. The general condition, neurological status, and wound healing of the pigs were examined twice daily for 2 weeks by a caretaker and the primary researcher (A.K.). All the pigs were weighed on arrival, day of surgery, and postoperatively, depending on the survival period. For their general condition, daily intake, defecation, and social behavior were noted. Neurological status was assessed by observing the alertness of the pig, the symmetry of the face and tongue, and the symmetric movement of the limbs without ataxia. The wound was inspected for leakage, redness, swelling, and warmth. Although any atypical behavior (decreased appetite, aggression, hyperexcitability, and being socially withdrawn) in pigs could be related to pain, head-shaking is specifically observed in piglets with pain after ear-notching. 4 Considering the nearness of the ear to the craniotomy site, we considered head-shaking as a sign of pain. Other behaviors suspected of pain were prostration, trembling, and/or stiffness. 4 Any suspicious behaviors of the pigs were discussed with the neurosurgeon and the veterinarian. If there was any suspicion of pain, additional intramuscular 0.05 mg/kg buprenorphine (Temgesic®, 0.3 mg/ml) was administered as rescue analgesia. If there was any sign of infection, blood samples were taken, and antibiotic treatment with amoxicillin/clavulanic acid was initiated.
Magnetic resonance imaging
Postoperative magnetic resonance imaging (MRI; Philips Ingenia 1.5 Tesla) was performed in all the pigs under anesthesia immediately before the scheduled euthanization. The cranial MRI included T1- and T2-weighted series, without and with contrast, a FLAIR series, and diffusion-weighted imaging (DWI)/apparent diffusion coefficient (ADC). Epidural hematoma, cerebrospinal fluid leakage, infection, or any intracerebral changes were assessed by a blinded radiologist at the Veterinary Department of the University of Utrecht. MRI was performed using Osirix (9.0, Pixmeo, Switzerland).
Results
Seven major and two minor complications occurred peri- and postoperatively in the 46 pigs. The major complications consisted of massive postoperative epidural hematoma (n = 3) and iatrogenic sagittal sinus rupture (n = 4). The minor complications included postoperative airway obstruction due to a sputum plug in one pig and a superficial wound infection at the jugular cannula in the second.
Epidural hematoma
Description
Massive postoperative epidural hematoma occurred in the three first pigs. On the second day after surgery, one pig developed a high fever, rhythmic movements of all the limbs, and decreased consciousness during the night. Blood samples for culture were immediately collected, and treatment with enrofloxacin 5 mg/kg (Baytril®, 100mg/ml) and dexamethasone 20mg (Rapidexon®, 2 mg/ml) was initiated. The pig died within 30 min after the animal caretaker initially noticed these signs. Postmortem MRI revealed a massive epidural hematoma with compression of the brain and cerebellar herniation (Figure 2). The CT-total body excluded other possible complications. Blood cultures remained negative. The pathologist confirmed the epidural hematoma as the cause of death.
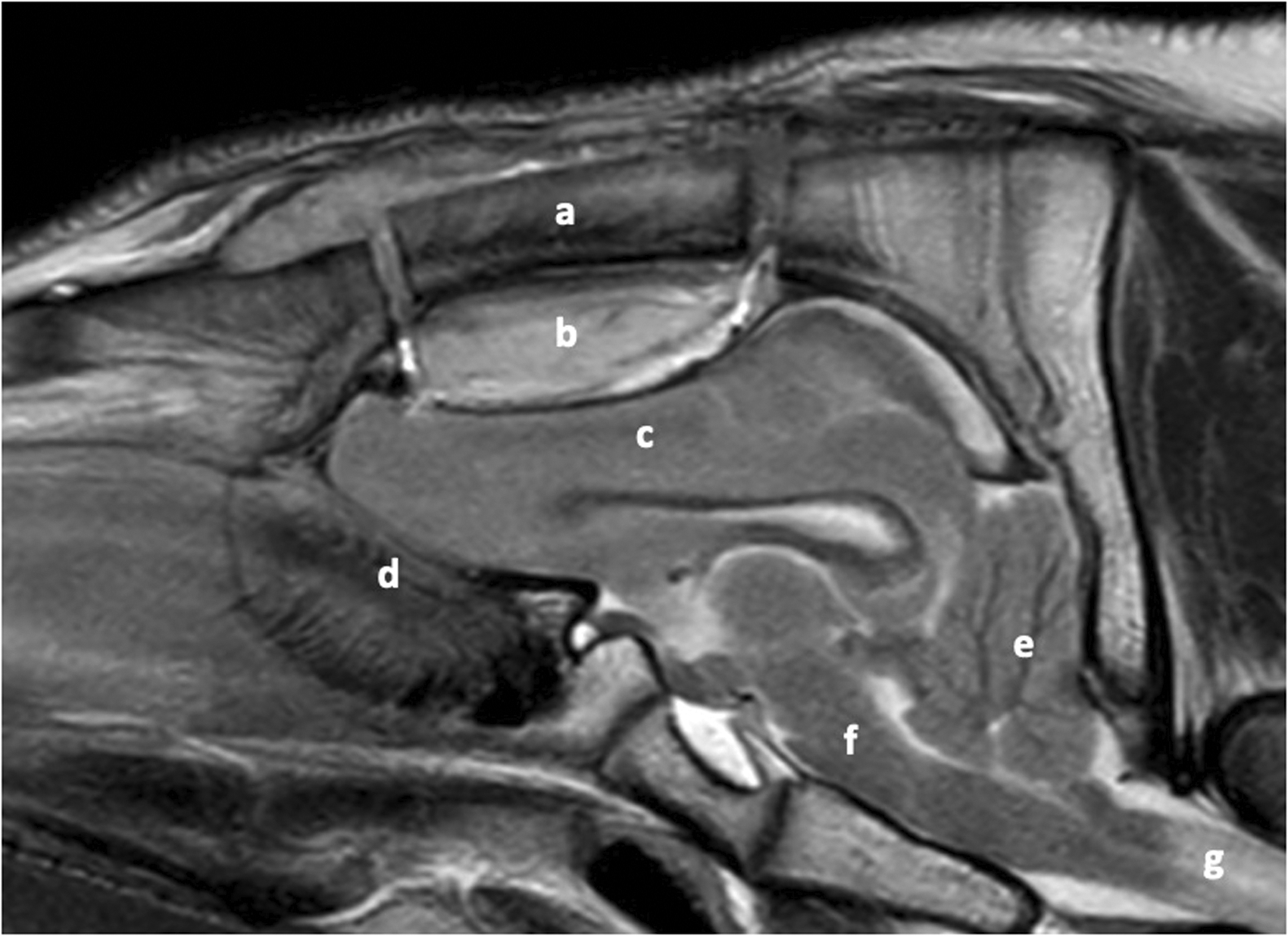
T2-weighted cranial magnetic resonance imaging (MRI) of the first pilot pig showing an extensive epidural hematoma with compression on the brain and herniation of the cerebellum into the foramen magnum. (a) Bone flap, (b) epidural hematoma, (c) cerebrum d) olfactory nerve, (e) cerebellum, (f) brain stem, (g) spinal cord.
The MRIs of the remaining two pigs also showed massive epidural hematoma before sacrifice on days 7 and 14. These pigs did not show any clinical signs of increased intracranial pressure. Remarkably, all three pilot pigs attempted to stand prematurely within 3 h, but they fell and hit their heads due to the after-effect of anesthesia. The epidural hematoma in these pigs may be caused by trabecular or dural vessel bleeding.
Prevention
Four measures were taken to minimize this risk. First, intraoperative blood pressure was maintained within normal postoperative values, and recovery time was shortened by changing the anesthesiology protocol. For maintaining anesthesia, midazolam (1.0 mg/kg/h) and sufentanil (0.01 mg/kg/h) were replaced by a first bolus propofol 3 mg/kg (Fresenius®, propofol 20 mg/ml (medium chain triglycerides/long chain triglycerides; MCT/LCT)) followed by intravenous propofol 4.5 mg/kg/h and remifentanil 0.0066 mg/kg/h (Ultiva® 2 mg lyophilized powder for solution).
Remifentanil was adjusted based on the parameters of pain during monitoring, such as an increase in blood pressure and/or heart rate with a maximum dose of 0.03 mg/kg/h. This change in protocol caused a change in the intraoperative mean arterial pressure (MAP) from 65 ± 37 mmHg to 104 ± 16 mmHg in the pigs during surgery.
Second, to detect the potential source of bleeding, blood pressure was shortly increased intraoperatively at the end of surgery above 150/100 mmHg with noradrenalin (0.03–0.06 mg iv). Subsequently, tranexamic acid (Sandoz® 100 mg/ml) 500 mg per pig was administered intravenously in all the cases before replacing the bone flap.
Third, bone wax (Ethicon, Inc.) was applied to the edges of the skull after the craniotomy to stop trabecular bleeding, and the bone flap was replaced using two sutures per side for strong fixation to the skull (Figure 3a).
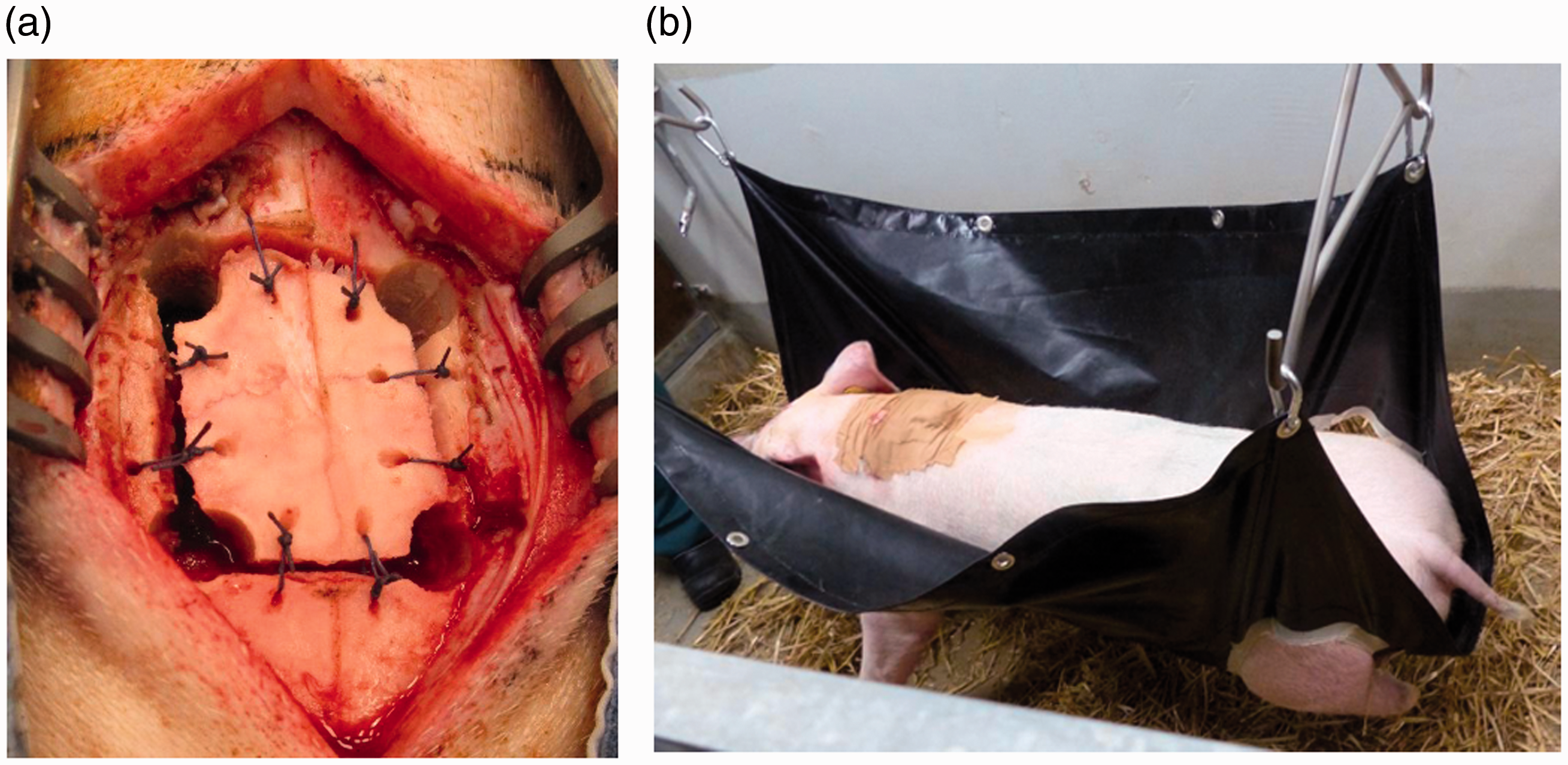
(a) Bone flap fixed to the skull using two sutures per side. (b) Postoperative course, pig laying in a hammock during recovery.
Finally, the pigs were placed in a hammock to avoid hitting their head during awakening (Figure 3b). When a pig could stand by itself, the hammock was lowered and freed. In addition, video recording of the pigs in the pens was introduced to evaluate the timing of events. After these measurements, the epidural hematoma did not recur, as observed on postoperative MRI of the first three pigs.
Sagittal sinus rupture
Description
Iatrogenic sagittal sinus rupture occurred in 4 of the 46 pigs (8.7%) during craniotomy (Figure 4). In these pigs, the dura footplate of the craniotome slipped under the dura, rupturing the dura and the sagittal sinus in the trajectory. The sagittal sinus rupture caused massive bleeding. These pigs were euthanized intraoperatively, as repair of the rupture carries a significant risk of venous infarcts and subsequent severe neurological deficits.
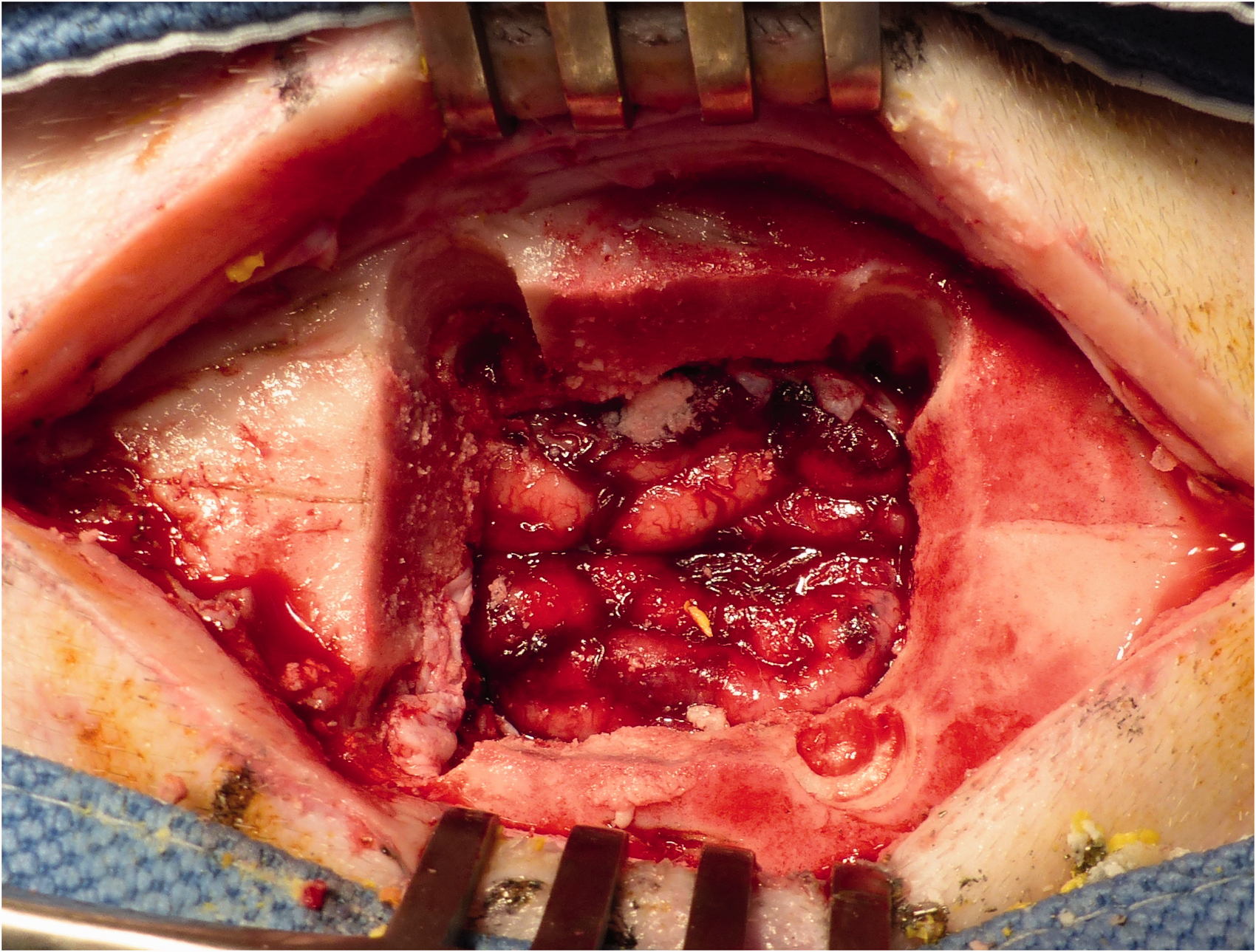
Complete rupture of the dura and sagittal sinus. The cortex is completely exposed.
Prevention
The cause of the complication seemed to be incomplete dural detachment from the skull. Standard dura dissectors and elevators could not sufficiently reach the dura through the drill holes because of the thicknesses of the skull of the pigs. As a solution, custom elevators from regular stainless steel kitchen knives were designed to fit the drill holes better and obtain the appropriate angle for reaching the dura through the drill holes (Figure 5). These elevators could also be used to free the dura after craniotomy through the craniotomy line and subsequently lift the bone flap.

Custom elevators made out of regular stainless steel kitchen knives. (a) 90° hook, 19-mm length and 7-mm width; (b) 45° 19-mm length and 7-mm width; and (c) 45°, 19-mm length, and 4-mm width.
Minor complications
In two pigs, a minor complication occurred: a postoperative airway obstruction in one pig and a superficial infection of the jugular cannula wound in another. The surgery of the pig with postoperative airway obstruction was uneventful. In the hammock, we observed that this pig had increasing airway obstruction. The pig was removed from the hammock and airway support, consisting of a chin lift and oxygen supply (9L), was provided. The pig coughed productively several times and recovered uneventfully within 15 min.
One pig developed superficial erythema and swelling at the location of the jugular cannula, which was suspected to be a superficial wound infection. Oral amoxicillin/clavulanic acid (Sandoz® 500/50 mg powder for solution) 500/50 mg was administered twice daily for 1 week, and the wound healed completely.
Discussion
This study describes learning encounters with craniotomy survival pig models. Although a craniotomy is technically not challenging, several measures had to be put in place to minimize the risk of major complications and improve animal welfare. During the study, we encountered two major types of complication: large epidural hematoma and sagittal sinus rupture. The measures taken affected anesthesia, surgical technique, and postoperative care.
Midazolam and propofol are the sedatives used most frequently in humans and animals. Human studies have shown that propofol has a faster postoperative recovery time.5,6 This was also observed in our pigs. Although propofol may have a higher potential to decrease blood pressure due to sympathetic activity reduction and intracellular calcium flux resulting in vasodilation, 7 this effect was not observed during surgery. The use of remifentanil instead of sufentanil also facilitated rapid recovery after surgery. This was consistent with findings of the study by Soltész et al., who showed a significantly faster response to commands in patients administered remifentanil than in those administered sufentanil for major surgery. 8
Custom elevators of sufficient length and angle were used to detach the dura through the burr holes. Despite this, the dura shifted into the craniotome and the footplate of the craniotome slipped under the dura in 4 of the 46 (8.7%) pigs. Detaching the dura from the skull may reduce the risk of iatrogenic sagittal sinus rupture, but it cannot completely prevent it. The preventive value of a burr hole above the sagittal sinus in pigs may be evaluated in future studies. 9
The length and angle of the elevators may also be adjusted to fit the size of the burr holes and the thickness of the skull in future studies performing craniotomies in animals. The same applies to the hammock, where the size and the distance between the limb gaps can be adjusted depending on the size of the animal.
Superficial wound infection occurred in 1 of the 46 (2.2%) pigs. Topical antibiotics were applied to all the pigs in addition to preoperative systemic antibiotics. Although debated, some human studies have shown that topical antibiotics may reduce surgical site infections relative to no antibiotics. 10 Because the surgical site infection rate in pigs is unknown, it was not possible to evaluate the added value of topical antibiotics in this study.
This study has several limitations. First, the study was rather a development of techniques aimed to increase chance of survival and minimize risk of complications. The methods developed were not compared with other alternative techniques; there was no match or comparison with other techniques for evaluation of their effectiveness. Thus, caution should be applied when considering use of the actions taken in the present study. Second, the MRI examination was performed only on the day of sacrifice, varying between 3 and 12 months post-surgery. No MRI examination was performed shortly after surgery. Latent intracranial bleeding without clinical consequences could therefore not be excluded in all the animals. Third, the analgesia in this study consisted of a transdermal buprenorphine patch for 7 days, intraoperative lidocaine and remifentanil, and meloxicam for 5 days postoperatively. Recent insights suggest that serum concentrations of transdermal buprenorphine are variable, and it is therefore not recommended. 11 Another limitation of this study was that it lacked a standardized scoring system for assessing and recording the signs of pain in the animals. The animals were observed mainly for deviant behavior that may indicate pain, including head shaking, prostration, trembling, and/or stiffness. 4 However, a standardized scale for pain, such as monitoring facial expression of the animals (Grimace Scale), allows the quantification of discomfort. 12 This scoring system was missing in this study.
The gross anatomy of the porcine brain and skull corresponds well to that of humans, and the porcine craniotomy model is well suited for medical research. Dogs have been used in previous craniotomy studies.4–6 However, they have become unsuitable, as ethical considerations and strong public feeling against the use of dogs as experimental animals highly limit scientific studies. The current study allows for a new standard in animal studies of intracranial devices.
A large craniotomy is feasible in pigs, and has an acceptable risk of complications if the appropriate anesthetic, surgical, and postoperative measures are taken into account.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772211009435 - Supplemental material for Minimizing complications in a porcine survival craniotomy model
Supplemental material, sj-pdf-1-lan-10.1177_00236772211009435 for Minimizing complications in a porcine survival craniotomy model by Ahmet Kinaci, Koen Vaessen, Saskia Redegeld, Albert van der Zwan and Tristan P.C. van Doormaal in Laboratory Animals
Footnotes
Acknowledgments
The authors would like to thank S. van Thoor BSc, Quality Manager of Brain Technology Institute, Utrecht, The Netherlands, for technical support and quality control. The authors acknowledge the encouragement, advice and constructive criticism of K. S. Han of the Department of Neurology and Neurosurgery Brain Center Rudolph Magnus, University Medical Centre Utrecht, The Netherlands.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.P.C. van Doormaal is a consultant for Polyganics B.V. and A. Kinaci’s PhD position is partially funded by Polyganics B.V.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the study was partially funded by Polyganics BV, Groningen, The Netherlands.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
