Abstract
Score sheets were first introduced 30 years ago to assess pain, distress and suffering in animals. To date, however, there is still no general agreement on their use in research practice, and only a few publications can be found on this topic. In the present work, we demonstrate the use of a special score sheet for severity assessment in the first three postoperative days in two showcased studies performed on Wistar and Lewis rats undergoing liver resection or orthotopic liver transplantation, respectively. Scoring of different criteria and the total score were evaluated within each intervention. Additionally, both procedures were compared regarding their degree of severity. Suitability of these score sheets was evaluated for assessing severity of the procedures and these showed a minor severity within each investigated study. A comparison of both studies showed slightly higher scores involving liver transplantation. In contradiction to the common classification of these procedures as a moderate severity grade the score sheets applied here indicates a minor severity grade within each investigated study. Also, limitations and possible improvements in the design of our score sheets for defined interventions are reconsidered.
The principles of replacement, reduction and refinement (3Rs) in animal research were first described by Russell and Burch in 1959. The idea behind this concept is to ensure the most humane treatment for laboratory animals as a prerequisite for successful experiments. 1 With the implementation of the EU Directive 2010/63 on the protection of animals used for scientific purposes, 2 the 3Rs are anchored within European law. One major point regarding the principle of refinement is the minimization of pain, suffering or distress. Therefore, the recognition of pain and distress is a crucial criterion for assessing the level of discomfort in animals.
Several techniques have been developed for grading the level of pain in humans based on self-report measures of patients such as visual rating scales or facial pain scales. 3 These techniques represent the gold standard as used in humans for over 30 years, but a translation to animals is impossible due to the fact that animals cannot report verbally. This necessitates a method to reliably recognize pain and distress based on observations by the person conducting animal experiments. Morton and Griffiths were the first who selected criteria describing deviations from the normal behavior of animals, which can be a sign of pain or distress. 4 This concept was taken up by various groups of scientists and adapted to their particular application, such as the assessment of discomfort in hepatomegaly in mice and rats, 5 the application of humane endpoints, 6 or the assessment of pain after laparotomy in rats.7,8
Up to today, scoring has rarely been reported as a refinement measure in procedures involving laparotomy in rats, only scarce evidence can be found on this topic. Roughan and Flecknell have compared the effects of different analgesic agents on the postoperative behavior of rats.9,10 They have improved their scoring system regarding behavioral peculiarities found in these studies, and used it to also test different analgesic protocols after laparotomy. 8 Another study performed by Sotocinal et al. in 2011 have introduced the rat grimace scale as a method for quantifying pain following laparotomy. 11 Nevertheless, according to the best of our knowledge, there are no studies available on a score sheet-based severity assessment after laparotomy in general or liver surgery specifically.
Therefore, the aim of the present paper is to report the results of severity assessment after liver resection and liver transplantation in rats using a score sheet and to give recommendations for the prospective use of this tool. In brief, we scored rats undergoing liver resection or orthotopic liver transplantation for three postoperative days, implementing a general score sheet based on the original reports. 4 A further aim of this article is to analyze the feasibility of using a score sheet, and to publicize its advantages and limitations regarding both surgical procedures.
Experimental procedure
Experimental animals and statistical analysis
All animal experiments were performed in accordance with the German Animal Welfare Law and the EU Directive 2010/63. The experimental protocol was approved by the governmental animal care and use committee (Landesamt für Natur, Umwelt und Verbraucherschutz (LANUV) Nordrhein-Westfalen, Recklinghausen, Germany). The rats were housed in groups of five per cage under specific pathogen-free conditions with a 12 h light and dark cycle in a temperature- and humidity-controlled environment according to the Federation of European Laboratory Animal Science Associations (FELASA, www.felasa.eu) recommendations. Water and standard diet for laboratory rodents (ssniff GmbH, Soest, Germany) were offered ad libitum. In accordance with the German Society of Laboratory Animal Science (GV–SOLAS) recommendation, ‘Pain management for laboratory animals 2015’, all the rats were treated with 0.1 mg/kg of buprenorphine subcutaneously as preoperative analgesia as well as postoperatively every 12 h for three days. 12 The number of animals per group was calculated via a power calculation using G*Power, version 3.1.9.2 (Freeware, Kiel University, Kiel, Germany). All the animals were allocated randomly to their groups, and they all completed the study (i.e. no dropouts). Data are expressed as mean ± standard error of the mean (SEM). Statistical analyses were performed using GraphPad Prism version 5.00 (GraphPad Software, San Diego, CA, USA). Data from the liver resection or the liver transplantation were analyzed by one-way analysis of variance (ANOVA) with Tukey’s post hoc test. For the comparison of both protocols repeated measures ANOVA were performed. Differences between groups were considered significant when P < 0.05.
Liver resection
For the liver resection experiments, 120 male Wistar rats (CS 4105; Janvier Labs, Le Genest-Saint-Isle, France) with a weight of between 290 and 400 g and aged 8–12 weeks were used. They were randomly allocated to 12 experimental groups (experimental unit) and liver resection was performed to investigate the efficacy of a tissue glue for bleeding control from the resection surface compared with the clinical gold standard (fibrin glue). The non-anatomic hepatic resection of the left lateral lobe was performed as described by Aysan et al. 13 and Henderson et al. 14 in 2010 (Figure 1). On days 14, 21 and 90 after intervention, animals were sacrificed to investigate the liver resection area as well as the histological, hematological and biochemical parameters. Because all the animals were scored for three days after intervention using identical score sheets, animals from all the experimental groups could be included in our study.
Liver transplantation
Whole-graft orthotopic liver transplantation (OLT) was performed on 40 male Lewis rats (LEW/OrlRj; Janvier Labs) with a body weight of between 260 and 320 g and aged 8–10 weeks, according to our protocol published previously.15,16 Briefly, total hepatectomy was performed on the donor animals and their livers were stored in a cold ischemic time for 8 h at 4°C in an organ preservation solution (Figure 2). Recipient animals were randomly assigned to the following experimental groups: (i) control group without remote ischemic conditioning, (ii) remote ischemic conditioning before liver excision and recipient hepatectomy (RIC1), and (iii) remote ischemic conditioning after graft reperfusion (RIC2). Remote ischemic conditioning treatment of the recipient animals was performed as described previously. 17 Cold ischemic time (478.2 ± 9.0 min) and anhepatic time (18.6 ± 0.7 min) were comparable for all groups. One-week survival rate was 100% in each group.
Score sheet
Design of the score sheet used.

Results
Liver resection
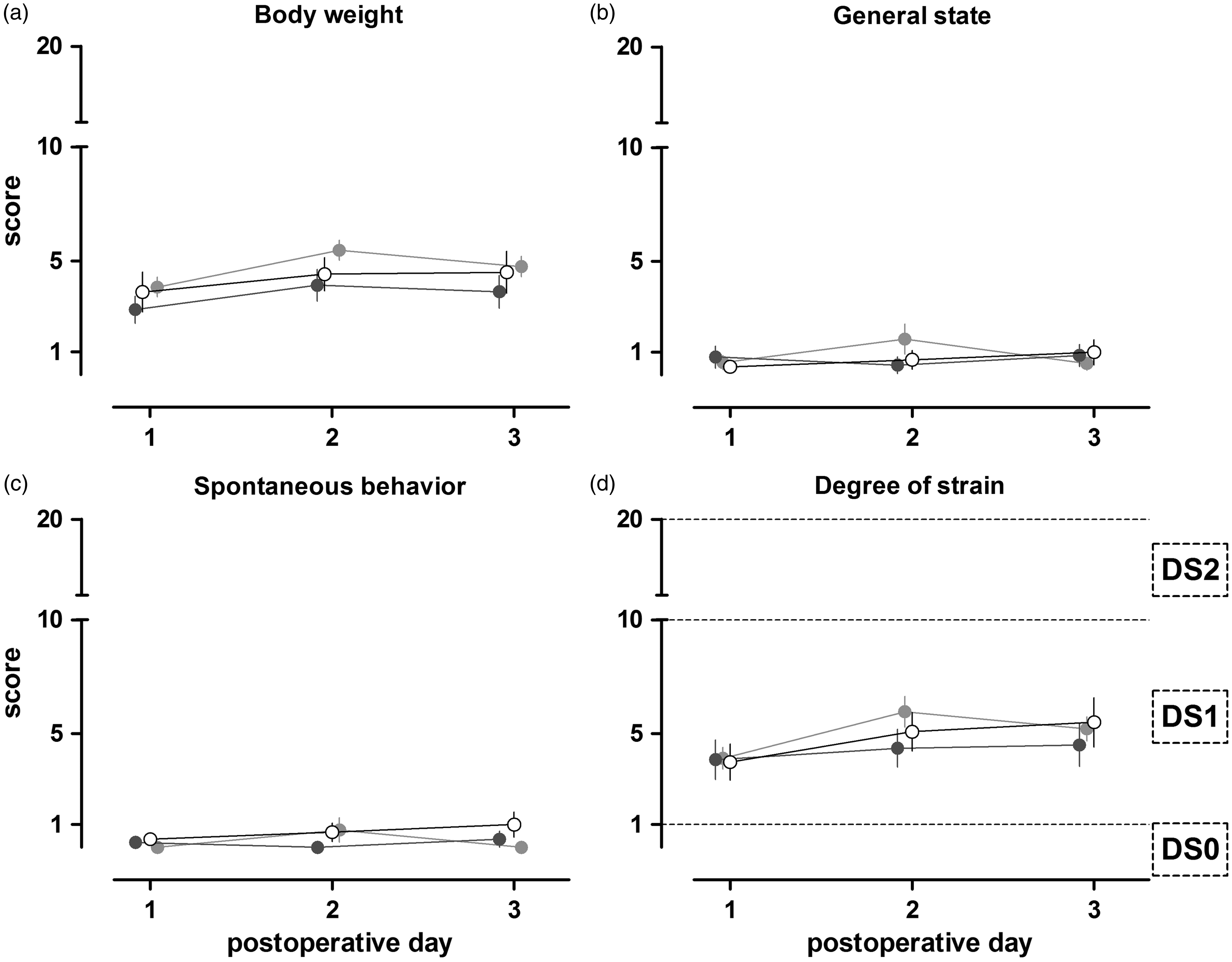
In Figure 3, the parameters of body weight, general state, spontaneous behavior and DS are shown. In general, the condition of all the rats was good and a total score of 6 points was not exceeded. No significant differences were detected between the rats treated with the test glue, gold standard or saline (Figures 3a–c). The score for body weight of all groups increased slightly on day 2, and decreased again on postoperative day 3. The scores for the parameters of general state and spontaneous behavior were constant, over the observation time, of around 1. All the animals’ scores ranged between 1 and 9 points (DS1), resulting in a classification of minor according to the score sheet used (Figure 3d).
Liver resection technique: A) After median laparotomy, the liver is positioned for resection B) The left lateral lobe is resected with a scissor C) The liver resection surface is sealed with a glue.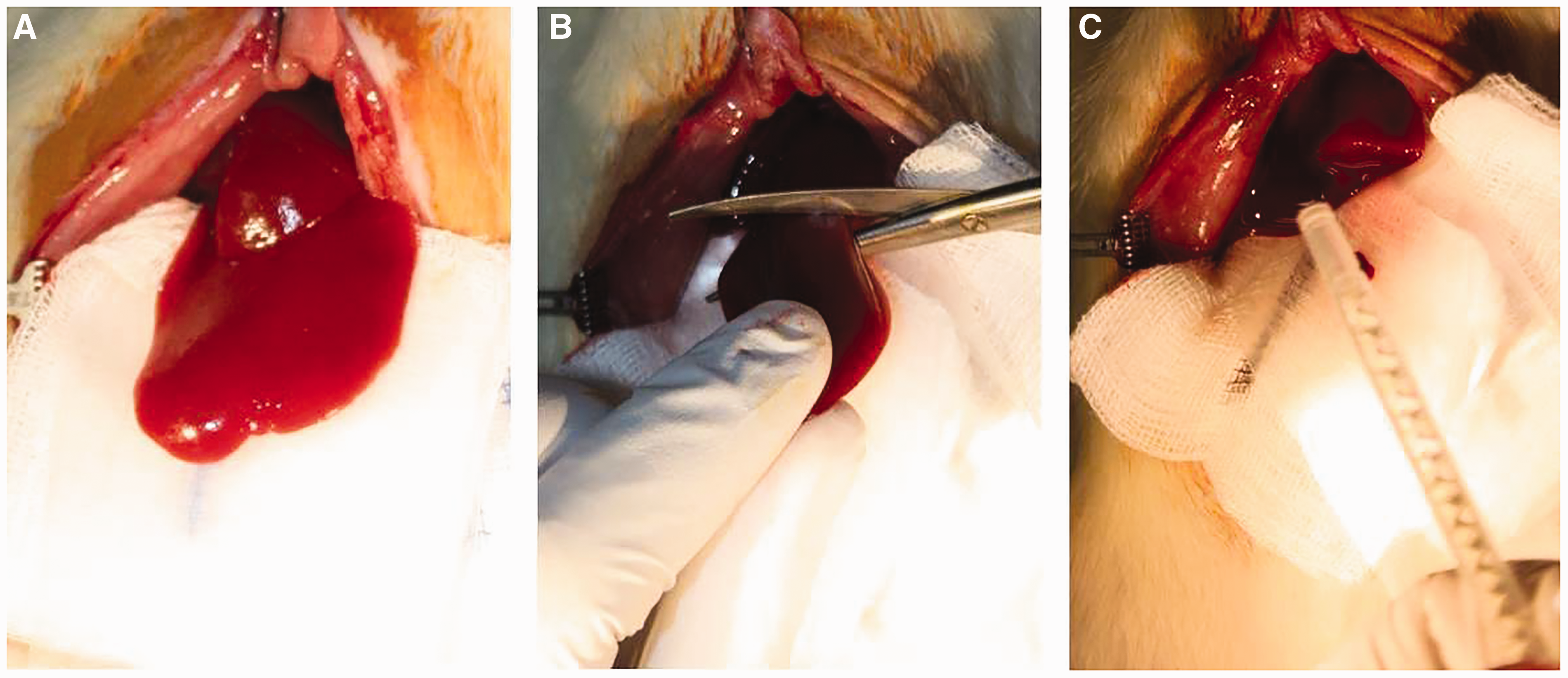
Liver transplantation
The changes in body weights of rats that underwent liver transplantation were scored as between 1 and 5 points over the examination period of three days. The parameters of general state, spontaneous behavior and clinical signs (data not shown) were slightly increased (1 point) on postoperative day 1 but were already decreased again on day 3 (Figures 2a–c). All animals that underwent liver transplantation were assigned DS1, which was defined as a minor strain according to the score sheet used (Figure 2d). No significant differences were found between treatment groups.
The timeline of the liver transplantation experiment. Scoring of rats that underwent liver resection for three postoperative days. The parameters body weight (a), general state (b), spontaneous behavior (c) and the degree of strain (d) of animals treated with saline (n = 12), clinical gold standard (n = 14) or a test glue (n = 25) are depicted. The degree of strain (DS) is defined as DS0 (no strain), DS1 (minor strain) and DS2 (moderate strain). More than 20 score points (DS3) which is associated with severe strain, were not reached in our experiments. Values are depicted as mean ± SEM.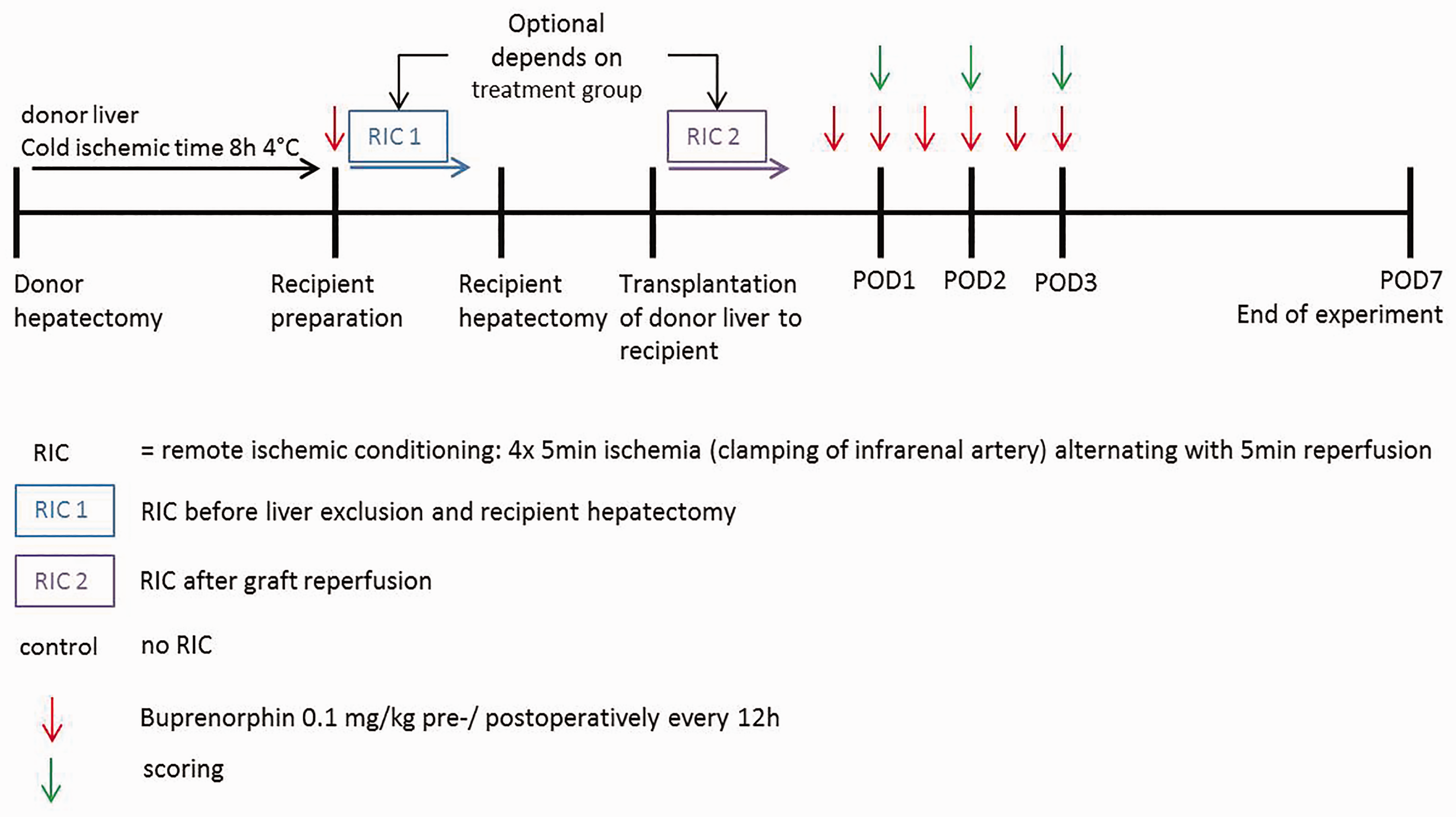
Comparison of liver resection and liver transplantation
Comparing both protocols (Figure 4), rats that underwent liver transplantation showed a slightly, but not significantly, higher score when the DS was analyzed (mean transplantation control: postoperative day [POd]1: 6.9 ± 0.7 SEM, POd2: 7.1 ± 0.6 SEM, POd3: 7.1 ± 1.3 SEM; mean resection gold standard: POd1: 3.9 ± 0.9 SEM, POd2: 4.4 ± 0.8 SEM, POd3: 4.5 ± 0.9 SEM). the scoring for the body weight of both interventions showed only slight but not significant differences (mean transplantation control: POd1: 3.9 ± 0.7 SEM, POd2: 4.4 ± 0.6 SEM, POd3: 5.1 ± 0.9 SEM; mean resection gold standard: POd1: 2.9 ± 0.6 SEM, POd2: 3.9 ± 0.7 SEM, POd3: 3.6 ± 0.7).
Comparison of both liver interventions. The parameters body weight (a) and degree of strain (b) of both rats that underwent liver resection (n = 14) or liver transplantation (n = 8) are shown. Rats showed a slightly higher but not significant degree of strain after liver transplantation compared with liver resection. This difference was not caused by their body weight alterations, which did not differ in score after both interventions. Values are shown as mean ± SEM.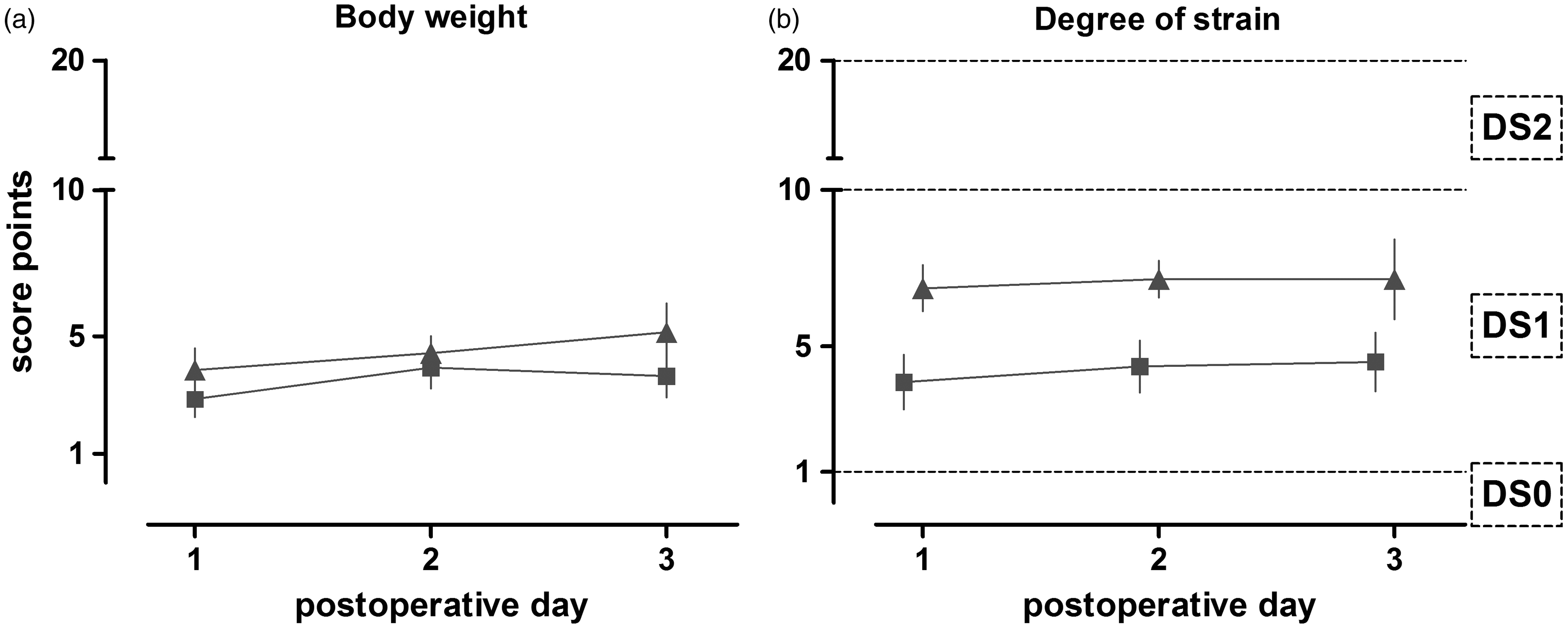
Discussion
The use of score sheets for assessing pain and distress was first suggested by Morton and Griffiths in 1985. 4 They introduced general parameters that can be altered in line with pain or distress in animals; and deviations of the normal behavior were categorized into four groups: no obvious deviation from normal, a minor change, a definite change, and a gross change from normal. Depending on the respective group, scores of 0 to 3 were assigned, with 0 indicating no deviation from the normal range. The scores from all groups were then summed up to a total score, and treatment recommendations were then prescribed according to this final value. Nevertheless, the authors emphasized that with increasing knowledge regarding special procedures, these parameters should be modified accordingly. To date only a few applications of score sheets can be found in the literature,5,6 all of which are modifications of the initial guidelines by Morton and Griffiths. Most studies from the pioneering era of animal welfare have focused on the effects of analgesia on animal behavior following procedures involving laparotomy but not on the effects of the protocol itself. 7 The present work implements the use of score sheets to estimate the degree of pain and distress of animals after liver resection or orthotopic liver transplantation, and also to retrospectively assess severity, focusing on the surgical procedure itself, while using appropriate analgesic treatment as a basic prerequisite for these major surgical interventions (see methods). As there are no recommendations for the design of score sheets for these specific procedures, the initial layout by Morton and Griffith was used. An advantage of this design is the good understandability of the described parameters and their alterations and, therefore, the easy assignment of scores to those. This is a crucial point in designing score sheets, as all personnel involved in the experiments should be able to clearly understand the score sheet. 18
When more than one person is involved in the assessment, between-assessor variations have to be taken into account, because such scoring is partly based on a subjective evaluation. In the described study, we reduced inter-observer objectivity within each protocol by appointing one senior technician for the scoring of all rats on all postoperative days. Nevertheless, a limitation is the fact that between both protocols (resection and transplantation), different personnel were responsible for the scoring of the rats. One possible way of avoiding personal bias is to have several observers, and to calculate average scores for each animal. 5 Furthermore, to evaluate subjective parameters, such as general state or spontaneous behavior, extensive knowledge of normal, healthy behavior of the animals is absolutely necessary. In order to improve personnel skills in better discriminating normal from abnormal behavior, Roughan and Flecknell have suggested using video-based training 8 to recognize relevant signs. As it is essential for their survival in a natural environment, rats often mask pain-related behavior; video analysis of undisturbed animals can also be used to observe alterations in spontaneous behavior. In a study by Roughan and Flecknell it was possible to identify uncommon criteria related to pain after laparotomy, such as back-arching, staggering or writhing. 7 The video-analysis technique could also be used to monitor the animals during the night, as rats are nocturnal and changes in behavior or activity could be observed more easily during the active time of the animal. 19 Another possibility for assessing masked behavioral changes in rats is to measure their motor coordination, i.e. the use of a Rotarod or parallel bars, where the animals have to solve a specified task, and scores could then be assigned to the time needed to fulfill that task. 20 A relatively new approach in detecting pain is the rat grimace scale, where facial expressions of the animal are detected using photographs from video-material and assigned to different states of pain. 11 This method is translated from the facial action coding system which is used for the detection of pain in non-verbal human patients. 21 The aforementioned techniques can be applied to minimize the subjectivity in assessing the spontaneous behavior of animals.
Besides the subjectively evaluated parameters (general state and spontaneous behavior), we also used objective parameters, such as body weight and clinical results. With liver resection and transplantation, body weight is not always a reliable parameter because the original body weight might change as a result of the intervention itself (resection of the liver, slight differences between recipient liver and the graft, weight gain from fluid resuscitation, etc.). This could be avoided by weighing all animals immediately after surgery to register the post-intervention body weight, and defined as the new baseline. Furthermore, analgesic treatment with buprenorphine should also be taken into account, because as an opioid it could possibly lead to strong obstipation, among other effects. Nevertheless, body weight can be evaluated as an objective parameter, although it must be interpreted very carefully after liver resection and transplantation as well as following any other major surgical interventions. Thus, it may be reasonable to supplement the scoring system with a body condition score. Using these scores, the amount of muscle and soft-tissue covering bony parts of the body, mostly the dorsal part of the hip, can be examined and estimated. These values should show a better correlation with the amount of subcutaneous fat stored. 22
Another limitation of using our score sheet to determine severity after liver resection or transplantation is that acute complications, such as shock or internal bleeding, cannot be detected with the present design. Due to the aforementioned deficiencies of the design, it is possible that animals scored with no or only a minor DS could die suddenly as result of a severe acute complication. Here we would like to emphasize the importance of performing thorough autopsies in all cases of sudden unexpected death in every research setting to underline that the scoring did not fail. This problem again highlights the necessity of adapting a general score sheet to an experiment. In doing so, performing a pilot study could be beneficial. Within the framework of a pilot project, animals could be observed intensively using continuous video recordings. The recorded materials could then be used to retrospectively evaluate the observed, maybe procedure-specific, alterations, which are not included on a general score sheet. For the main study a score sheet, preferably containing the identified important parameters, could be created based on the observations of the pilot study. This would result in an easily understandable scoring system, which could be used to observe large numbers of animals. The establishment of a web-based database of a pool of specific alterations observed after certain procedures might help researchers to select the most relevant parameters from this pool, and export them to an adapted score sheet which best fit their study design (measures for refinement and reduction – in line with the 3Rs).
In the studies described, we observed a slightly higher DS in rats that underwent liver transplantation compared with animals that underwent liver resection, however this was not statistically significant. This difference in DS was not caused by body weight changes, as it was comparable in both interventions. However, as two independent protocols were compared in the present study, it has to be mentioned that the scoring was performed by different observers. Therefore it cannot be excluded if a real difference in severity of both procedures was detected, or if the difference resulted from inter-observer deviation.
All animals in the studies described were assigned to a DS of 1, which was defined as a minor strain in our score sheet. This is in contrast to the severity classification in EU Directive 2010/63/EU Appendix VIII, 2 where laparotomy and organ transplantation are both assigned to the moderate severity category. This strongly indicates the need for further evaluation of our scoring system with special attention to the sensitivity and comparison with other scoring systems. The consequent use of precisely evaluated score sheets after all interventions, such as liver surgeries, can lead to a better and more precise evaluation of severity. Therefore, the use of score sheets should be obligatory in order to obtain detailed information about the severity of different interventions based on empirical data. By this means the score sheet could also be used as a refinement tool to improve the health and well-being of animals after interventions such as liver resection or transplantation, respectively. Using analgesia as one example, the type and dosage of analgesics used can possibly lead to severe postoperative complications due to numerous side-effects. 23 It would be beneficial to adapt the analgesic dosage to each animal depending on its individual condition, which would be possible by using the precisely-evaluated score sheet to detect the actual condition. In the present study a standard analgesic therapy was applied according to the GV–SOLAS recommendations for pain management of laboratory animals, meaning that the dosage of analgesia was not tailored individually to each animal. Nevertheless, the animals did not show any strong alterations in their spontaneous behavior, leading to the assumption that no or only few side-effects, as a result of an overdosage or signs of pain due to an inadequate dosage, occurred.
However, keeping all points in mind, more score sheet data have to be collected from different protocols in the field of liver surgery to ‘fine-tune’ severity assessment after these interventions in order to establish a robust basis for welfare assessment. This reliable data-collection would also allow the scientific community to perform and publish preclinical meta-analyses for severity assessments in specific procedures, which again contributes to the possibility of reducing the numbers of animals used which is in line with the principle of the 3Rs. 1
Conclusion
Within this article we were able to show that use of a score sheet is a simple and effective way of assessing severity following major liver surgery. The general condition of the animals was recorded, and slight but not significant differences were shown between two different interventions. Although the initial idea of a score sheet was first implemented in 1985, there are still ongoing discussions within the scientific community on the reliability of these score sheets. Based on the present work, we can conclude that the use of score sheet is in general a suitable tool for evaluating severity after liver resection and transplantation. However, limitations are identifiable in our score sheet, and several improvements might be needed in the future to assess the condition of the rats in an adequate way. Additional measures such as pilot operations, video recordings, thorough autopsies in all cases of sudden death of animals and modifications in the scoring system to meet the special needs of certain procedures, might increase the overall sensitivity and specificity in detecting complications. In general, due to the lack of verbal communication of animals, we as humans have to be able to recognize and evaluate signs of pain and distress, and the method presented is a good way of fulfilling this task, at least in part.
Footnotes
Acknowledgement
The authors would like to thank Pascal Paschenda for his excellent technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
