Abstract
According to European Union directive 2010/63/EU a severity classification of experimental procedures performed on laboratory animals is mandatory. This includes a prospective evaluation of all interventions performed within the experiment, as well as an assessment of the actual burden of each animal during the experiment. In this regard, the evaluation and scoring of defined criteria regarding the health state of animals could help to early identify deteriorations in animal health and facilitate the application of humane endpoints. This article discusses the applicability of an adapted score sheet in BALB/cAnNRj mice receiving either cisplatin, doxorubicin or busulfan, three chemotherapeutic agents with different toxicological profiles and longitudinal non-invasive molecular imaging. The health state was investigated by score sheets documenting general state, body weight, spontaneous behaviour and treatment specific parameters (e.g. anaemia, neurotoxicity, persistent diarrhoea). Although blood and serum analyses clearly indicated various organ damage, most scoring parameters except for body weight did not report on the deceasing animal health state. Thus, there is need for more sensitive observational parameters to judge the animal's health state and welfare.
The severity classification defined by EU directive 2010/63/EU includes a prospective evaluation of all interventions carried out on a single animal and an assessment of the actual burden during the experiment. 1 The prospective severity classification estimates the highest level of suffering any animal has to experience during the procedure and should assist in the planning of welfare monitoring intervals and proper humane endpoints. 2 Furthermore, the actual severity each animal experiences during a procedure has to be determined and documented using appropriate observation techniques. This is facilitated by score sheets ideally including easy objective measurements to detect pain, distress and suffering.2,3 The first concept of a rating system that describes parameters assessing pain and distress in animals to evaluate severity of behavioural or physiological alterations was introduced by Morton and Griffith. 4 Although this concept is still in use for the basic design of score sheets, a modification with experiment-specific parameters is necessary to prevent the risk of scoring insignificant parameters or missing essential symptoms that could result in a false evaluation of animals' health state.3,5 In the present study a score sheet designed to assess the well-being of mice during chemotherapy and longitudinal non-invasive imaging was re-evaluated regarding the suitability of the applied parameters to detect signs of discomfort. Furthermore, parameters are discussed that may detect animal well-being more sensitively and thus, improve the quality of score sheets.
Material and methods
Animals
Animal experiments were performed in accordance with the German Animal Welfare Law and the EU Directive 2010/63/EU. The experimental protocol was approved by the governmental animal care and use committee. Female BALB/cAnNRj mice (age 10–12 weeks; Janvier Labs, Saint Berthevin, France) were housed in groups of 3–5 animals on spruce granulate bedding (Lignocel, JRS, Germany) under specific pathogen free conditions in type II long individually ventilated cages (Tecniplast, Germany) with a 12 h light and dark cycle in a temperature (20–24℃) and humidity controlled (45–65%) environment according to the guidelines of the ‘Federation for Laboratory Animal Science Associations’ (FELASA, www.felasa.eu). One nestlet per cage was provided to enable nest building. Water and standard pellets for laboratory mice (Sniff GmbH, Soest, Germany) were offered ad libitum. Group-housed animals were assigned individual earmarks for identification. The present study is a subgroup analysis of an experiment for evaluation of a new radiotracer for detection of chemotherapy side effects. 6 No additional animals were used for evaluation of score sheets.
Chemotherapy
Mice were allocated randomly to the following experimental groups: 11 mice received 20 mg/kg BW (body weight) cisplatin (Teva, Ulm, Germany) in 0.9% sodium chloride intraperitoneally (ip) on day 0. Longitudinal PET-CT (positron emission tomography-computed tomography) measurements were performed on day 1, 2 and 3 after injection in five animals. On days 1 and 2, three animals were sacrificed by cervical dislocation for further analyses, respectively. Well-being was assessed for 3 days after injection.
Doxorubicin (4 mg/kg BW in 0.9% sodium chloride, LC Laboratories, Woburn, USA) was injected ip in 11 animals on days 0, 7 and 14. Longitudinal PET-CT measurements were performed on days 8 and 15 after injection in five mice. On days 8 and 15, three animals were sacrificed by cervical dislocation for further analyses. Well-being was assessed for 17 days after the first injection.
On days 0, 3, and 5, 11 mice received 18.8 mg/kg BW busulfan ip. Longitudinal PET-CT measurements were performed on days 3, 5 and 10 after injection in five animals. On days 3 and 5, three animals were sacrificed by cervical dislocation for further analyses. Well-being was assessed for 10 days after the first injection. Additionally, nine untreated control mice were examined once by PET-CT.
Blood sampling and analysis
Before euthanasia, blood and serum were collected from the retro bulbar venous plexus under inhalation anaesthesia with 2 vol% isoflurane (Forene, Abbott, Wiesbaden, Germany) in 1 l/min oxygen. Red blood parameters were assessed using a haematology analyser (Celltak alpha MEK-6550, Nihon Kohden, Tokyo, Japan). The following serum parameters were measured using the Vitros 350 Chemistry system (Ortho Clinical Diagnostics, Neckargemünd, Germany): aspartate transaminase (AST), alanine aminotransferase (ALT), urea, creatinine, creatine kinase and albumin.
Imaging
PET-CT imaging was performed using the Triumph II small animal PET/SPECT/CT (TriFoil Imaging, Dijon Cedex, France). 5–10 MBq of a cell death specific radiotracer 7 was injected intravenously in a maximal volume of 10 ml/kg BW. Two hours after injection mice were anesthetized using 2 vol% isoflurane (Forene, Abbott, Wiesbaden, Germany) in 1 l/min oxygen and placed on a tempered animal holder.
Welfare assessment
Score sheet design.

Statistical analysis
The number of animals per group was calculated using a power calculation. 8 Data are expressed as mean ±SD (standard deviation). Statistical analyses were performed using Graph Pad Prism 5.0 (Graph Pad Software, San Diego, USA). Data were analysed by One-way ANOVA with Tukey's post-test. Data were considered significant when p < 0.05.
Results
Cisplatin treatment
Cisplatin-treated mice decreased in body weight resulting in a score of 2.2 ± 2.5 (day 1) and 2.8 ± 3.6 (day 2). On day 3 body weight was decreased to approximately 10% of baseline and scored with 9.0 ± 2.2 points. Alterations of general health state could only be detected on day 3 when the fur showed a slightly scrubby appearance, and mice were scored with 1 point. All other parameters did not show alterations. The overall degree of strain defined by the total score could be allocated as a mild strain throughout the experiment (Figure 1(a)).
Severity assessment after cisplatin treatment. (a) Cisplatin treatment resulted in a mild severity in mice. A reduction in body weight was the main parameter that could be scored. On the last day slight changes in the fur appearance became visible. • timepoint of chemotherapy injection. (b) Body weight changes after injection of cisplatin. Data presented as mean ±standard deviation, *p < 0.05, n = number of animals.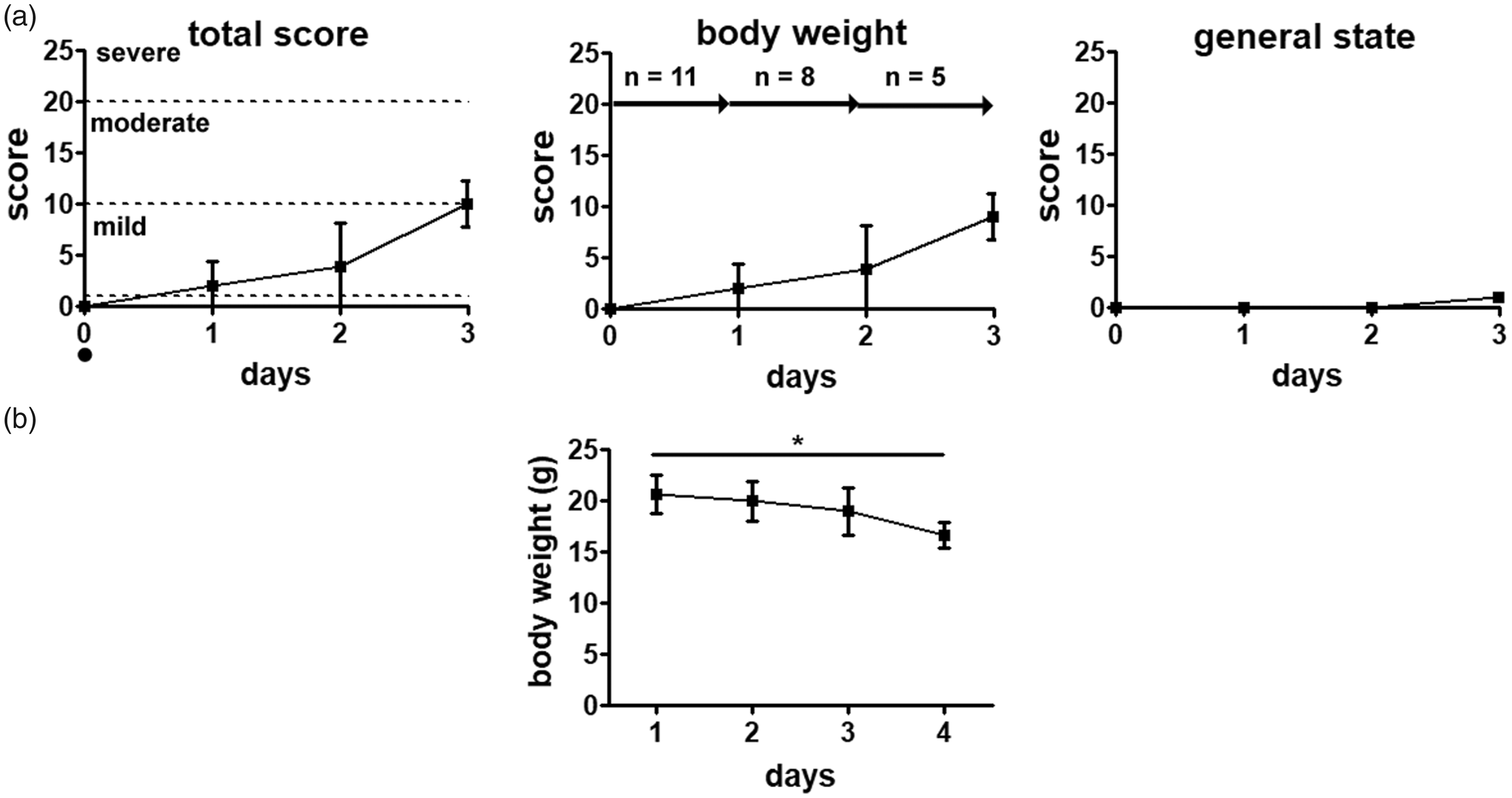
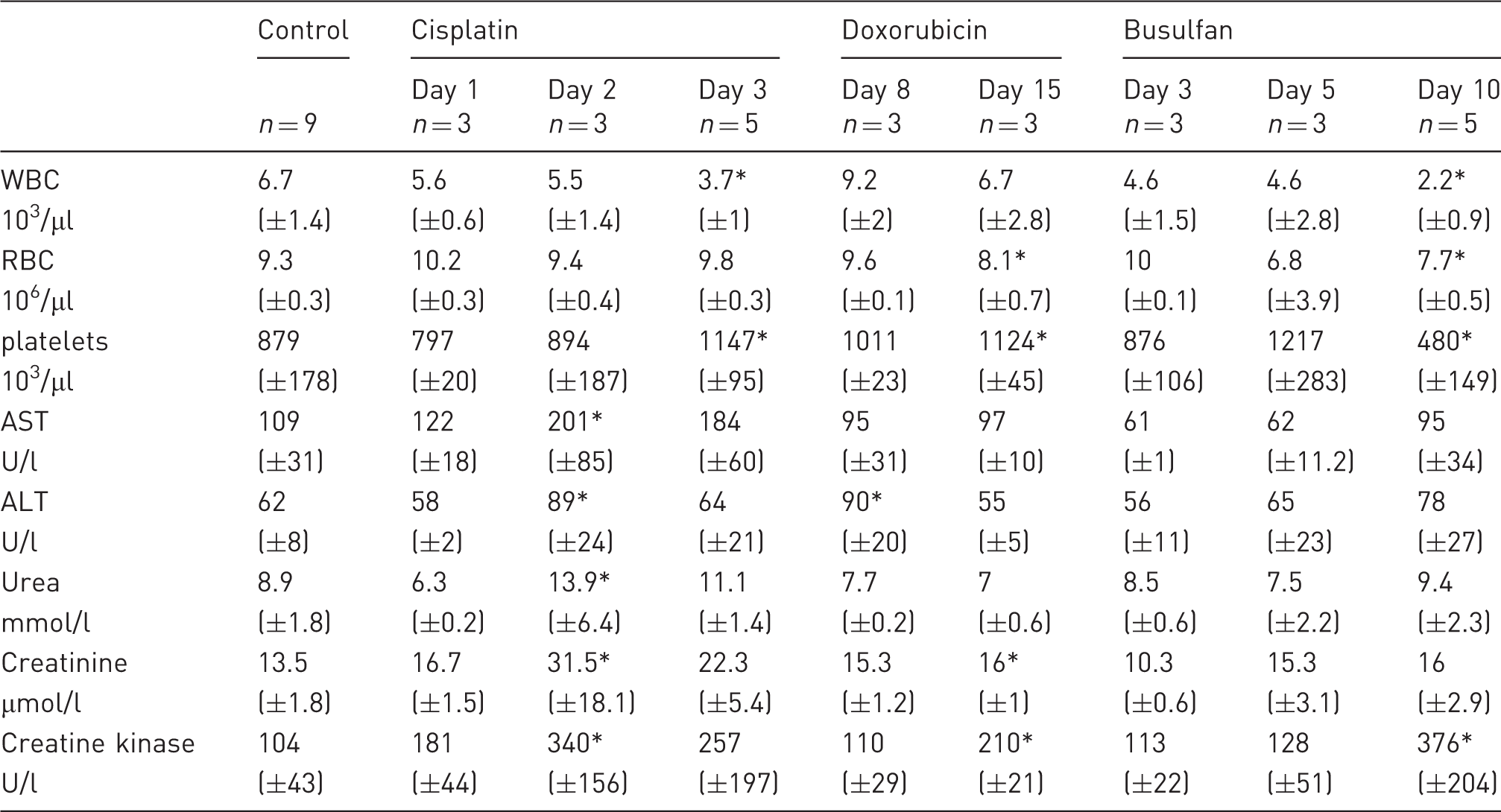
Blood count and serum parameters of control, doxorubicin-, busulfan- and cisplatin-treated mice. Cisplatin and busulfan decreased the number of white blood cells (WBC). The number of red blood cells (RBC) was significantly lower after doxorubicin and busulfan treatment. While cisplatin and doxorubicin increased the number of platelets, busulfan treatment resulted in a reduced platelet count. Cisplatin was the only chemotherapeutic agent that increased serum aspartate transaminase (AST), whereas alanine aminotransferase (ALT) was elevated after both cisplatin and doxorubicin treatment. Furthermore, cisplatin increased urea and creatinine values, whereas doxorubicin treatment only resulted in higher creatinine values. All chemotherapeutics increased serum creatine kinase. Data are presented as means ( ± standard deviations). *p < 0.05.
Doxorubicin treatment
Body weight of doxorubicin-treated mice was almost stable during the first 13 days of treatment and started to decrease from day 14 onwards, resulting in a decrease of approximately 11% and a scoring of 6.7 ± 5.8 points. According to the score sheet, doxorubicin only had a mild strain (Figure 2(a)).
Severity assessment after doxorubicin treatment. (a) Doxorubicin treatment lead to a slight decrease in body weight resulting in a mild severity for the animals. Although the score sheet gives no indication, all mice died suddenly on day 16 and 17. • timepoint of chemotherapy injection. (b) Body weight changes after repetitive injection of doxorubicin. Data presented as mean ±standard deviation, *p < 0.05, n = number of animals.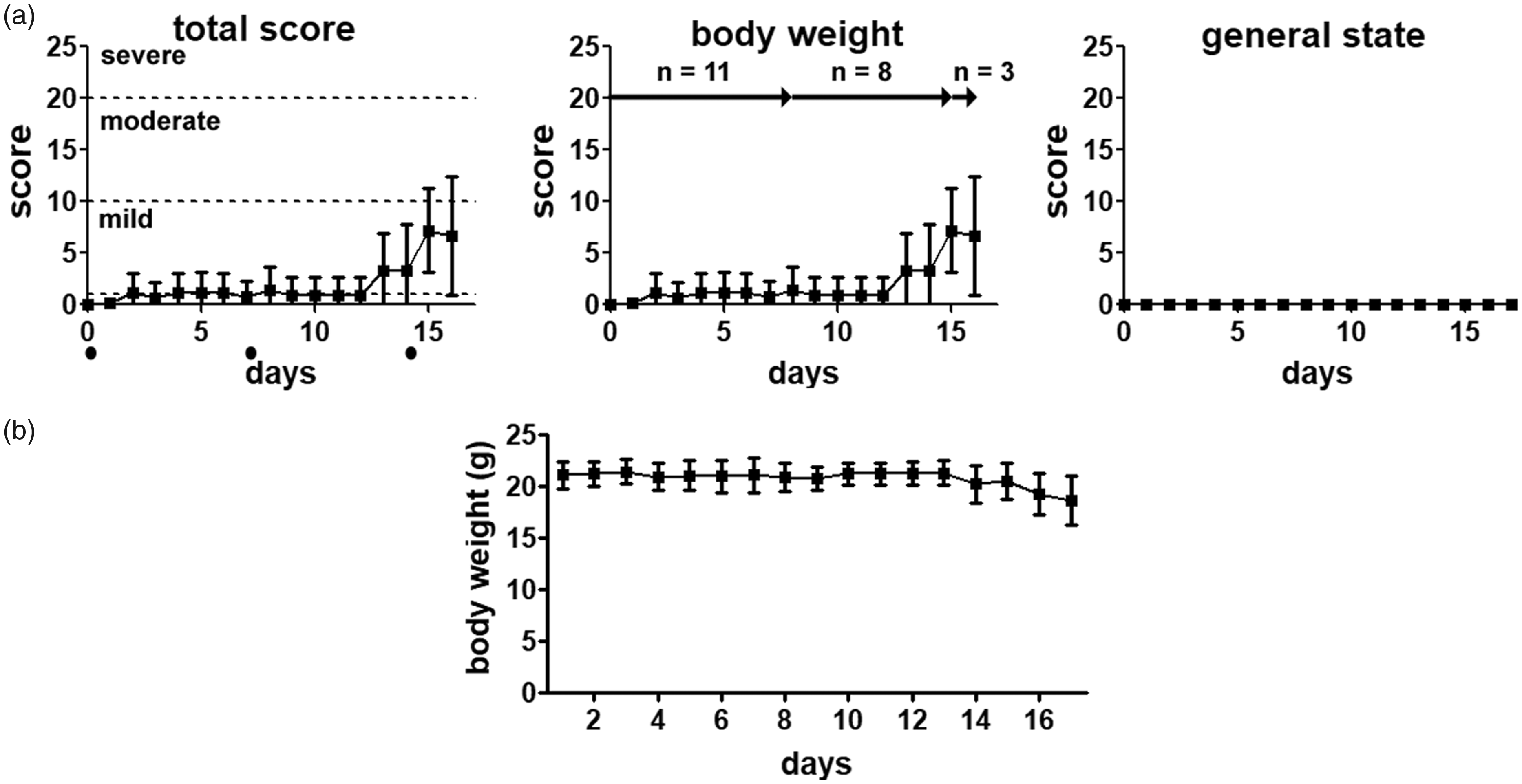
Blood analyses showed no change in white blood cell count, whereas red blood cell count was significantly reduced at day 15 (dF = 2, p = 0.0005). In contrast, the number of platelets was significantly higher (dF = 2, p = 0.029) (Table 2). Serum analyses showed an elevation of ALT in mice sacrificed on day 8 (dF = 2, p = 0.0016), whereas no change could be observed on day 15. Creatinine (dF = 2, p = 0.0015) and creatine kinase (dF = 2, p = 0.0037) were significantly higher on day 15. All other parameters were unaltered (Table 2).
Although score sheets only showed slight deviations from physiological values, all mice died before the end of the experiment (two mice were found dead in their cage at day 16 and three on day 17). A dissection of animals did not show any abnormalities. However, an increase in serum creatinine and creatine kinase in animals sacrificed on day 15 indicated an impaired function of heart and kidneys.
Busulfan treatment
Busulfan slightly decreased body weight of mice with a weight loss of approximately 8% at day 10 resulting in a score of 4.7 ± 4.1 points. The fur appearance led to a short-term increase in the score of the general state between day 3 (5.0 ± 0.0 points) and day 5 (3.1 ± 2.6 points) and the total score indicated a mild strain on the mice during the experiment (Figure 3(a)).
Severity assessment after busulfan treatment. (a) Busulfan treated mice showed the lowest degree of severity in this experiment with a low degree of weight loss and some abnormalities in the fur appearance • timepoint of chemotherapy injection, (b) Body weight changes after repetitive injection of busulfan. Data presented as mean ±standard deviation, *p < 0.05, n = number of animals.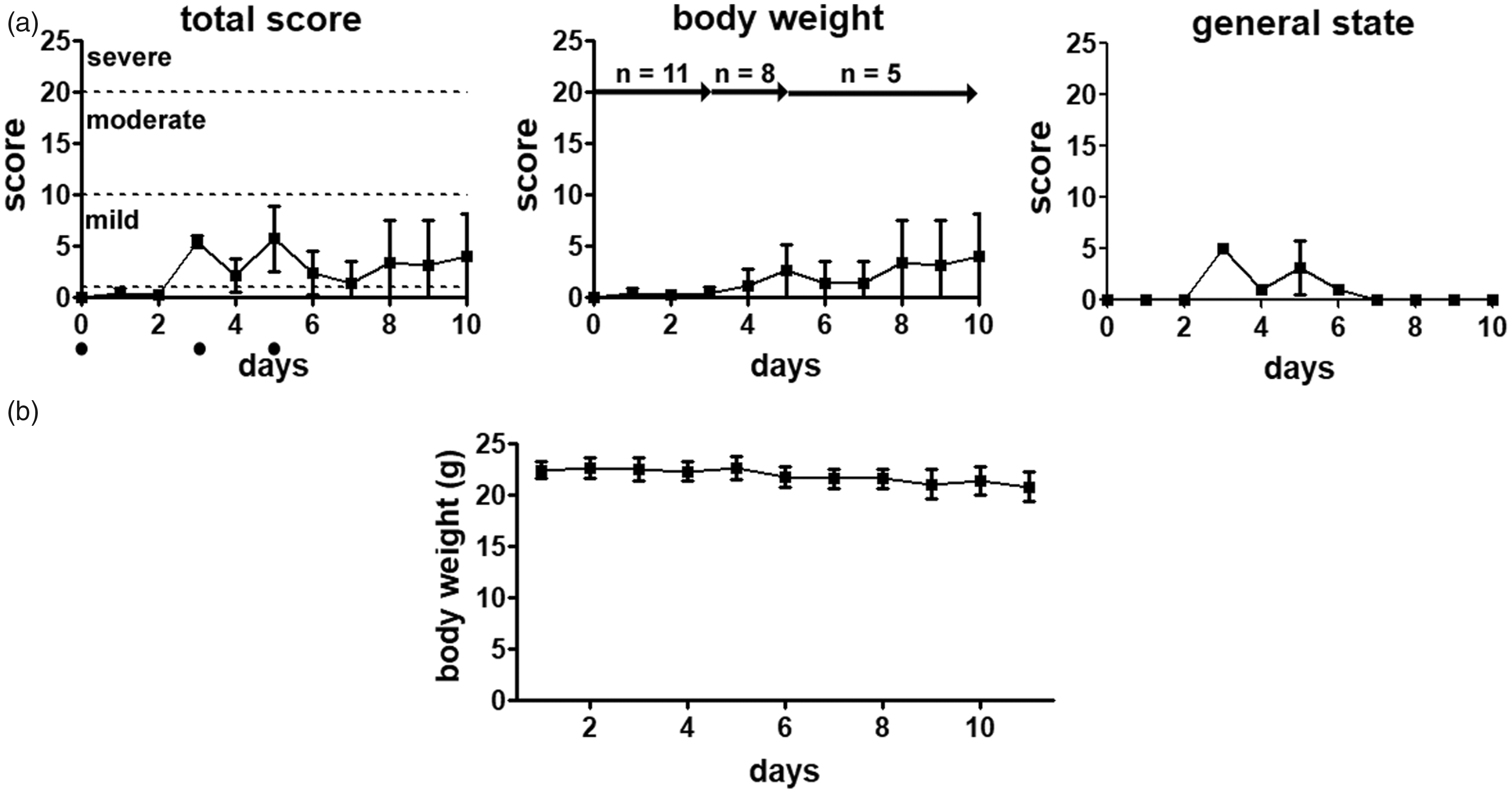
Busulfan significantly reduced white (dF = 3; p = 0.0012) and red blood cell (dF = 3; p = 0.0287), and platelet counts (dF = 3; p = 0.0004) at the last time point (Table 2). Serum analysis did not show alterations except for an increased creatine kinase value in mice sacrificed on day 10 (Table 2).
Influence of imaging on animal welfare assessment
No differences in the actual severity using these measures could be detected between animals subjected to chemotherapy with PET-CT imaging and animals only receiving chemotherapeutics (Figure 4).
Influence of imaging on severity assessment. A comparison of mice that received only chemotherapy and mice that received chemotherapy and additional imaging did not show any difference in welfare assessment. Data presented as mean ±standard deviation, *p < 0.05; 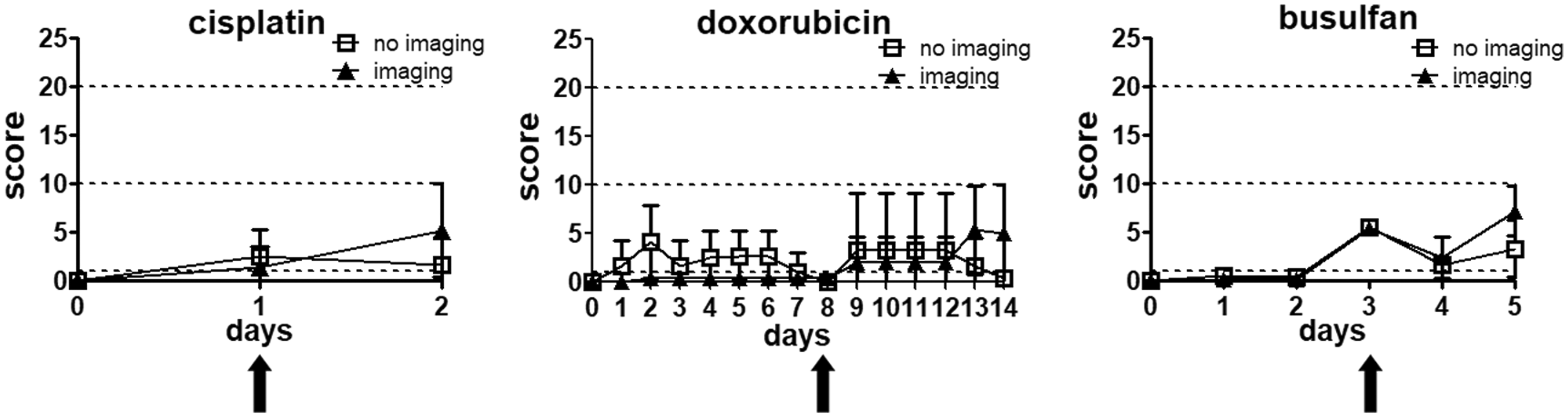
Discussion
The EU directive 2010/63/EU prescribes a prospective and an actual severity classification of procedures applied on animals. 2 In the present study, the prospective evaluation classified the procedures as moderate severity resulting from a longer-lasting impairment of welfare after injection of chemotherapy and repeated anaesthesia during imaging. Although imaging procedures are classified as mild severity, 1 it is assumed that repeated anaesthesia, a necessity for immobilization of animals during imaging, could lead to a higher severity. 9 Nevertheless, a comparison of animals subjected to chemotherapeutics with those subjected to chemotherapeutics and longitudinal imaging revealed no differences in the actual severity during the experiment. This is in line with a recent publication, where the impact of repeated isoflurane anaesthesia on C57BL/6JRj mice induced a short-term mild distress in the immediate post-anaesthetic period. 9 Although all animals experienced only mild strain during the experiment the duration of up to 15 days assigns the procedure to a moderate severity as defined by Annex VIII of the EU directive. 1 Therefore, the prospective severity classification of the present study was in line with the governmental requirements.
In the current study, body weight was the most sensitive parameter for detecting alterations in animals' well-being. Nevertheless, the current scoring systems assigns 1 point for any alteration in body weight below 5%, not considering normal fluctuations of weight due to defaecation or urination. Weight measurements of 25 healthy female BALB/cAnNRj mice over 6 days revealed a variation of 2.4 ± 1.8%. Therefore, the threshold for scoring changes in body weight should be adapted.
The body weight of busulfan-treated animals was slightly reduced, and changes in serum parameters were less pronounced in these animals, whereas a strong reduction in blood cell numbers could be observed that was in line with changes found in the literature. 10
In contrast, cisplatin-treated animals showed the strongest decrease in body weight and multiple changes in blood and serum parameters. A single high dose of cisplatin is reported in the literature to increase serum creatinine and blood urea nitrogen levels 3 days after injection. 11
Although doxorubicin-treated mice showed the lowest alteration of body weight and blood and serum parameters, all mice unexpectedly died before the end of the experiment. The applied dose of 4 mg/kg BW doxorubicin weekly for 3 weeks was chosen as it is known to induce cardiotoxicity in mice, 12 whereas no spontaneous deaths were reported in the literature with comparable dosages and treatment duration.12–14 No alterations of the animals' well-being could be observed one day before the animals died that would motivate a more frequent monitoring. An elevation of creatine kinase in animals euthanized on that day for histological analyses indicated a possible impairment of the heart function. Furthermore, increased serum creatinine levels pointed to the development of a nephrotic syndrome, which was reported after doxorubicin injection. 15 However, it could not be figured out whether the parameters we used to assess well-being did not accurately reflected the degree of the impairment of the animals' health states, whether their health condition deteriorated too rapidly to be captured after the last monitoring, or whether the animals had an unknown previous illness that resulted in the unexpected death.
From the score sheet only body weight scoring could be identified as an objective parameter of the animal's health state. All other parameters rather failed to detect changes, especially in the mice that died spontaneously. This may be due to the fact that the selected experiment-specific parameters mostly represent humane endpoints. Therefore, there is a need for a better adaptation of the score sheet for rodent-specific strain.
In this study, the body-weight reduction could be a result of reduced food intake possibly caused by fatigue, loss of appetite or nausea, common side effects of chemotherapy. Measuring food and water intake could be a simple method to assess alterations in eating behaviour. 16 Another sign for nausea could be ingestion of non-nutritive substances like bedding or cage enrichment material and should be observed. 17 Furthermore, alterations in locomotion can be a result of nausea or fatigue. One method described to assess these changes is the voluntary wheel running test, where mice are housed in cages equipped with running wheels to assess activity parameters.18,19 A reduction of time spent on the wheel 18 or distance travelled 19 could be observed after administration of chemotherapeutics. Although the before mentioned parameters can be assessed easily without disturbance of the animals, a drawback is the fact that animals need to be single housed to detect changes in an individual animal. The impact of single versus group housing of mice on their well-being is still an ongoing discussion.20,21 Furthermore, voluntary wheel running, a model of exercise in mice, could also alter the amount of chemotherapy-related measurable fatigue, as exercise is described to be beneficial for humans suffering from cancer related fatigue.19,22
Another option to assess locomotion is the open field test, where mice are placed in a box of defined size and a camera located above the arena records the animals’ behaviour for a defined time. Outcome measures can give locomotion information of the animal, like travelled distance in a defined time and information about anxiety related behaviour detected by time spend in a certain area. 23 It has been reported that mice showed reduced activity and a reduced time spent in the outer sections after chemotherapy. 23 Although the open field test is an additional procedure, the severity of this procedure is defined by Annex VIII of EU directive 2010/63/EU as ‘below the threshold for regulation’. 1
In conclusion, body weight was the main parameter detecting physiological changes in mice during chemotherapy. Regarding side effects of chemotherapy, more focus should be set on signs of nausea and fatigue as body-weight reduction could be a result of these signs. Useful measures to assess nausea and fatigue could be measuring food and water intake. Furthermore, changes in locomotion can be detected by voluntary wheel running or open field test, with the latter additionally enabling the assessment of anxiety relating behaviour.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was supported in part by the German Research Council (Deutsche Forschungsgemeinschaft DFG FOR 2591 to FK Grant No. 321137804 and RT TO 542/5-1) without any involvement of the funders in the study design, data collection and analysis, decision to publish, or the preparation of the article.
