Abstract
Although the recognition of pain, distress and discomfort has already been described in 1985 by Morton and Griffiths there is still very little known about the establishment of score sheets especially, regarding post-surgical pain and severity assessment for laboratory animals such as rabbits. In this paper we describe the estimation of severity and recovery status of 36 female New Zealand White rabbits (NZW) in a standardized liver resection model using two different adhesive treatments and one control group. Welfare was assessed at 3–4 consecutive days after surgery using a scoring system which included the following criteria: body weight, general state, clinical results, spontaneous behavior and clinical examination. Values could range from 0 to 20 where increasing values indicated increasing severity with a predefined humane endpoint for a score ≥20 points. Documented score points were almost exclusively a result of body weight loss, whereas clinical signs and general health status had no influence on the overall sum of points scored. Behavioral variation was solely observed postoperatively, within the first 24 h, with an average score ≤1. In contrast to the classification of a laparotomy as a moderate procedure in the EU Directive 2010/63 (annex VIII) the assessment herein presented showed a mild burden in all groups according to the scoring system used. The partial hepatectomy itself, as well as the adhesive treatment using either synthetic glue VIVO-107 or fibrin glue, were well tolerated.
Historical background
Partial hepatectomy in rabbits has been practiced since the early 19th century; whereas the first operation, even though performed initially in the 1830s by Cruveilhier and Andral, was only described in 1879 by Tillmans et al. as a proof of liver regeneration.1,2 Surgical procedures in (laboratory) animals are well known and have been established for centuries, while animal welfare as a concept has only recently been implemented in national and international law. In 1959 Russell and Burch postulated the concept of replacement, reduction and refinement (the 3Rs) that has underpinned the ethical framework for animal experimentation ever since. 3 Today the 3R concept is incorporated in national and international law as well as in published guidelines on animal experimentation.4,5 Severity assessment as an aspect of refinement is emphasized in the EU Directive 2010/63, where refinement is described as methods that avoid, alleviate or minimize the potential pain, distress or other adverse effects suffered by the animals involved, or which enhance animal well-being. A surgical procedure involving laparotomy such as the partial hepatectomy discussed in this paper under general anesthesia and appropriate analgesia, associated with post-surgical pain, suffering or impairment of general condition is classified as a moderate intervention (annex VIII of EU Directive 2010/63). A procedure is considered to be moderate if the animals are likely to experience only short-term moderate, or long-lasting mild pain, distress or suffering, or if the animals’ well-being or general condition is impaired. 4
The first guidelines on the recognition of pain, distress and discomfort in laboratory animals using a score sheet system were published by Morton and Griffiths in 1985 who described the relationship between appearance, body weight, clinical and behavioral signs and the degree of pain or distress. It is possible with an overall assessment, if well established, to categorize the severity of a given procedure. 6 .Ideally a score sheet should be modified according to specific species, and optimized for each procedure and animal model. A score sheet should include general as well as tailored parameters for any given protocol. 7
However, there is an ongoing discussion regarding the limitation of a score sheet estimation to classify the severity of an experiment or to predict pain and distress for the laboratory animal, and to clearly identify at what point human intervention is required.
In this study, a standardized liver resection model in rabbits was used to test and compare the sealing with a novel synthetically medical adhesive, VIVO-107, to the clinical gold standard of fibrin glue (Tissucol Duo S), and to a control group treated with 0.9% sodium chloride (NaCl). All the groups were part of a severity assessment with a semi-quantitative score as a sum of changes in body weight, general health status, spontaneous behavior and/or clinical signs. The aim of the study was to assess and to analyze data for severity scoring after liver resection in rabbits. We discussed and compared our results with published data on the recognition of postoperative pain. Furthermore, we highlighted differences in the scoring method as well as limitations, and compared the results of our suggested score sheet system with the classification of comparable procedures as stated in the EU Directive 2010/63.
Animals and statistical analysis
In total 36 female, 12–16-week-old rabbits (New Zealand White; specific pathogen-free according to FELASA recommendations; Charles River Laboratories, Saint-Germain-Nuelles, France) with a body weight range of between 2800 and 4446 g were used in this study. The rabbits were acclimatized for at least one week after shipment. They were housed individually in type 4421X or 4541P cages (Tecniplast, Buguggiate, Italy) under standardized conditions with a 12 h/12 h light/dark cycle at 22 ± 2°C, a relative air humidity range of 30 to 70% and 15 air changes per hour (air movement <0.2 m/s). The animals were bedded on Lignocel hygienic animal bedding, type 3/4-S (Rettenmaier & Söhne GmbH + Co KG, Rosenberg, Germany), and fed with a ssniff diet (K-H; ssniff, Soest, Germany). Water was provided via supply bottles filled with sterile water which was ultraviolet-treated, ozoned and pH-reduced.
The number of animals per group was calculated via a power calculation using G*Power, version 3.1.9.2 (Freeware, Kiel University, Kiel, Germany). Results are expressed as mean ± SD. All statistical calculations were performed using GraphPad Prism version 5.00 (GraphPad Software, San Diego, CA, USA) for Windows. One-way analysis of variance (ANOVA) with post hoc comparisons were performed with Tukey’s test. The results were considered to be statistically significant if P ≤ 0.05.
Ethical statement
The experiments were performed in accordance with the German legislation governing animal studies according to the ‘Guide for the care and use of laboratory animals’ (NIH publication, 8th edition, 2011) and the EU Directive 2010/63/EU on the protection of animals used for scientific purposes (Official Journal of the European Union, 2010). Official permission was granted by the governmental animal care and use office (Landesamt für Natur, Umwelt und Verbraucherschutz (LANUV) Nordrhein-Westfalen, Recklinghausen, Germany).
Experimental procedure
Liver resection
The rabbits were randomized to receive either VIVO-107(Adhesys Medical GmbH, Aachen, Germany), Tissucol Duo S fibrin glue (Baxter, Unterschleißheim, Germany) as the clinical gold standard, or saline as the control. The non-anatomic hepatic resection of the main left lateral lobe was performed according to the method described by Kroez et al. 8 Buprenorphine 0.5 mg/kg (Temgesic; Essex Pharma GmbH, Munich, Germany) was administered subcutaneously as preoperative analgesia. An indwelling cannula was placed into the marginal ear vein for induction and to maintain anesthesia. Anesthesia was induced using a mixture of medetomidine (Domitor 0.1 mg/kg; Zoetis GmbH, Berlin, Germany) and ketamine 10%, 0.2 mg/kg (Medistar Arzneimittelvertrieb GmbH, Ascheberg, Germany).
All preparations were conducted under aseptic conditions (see Figure 1). Animals were placed in a supine position. A ventral midline incision of approximately 20 cm long was made, and bleeding was controlled by electrocautery. Gauze swabs (10 × 10 cm) soaked with lactated Ringer’s solution were used to retract the liver or intestines, and to keep the organs moist. The liver was exposed and standardized hepatic trauma (about 3 cm) was induced by sharp resection with a precise incision of the elevated left lobe of the liver. VIVO-107, Tissucol Duo S or saline (about 2 mL) were immediately applied to the cut edge. The liver was repositioned into the abdomen and the abdomen irrigated with 20 mL of sterile saline solution. The abdominal wall was closed with a continuous suture (Ethicon Vicryl 2-0, #V443). The skin was closed as a separate layer with interrupted suture (Ethicon Prolene 2-0, # EH7038. Ethicon Johnson & Johnson Medical GmbH, Norderstedt, Germany). The animals recovered in a special intensive care unit (Vetario; Brinsea Products Ltd, North Somerset, UK) with warmed air (30–35°C) for at least 20 min. The resected part of the liver was weighed to calculate the percentage of the resected liver. The calculation of the relative weight of the resected liver was based on an average liver weight of 102.8 ± 13.5 g of 12–16-week-old rabbits with a weight range of 2356 to 3740 g (historical data of the breeder Charles River). Buprenorphine 0.1 mg/kg (Temgesic, Essex Pharma GmbH) was administered subcutaneously every 12 h for three days as postoperative analgesia.
9
Surgical technique of partial hepatectomy in rabbits. (a) Preparation of anesthetized rabbit. (b) Midline laparotomy and exposure of liver. (c) Induction of hepatic trauma. (d) Resected liver. (e) Application of adhesive glue or NaCl. (f) Repositioning of organs into the abdomen.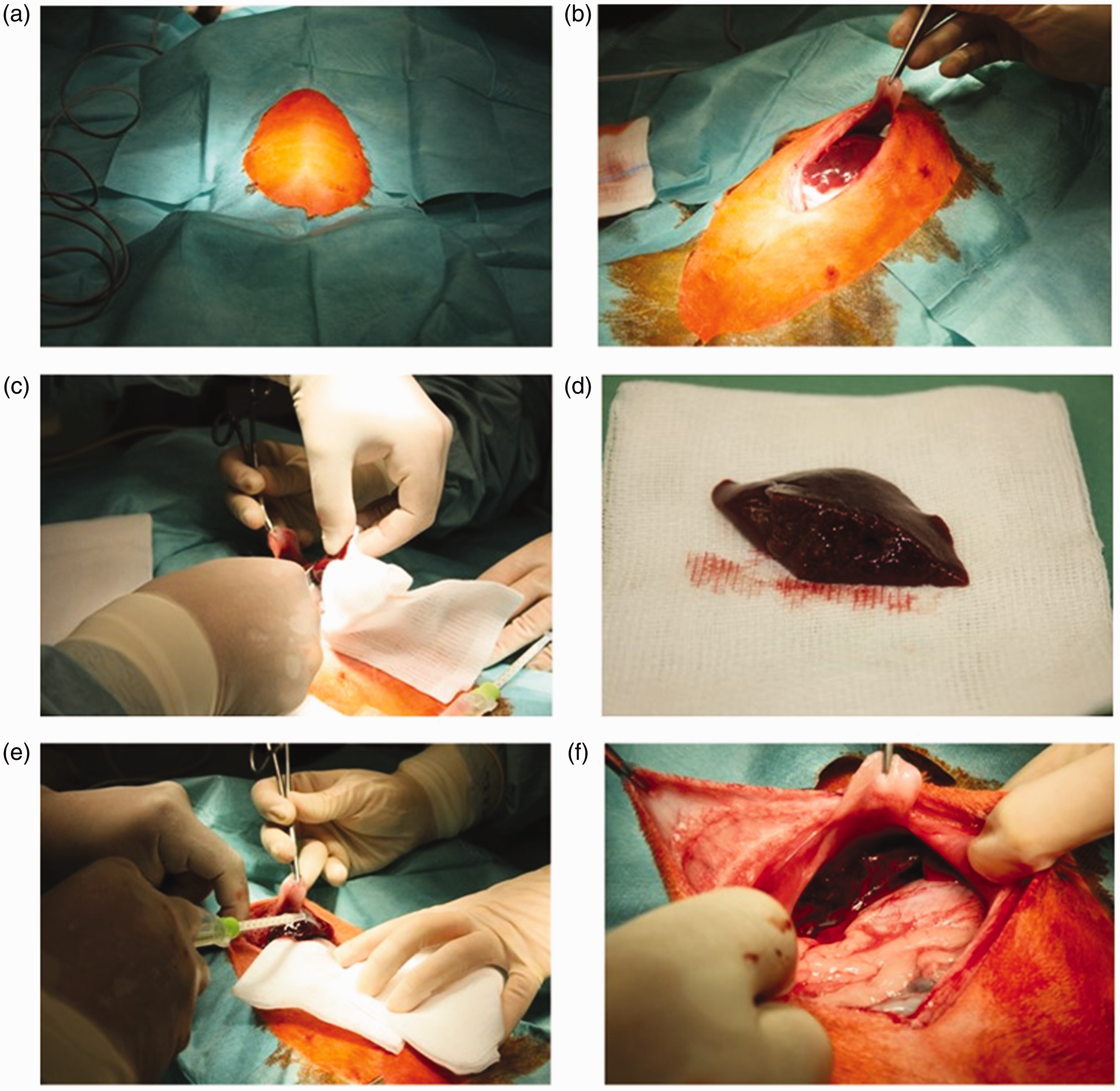
Severity assessment
Full score sheet used for severity assessment after partial hepatectomy in rabbits.

PO: postoperative.
Results
The amount of liver resected was comparable in all groups. The relative amount of resected liver on the day of surgery was 9.12 ± 2.45% in the VIVO-107 group, 10.05 ± 2.34% in the Tissucol Duo S group and 11.62 ± 2.63% in the 0.9% NaCl control group. The survival rate was 100% for seven days in all groups after surgery. In the VIVO-107 group, 2 out of 16 rabbits died during surgery due to technical complications (air emboli via central liver veins due to anatomical variation). These animals were excluded as technical failures.
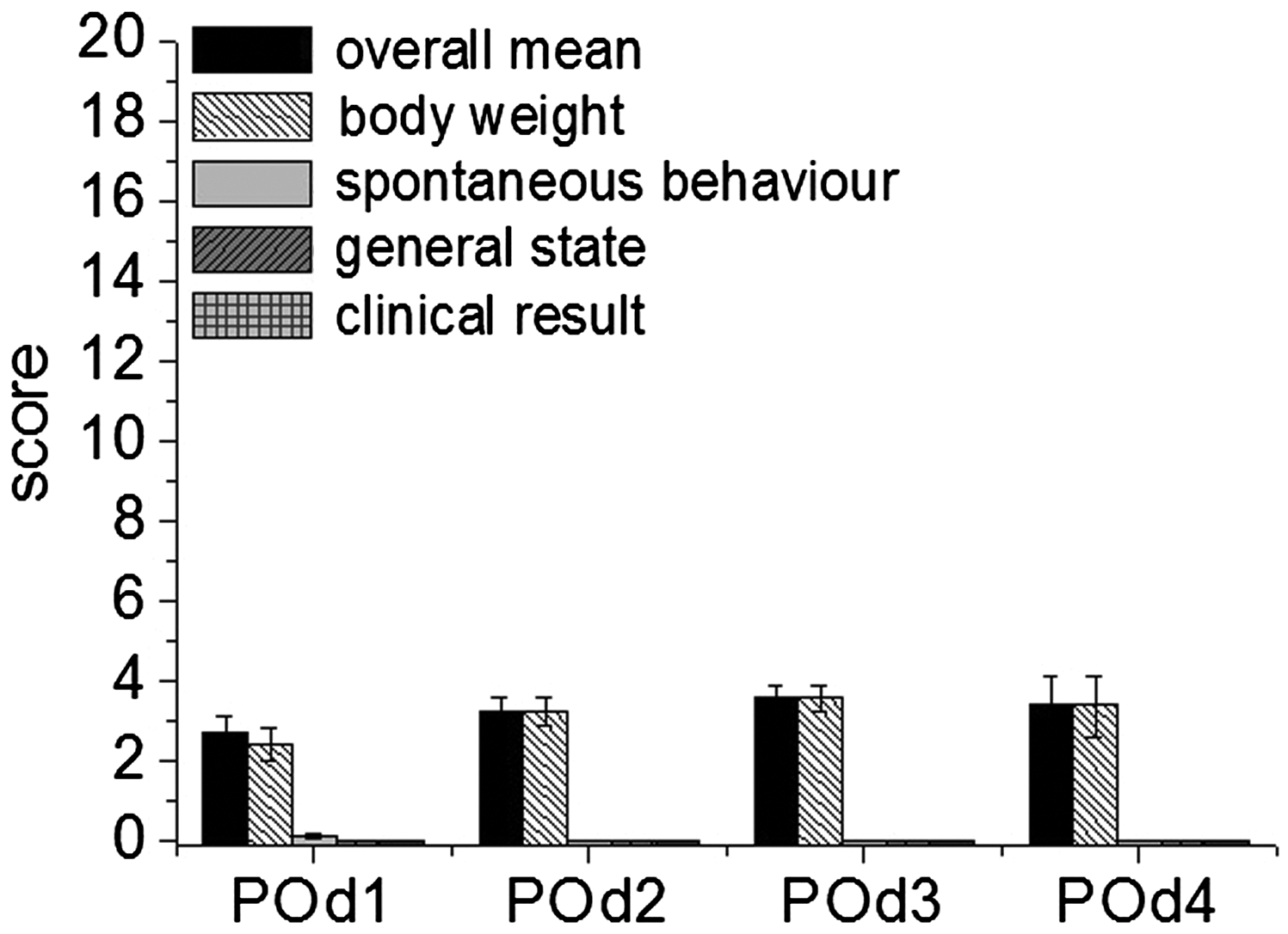
The severity assessment did not reach the predefined endpoint (score ≥ 20) in any of the animals. The overall assessment and sum of score points of the severity scoring showed a burden, defined as mild in our score sheet, in all groups with a maximum of 4 out of a maximum of 20 points (Figure 2). There were no significant differences within the groups (VIVO-107, fibrin glue or NaCl control). Nevertheless, a slight increase in the overall score was observed in that for some animals (n = 20; random number out of each group: n = 10 (VIVO-107); n = 4 (fibrin glue); n = 6 (NaCl control)) the score sheet was, as a direct consequence of scoring, repeated on day 4 for further evaluation of the well-being of the animals. The results are presented in Figure 3 and showed a slight decrease on day 4 after the analgesic treatment was stopped.
Overall score points at three days post partial hepatectomy are given as mean ± SD and showed no significant difference between the adhesive treatment with VIVO-107 (n = 14) (black sqaures), clinical gold standard (n = 10) (open circles) or with the NaCl control (n = 10) (grey triangle) groups (one-way ANOVA, Tukey’s test). Degree of strain (DS) = 1 (minor). POd: postoperative day. Mean scores of overall sum (of all groups) of scoring points with body weight, spontaneous behavior, general health status and clinical signs having a part in the degree of strain (mean ± SEM; [n = 34 (POd1–POd3)]; [n = 20 (POd4)]). POd: postoperative day.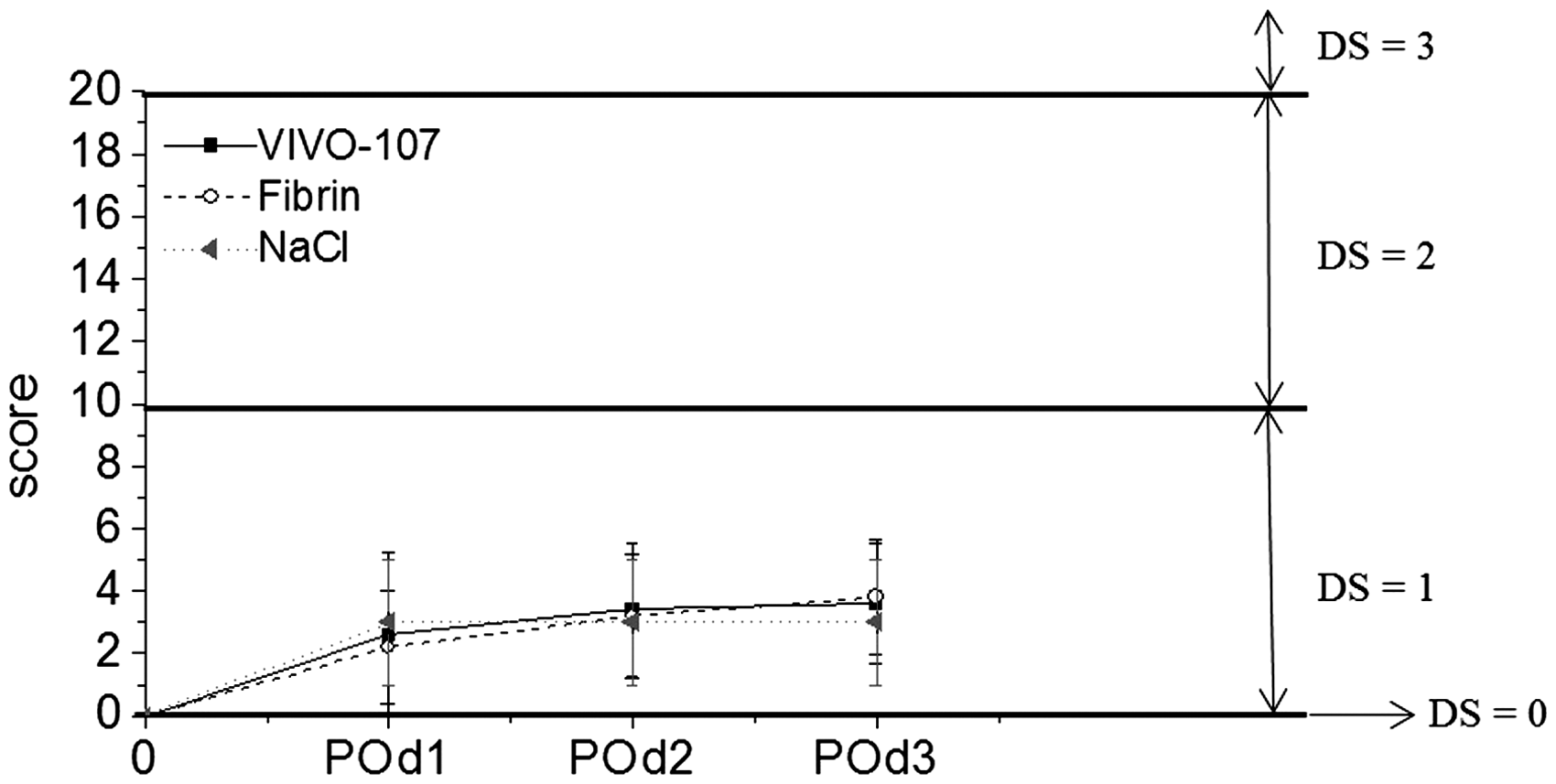
The body weight changes were comparable for the groups treated with VIVO-107 and Tissucol Duo S. The decrease in body weight in the first three days was <10% in the adhesive-treated groups as well as in the NaCl control. Numerically, on average this accounted to 3.1 score points for the adhesive treatment and 3.0 score points for the control group. The body weight was not corrected for the weight of the resected liver. Over seven days the 0.9% NaCl group showed the smallest change of −2% body weight compared with −4.0 to −5.3% weight changes for the VIVO-107 and fibrin glue, respectively. However, these differences were not statistically significant. Mean absolute values for the different groups are shown in Figure 4.
Illustration of the mean body weight (±SD) with equivalent weight loss (±SD) at day 7 post-surgery in the three different groups. POd: postoperative day.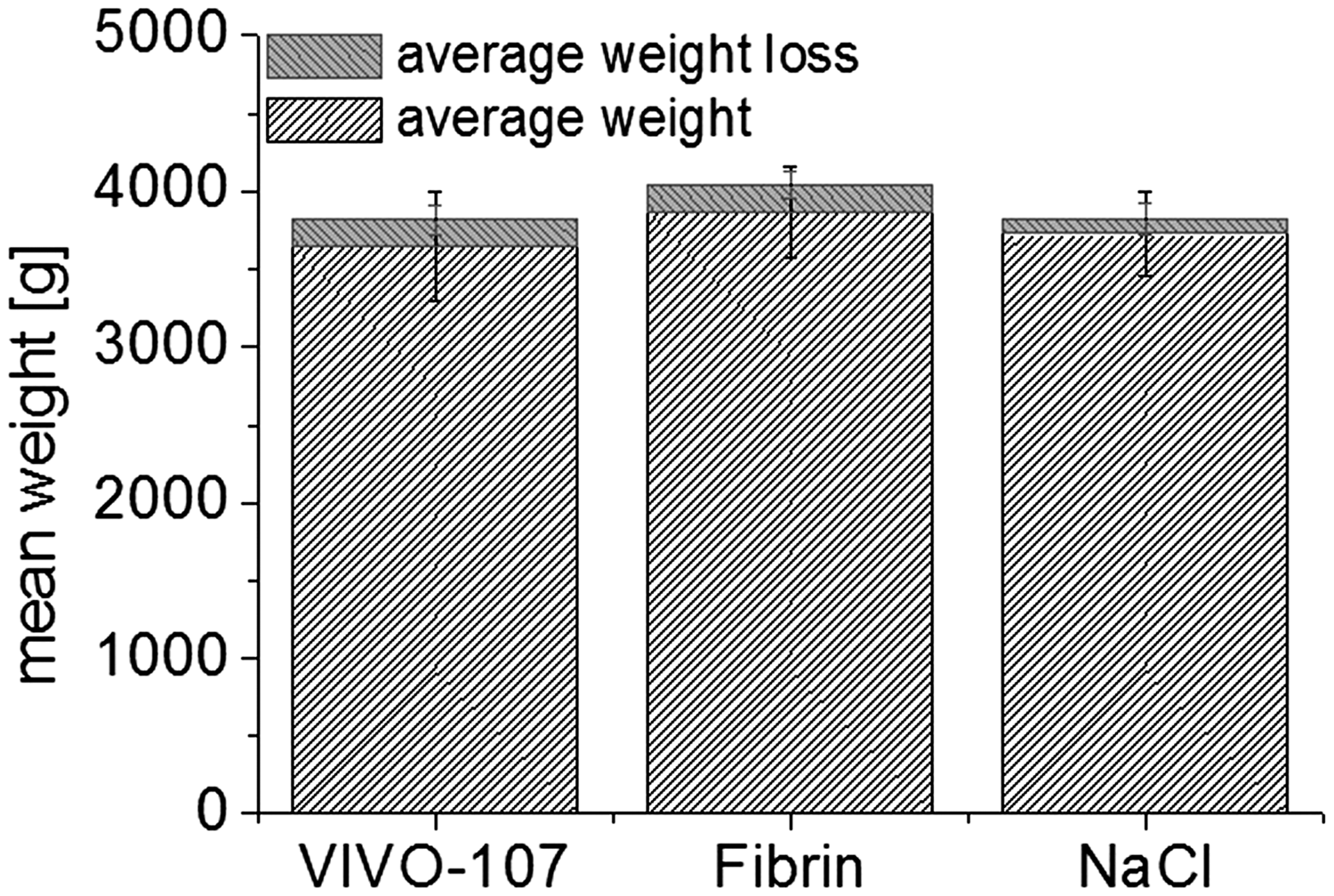
In addition to suffering loss of body weight the spontaneous behavior of the animals was observed to be different, except in the case of the control group. However, these behavioral changes were exclusively visible postoperatively within the first 24 h, and furthermore exclusively scored as slight variation if any to the normal behavior (score ≤ 1) (Figure 5 and Table 1).
Score points of spontaneous behavior in the three different animal groups at three consecutive days post-surgery. Score points are presented as mean values ± SD. POd: postoperative day.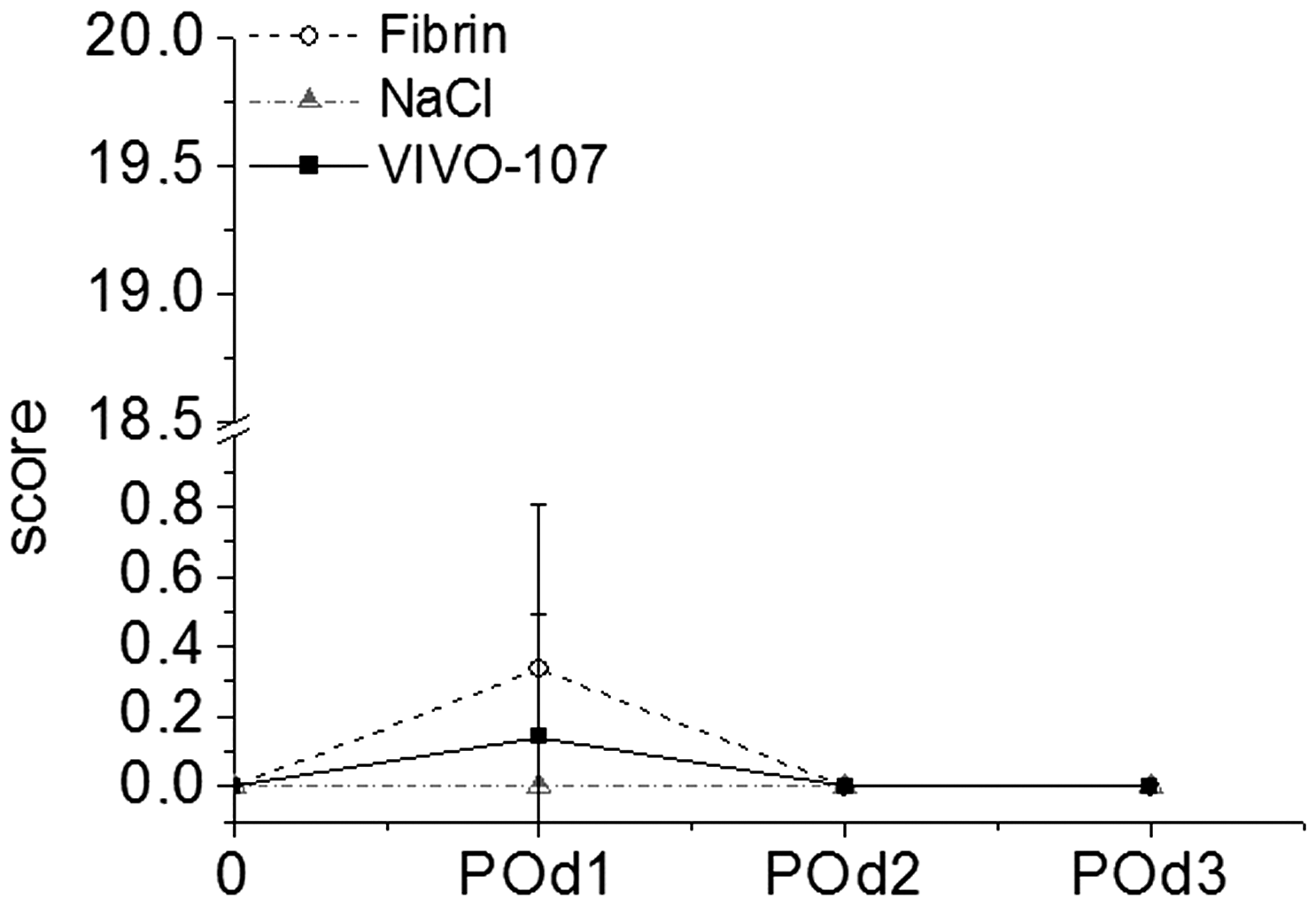
No changes were documented regarding either the general health state of the animals or clinical results such as body temperature, respiration and pulse via pulseoxymetry (Nellcor; Medtronic Corp, Fridley, MN, USA) (Table 1 and Figure 5).
Discussion
The aim of this study was to evaluate the severity of an established rabbit liver resection model with a survival time of seven days and a scoring system with daily observation on three to four days post-surgery. Two different adhesive treatments as well as an NaCl control group were compared. Analgesia was given pre-surgery as well as for three days post-surgery. The severity assessment of the animals was performed using a score system based on body weight, general health status, spontaneous behavior, and clinical examination. The analgesic treatment for this first evaluation was identical for all the animals. However, in future studies subsequent doses could be adjusted according to the results of the score sheet. Increasing values indicated increasing severity and correlated to an impairment of the general condition. The data indicated that the partial hepatectomy itself as well as the products used (synthetic glue VIVO-107 and fibrin glue) were well tolerated. Within the different groups the synthetic glue VIVO-107 showed no significant differences with respect to the score sheets when compared with either the clinical gold standard of fibrin glue or the NaCl control group. The results of the severity assessment with our score sheet indicated a mild burden in all groups without any score point deriving from the section on general health status or clinical estimation on the score sheet. The data showed the liver resection herein described (with analgesia given postoperatively for three days) to be a minor intervention, in contrast to the EU Directive 2010/63 which states that such a procedure, even a laparotomy itself, is moderate. This should not be seen as an overestimation of the severity of the procedure in the EU Directive 2010/63, because it might even be an underestimation due to the non-optimized scoring in this context. This could be evaluated in more detail, e.g. by comparing different score sheet systems as well as by taking more objective data via telemetry, e.g. blood pressure, heart rate, etc.
The rabbits from all groups lost weight over the time course of the study. The weight loss documented during the first three days was not corrected for the resected part of the liver. At day 7 the change in body weight was on average −4.0 to −5.3% in the adhesive-treated groups, and −2% in the NaCl group (for absolute values, see Figure 2). A body weight change of up to −5% is rated as a low burden for animals (and is thus scored with a 1 score point), according to the modified score system of Morton DB et al., which is recognized by the governmental animal care and use committee in the state of North Rhine Westphalia in Germany and is used to evaluate the burden of animals in experimental projects. 10
However, when considering body weight one has to keep in mind that the change in weight may be delayed, reducing its usefulness as a criterion for detecting acute causes of pain/distress in an animal. It might also be misleading, as for example in the case of tumors which can even lead to an increase in body weight. Before drawing conclusions from a decrease in body weight, the water and food intake of the animals could give meaningful information on their well-being.
Another point that could be optimized is the monitoring of clinical results, even though no variation was seen in the present study. In general, a pilot study could help to estimate and identify objective clinical signs that lead to severe pain/distress in an animal. Early clinical signs can thus be used and listed in the scoring procedure to predict later ones, and thereby prevent suffering. 7 Overall blood loss as well as bleeding time during the procedure could be included in the score sheet as a separate section or as a clinical sign; thus for example if a critical loss of blood is reached during surgery the animal can be euthanized immediately instead of being awakened again.
Content from Farnworth et al. 2011. 13

Even though Farnworth et al. focused exclusively on the first 0–6 h post-surgery, their conclusions about behavioral changes are in good agreement with the point scoring of our study where behavioral changes were seen within the first 24 h but disappeared afterwards. In line with the works discussed, the evaluated score sheet system shows the importance and the high value of behavioral assessment related to pain and distress in animals. Score sheets should thus be precise regarding the questions of which behavioral changes need to be followed that really are related to pain, and how they should be. Video observation should ensure that changes are assessed correctly.
In summary, a score sheet for the assessment of the severity of a surgical procedure is most likely to fail in predicting acute failure (e.g. re-bleeding), especially where symptoms occur only at a very late stage and where the animal’s welfare is already compromised. But it is unlikely that each different variable will be scored wrongly, and so there is a certain stability in this type of scoring system.
Conclusion
The recovery status (estimation of severity) of rabbits in a standardized well-established liver resection model with two different adhesive treatments and one control group was assessed over three consecutive days after surgery using a score system which included the criteria: body weight, general health status, spontaneous behavior and clinical examination. In contrast to the classification of a laparotomy as a moderate procedure in the EU Directive 2010/63, our study categorized the procedure as a mild intervention in all groups. However, observations especially during the first 24 h are largely missing in the score sheet used herein and, as already discussed, an expansion of the score system might increase its sensitivity and thus might change the final classification. It is also necessary to correlate the score sheet data in the future with more objective data, e.g. blood pressure, heart rate, cortisol levels, etc. The procedure (partial hepatectomy) itself, as well as the adhesive treatment using either synthetic glue VIVO-107 or fibrin glue, was well tolerated. Score points were almost solely a result of body weight loss, whereas clinical signs and general health status had no influence on the overall sum of score points. Behavioral variation was observed exclusively within the first 24 h post-surgery, which is in line with results from other groups where behavioral changes were most frequently observed close to the surgical procedure. The direct comparison with other studies of postoperative pain expression in rabbits suggests that behavioral indicators of pain have to be evaluated very precisely and with detailed knowledge of the undisturbed species-specific behavior. 14
Within this study we were not able to introduce a generalized scoring system but discussed limitations and considered how to make improvements. The less objective the parameters scored and the less precise these parameters are defined, the more the outcome of a score sheet relies on the skill and experience of each observer and the more it might differ from others.
In summary, score sheets need to be re-evaluated and tailored to each procedure and each species undergoing the procedure. Even with its limitations (e.g. an inability to predict acute failure) it remains a helpful tool in assessing and safeguarding animal well-being, and is thus acknowledged as good scientific practice, and a valuable refinement tool in laboratory animal science.
Footnotes
Acknowledgement
The authors would like to thank Pascal Paschenda for his excellent technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
