Abstract
Summary
In vivo models of hepatic ischaemia/reperfusion injury (IRI) are widely used to study both the mechanisms of hepatic ischaemic injury and to seek means of hepatic protection. Achieving high-quality reproducible data are essential if the results of multiple studies are to be compared and reconciled. This paper presents our findings concerning the effect of intraoperative thermoregulation upon signal to noise ratios of hepatic IRI experiments in mice. Four experiments were conducted, using three different strategies for core temperature maintenance. Animals underwent hepatic IRI and euthanized 24 h postoperatively for measurement of plasma alanine aminotransferase (ALT). Duration of ischaemia was used to adjust the severity of injury. Experiment 1 utilized a constant output heating system and resulted in rising postoperative ALTs following increasing durations of hepatic ischaemia. Experiment 2, using the same constant output heating system confirmed a difference between ischaemic and sham-operated animals. Experiment 3 used a thermostatically controlled heating system and resulted in highly variable results with a small, but statistically significant correlation between ALT levels and rectal temperature readings. Experiment 4 used a homeothermic warming system and demonstrated highly reproducible data from increasing durations of ischaemia. High-quality data from hepatic ischaemia/reperfusion models are dependent upon careful control of intraoperative temperature. The use of homeothermic warming systems is recommended and conversely, the use of thermostatically controlled warming mats is to be avoided in these models.
Human liver transplantation surgery necessitates the interruption of hepatic blood supply while the organ is retrieved from the donor, transported to the transplant centre and implanted in the recipient. Similarly, liver resection surgery often involves occlusion of the hepatic blood supply in order to limit blood loss from the cut surface of the liver. In both cases, the resulting ischaemia/reperfusion insult leads to impaired hepatic function postoperatively. Ischaemia/reperfusion injury (IRI) most severely affects the livers from older donors and those with fatty infiltration (steatosis) arising from obesity or excessive alcohol intake. In the transplant setting, where the risk of IRI is judged excessive, such marginal livers may be discarded, placing further demands on an already overstretched donor pool. Conversely, overcoming hepatic IRI might alleviate postoperative liver dysfunction and expand the donor pool by recruiting back currently unusable organs (Patel et al. 2004, Devey et al. 2007).
Various strategies intended to overcome hepatic IRI are under investigation. Among the most promising is ischaemic preconditioning (IP). This is a physical manoeuvre, which involves a brief interruption of the hepatic blood supply prior to the index ischaemic event. Although the value of this technique has been proven beyond doubt, its molecular and cellular underpinnings remain to be elucidated (Clavien et al. 2003). Greater understanding of both the mechanisms of IRI and IP would provide an opportunity to develop pharmacological preconditioning drugs (Patel et al. 2004).
Manipulations of putative molecular mediators of IRI and IP can be achieved using pharmacological interventions or targeted gene deletions to assess the roles of various genes and pathways. We chose to establish our model in mice to exploit the availability of a wide range of genetically-altered strains.
An appropriate model would simulate with high fidelity the insults arising in human surgery, with minimum severity of insult in the minimum number of animals.
A wide range of model designs have been proposed in the literature to date. The human Pringle manoeuvre involves occlusion of the blood supply to the whole liver via the portal triad at the free edge of the lesser omentum (Pringle 1908). Necessarily, this results in upstream mesenteric venous congestion and ischaemia. Owing to increased bacterial translocation during mesenteric congestion in mice, it is argued that the total hepatic ischaemia models do not provide easily interpretable data unless combined with portosystemic shunting manoeuvres.
An alternative is to exploit the discrete lobar anatomy of rodent livers by selective occlusion of the vessels feeding only part of the liver, leaving the remaining lobes to act as portosystemic shunts. This causes less intraoperative haemodynamic disturbance and is the model we have chosen to use here.
Variables previously shown to affect performance in response to hepatic IRI are animal sex (Harada et al. 2004) strain (Burne et al. 2000, Shireman & Quinones 2005, Dodd et al. 2006) and intraoperative temperature regulation (Biberthaler et al. 2001, Kato et al. 2002, Khandoga et al. 2003). These factors have been tested in heterogeous models of hepatic IRI, and as a result, experiments cannot be directly compared; their results are difficult to apply directly to the development of new experimental models.
The data herein represent our experience gained from relocating our laboratory work between two university locations in the UK. The redesign of heating mats arising from European directives requiring thermostatic controls led to an unexpected deterioration in the signal to noise ratio of a previously reproducible model. Careful analysis of the potential causes and solutions for this problem led us to determine the importance of the mode of intraoperative thermoregulation.
Materials and Methods
Animals
All experiments followed the guidelines of the Animals (Scientific Procedures) Act 1986 and were subject to ethical review at both UK universities involved, and appropriate UK Home Office Licensing. Male and female eight-week-old specific pathogen free (SPF) BalbcO1aHsd and C57BL/6J01aHsd mice were purchased from Harlan, UK (Shaw's Farm, Blackthorn, Bicester, Oxon, UK). Mice were acclimatized for 4–5 days in groups of four animals per cage, and fed SDS-RM1 ad libitum.
Surgical technique
Following administration of buprenorphine analgesia (0.06 mg/kg), animals received isoflurane anaesthesia via face-mask inhalation. Body temperature was maintained as described below. Full sterile technique was employed, including sterile instruments, drapes and gloves. Following skin cleaning with alcohol, a midline laparotomy was performed. Using a cotton bud and dissecting forceps, the vascular pedicle supplying the left lobe of the liver was isolated. Occlusion of the vessels was achieved with a micro-serrefine clip (Fine Science Tools, USA). Sham-operated animals underwent the full procedure as described above, but without vessel occlusion. Duration of ischaemia was altered to dictate severity of injury (see below). While ischaemia was ongoing, a sterile absorbent paper pack was placed in the abdominal cavity kept wet with saline. Following removal of the clamp, the laparotomy was closed, the animal recovered and saline 0.5 mL was administered subcutaneously. Mice were euthanized 24 h postoperatively by exsanguination under isoflurane anaesthesia. Blood was taken for plasma alanine aminotransferase (ALT) determination and the liver collected for histological evaluation of injury.
Temperature maintenance
Experiments 1 and 2 were done in location A. Animals were placed on Cozee Cumfort heat electric pad model – hp 220–240 v ~3441 w 50 hz (Vet tech, Cheshire, UK) consisting of a simple two level heat control such that the mat warmed to a constant heat output. Experiments 3 and 4 were performed in location B. The animals in experiment 3 were placed on a thermostatically controlled heating blanket (Morphy Richards SR090, Vet tech) containing a simple on/off thermostat. In experiment 4, body temperature was maintained with a homeothermic warming system (Harvard apparatus, Edenbridge, Kent, UK) consisting of a warming blanket (set on a curved operating table to increase surface area in contact with the animal) connected to a control unit which continuously varied heat output in order to maintain core body temperature of 36°C (± 0.3°C).
Alanine aminotransferase measurement
Terminal cardiac puncture blood samples were measured by the hospital biochemistry laboratory in accordance with local neonate protocols adapted to small sample volumes. In location A, ALT was measured on the Olympus AU2700 (Olympus Optical Co, UK), while in location B, an Olympus AU400 was used. In both locations, methodology was as recommended by the International Federation of Clinical Chemistry (IFCC).
Statistical analysis
ALT levels were analysed by an analysis of variance using MINITAB (Minitab Inc, USA), with residual plots being used to check the assumptions of equal variances in each group and a normal distribution of the residuals. Based on experiment 1, involving two C57BL/6 mice at each ischaemia time of 20, 30, 40, 50, 60 and 70 min, we determined that plasma ALT levels had an approximately log normal distribution. Accordingly, all subsequent calculations of plasma ALT levels were done on log-transformed data.
Experimental design
In all experiments, the order in which the various treatments were applied was formally randomized. Experiments were either single factor or factorial designs. Experiments 1 and 2 were performed in location A with the aim of simple calibration of our model. Experiments 3 and 4 were performed in location B. The aims of the four experiments reported here were as follows:
Experiment 1 This was a small study using C57BL/6 mice, the aim of which was to gather preliminary information on plasma ALT levels following partial liver ischaemia for 20, 30, 40, 50, 60 or 70 min, using two mice at each time point. This design was employed in order that the threshold above which the injury would occur could be established while minimizing the number of animals used. In this design, it was intended that effect size could be calculated by comparison of all injured animals against all non-injured animals.
Experiment 2 This was a small-scale study to compare ALT levels in 10 sham-operated and 11 mice with partial ischaemia for 60 min.
Experiment 3 Aimed to re-establish our IRI model in a different centre, and to study the effects of ischaemia time, sex/strain of the mice on ALT levels. However, this experiment gave inconsistent results. This technical issue appears to have been caused by erratic intraoperative temperature control of the mice (see Results). It is this issue and its resolution, which form the focus of this report.
Experiment 4 In light of the unexpectedly inconsistent results of experiment 3, the protocol was simplified to focus only upon sex differences. The aim of this experiment was to confirm that the repeatable and consistent results could be obtained once core temperatures were correctly controlled by looking at the effect of time and sex on ALT levels.
Results
Experiment 1
The mean log ALT levels at 20, 30 and 40 min were approximately the same, averaging 2.12 log units (132 units), and for the three more severe injury durations (50, 60 and 70 min ischaemia) the means were also similar, averaging 3.17 log units (1479 units), with a pooled standard deviation (SD) of 0.34 log units. Using these data, power calculations suggest that a reduction in log ALT levels from 3.17 to 2.67 (i.e. 0.5 log units) would require sample sizes of about 11 mice per group, assuming a 90% power, a 5% significance level and a two-sided Student's t-test.
Experiment 2
One mouse died in each group from which causes were undetermined. The mean plasma ALT levels in the sham-operated group was 2.17 log units (149 units) and in the ischaemic mice it was 3.57 log units (3758 units) with a pooled SD of 0.36 log units. These differences were statistically highly significant (P < 0.01). Thus in the sham-treated animals the ALT levels were comparable with mice with partial ischaemia of 20–40 min. The ALT levels in the 60 min ischaemia mice were slightly higher than the average of the 50–70 min ischaemic mice in experiment 1, and the SD were very similar in the two experiments.
Experiment 3
The aim of this experiment was to determine whether there were strain (BALB/c versus C57BL/6) and sex differences in response to six different ischaemia times from 20–70 min. However, entirely unexpected and inconsistent results were obtained. The analysis of variance showed that ALT levels were not significantly related to the degree of ischaemic insult (Figure 1a) and the pooled within-group SD of log ALT levels was substantially higher at 0.68 than in the previous two experiments. This contradicted the results from the previous studies, which had shown increasing durations of hepatic ischaemia to result in increasingly high ALT rises. Of concern was the highly variable end-procedure rectal temperature. Analysis of ALT release compared with end procedure core body temperature showed that no ALT release occurred when it was lower than 33.5°C (Figure 1b), and there was a low but statistically significant correlation of 0.32 (P = 0.03) between log ALT levels and end-procedure rectal temperature. In later experiments, continuous monitoring of body temperature showed that heating mats warmed progressively until they were hyperthermic, then switched off to become excessively cool, which triggered the thermostat to recommence heating the pad, thereby restarting a similar cycle of heating and cooling. Interaction plots showed that data were most reliable using a core temperature of 36°C (above which temperature it was difficult to maintain core temperature due to heat loss). Therefore for subsequent experiments, we used a homeothermic warming system to maintain core temperature at a set value of 36°C.
Using a thermostatically controlled warming mat: (a) Log alanine aminotransferase (ALT) levels were unrelated to duration of ischaemic injury. (b) No significant ALT rise occurred when end procedure rectal temperature was 33.5°C or lower. There was a weak correlation (0.32 [P = 0.03]) between end procedure rectal temperature and log ALT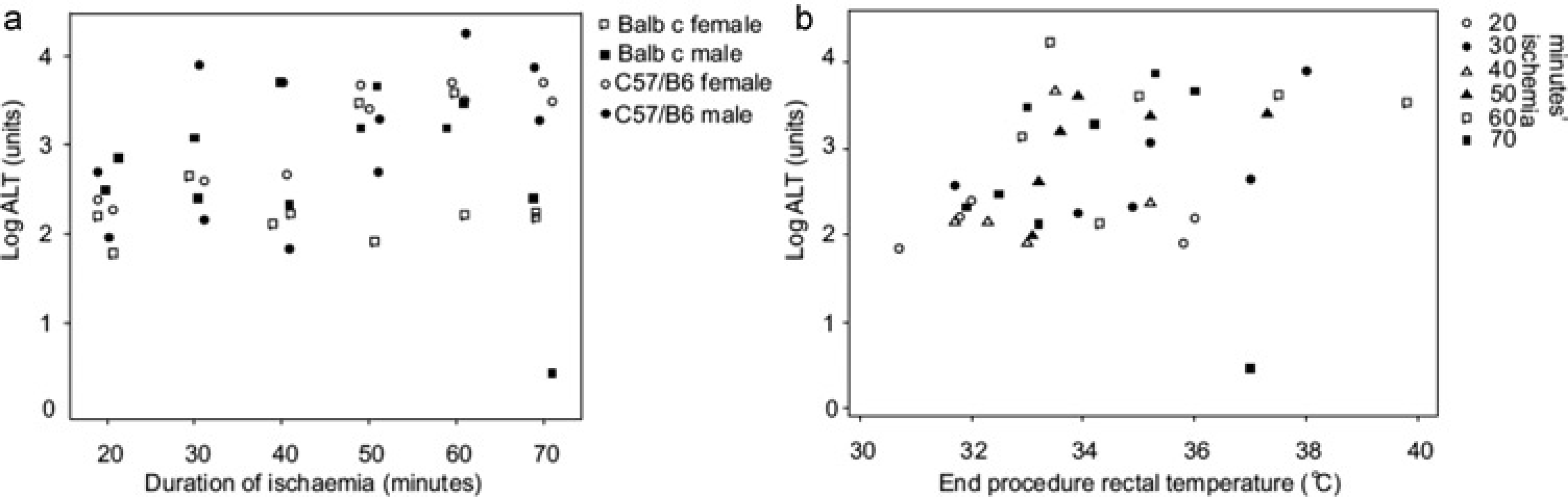
Experiment 4
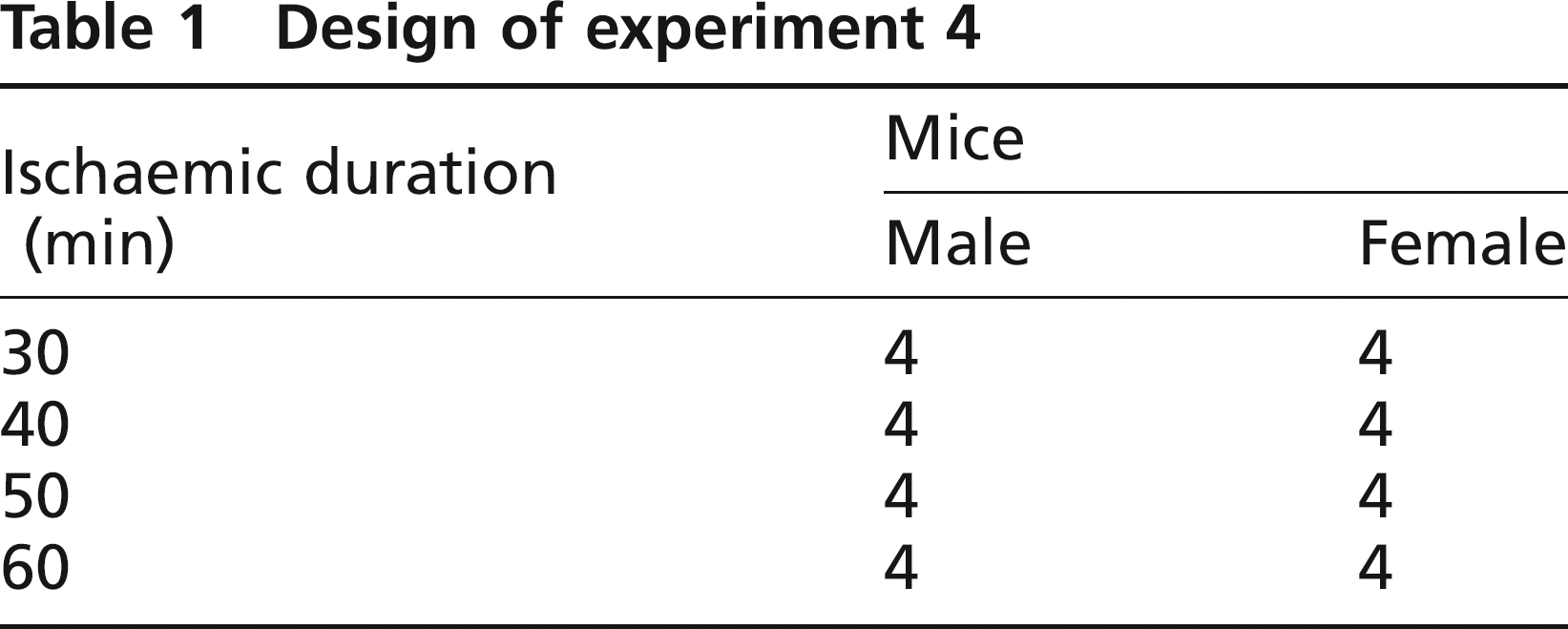
A simplified version of experiment 3, addressing only sex and not strain differences between mice, was repeated using a homeothermic warming system which used a rectal thermometer to control the heat output of a dedicated warming unit. Four C57BL/6 mice of each sex were randomized to 30, 40, 50 and 60 min groups (Table 1). Using homeothermic warming, procedural core temperature was maintained stable at 36°C, and again, ALT was related to duration of ischaemic injury (Figure 2).
Using a homeothermic warming system, log alanine aminotransferase (ALT) was related to duration of ischaemia. Injuries of 30 min resulted in mean log ALT of 2.17 while longer injuries of 50 and 60 min resulted in mean log ALT of 3.61. The intermediate injury of 40 min duration led to more variable log ALT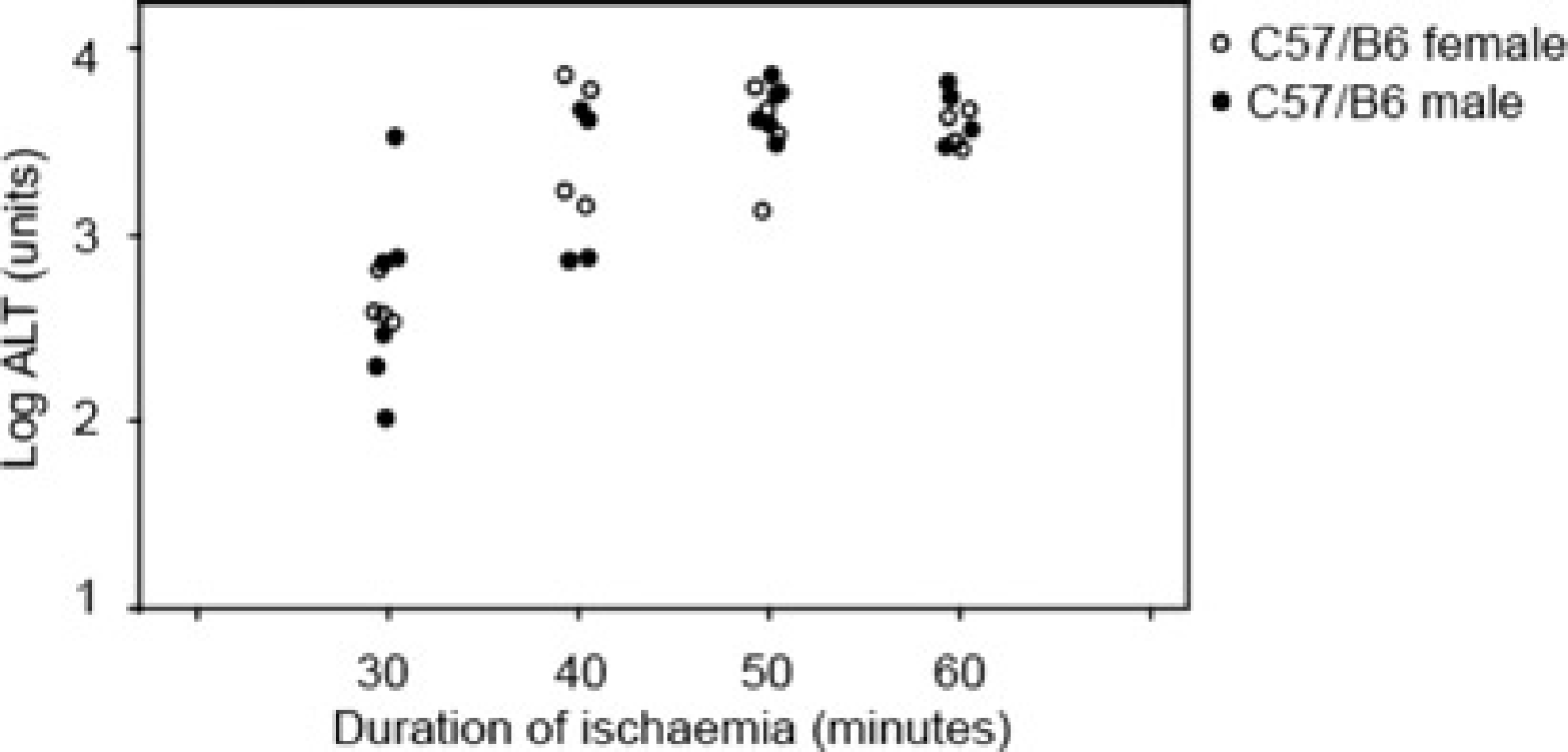
Design of experiment 4
Statistical analysis showed no evidence of a difference between males and females (P = 0.92), but highly significant differences between time points (P < 0.001). The mean ALT levels were:
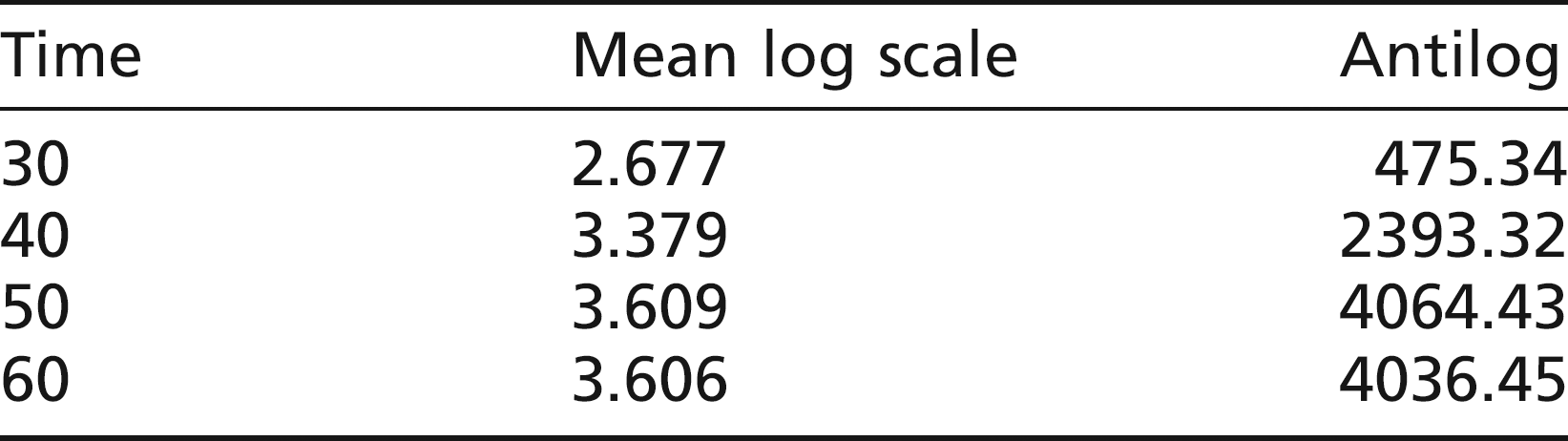
The pooled SD was 0.34, which is comparable with experiments 1 and 2.
Discussion
Increasing interest in hepatic IRI and preconditioning has prompted greater use of animal models for the analysis of cellular and molecular mechanisms of injury and protection. An important generic experimental design involves subjecting wild-type and genetically-altered animals to standardized ischaemia/reperfusion insults and comparing their degree of injury, thereby inferring whether the affected gene product confers susceptibility to or protection from injury (Camargo et al. 1997, Harada et al. 2004, Hasegawa et al. 2005, Ke et al. 2005).
Ethical considerations encapsulated in the 3Rs (Reduction, Refinement and Replacement) require us to maximize data output from well-designed animal experiments which use the minimum number of animals subjected to the mildest possible insults.
Unfortunately, data from diverse experiments are difficult to compare and reconcile due to differences in the model design used. We have shown that significant hepatic IRI occurs following a 50 min inflow occlusion time, yet many papers using such models employ much lengthier injuries, often of 90 min. Our own experience demonstrates that following such extreme injury, detection of susceptible phenotypes would be almost impossible since injury to the wild type is so severe already. Protected phenotypes would need to be extremely potent to protect the animal from such a massive injury.
Furthermore, where phenotypic differences are detected, we suggest that the effect sizes are rendered smaller than necessary thus reducing statistical power and increasing sample sizes. It is likely, therefore, that use of long ischaemic injuries both increases the severity of the experimental procedure, and the number of animals needed to achieve statistical significance.
Many papers use mortality to distinguish protected from susceptible phenotypes. Such experiments generally employ total hepatic ischaemia models, using intraoperative portosystemic shunting (Camargo et al. 1997), or partial hepatic inflow occlusion with end-procedure excision of the non-affected lobes such that the animal is relying upon the injured lobes for its survival (Harada et al. 2004). Not only does this result in considerable suffering to the animals, it also limits data production from the experiment, since it precludes other tissue analysis from the animal.
Although temperature control has long been known to affect performance in response to ischaemia/reperfusion insults, its practical affect upon model performance has not been previously reported. We have shown that temperature control is critical in optimizing signal to noise ratio in IRI experiments. Total systemic cooling leading to reduced enzymatic activity and oxygen consumption is the principle exploited in human surgical techniques such as cardiopulmonary bypass and the cooling of organs in transit prior to transportation. The extremely high body surface area to weight ratio of mice (13 times that of an adult human, US Food and Drug Administration) renders them particularly prone to heat loss during surgery, which we have shown in itself to be capable of abrogating injury.
Older generation heating mats maintained constant heat output and reached stable temperatures after equilibration between energy input and heat loss. Our initial experiments using such older pads produced consistent data in IRI experiments. New European regulations demanding the introduction of thermostatic controls into heating mats appear to have resulted in warm up–cool down cycling resulting in unpredictable temperature maintenance in some of our experiments. The use of a homeothermic warming system has proven capable of generating reproducible data with excellent signal to noise characteristics.
Footnotes
Acknowledgements
Luke Devey is funded by an MRC/Royal College of Surgeons of Edinburgh Clinical Research Training Fellowship. We would like to thank the staff of the Biomedical Services Unit of the University of Birmingham for their help in developing this model.
