Abstract
The most important acute neurological diseases seen at neurosurgery departments are traumatic brain injuries (TBI) and subarachnoid hemorrhages (SAH). In both diseases the pathophysiological sequela are complex and have not been fully understood up to now, and rodent models using rats and mice are most suitable for the investigation of the pathophysiological details. In both models, surgery is performed under anesthesia, followed by assessment of their functional outcome and behavioral testing before brain tissue analysis after euthanasia. Postoperative analgesia is mandatory, and supplementary care is highly recommended for refinement purposes. Pain and stress assessment is mainly based on clinical and behavioral signs, and further research is needed to improve the evaluation of severity in these models.
Besides spinal injury, the most important acute neurological diseases that neurosurgery departments have to deal with are traumatic brain injuries (TBI) and hemorrhaging into the subarachnoid space from acute ruptures of cerebral aneurysms (aneurysm-induced subarachnoid hemorrhages, SAH). In both diseases the pathophysiological sequela are complex and have not been fully understood up to now.1–3 For targeted therapy we have to remedy this lack of knowledge by thoroughly investigating the pathophysiological cascades using well characterized animal models that mimick the human diseases in total or in well-defined separate components. The most commonly used species for TBI and SAH research are small rodents such as rats and mice, 4 with rats being used more often in SAH studies than mice (evaluation up to 2007 5 ). We will therefore focus our review on the severity assessment and scoring for TBI and SAH models in rats and mice.
Animal models of TBI and SAH
Both diseases are characterized by an initial impact to the cerebral tissue due to a physical insult (TBI) or a massive bleeding from a ruptured artery (SAH), and the development of secondary damage which evolves over hours and days or even weeks after the initial insult, leading to the growth of a lesion. Depending on the area of the damaged tissue in the brain, specific functional impairment occurs. To evaluate the degree of cognitive, sensory or motor deficits, a battery of functional tests are available which have been established in small rodents. By using the modified neurological severity score, the grade of impairment can be quantitatively assessed. Both basic reflexes (pinna reflex and corneal reflex) and somatomotor functions (paw flexion, head support, spontaneous and initiated locomotion), as well as postural responses and startle responses are usually tested in mice and rats.6,7 In addition to these simple tests, more specific assessments are possible, though these are highly demanding of time and experience (a review and descriptions of these tests are beyond the scope of this paper but are provided in detail elsewhere8–13). As a final outcome measure, the brains are usually analyzed by histomorphological and molecular biological methods in detail after sacrifice of the animals.
Induction of TBI
TBI is the leading cause of mortality and disability in the younger population (up to the age of 45) worldwide. 14 Within the pathophysiological course of TBI, we can differentiate between the primary damage, which rapidly evolves as a direct result of the physical impact on brain tissue, and secondary damage, which enlarges the initial injury to the tissue. 15
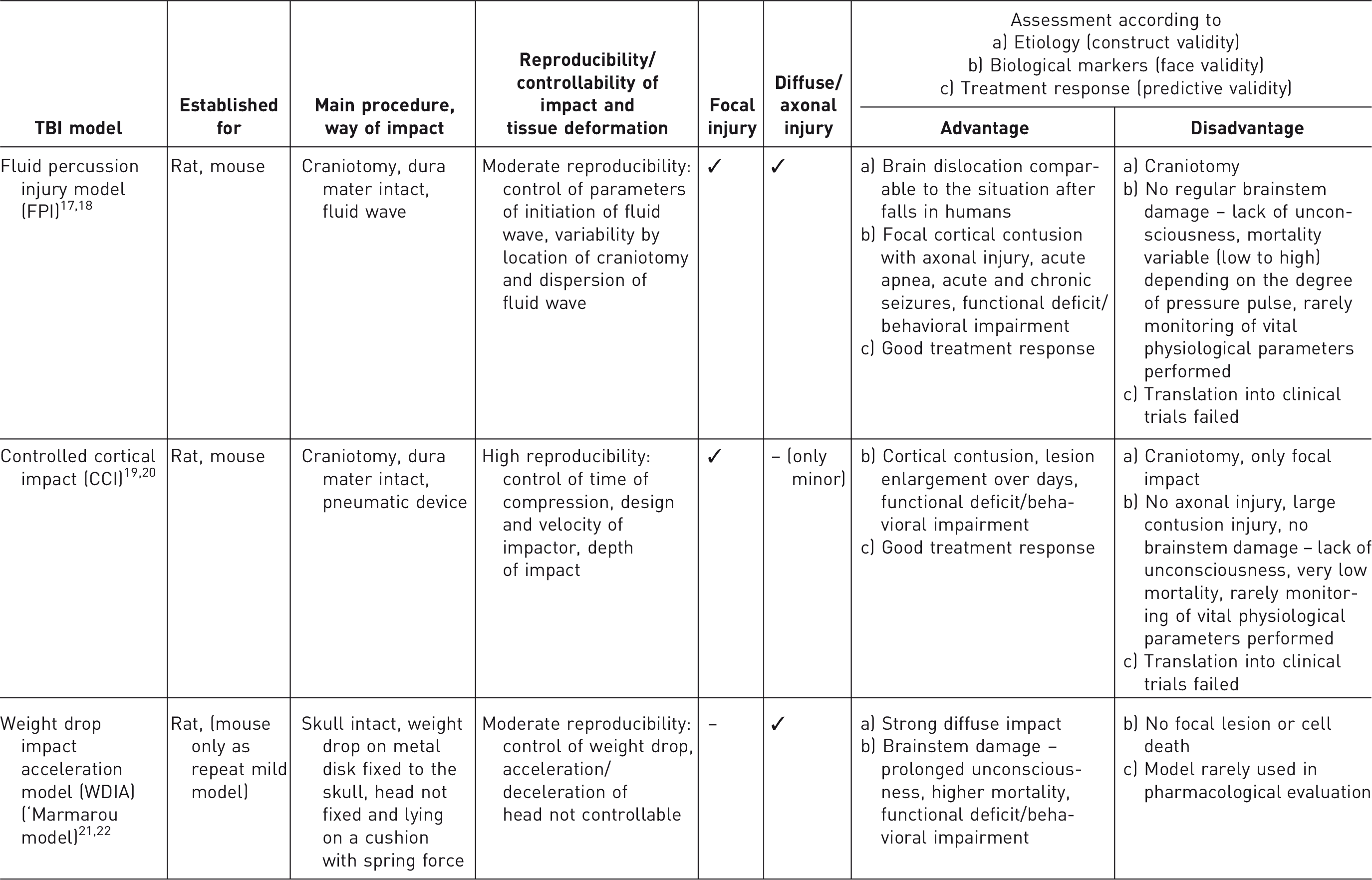
Most commonly used traumatic brain injury (TBI) models in rats and mice.
Induction of SAH
Most commonly used subarachnoid hemorrhage (SAH) models in rats and mice.
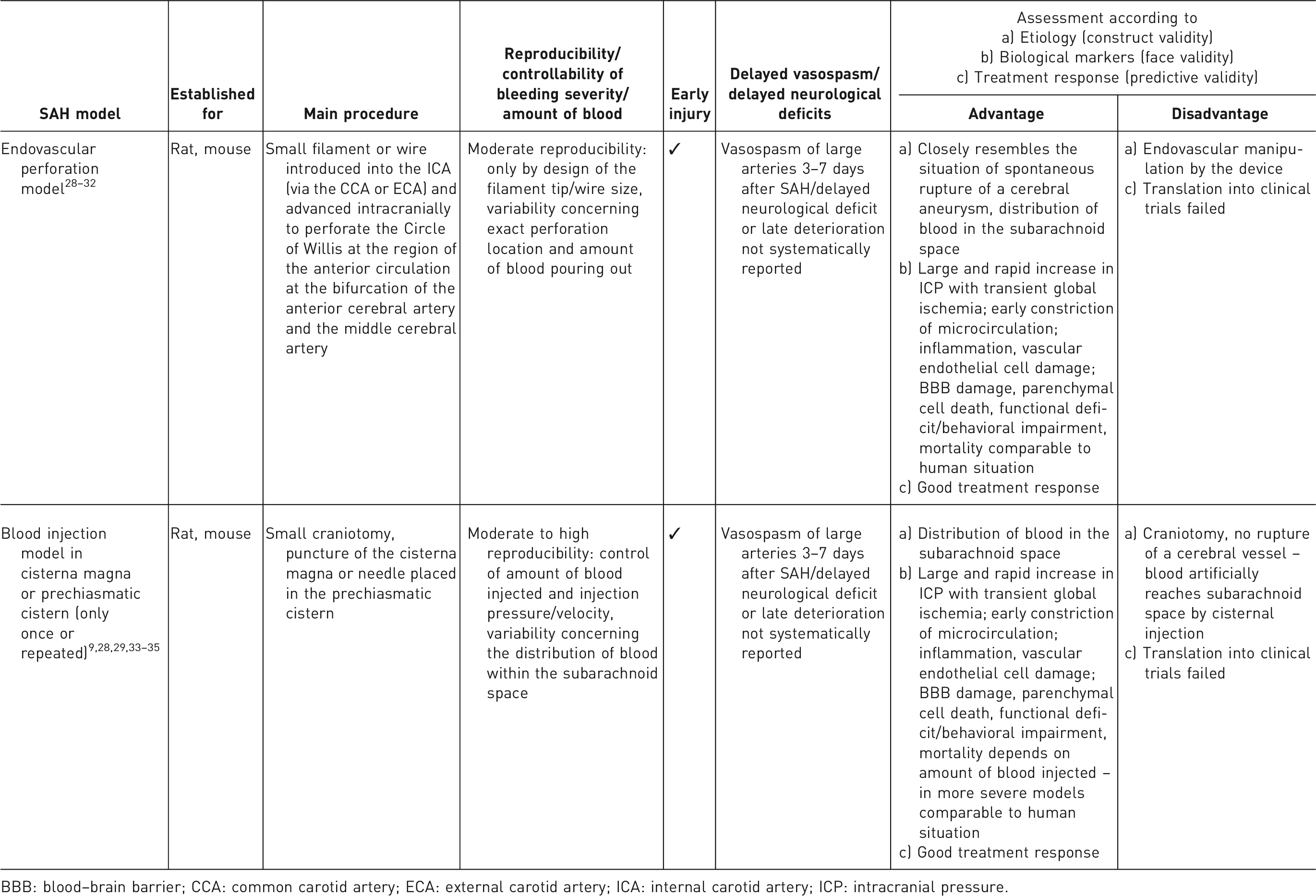
BBB: blood–brain barrier; CCA: common carotid artery; ECA: external carotid artery; ICA: internal carotid artery; ICP: intracranial pressure.
Additional procedure (both TBI and SAH)
Despite TBI or SAH in humans occurring while they are awake, anesthesia is obligatory when performing TBI or SAH surgery in animals.
In both models, during the initial surgery (SAH) and/or at different time points after the insult (SAH, TBI), it is general practice to measure the ICP as an important parameter of disease severity. 36 This can be achieved by insertion of a cannula in the cisterna magna connected to a pressure transducer, or by using fiber-optic-based systems, where a small pressure transducer is placed between the skull and the dura mater. In both cases, a small craniotomy has to be performed.
Anesthesia and surgical procedures may induce hypothermia in rodents, not only during the surgery but also within hours after awakening. However normothermia has to be ensured, since hypothermia provides neuroprotection,37,38 and may also impair animal welfare. 39
Reasons for choosing the rodent model for TBI and SAH
Most TBI and SAH animal models were first established in larger animal species, however in recent decades rodent models have most often been used. Besides the advantages of low cost (purchase and housing), easy handling and high level of standardization, rodents provide the possibility of advanced assessment of functional outcome and analysis of the underlying complex molecular cascades of cellular damage in the tissue after euthanasia. In addition genetically-modified mouse models can advance our understanding of molecular pathways. 40 As suggested in Tables 1a and 1b, the validity in all three common categories (construct, face and predictive) is not perfect for either TBI or SAH. This is not surprising since TBI in humans is a highly heterogeneous and complicated disease. The same is true for SAH, where size and location of the aneurysm, and the amount of blood and pressure under which it is released during rupture, depend on each individual situation. In addition, comorbidities (such as systemic hypertension or diabetes) make the situation even more complex. It is therefore not possible to model all aspects of the diseases in a single animal model,4,16 and the selection of the model has to be based on a clear definition of the respective aim to be followed and hypothesis to be proven. Accordingly rodent models for TBI and SAH are of extraordinary high value, and have remained indispensable to advancement of our yet incomplete understanding of the pathophysiological cascades in TBI and SAH, by elucidating important parts of the complex puzzle.
Current knowledge shows that the pathophysiological events in TBI and SAH are substantially comparable between small rodents and humans; yet the human brain is structurally and functionally more complex, with significant differences in the white-to-grey matter ratio. Therefore, in order to close the translational gap for preclinical drug development, studies in larger animal species with brains more closely resembling those of humans are recommended in order to make comprehensive use of the findings on pathophysiological cascades achieved in rodent studies.
Severity of the model
As stated in the ‘Working Document on a Severity Assessment Framework’, 41 a standard procedure should be followed for severity assessment, based on the thorough consideration of each step within the model procedure, to result in an assessment tailored for each respective species and strain, and for the individual laboratory situation. Therefore we can only outline the most basic considerations here and not refer to details within the models and species.
In both TBI and SAH, ICP increase occurs acutely or develops within hours. To measure ICP, which is highly recommended, a small craniotomy has to be performed. In addition, the skin and the underlying soft tissue at the head and/or the neck have to be cut. The brain itself has no pain receptors, but the neurocranium with its overlying periost and the meninges that are mainly affected during physical impact, and/or the elevated ICP, are highly algesic. The level of pain induced by the increased ICP correlates with the degree of the physical impact in TBI (which can be standardized) or the amount of blood in SAH (which can be standardized only in the blood injection model). After awaking, animals develop neurological deficits, which may reduce their ability to take food and water (with consequent risk of weight loss and dehydration) and inhibit normal behavior within their social environment. Assessment of functional deficits will probably also be associated with stress for the animals affected by the disease, particularly if they are not used to the tests. In addition, rodents are by nature prey animals, and as such fugitive, 42 and it is not known whether being unable to escape due to motor and/or cognitive deficits itself induces stress in rats and mice. Post-surgical housing is typically individual housing, so as to avoid wound manipulation by cage mates; but isolation can also be stressful for social species.43,44
Mortality in the controlled cortical impact (CCI)–TBI model is usually low (<10%, while controlled by the degree of impact) because no damage to the brainstem occurs. Mortality in fluid percussion injury (FPI) models depends strongly on the size of the pressure pulse, with mild to moderately severe impact resulting in mortality rates of 10–22% in rats 17 and zero mortality in mice. 18 In the ‘Marmarou’ (weight drop impact acceleration [WDIA]) model, the originally described mortality of up to 60% in spontaneously breathing rats is reduced to <10% by intubation and mechanically-assisted ventilation, which is recommended as the standard procedure for this model. 22 Very recently, the WDIA rat model has been redesigned for application in mice as a repeat mild TBI model, with zero mortality. 45
In SAH, mortality depends strongly on the model. It can reach up to 60% in the endovascular perforation model in rats,9,46 while most of the animals usually die immediately after the perforation (while still under anesthesia) or within the first few hours after the bleeding, 46 which resembles the early mortality rate in human patients. Recently, a modification of the endovascular perforation model in rats has been described, using a tungsten wire instead of a polypropylene filament, which induces a vessel perforation with a smaller diameter, leading to reduced mortality rates of around 20%.47–49 In mice, the endovascular perforation model is accompanied by a mortality rate of around 30%.5,30 In the blood injection models, mortality depends on the amount of blood injected, and usually remains ≤10%. 5
In summary, besides ensuring a thorough assessment of pain and distress, the definition of humane endpoints is especially necessary when using the more clinically related models of TBI and SAH. Overall, based on the cumulative consideration of the above-mentioned aspects, severity is usually graded as moderate for TBI (depending on the degree of physical impact) as well as for SAH when using the modified versions of the models with reduced mortality rates, and when implementing all the refinement opportunities mentioned in the text and table 3 below.
Assessment of welfare
Expected signs of pain, stress, disease, constraint or signs of reduced welfare
In rats and mice, pain and suffering cannot be easily detected. Generally, the burden induced by the pre- and postoperative handling, the surgery and the local effect of the lesion (most important of which is the increase in ICP) on the one hand, and the impairment of normal behavior caused by the lesion-induced functional deficit on the other hand, cannot be distinguished by common measures of reduced welfare. Therefore, up to now, signs of reduced well-being in TBI and SAH studies have had to be considered as unspecific as well as model-specific. Rats and mice are social animals, and reduced welfare rapidly leads to reduced spontaneous activity and isolation of the animals from their cage mates, which can be easily detected by observing the animals in their home cages. Furthermore, reduced activity can be tested by stimulating movement with a gentle push. Unusual body posture (crooked back) or an ungroomed appearance indicate that the animal is reluctant to move. Following TBI and SAH, any disregard of grooming (resulting in porphyrin deposits around the eyes and nose in rats) may be a sign of discomfort as well as the result of functional impairment of the paws or a deficit in motor coordination. Unusually aggressive behavior is another, yet more unspecific, sign of reduced well-being or stress and pain. Another important marker of constraint, and possibly the most important and scalable one, is reduced body weight, which is measured by weighing the animals daily. This indicates reduced food and water intake, which is caused by both unspecific illness as well as by functional impairment. More model-specific symptoms are the development of convulsions and epileptic seizures, which add to the burden for the animals. Isolation and sitting with the head towards the corner of the cage may be signs of head pain caused by increased ICP.
All these signs can be monitored by clinical examination. Routine home cage observation is therefore obligatory for welfare assessment, which however requires thorough knowledge of the normal appearance and behavior of the respective species. Strain-dependent characteristics also have to be considered. Besides body weight, these measures are not scalable and are, at least to some extent, observer-dependent.
This basic clinical observation and scoring system has been developed according to the standard procedure defined in the ‘Working Document on a Severity Assessment Framework’, 41 and is frequently used and accepted as a routine procedure for experimental studies of TBI and SAH. The main disadvantage of clinical scoring systems is the lack of scalability and the necessity for the observer to be sufficiently experienced; and it is not clear to what extent minor stress and pain or even subtle deviations from well-being can be detected using these tests. In addition, illness-related deviation from normal behavior cannot be discriminated from functional impairment-induced deficits, and it is not known whether (and if so, to what extent) the sole occurrence of functional impairment affects the subjective well-being of the animals. Further research is needed on this issue.
The same is true for more advanced tests assessing spontaneous locomotion (for example from home cage video recording over longer time periods or performance of home cage wheel running), which are increasingly being used for assessing pain in rodent models. 50 Overall, when applying tests requiring normal sensorimotor and cognitive functions, it must be thoroughly investigated whether abnormal test results are indeed induced by pain and stress, or rather by cognitive, sensory or motor impairment induced by the brain lesion following TBI or SAH, which could be independent of pain and stress.
Score sheet
Score sheet for traumatic brain injury (TBI) and subarachnoid hemorrhage (SAH).
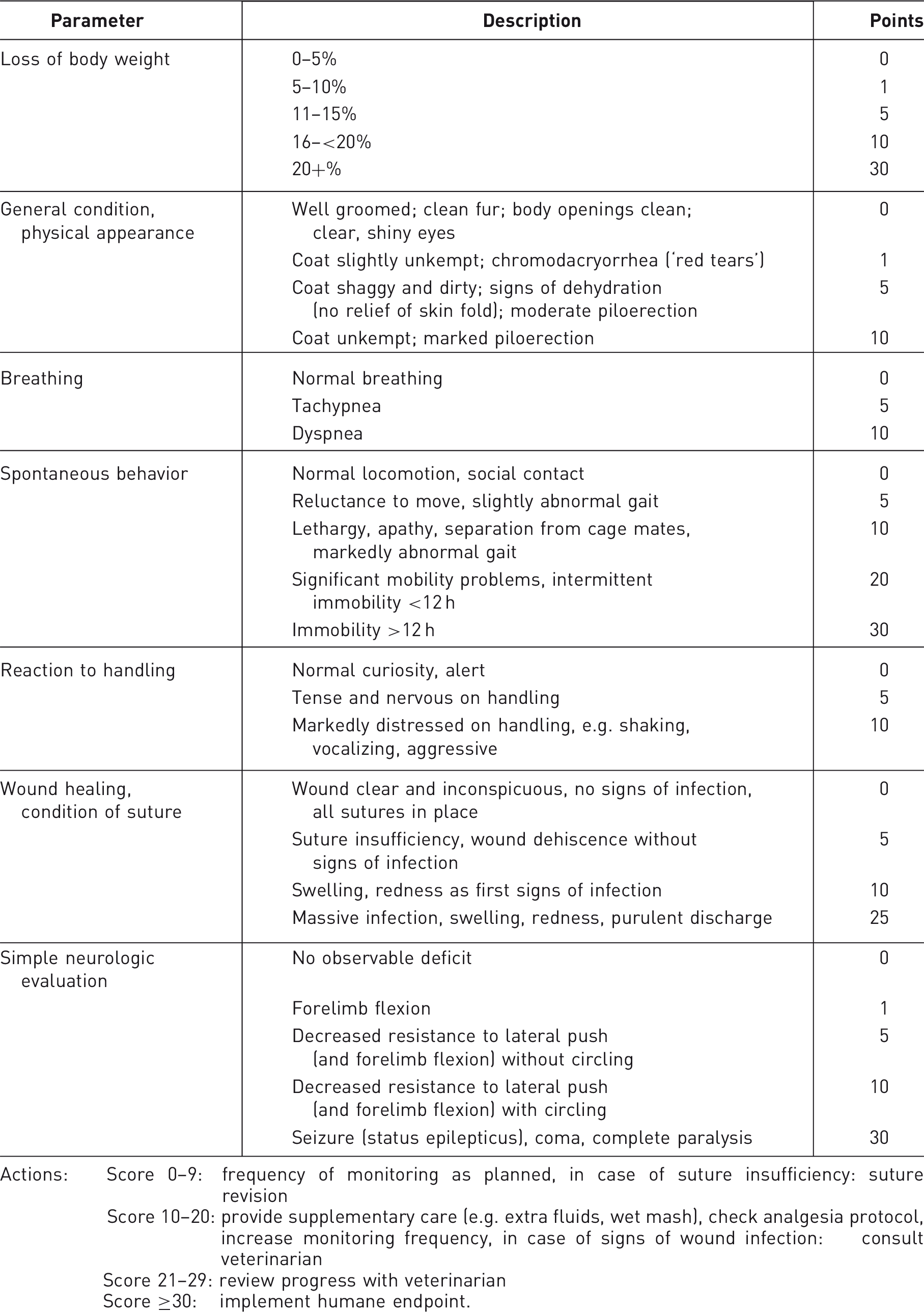
Actions: Score 0–9: frequency of monitoring as planned, in case of suture insufficiency: suture revision Score 10–20: provide supplementary care (e.g. extra fluids, wet mash), check analgesia protocol, increase monitoring frequency, in case of signs of wound infection: consult veterinarian Score 21–29: review progress with veterinarian Score ≥30: implement humane endpoint.
Assessment of the ‘General condition, physical appearance/Breathing/Spontaneous behavior/Reaction to handling/Wound healing, condition of suture’ topics and of body weight is standard practice for an experienced staff member familiar with normal rodent behavior.
The simple neurological evaluation can be briefly explained. It serves as a rough measure of the lesion-induced functional deficit, which may lead to impaired food and water intake and to deficits in grooming and activity, and therefore adds to the summed score. To evaluate forelimb flexion, the animals are carefully lifted by the tail with support of the lower body and advanced to the surface of a table. Healthy animals symmetrically stretch their limbs towards the ground, and animals with deficits in motor function and coordination of the limbs show asymmetric stretching or failure of stretching. Asymmetric limb function also leads to circular locomotion instead of straight movement.
Humane endpoints
In TBI and SAH models, pain or distress are unavoidable, therefore humane endpoints must be obligatorily defined by an upper limit of maximally tolerable score points. Close-mesh monitoring of the animals throughout the experiment is mandatory for checking if the endpoint is reached, and reaching the humane endpoint score in TBI and SAH studies leads to euthanasia of the animal.
In TBI and SAH models, the most likely problems expected to lead to endpoint scoring are:
weight loss; pain due to headache (craniotomy/ICP increase) or surgical manipulation; infection of surgical wound; immobility.
Thus, the most important symptoms, any one of which can by itself lead to euthanasia, are:
weight loss exceeding >20%; extreme pain which cannot be relieved by analgesics; immobility >12 h (assessed by close-mesh observation in the home cage); status epilepticus, coma, complete paralysis.
In addition, a combination of several signs of pain and stress may also lead to euthanasia, any one of which on its own may not require euthanasia.
Refinement opportunities
According to the ‘ARRIVE – Animals in Research: Reporting In Vivo Experiments’ guidelines, 51 all relevant refinement strategies should be described in the Methods sections when publishing results. In addition it should be kept in mind that every strategy to reduce (avoidable) stress in animal experiments is not only a matter of animal welfare but also leads to more stable and reliable results.
Refinement opportunities.
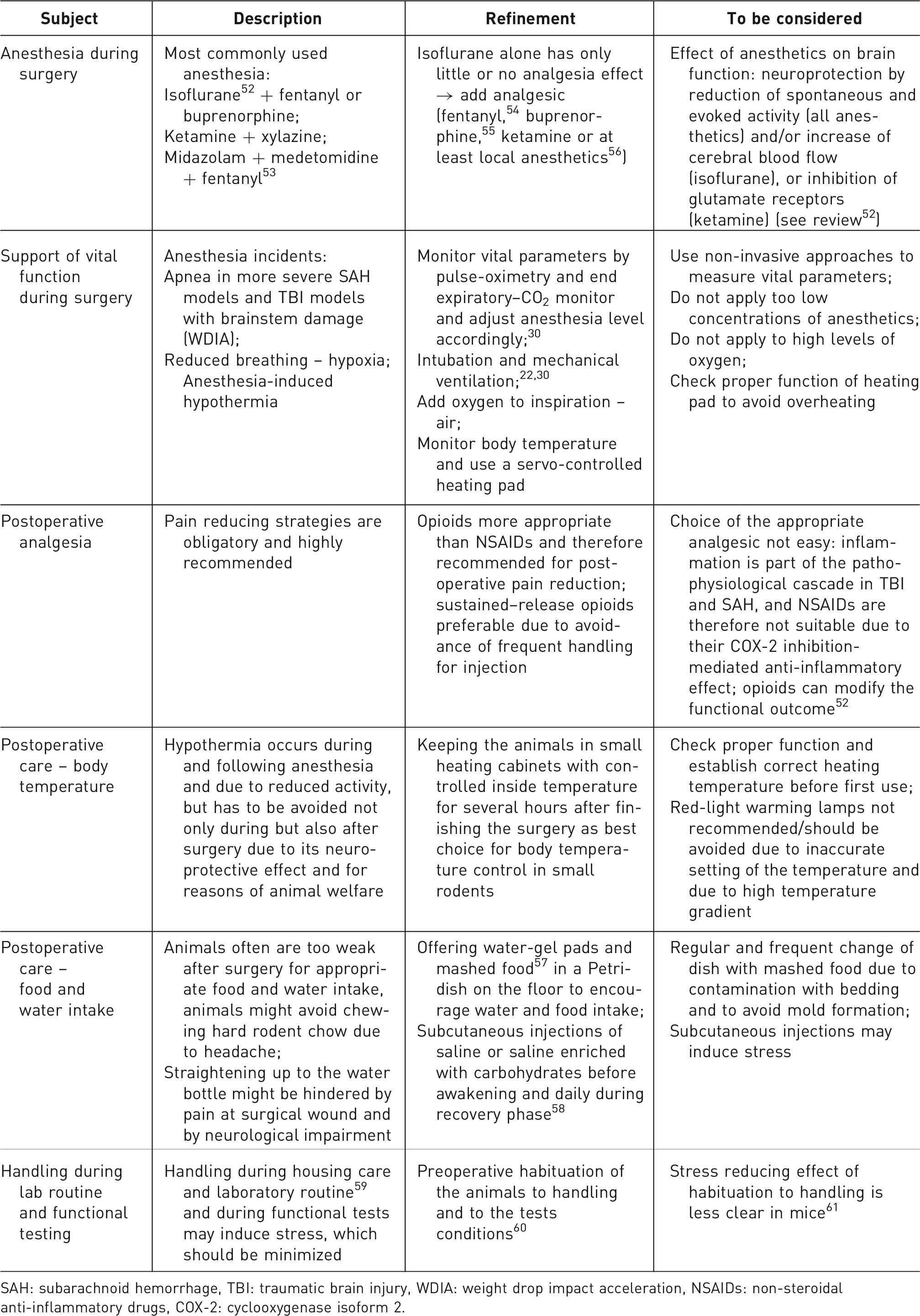
SAH: subarachnoid hemorrhage, TBI: traumatic brain injury, WDIA: weight drop impact acceleration, NSAIDs: non-steroidal anti-inflammatory drugs, COX-2: cyclooxygenase isoform 2.
Recommendations and further 3R research
As described above, the evaluation of pain and stress following TBI and SAH is not easy and up to now has mainly been based on the observance of behavioral and clinical signs of reduced welfare. More advanced evaluation is not commonly performed, but further research is highly recommended for enhancing our knowledge of suffering by the models, and for refining the parameters monitored within the score sheet applied here.
By thoroughly assessing facial expressions in rats and mice, the recently developed ‘grimace scale’ can in general be used to quantify pain.62,63 Whether it can be applied to TBI and SAH has yet to be proven. 64 Measurement of hormonal parameters such as prolactin and corticosterone in the blood can be useful for stress assessment.65,66 However it has to be considered that these hormones underlie circadian variations and show a high variance of basal levels, and sampling itself can be stressful. For reliable assessments, basal preoperative values have to be compared with postoperative values, and blood sampling has to be conducted under similar circumstances (time of day, experimenter and sampling method). It has to be investigated whether analysis of corticosterone content in feces may be a useful alternative to assessment in the blood. This provides an index of stress reaction over several hours, in contrast to blood sampling which provides an index of stress at one brief moment in time. 67 Monitoring of ultrasonic vocalization may also be considered. While experiencing fear or anxiety, adult rats emit alarm calls at a frequency of 22 kHz. 68 Yet in a recent study the suitability of using 22 kHz vocalization for pain assessment has been questioned. 69
Compiling standard operating procedures for the most common models of TBI and SAH with implementation of the above-mentioned refinement strategies, as is already available for the focal cerebral ischemia model of filament-induced middle cerebral artery occlusion in the mouse, 57 would further help to enhance standardization, reproducibility, and animal welfare in these models.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
