Abstract
Severe traumatic brain injury (TBI) is a multifactorial injury process involving respiratory, cardiovascular and immune functions in addition to the brain. Thus, live animal models are needed to study the molecular, cellular and systemic mechanisms of TBI. The ethical use of laboratory animals requires that the benefits of approaches be carefully weighed against potential harm to animals. Welfare assessments adapted to severe TBI research are lacking. Here, we introduce a scoresheet to describe and monitor potential distress in animals, which includes general welfare (body weight, general appearance and spontaneous behaviour) and TBI-specific indices (respiratory function, pain, locomotor impairment, wound healing). Implementation of this scoresheet in Sprague–Dawley rats subjected to severe lateral fluid percussion TBI revealed a period of suffering limited to four days, followed by a recovery to normal welfare scores within 10–15 days, with females showing a worse impact than males. The scores indicate that animal suffering in this model is transitory compared with TBI consequences in humans. The scoresheet allows for the implementation of refinement measures including (1) analgesia during the initial period following TBI and (2) humane endpoints set (30% weight loss, score ≥90 and/or respiratory problems). This animal scoresheet tailored to TBI research provides a basis for further refinement of animal research paradigms aimed at understanding or treating the sequelae of severe TBI.
Introduction
Traumatic brain injury (TBI) is a leading cause of death and disability worldwide. In humans, the Glasgow Coma Scale is used to classify patients’ state of consciousness and TBI severity, ranging from mild to moderate and severe. Among the 69 million people affected by TBI each year worldwide, an estimated 5.48 million suffer from severe TBI. 1 Such patients suffer from complex and multifactorial injuries and they have a high mortality rate. 2 Therefore, modelling this pathology in biomedical research is a priority.
Primary injuries are apparent immediately after a TBI has been sustained, and then evolve over hours, weeks or even years.3,4 In addition to brain changes, TBI can also affect respiratory, cardiovascular and immune functions. 5 Consequently, TBI is difficult to model in vitro, in cell cultures or in brain explants, and in vivo animal research is warranted. 6 The ethical use of laboratory animals requires that the benefits of the approach be carefully weighed against potential harm to animals. In TBI research, ethical concerns are especially relevant to severe TBI, after which animals may experience suffering. Animal research laboratories worldwide have committed to the three Rs – replacement, reduction and refinement7 – and they are additionally required to abide by national regulations.8,9 Moreover, animal welfare can impact on scientific outcomes in terms of reliability and reproducibility. 10 Thus, for both ethical and scientific reasons, it is important to assess accurately animal welfare after TBI. The first attempts to understand and evaluate general animal welfare in research laboratories were developed in the 1980s based on behavioural and physiological criteria, such as body weight, cardio-respiratory function and general appearance.11,12 Since then, scoresheets have become increasingly specific to species and the pathology being modelled. Severity scoring rubrics have been developed for use in welfare assessment and to define humane endpoints in toxicology or cancer research. Although a brain injury-specific scoresheet has been proposed, 13 specific scoring grids adapted to TBI research are lacking.
Here, we introduce a scoresheet designed to monitor animal welfare following TBI that includes both general welfare indices and TBI-associated characteristics. This scoresheet was used to describe animal welfare for a period of 160 days after severe TBI induced by the lateral fluid percussion (LFP) method (5–6 months and up to one year is considered a long-term timeframe for TBI consequences in rodents). 14 Following this descriptive approach, observations documented via the scoresheet provided a basis for the definition of humane endpoints, and refinement of animal care after severe TBI.
Materials and methods
Animals
Experimental protocols were approved by the CELYNE (CEA-042) committee on animals in research (APAFIS#10019-201704211705870), according to European Directive 2010/63/UE, and abide by the ARRIVE guidelines. Forty-two female and male adult (10-week-old) Sprague–Dawley rats (Envigo, Gannat, France) were randomized into four groups: 10 sham females; nine sham males; 11 TBI females; and 12 TBI males (the TBI group was designed to be larger than the sham group because of possible deaths that could be expected following severe TBI). There was no significant difference in body weight between the sham and TBI groups (females 219 (212.5–223.5) g, males 264.0 (251.5–279.5) g). The rats were housed in pairs, a sham rat and a TBI rat, on a 12-h light/dark cycle with controlled temperature (23 ± 3°C) and hygrometry (50 ± 5%). Enrichment included one wooden stick and a sizzle nest. Rats had ad libitum food and water access. After one week of acclimatization, the animals were handled gently for 5 min/day for one week to habituate them to the laboratory conditions and the experimenter.
Surgery and TBI induction
TBI was induced using the LFP model.15,16 Rats were anaesthetized with isoflurane (induction, 4.5%; maintenance, 1.8–2% in 30% oxygen/70% air) and received a subcutaneous infiltration of buprenorphine (0.02 mg/kg, Buprecare®, Axience, Pantin, France) and glycopyrronium bromide (0.1 mg/kg, Robinul-V®, Centravet, Lapalisse, France). After 25 min, animals were intubated with an endotracheal tube coated externally with pramocaine (Thronothane®, Biocodex, Gentilly, France) and anaesthesia was maintained via the artificial ventilation system (Ispra MA1 55-7058, Harvard ApparatusR, Edenbridge, UK; tidal volume = 6 mL/kg, breath rate = 60/min, positive end expiratory pressure = 5 cm H2O). Animals received an additional subcutaneous injection of ropivacaine (3.75 mg/mL, Naropeine, AstraZeneca LP, Courbevoie, France) and lidocaine (5 mg/mL, Xylovet®, Ceva, Libourne, France) in the scalp 10 min prior to incision. A circular craniotomy (4 mm diameter) centred 3.8 mm caudal to Bregma and 3.00 mm lateral to the midline was performed over the right hemisphere. A luer-lock syringe tip was attached to the skull and over the cranial window with cyanoacrylate and dental acrylic cement. Tubing (100 cm in length and 2.5 mm inner diameter, model TU0425, SMC) filled with saline was connected on one side to the LFP device (Custom Design and Fabrication, Virginia Commonwealth University, Richmond, VA, USA), and on the other side, to the luer-lock tip cemented to the cranial window. Anaesthesia was stopped and TBI was induced upon recovery of the hindpaw withdrawal reflex. This lighter anaesthesia corresponded to the transition from plane 2 to plane 1 of stage 3 (moderate) anaesthesia as defined by Siddiqui and Kim (2021), 17 and was shown to limit the loss of animals by respiratory arrest after TBI. The pendulum was released from its hook and fell down until it struck the water column, inducing a single 20 ms–5 atm fluid percussion pulse. The pressure of the shockwave usually decreased in intensity when it reached the dura mater, and was also measured at that location using an MLT0699 Disposable Blood Pressure Transducer (AD Instruments, Oxford, UK). A few drops of doxapram hydrochloride (Dopram-V®, Centravet) were applied on the tongue before injury and mechanical ventilation was resumed without anaesthesia. The scalp was sutured and the rat received 2 mL of saline in the flank subcutaneously. The same procedure, including a scalp incision and cranial window, was used for the sham group, but without TBI induction. Body temperature was set at 37°C with a temperature-controlled heating pad (LSI Letica, Barcelona, Spain). Breathing and heart rate were monitored with a MouseOx (STARR Life Sciences Corp., Oakmont, PA, USA) or Etisense system (Etisense, Lyon, France). After surgery, animal consciousness was assessed with the Tübingen–Boston Rat Coma Scale (RCS). 18 The RCS was completed every 5 min for a total score ranging from 0 (coma) to 16 (normal full consciousness).
Rats were injected with buprenorphine at 8 h and 16 h after surgery, and during the first 24 h they were heated with a red lamp. Moistened food was given for 1–2 weeks to facilitate food intake.
Post-TBI welfare scoresheet
The 11-item welfare scoresheet (Figure 1) was completed daily (08:00 h to 09:00 h, 3 min by cage) for the first 28 days post-TBI by three experimenters, then by one, until 160 days. The experimenters were one PhD student with six years of laboratory experience with rodents, a zootechnician with 10 years’ experience, and a master’s degree student with one year of experience. The three scorers examined the animals independently and were alone in the room during scoring. Blinding was not possible because the difference in animal behaviour after TBI was obvious. Animals were first observed remotely through transparent cages to determine their general condition, grimace score, breathing rate and spontaneous behaviour. The experimenter then opened the cage and scored each animal’s reaction to handling.
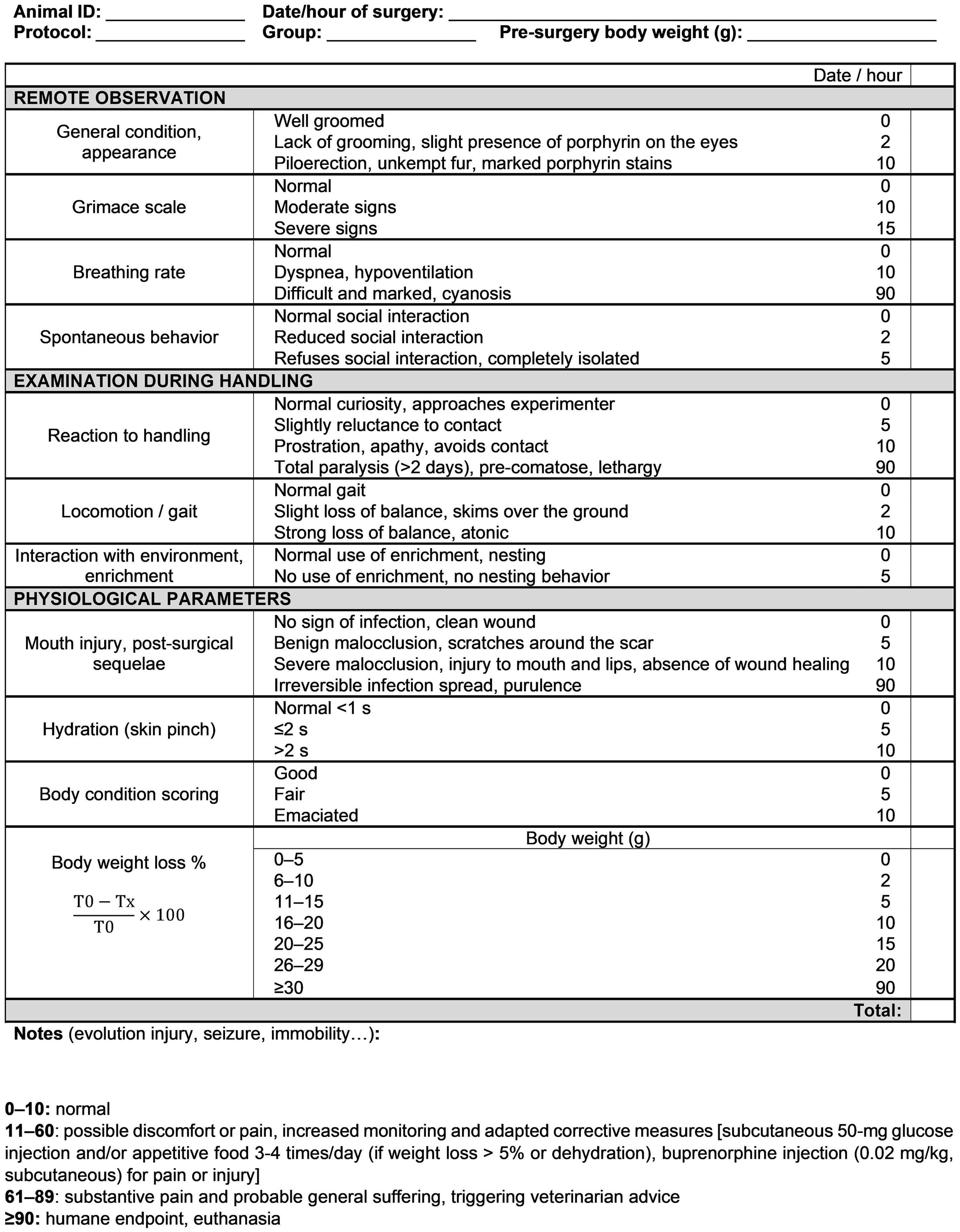
Scoresheet for severe traumatic brain injury. 0–10: normal physical condition, no pain; 11–60: presence of discomfort indicating a need for sustained observation and increased monitoring, moistened, palatable food, analgesia and subcutaneous saline or glucose; 61–89: significant suffering and pain indicating a need for compensatory measures, subcutaneous glucose, veterinary advice and consideration of euthanasia; ≥90: severe suffering indicative of a humane endpoint. T0 = pre-surgery weight; Tx = daily weight.
General condition, physical appearance
Included were coat (presence of piloerection, unkempt fur), eyes (presence of porphyrin), forelimbs and nose (clean or stained). We should note that adult animals have a tendency for a rougher, less silky coat than adolescent animals, and this should be differentiated from suffering.
Grimace scale
Pain and discomfort were indexed based on facial expressions with the Rat Grimace Scale developed by Sotocinal et al.; 19 rated expressions include orbital tightening, nose/cheek flattening, and ear and whisker changes.
Breathing rate
Breathing rate was observed visually, with attention to potential breathing problems (open mouth, wheezing, vocalizations or jerking) and cyanosis (paleness or blue coloration of the limbs, tail, ears or nose).
Spontaneous behaviour
Animals must be housed in pairs to enable observation of social behaviour and note any isolation from the congener. Subsequently, the experimenter moved each cage to a bench and opened it while watching the animal’s behaviour.
Reaction to handling
Prostration, isolation, absence of movement or escape from the experimenter were considered signs of poor welfare.
Locomotion/gait
Locomotion/gait were scored while the animal was on a table, with specific attention to the signs of atonia, loss of balance and/or weakness.
Interaction with environment, enrichment
Evidence of interaction with enrichment items, such as nibbling of the wooden stick or nesting.
Injury or sequelae
The surgical scar and incisor growth were examined daily. Because malocclusion can reduce food intake and lead to weight loss, incisor filing was performed after isoflurane induction (4.5%, 2–3 min procedure) with miniature saw (Dremel, France) whenever necessary until resumption of normal wear.
Hydration
Hydration was determined by pinching the upper back skin and measuring its relaxation time. 20
Body condition and weight
Each animal was observed visually and palpated on its back and flanks to feel the prominence of its bones. 21
Four score-based welfare stages were defined: 0–10, good physical condition and absence of pain; 11–60, possible discomfort or pain, triggering increased monitoring and adapted corrective measures, including subcutaneous 50 mg glucose injection and/or appetitive food 3–4 times/day (if weight loss >5% or dehydration), buprenorphine injection (0.02 mg/kg, subcutaneous) for pain or injury; 61–89, substantive pain and probable general suffering, triggering veterinarian advice; and ≥90, humane endpoint triggering euthanasia. Grades assigned on the scoresheet increased exponentially as the symptoms worsened, following a similar approach to Pinkernell et al. 13
Pinkernell et al. 13 proposed a scoresheet for both TBI and subarachnoid haemorrhage models that was used here to evaluate animal welfare independently of our own scoresheet for comparison during 20 days.
Endpoints
Two peri-operative endpoints were defined: dural tear; and respiratory failure requiring mechanical ventilation for >45 min. Subsequently, a veterinarian indicated whether euthanasia was warranted based on daily interaction. When necessary and at the end of the experiments, euthanasia was performed with 5% isoflurane anaesthesia followed by lethal intraperitoneal injection of barbiturates (100 mg/kg, Euthasol®, Centravet). Finally, the brain was removed for further histological analysis.
Data analysis
Data are expressed as median (interquartile range). Data were analysed in Prism® version 8.4.3 (Graphpad Software LLC, San Diego, CA, USA) or R software 4.0.3. 22 Raw data were first analysed for normality with the Shapiro–Wilk test. For two-group comparisons, normally and non-normally distributed values were analysed with Student’s unpaired t-tests and Mann–Whitney U tests, respectively. Including both male and female animals in the study warranted statistical analysis with a two-way analysis of variance (ANOVA) for sex and TBI. Before the two-way ANOVA was performed, data samples were tested for homoscedasticity with the Levene test and normality of residues. For non-normally distributed residues or heterogeneity, raw data were transformed with the Box–Cox power transformation. 23 If the two-way ANOVA revealed an interaction between sex and TBI, a Mann–Whitney U post-hoc test adjusted by the Bonferroni method was applied to identify specific differences. If not, no post-hoc tests were performed. Differences were considered significant at p < 0.05 for ANOVAs and at p < 0.025 for repeated Mann–Whitney U tests (two repetitions for males and females).
Inter-experimenter variability was studied by calculating intraclass correlation coefficient, expressed with 95% confidence intervals based on a mean-rating (k = 3) absolute-agreement/two-way mixed-effects model, and interpreted based on Koo and Li. 24
Results
Severe TBI induction and acute recovery
Although LFP induction produced a similar 5 atm shockwave in the water column, dural peak pressure diminished to a lower value in males (2.5 (2.4–2.6) atm) than in females (2.8 (2.6–2.8) atm; t(21) = 3.04; p = 0.006). Eights rats placed in the TBI group were not included in the analysis: five were euthanized due to a dural tear; one died spontaneously in the first hour following TBI; and two did not show evidence of TBI from RCS.
Relative to sham animals (n = 19; 10 females), the remaining TBI animals (n = 15; eight females) required an extended surgery-recovery time, as indicated by hindpaw withdrawal reflex (36.0 (18.0–72.0) min vs. 0 min in sham animals), righting reflex recovery (84.0 (51.0–90.0) min vs. 3.0 (0.0–30.0); F(1,30) = 134.40; p < 0.001) and RCS normalization (100.0 (75.0–115.0) vs. 5.0 (5.0–10.0) min; F(1,30) = 161.4; p < 0.001) (Figure 2). Immediately after TBI, we observed a 10 to 30-s respiratory arrest and abnormal heart rhythms (tachycardia and bradycardia) with cyanosis for about 5 min.
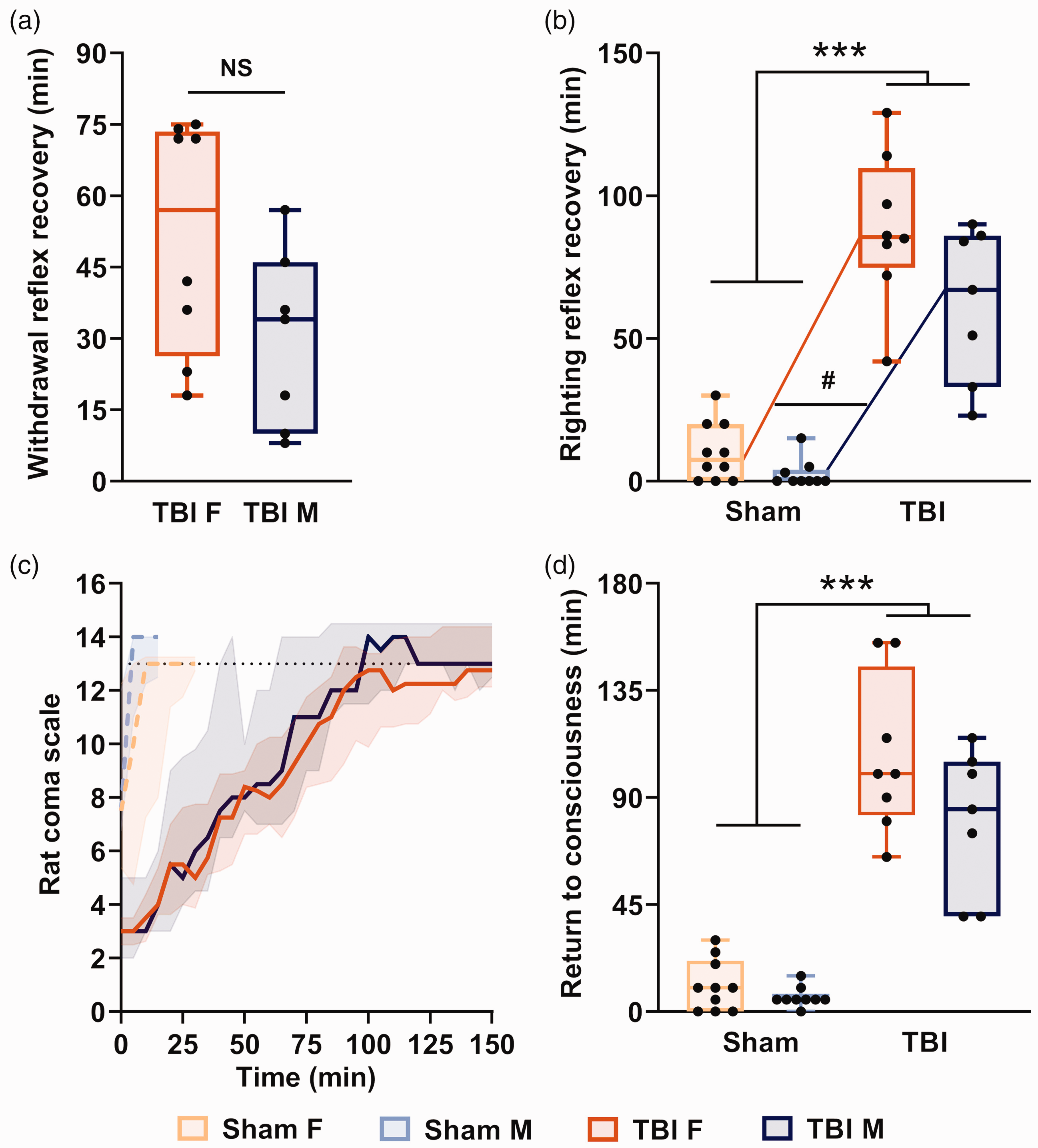
Return to consciousness after severe traumatic brain injury (TBI). Hindpaw withdrawal reflex (a) and righting reflex recovery (b). (c) Evolution of the Rat Coma Scale (RCS) scores. The dotted lines are the mean scores for sham rats, and solid lines are the mean scores for TBI rats. The black dotted line is the maximum score of sham rats. (d) Time to return to full consciousness assessed by the RCS. Values are shown as median and inter-quartile range. Sham F n = 10, TBI F n = 8, sham M n = 9, TBI M n = 7. NS, non-significant (Student’s t-test); # represents a significant interaction between sex and recovery time, p < 0.05, F vs. M comparison; ***p < 0.001, TBI vs. sham comparison (two-way analysis of variance followed by Mann–Whitney U post-hoc test).
Female rats took more time to recover their righting reflex (100.0 (82.5–145.0) min) than males (85.0 (40.0–150.0); F(1,30) = 7.44; p = 0.011) and showed a trend toward a longer hindpaw withdrawal reflex recovery (females, 57.0 (26.25–73.50) min; males, 34.0 (10.0–46.0) min; t(13) = 1.91; p = 0.088). Finally, 11/15 TBI rats (four females and seven males) had a subdural haematoma. These observations indicated severe TBI.
Applying our scoresheet to rats with severe TBI
Score of TBI animals worsened dramatically during the first 1–2 days postoperatively and then improved gradually thereafter until reaching stable basal values (<0.001; post-hoc Mann–Whitney U sham vs. TBI female p < 0.001) with an interaction of sex and TBI variables (F(1, 30) = 7.19; p = 0.02) but no significant difference between female and male groups (F(1, 30) = 1.577) (Figure 3(b)).
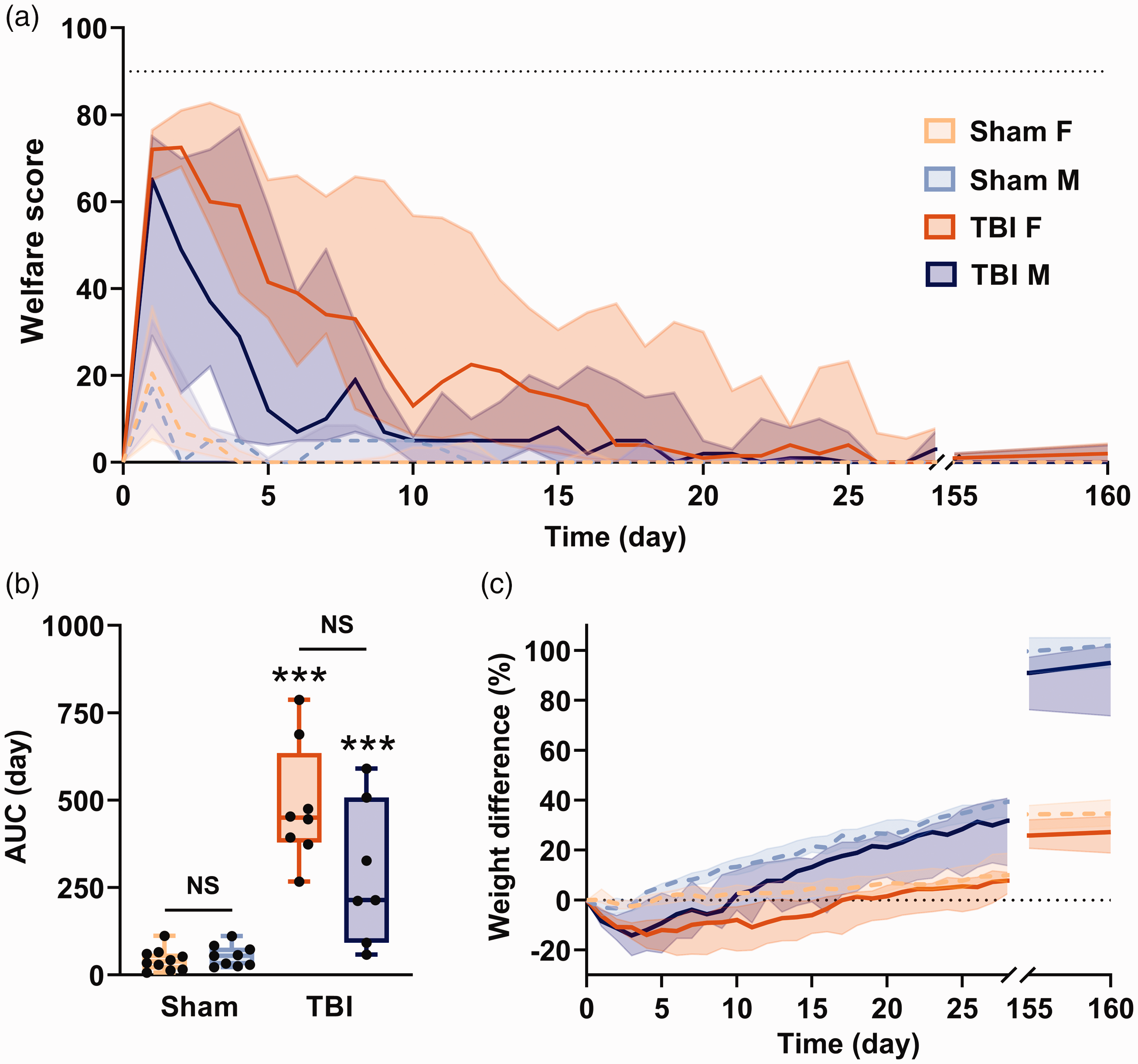
Animal welfare after severe traumatic brain injury (TBI). (a) Evolution of welfare scores in sham and TBI animals. The dotted lines are the mean scores for sham rats, and solid lines are the mean scores for TBI rats. The black dotted line is a humane-endpoint threshold. (b) Area under curve (AUC) of welfare scores during the first 12 days. (c) Per cent changes in body weight during 160 days after TBI. The black dotted line is the basal value. Values are shown as median and inter-quartile range. Sham F n = 10, TBI F n = 8, sham M n = 9; TBI M n = 7; NS F vs. M; ***p < 0.001 TBI vs. sham comparison (two-way analysis of variance followed by Mann–Whitney U post-hoc test).
Welfare worsening following TBI was attributable to virtually all scoresheet items. Compared with sham animals, TBI animals took longer to recover a silky coat (female (F), p < 0.001; male (M), p = 0.004) and had more days with probable pain (grimace scale ≥10; F, p < 0.001 and M, p = 0.008), dyspnoea (F, p < 0.001 and M, p = 0.012), abnormal responses to handling (F, p < 0.001 and M, p = 0.002) and impaired locomotion (both p < 0.001). TBI rats had greater weight loss than sham rats (F, p < 0.001 and M, p = 0.005) and took several days to normalize (female 16 days, p < 0.001 and male 8 days, p = 0.016) (Figure 3(c)). Finally, dehydration paralleled the changes observed in body weight, lasting 8–16 days in TBI rats versus 0–1 day in sham animals (F, p < 0.001 and M, p < 0.005). Detailed results for each scoresheet item are shown in Table 1.
Return to basal levels of scoresheet criteria and minimum weight after the severe traumatic brain injury.
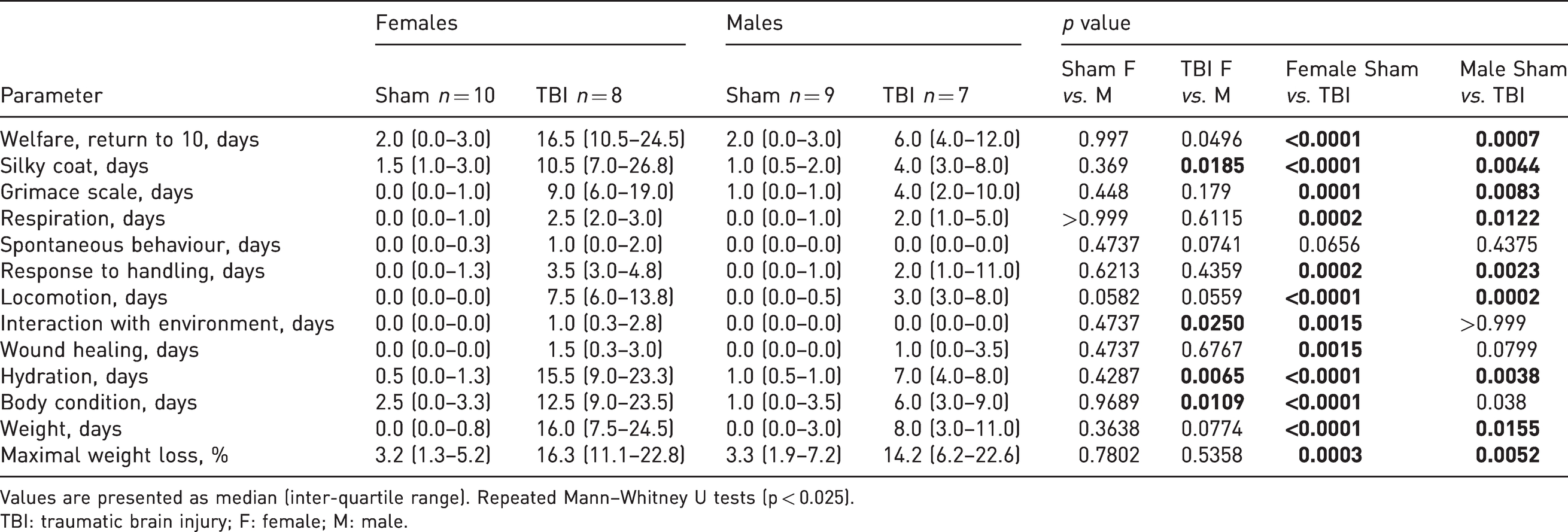
Values are presented as median (inter-quartile range). Repeated Mann–Whitney U tests (p < 0.025).
TBI: traumatic brain injury; F: female; M: male.
TBI female rats took longer than males to recover a normal coat (10.5 (7.0–26.8) vs. 4.0 (3.0–8.0) days, p = 0.019), were dehydrated longer (15.5 (9.0–23.3) vs. 7.0 (4.0–8.0) days, p = 0.007) and took longer to recover their normal body condition (12.5 (9.0–23.5) vs. 6.0 (3.0–9.0) days, p = 0.01). Body condition (thinness, 12.5 (9.0–23.5) days) and interaction with the environment (1.0 (0.3–2.8) day) were significantly impacted compared with sham animals in females only (p < 0.001 and p = 0.002, respectively) (Table 1). At 160 days post surgery, no significant differences between sham and TBI animals were evident. Notwithstanding these sex differences suggesting a greater impact on female well-being, our scoresheet detected suffering or pain during less than two weeks in both sexes. Welfare scores were then similar between sham and TBI animals, indicating that suffering, if present at these later time points, was not detectable by our scoring method. Results of scores from each item can be found in Supplementary material Table S1 online.
The scoresheet was also used to decide on animal care. For example, analgesic treatment with buprenorphine was administered during the first 24 h after injury, and was prolonged or reinstated if reaction to handling, grimace scale and/or mouth injury scored ≥10. This happened during the first week for 12/15 animals with severe TBI (all of the females, four males), but only for one sham female. In addition, subcutaneous saline or glucose injections were decided if dehydration or body weight scored ≥10, which happened in 12/15 TBI animals (all of the females, four males) and never for sham animals in the two weeks after surgery. Rats in which visual inspection revealed abnormally long incisors and displaying a welfare score ≥5 had their incisors filed. This happened in 13/15 animals (all of the females, five males) at four days, 9/15 animals (six females, three males) at one week, 7/15 animals (four females, three males) at 28 days and 3/15 (one female, two males) at 160 days after the TBI.
Inter-rater reliability and an alternative scoresheet
Our scoresheet was validated across three evaluators with different levels of education and lab experience. Scoring took about 3 min per cage of two animals, and was very consistent among the three experimenters for 22/23 TBI animals, with only one female receiving high scores >90 by two experimenters, but not the third, during the first four days post surgery. Overall, even taking this animal into account, the intraclass correlation coefficient was 0.89 (0.88–0.91) (Figure 4(a) and Supplementary Figure S1). 24
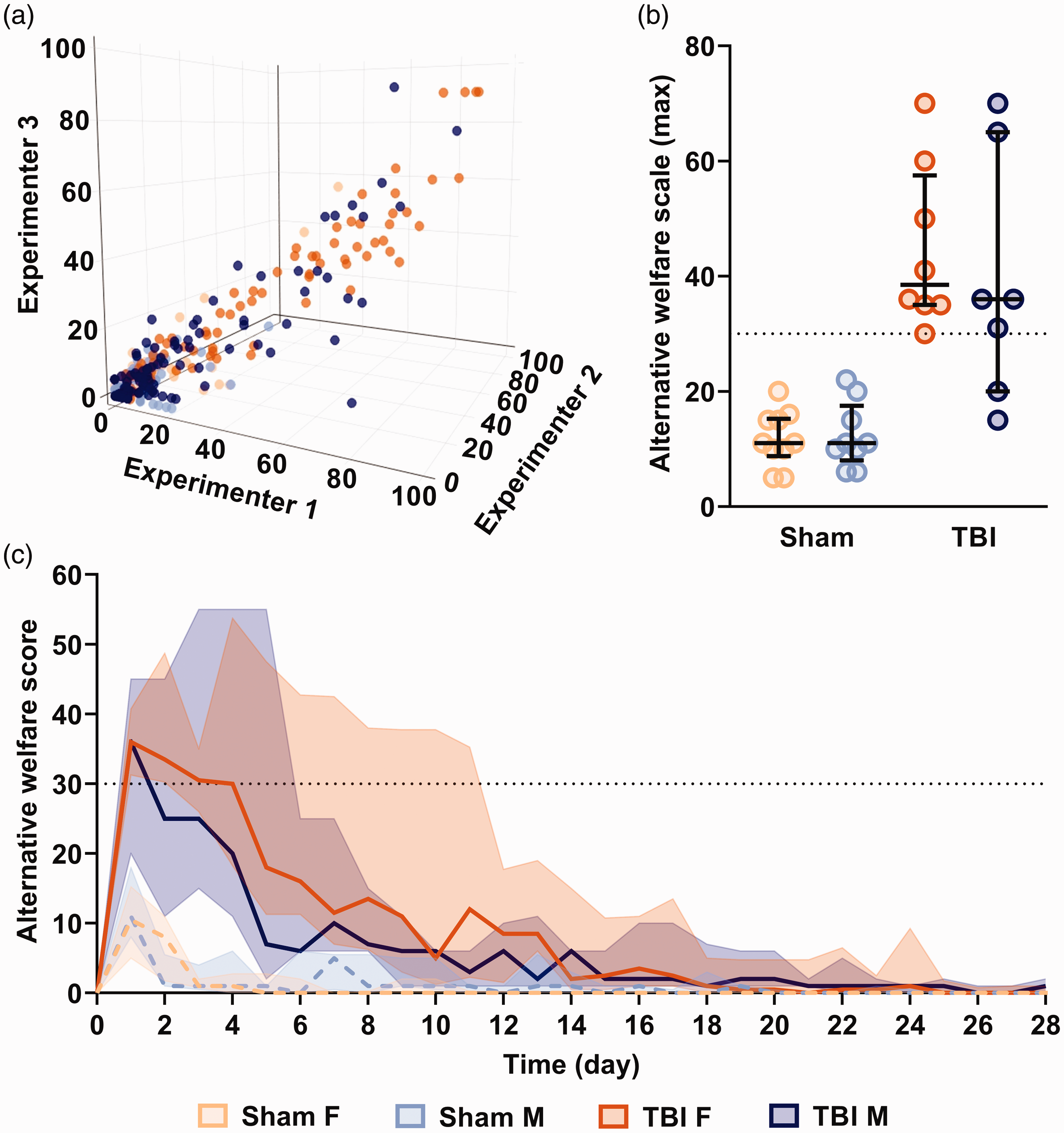
Inter-rater reliability and excess mortality with alternative welfare scoresheet. (a) Comparison between three independent experimenters. Each point corresponds to an animal’s daily score (x: experimenter 1, y: experimenter 2, z: experimenter 3). (b) Maximal scores with an alternative welfare scale 13 and (c) temporal evolution obtained with an alternative welfare scoresheet. 13 The dotted lines are the mean scores for sham rats, and solid lines are the mean scores for TBI rats. The black dotted line represents the humane endpoint triggering euthanasia in the alternative scoresheet. Values are shown as median and inter-quartile range. Sham F n = 10, TBI F n = 8, sham M n = 9, TBI M n = 7.
We also applied an alternative scoresheet used for brain injury. 13 Using this scale, 13/15 TBI animals (eight females, five males) that recovered a normal condition within two weeks reached a total score that would have triggered euthanasia from 1–4 days after surgery (Figure 4(b) and (c)) due to maximal score for spontaneous behaviour (one female), and weight loss (two females, two males) and/or maximal overall score (12 animals: seven females, five males). Among these animals, all but two recovered normal scores ≤10 28 days after TBI (except one male that scored 12 and one female at 30) and all were ≤5 after 160 days, indicating that suffering was no longer detected in these animals in the long term. Results from each item can be found in Supplementary Table S2.
Discussion
The currently introduced welfare scoresheet was developed specifically for assessing animal suffering after severe TBI. It includes general wellbeing items, such as weight, hydration, fur condition and interactions with conspecifics, experimenters and toys, as well as TBI-associated items, such as respiratory function, gait and the presence of wounds. The LFP method often induces transient respiratory dysfunction and mimics systemic complications in human patients. Gait deficits in TBI models parallel motor deficits25–27 and immobility/paralysis 26 in human patients. These deficits are not apparent in many rodent models of human disease and should be specifically scored after TBI. In addition, TBI model rats should be monitored for the presence of wounds, excessive incisor growth, reduced food intake, weight loss and a poor general condition. This scoresheet took only a few minutes to complete, and was implemented with a high degree of correlation across evaluators, even with different backgrounds in education and lab experience, suggesting high robustness.
Welfare scores took longer to normalize in females than in males, including a longer lasting impact on grooming, dehydration and interaction with environmental enrichment products. In humans, a meta-analysis suggested that women may fare worse than men following mild–moderate TBI, but may fare better than men following severe TBI. 28 Females have been reported to fare better than males in some animal models.28–30 Prior examination of sex differences in outcomes of moderate-injury LFP model rats have also revealed higher mortality but smaller contusions in females.31–33 Here, worse outcome in females was observed, but was also associated with a higher pressure applied to the dura. This result suggests that biomechanical factors, such as a smaller intracranial volume in females, 34 should be taken into account when assessing sex differences in TBI outcome, for example by comparing female and male rats of similar body weight (but different ages). Further studies are therefore required to better understand the sexual dimorphisms that may contribute to differences in TBI outcome.
To our knowledge, there are no specific studies applying a welfare scoresheet to a severe TBI model. An alternative scoresheet by Pinkernell et al. was designed for brain injury in general, but led to high scores and would have led to euthanasia in most of our animals, due to weight loss (>20%), immobility scores and/or epileptic seizures.35,36 Unspecific scoresheets and poorly adapted endpoints therefore have the potential of leading to unnecessary euthanasia of the animals.
Notably, we observed rapid recovery to normal welfare scores within two weeks of LFP. Severe TBI was demonstrated by loss of consciousness for >1 h, respiratory arrest lasting 10–30 s with cyanosis, and heart rate modifications lasting ∼5 min. The transience of effects on welfare scores in our rats differs from the long-term cognitive problems and disability found in 60–65% of patients with severe TBI,2,37 perhaps due to a higher degree of neuroplasticity and neurogenesis38,39 as well as accelerated timescales of metabolic, inflammatory and axonal dysfunctions in rodents. 40 Our study was limited to welfare assessment and thus we did not track behavioural deficits or neurological lesions that might persist beyond 160 days.
Most importantly, data collected with this welfare scoresheet can provide information relevant to the ethical justification of animal use in TBI research. Careful rating of procedures in terms of animal welfare and injury severity is needed to clarify benefits to society versus hardship for animals. The present data demonstrate that LFP-induced severe TBI does not induce long-lasting distress or suffering that could be detected by our scoresheet. By principle, any unnecessary suffering should be avoided in animal research, and when suffering is inevitable, its impact should be carefully weighed against expected benefits to society. Careful characterization of animal welfare using a scoresheet precisely adapted to TBI is therefore a pre-requisite for accurate judgement of the costs of animal research on severe TBI by ethical committees, and supports a justified use of rodents in such research projects. Our scoresheet is also helpful for defining precise humane endpoints for animals with severe TBI in accordance with the 3Rs criteria. 7 We propose that a weight loss trigger for euthanasia be 30% rather than the common 20% limit13,41 because 20–30% weight loss is common in moderate-to-severe TBI, 35 and in our study, animals that lost up to 30% of their body weight recovered their normal weight within two weeks if other welfare indicators were improving. This higher limit would prevent unnecessary euthanasia and thus could reduce the total number of animals used in TBI projects. This study can provide experimental evidence for discussions with ethical committees worldwide, and possible adjustments of endpoints for animals with severe TBI. Other humane endpoints are respiratory dysfunction, total paralysis, irreversible infection, untreatable wounds and a total score of ≥90 on our welfare scoresheet. Optimal adjustment of humane endpoints is key to the reduction of the number of animals included in experimental groups, as well as the refinement of procedures that may induce suffering.
Finally, welfare scores obtained during the first week following TBI could also provide an indication of the severity of the trauma, in addition to shockwave intensity, loss of consciousness indicated by RCS, withdrawal and righting reflexes recovery, and possible neurological lesions apparent on magnetic resonance imaging or histological images. Mild, moderate and severe TBI are still poorly defined in animals, and it is possible that this scoresheet could facilitate a better characterization of TBI severity, by taking into account animal welfare.
Study limitations
This study was performed using a limited sample size and the scoresheet presented here has not been validated by multiple research centres. Blinding the scorers was not possible due to the large difference in welfare between sham and TBI animals during the first weeks following surgery. This could have potentially impacted the reliability of our scoring at later time points. We should also mention possible neurological effects on repeated anaesthesia in animals requiring frequent incisor filing. We are aware that some but not all ethical committees might allow an endpoint definition at 30% weight loss, depending on national regulations and local ethical sensitivities.
Conclusion
This study proposes a reproducible and easy-to-use welfare scale adapted to severe TBI in rodents. In addition to providing semi-quantitative estimates of animal welfare to refine TBI models, this scoresheet demonstrated that suffering is apparent during a transitory period following TBI, but no longer detected in the long term, with animals recovering normal welfare within less than two weeks.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772221107269 - Supplemental material for Animal welfare assessment after severe traumatic brain injury in rats
Supplemental material, sj-pdf-1-lan-10.1177_00236772221107269 for Animal welfare assessment after severe traumatic brain injury in rats by Clélia Allioux, Laëtitia Achaintre, Fatima Cheataini, Baptiste Balança and Stéphane Marinesco in Laboratory Animals
Supplemental Material
sj-pdf-2-lan-10.1177_00236772221107269 - Supplemental material for Animal welfare assessment after severe traumatic brain injury in rats
Supplemental material, sj-pdf-2-lan-10.1177_00236772221107269 for Animal welfare assessment after severe traumatic brain injury in rats by Clélia Allioux, Laëtitia Achaintre, Fatima Cheataini, Baptiste Balança and Stéphane Marinesco in Laboratory Animals
Footnotes
Acknowledgements
CA is the recipient of a doctoral fellowship by Région Auvergne-Rhône-Alpes. We thank Mélodie Borel, Ellora Sequeira and Jenya Dursun for help with experiments and for their assistance in data collection. Thanks to Yannick Tholance for the discussions on statistics, Sophie Picavet and Manon Dirheimer for veterinarian advice.
Author contributions
CA and SM designed the welfare scoresheet. CA performed in vivo experiments. CA, LA and FC scored animals and retrieved data. CA, SM and BB contributed to the design of statistical analyses. CA and SM wrote the manuscript, with contributions made by all of the other authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Foundation Gueules Cassées ‘Sourire Quand Même’ (grant numbers FGC57-2020 and FGC34-2018).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
