Abstract
In many animal experiments scientists and local authorities define a body-weight reduction of 20% or more as severe suffering and thereby as a potential parameter for humane endpoint decisions. In this study, we evaluated distinct animal experiments in multiple research facilities, and assessed whether 20% body-weight reduction is a valid humane endpoint criterion in rodents. In most experiments (restraint stress, distinct models for epilepsy, pancreatic resection, liver resection, caloric restrictive feeding and a mouse model for Dravet syndrome) the animals lost less than 20% of their original body weight. In a glioma model, a fast deterioration in body weight of less than 20% was observed as a reliable predictor for clinical deterioration. In contrast, after induction of chronic diabetes or acute colitis some animals lost more than 20% of their body weight without exhibiting major signs of distress. In these two animal models an exclusive application of the 20% weight loss criterion for euthanasia might therefore result in an unnecessary loss of animals. However, we also confirmed that this criterion can be a valid parameter for defining the humane endpoint in other animal models, especially when it is combined with additional criteria for evaluating distress. In conclusion, our findings strongly suggest that experiment and model specific considerations are necessary for the rational integration of the parameter ‘weight loss’ in severity assessment schemes and humane endpoint criteria. A flexible implementation tailored to the experiment or intervention by scientists and authorities is therefore highly recommended.
The importance of animal welfare is increasingly appreciated by the general public as well as scientists. At the same time animal experiments are necessary for basic research and often provide the basis for subsequent clinical studies. For example, one publication describes that from 76 animal studies, which were published in highly cited journals, the results of 28 animal studies (37%) were replicated in human randomized trials, finally yielding eight interventions, which were subsequently approved for patients. 1 This suggests that well performed animal studies can provide a solid basis for the development of novel therapies. To facilitate animal experiments while improving animal welfare, it is necessary to develop objective measures to evaluate distress of animals and to define humane endpoints. Already in 1985 Morton and Griffiths published that the body weight of animals can be an indicator of animal distress and that this criterion has the distinct advantage that it can be objectively measured. 2 In this publication more than 20% reduction of body weight plus no consumption of water or food was defined as starving condition. In subsequent publications it was suggested that animals, which lose more than 20% of body weight should be killed in order to avoid suffering. 3 Consequently, many institutional animal care and use committees in the USA, but also in other countries, adopted the recommendation to consider euthanasia for animals, which lost 20% of their body weight unless a severe outcome for the animals was predicted and approved. Legislation has also embraced the idea of defining humane endpoints for experimental animals. One example is the directive 2010/63/EU of the European Parliament, which demands the use of humane endpoints without defining at which percentage of body weight loss an animal must be killed. 4 Supplementary information relating to this directive, recommends euthanasia of animals, when more than 20% or more than 35% of body weight is lost. 5 However, a weight loss of 35% is considered to be an extreme endpoint that requires sound scientific justification. In some toxicological studies, feed restriction studies, or animal models of colitis a weight loss of respectively, 20-30, 50 or about 40% can be observed, when compared to the starting weight or the body weight of a control group.6–8 Moreover, some mutant mouse strains can have up to 50% reduction in body weight when compared to wild-type mice. 9 However, absolute boundary values for euthanasia are also criticized. 10 Thus, the purpose of this study was to evaluate if 20% body-weight reduction is a valid parameter for defining the humane endpoint in different animal studies. In order to assess the validity of the parameter in various ‘real-life’ experimental conditions, we analyzed data from animal models with varying group designs and time schedules, which have been completed in different research facilities.
Materials and methods
Data and computation
Body weight data of rodents throughout distinct animal experiments were collected from different consortium members and pooled in an online repository (see online Supplementary file 1). Some data have been also used for other publications (see online Supplementary file 2). The absolute body weight or the percentage of body weight change was assessed centrally. All experiments were executed in accordance with the German legislation, the EU directive 2010/63/EU and approved by local authorities (for reference number see online Supplementary file 1).
Animal models
Restraint stress model in mice
Female C57BL/6 J mice were obtained from the Central Animal Facility (Hannover Medical School, Hannover, Germany). For restraint stress mice were placed into restraint tubes on 10 consecutive days (days 1–10) for 60 min (from 09.00 to 10.00 am). Restrainers (tubes with 23 mm internal diameter and 93 mm length) consisted of clear acrylic glass with ventilation holes (8 mm diameter) and a whole-length spanning, 7 mm-wide opening along the upper side of the tube. Following the restraining procedure mice were removed to their home cages. 11
Streptozocin model in mice
For this study female MRL/MpJ mice from the Central Animal Facility (Rostock Medical School, Rostock, Germany), which are prone to spontaneously develop autoimmune pancreatitis, were used. Diabetes was induced by ip (intraperitoneal) injection of 50 mg/kg STZ (Sigma-Aldrich, Steinheim, Germany) on five consecutive days (days 1–5). All control mice were sham-treated with the appropriated vehicle (50 mmol/l sodium citrate pH 4.5).
Dextran sodium sulfate-induced colitis in mice
Female C57BL/6 J mice were obtained from the Central Animal Facility (Hannover Medical School, Hannover, Germany). For induction of an acute colitis dextran sulfate sodium (DSS, mol wt 36,000–50,000; MP Biomedicals, Eschwege, Germany) was used. Mice were exposed to 0% (control group, H2O only), 1% and 1.5% DSS in drinking water for five consecutive days (days 1–5) and had access to a running wheel. 11
Chemical status epilepticus model in rats
In female Sprague Dawley rats with an electrode in the right hippocampal dentate gyrus, status epilepticus (SE) was induced by fractionated lithium-pilocarpine injections (as described by Glien et al., 2001 12 ). SE was pharmacologically terminated after 90 min. Post-SE rats were injected sc (sub-cutaneous) for two days with Ringer lactate solution and fed with baby food until they resumed normal feeding behaviour.
Intracranial rat glioma model
BT4Ca cells were stereotactically implanted into the right frontal cortex of male BDIX rats. Post-operatively, weight and general health condition were scored on a daily basis. Whenever a rat reached score 4 (severe neuronal symptoms, apathy) or lost >20% body weight as compared to the pre-surgical weight, the animal was sacrificed. In that condition the animal usually dies within the next few hours. Therefore, this criterion was defined as humane endpoint (please see Wu et al., 2018 for more detail). 13
Pancreatic resection model in rats
Wistar rats underwent a left sided pancreatic resection. The resected area was sealed with either a recently developed glue, fibrin or rinsed with NaCl as control group. The animals were observed for 14 post-operative days (POD) and body weight as well as severity score has been measured.
Liver resection in rats
50% of the liver was resected in Wistar rats. Resected area was sealed with Vivo 100, Fibrin glue or rinsed with NaCl. The animals were observed postoperative for three days and body weight as well as severity score has been determined. In another experiment a 50% liver resection has been performed using Wistar rats (Janvier, France) and the resected area was sealed with a polyurethane-based glue, fibrin glue or rinsed with NaCl.
Kindling model with Celastrol administration in mice
Male wild-type mice and HSP70-knockout mice were subjected to repeated kindling stimulations via an electrode in the right amygdala (as described by von Rüden et al., 2015). 14 Celastrol (1 mg/kg ip; Sigma Aldrich Chemie GmbH, Taufkirchen, Germany) or vehicle (5% ethanol, 0.1% Cremophor EL® in saline) was injected once daily 6 h before the electrical kindling stimulation.
Restrictive feeding in mice
For this study female C57BL/6J and ApoE deficient (ApoE-/-) mice were fed either ad libitum (AL) or caloric restricted (CR, 60% of AL) for 74 weeks (C57BL/6 J) or 60 weeks (ApoE-/-).
Dravet syndrome in mice
Conditional knockin male mice carrying mutation A1783V in exon 26 (B6(Cg)-Scn1atm1.1Dsf/J; #026133) were crossed with heterozygous female mice that express cre recombinase (X-linked to neuronal promotor Hprt gene; 129S1/Sv-Hprttm1(CAG-cre)Mnn/J; #004302). The offspring resulted in heterozygous Dravet mice expressing the A1783V mutation with/without Cre or wild-type mice with/without Cre presence. Animals received food (ssniff® R/M-H, Sniff, Soest, Germany) and water AL while Dietgel76A was offered as a supplement (Sniff, Soest, Germany) from P14 until P26.
Statistics
Statistical computing was performed with the R software (version 3.4.1) on a 64-bit machine. 15 The following packages were used: readxl for data extraction from MS-Excel files, reshape2 for data restructuring to the long format.16,17 Further, some plots were generated using Prism Software (version 6.1, GraphPad Prism Inc.). Group and/or time differences were analysed either by t-test (with Welch correction in case of unequal variances) or Mann-Whitney U test (Wilcoxon Rank Sum Test), depending on data distribution. The Shapiro-Wilk test was used to test against the null hypothesis of normality. In case of a rejection, the non-parametric test was used. Results were considered to be significant at the α = 0.05 threshold.
Results
Adult animals: impact of distress exposure, disease models, surgical procedures and drug treatment
Restraint stress model in mice
Mice exposed to 10 days of restraint stress lost weight during the early exposure phase resulting in a significantly reduced body weight (Figure 1(a)). However, towards the end of the stress model procedure animals started to regain weight thereby reducing the difference to control animals.
Stress paradigms and disease models. (a) Restraint Stress model in mice. Animals exposed to restraint stress show a significant loss in body weight compared to control animals on days 1–6, 8, 9, 13 and 14 (t-test with p ≤ 0.05 and Welch correction in case of unequal variances, n = 8, error bars are standard deviation). The maximum drop of body weight in single animals compared to starting conditions occurred on days 2 and 4 with 19%. No animal violated the 20% loss in body weight threshold. (b) Diabetes in MRL/MpJ mice. After induction of diabetes by streptozocin (STZ), mice lost significantly more body weight than control animals (Sham). Weight loss in animals with streptozocin-induced pancreatitis compared to untreated control animals was significant at day 22 and stayed so until day 113 (t-test or Mann-Whitney U test depending on data distribution, p ≤ 0.05, nctrl = 19, nmodel = 13, error bars are standard deviation). In one mouse a maximum loss of body weight of 32% was observed on day 89. Nine animals dropped below the 20% body weight threshold. (c) Dextran sulfate sodium (DSS)-induced colitis in mice. 0% DSS v. 1% DSS. Mice reached a body weight that did not differ significantly from control animals at day nine (t-test with Welch correction, p ≤ 0.05, nctrl = 7, nmodel = 8, error bars are standard deviation). (d) DSS-induced colitis in mice. 0% DSS v. 1.5% DSS. Day 5 shows a significant drop in body weight compared to control animals (t-test, p ≤ 0.05, nctrl = 7, nmodel = 8, error bars are standard deviation). Two animals dropped below the 20% body weight threshold and had to be euthanized. (e) Chemical status epilepticus in rats resulted in a rapid significant weight loss. This loss can be seen one day after SE compared to electrode-implanted sham rats without induced SE (t-test, p ≤ 0.0001, nctrl = 12, nmodel = 15, error bars are standard deviation). (f) Intracranial glioma model. Change in body weight (%) of rats with intracranial tumor 8 days before perfusion (P). Body weight on day 8 before perfusion was set as 100%. Data are shown as mean ± standard deviation. Significant differences of days compared to day 8 are indicated with an asterisk (one-tailed, one-sample t-test, p ≤ 0.01, n = 10).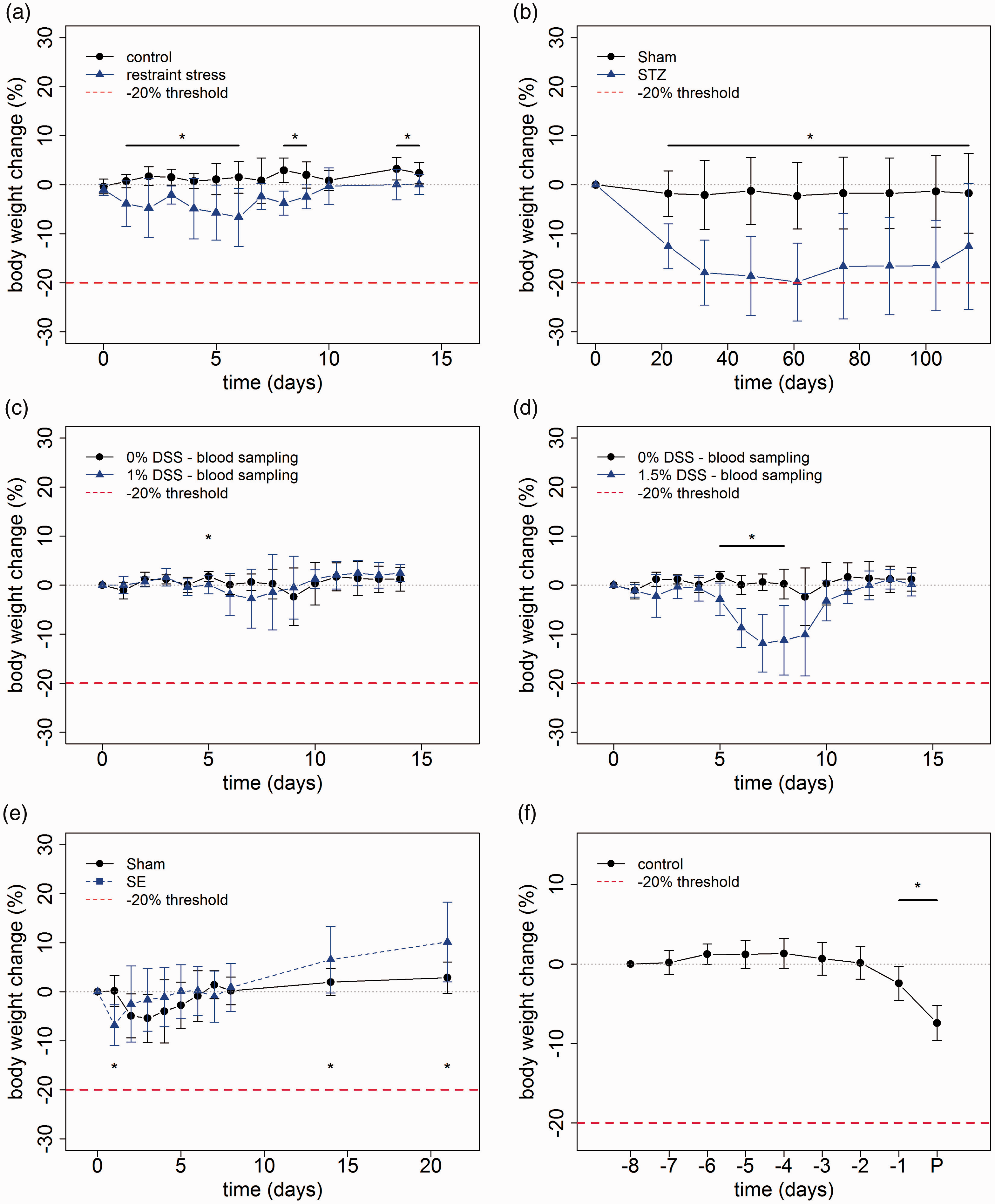
Streptozocin model in mice
Streptozocin administration in mice induced damage of pancreatic β cells resulting in a type 1 diabetic phenotype with hyperglycemia. The metabolic alterations in this model caused a progressive loss of body weight resulting in a significantly reduced body weight in comparison with control mice (Figure (a)). In eight animals (8 out of 13) weight loss exceeded 20% during the first two months after streptozocin exposure. These animals had a clinical score of not higher than 5 on a scale from 0 to 37 (for details of the clinical score: see online Supplementary file 3). Interestingly, mice that were not euthanized due to weight loss started to regain weight from the third month onwards (Figure 1(b)). This weight gain was not caused by reduced hyperglycemia, since the mean blood glucose concentration in these mice did increase from 21.8 mmol/l (standard deviation (SD) 3.4) on day 61 to 24.9 mmol/l on day 89 (SD 4.3) and further slightly increased until day 113.
DSS-induced colitis in mice
DSS-induced colitis represents a widely applied model of intestinal inflammation. Oral administration of 1% DSS via drinking water remained without relevant impact on body weight (Figure 1(c)). In contrast, mice exposed to 1.5% DSS in the drinking water for five consecutive days showed a pronounced drop in body weight. The weight loss exceeded 20% in two animals (2 out of 8), which were then euthanized. These animals had a clinical score of 0 and 3 on a scale from 0 to 17 (for details of the clinical score: see online Supplementary file 3). Five days following the end of oral DSS administration animals started to recover. Mice reached a body weight that did not differ significantly from control animals at day nine (following the termination of DSS exposure (Figure 1(d)).
Chemical SE model in rats
Pilocarpine is frequently used to induce a SE in laboratory rodents. SE with administration of the anticonvulsant diazepam 90 min after onset of SE resulted in a rapid weight loss. One day following SE the body weight proved to be significantly reduced as compared to naïve rats and electrode-implanted rats without induction of SE (sham) (Figure 1(e)). However, during the next two days the animal regained the lost weight. Subsequently, a higher weight gain was evident in SE-exposed animals resulting in a mean body weight exceeding that in both control groups 14 and 21 days following SE (Figure 1(e)).
Intracranial rat glioma model
After initial surgery for stereotaxic injection of BT4Ca glioma cells rats were in good health condition until shortly before finalizing the experiment with a mean survival time of 16 days. A minor loss of body weight (mean weight loss of 2.1% compared to the previous day), together with a slight deterioration of the general health condition was found about 2 days before reaching endpoint criterion. This was followed by a severe deterioration of the clinical score and more pronounced weight loss (mean weight loss of 5.2% compared to the previous day) on the following day (Figure 1(f)).
Pancreatic resection in rats
In a rat pancreatic resection model the resection surface was sealed with three different synthetic tissue glues or fibrin. In control animals, saline was administered instead of the adhesive material. Regardless of the treatment of the resection surface, a minor transient body weight loss was observed in the early post-surgical phase in the majority of animals (Figure 2(a)). None of the animals lost more than 20% of the body weight. On POD8 animals exceed their initial operation weight.
Surgery. (a) Pancreatic resection model in rats. Following a left sided pancreatic resection in Wistar rats the resected area was non sealed (NaCl group) or sealed either with a fibrin glue (TISSEEL) or polyurethane-based glues (PUG) (AM 110, 115 or 107). The animals experience a rapid weight loss up to day 4. After this time point the animals recovered and the weight gain to preoperative values was reached at day 7. Significant differences (*) indicate that data points are different from zero (one-tailed t-test, p ≤ 0.01, n = 3–9, error bars are standard deviation, n = 4). (b) 50% liver resection and comparison of recently developed glue. Following a 50% liver resection in Wistar rats, the resected area was non sealed (NaCl group) or sealed either with a fibrin glue (TISSEEL) or PUG (Vivo 100). The animals experience a rapid weight loss in the first 3 post-operative days. This weight loss is significant different to the pre-operative weight. A one-sample t-test was used in testing the samples whether they differed from zero body weight change (%). After day 0 all samples were significantly different from zero (one-tailed t-test, p ≤ 0.01, n = 3–9, error bars are standard deviation). (c) 50% liver resection and severity assessment. 50% liver resection in Wistar rats has been performed. The resected area was either glued with fibrin, PUG or rinsed with NaCl. The animals experience a rapid weight loss in the first 3 post-operative days. This weight loss is significantly different to the pre-operative weight. After this time point the animals recovered and the weight gain to preoperative values was reached at day 4. A one-sample t-test was used to test for significant differences to zero body weight change (%). On day 1–3 values are significantly different from zero (one-tailed t-test, p ≤ 0.05 n = 21, error bars are standard deviation).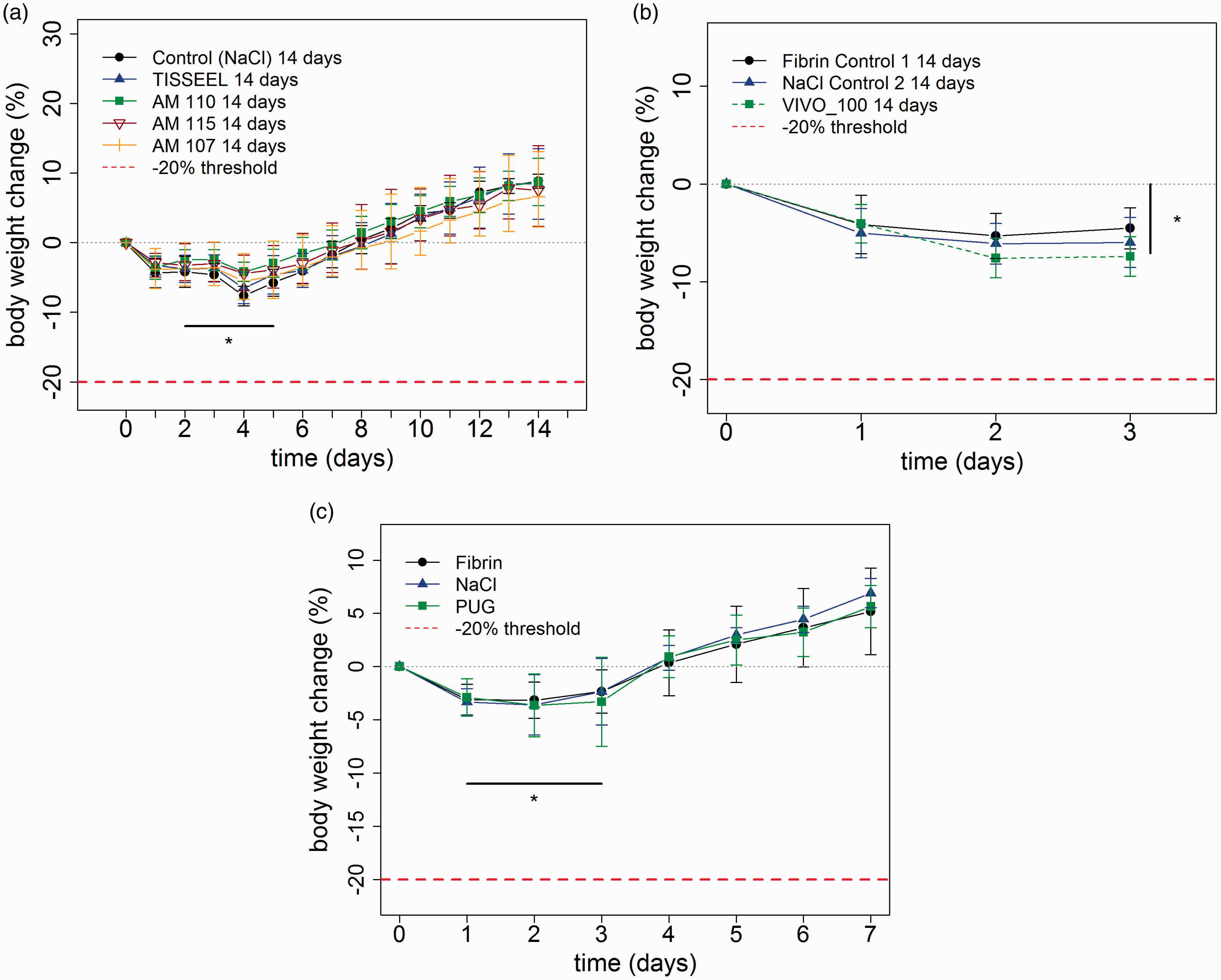
Liver resection in rats
During partial resection of the liver a recently developed glue (VIVO-100) was tested in comparison with fibrin for adhesion of the resection surface (Figure 2(b)). In control animals, saline was administered instead of the adhesive material. Regardless of the treatment of the resection surface animals exhibited only a slight drop in body weight following surgery which did not exceed 20%. In another experiment a 50% liver resection was performed on rats with a sealing of the resection site with PUG, fibrin or saline. Regardless of the groups, animals exhibit a significant weight loss on POD1–3 compared to their operation weight (Figure 2(c)). This was followed by an increase of weight at POD4 exceeding the initial weight.
Kindling model with Celastrol administration in mice
The kindling model with once daily seizure induction represents a frequently used model of temporal lobe epilepsy. The consequences of genetic and pharmacological targeting of an inducible heat shock protein were assessed in this paradigm. Daily injections of Celastrol significantly lowered overall body weight in wild-type mice and HSPA1 knockout mice in the kindling model of temporal lobe epilepsy (Figure 3(a) and (b)). The treatment protocol has been slightly adjusted introducing two interim phases without treatment in order to avoid a too pronounced weight loss. During these phases without drug administration, animals regained weight (Figure 3(a) and (b)).
Drug treatment. (a) Kindling model with celastrol administration in wild-type mice. Daily injections of celastrol significantly lowered overall body weight in wild-type mice (WT) and HSPA1 knockout mice (KO) in the kindling model of temporal lobe epilepsy (t-test, p ≤ 0.01 from day 1 to 17, nctrl = 8, nmodel = 9, error bars are standard deviation). (b) Kindling model with celastrol administration in knockout mice (t-test, p ≤ 0.05 from day 1 to 13 and 17, nctrl = 11, nmodel = 14, error bars are standard deviation).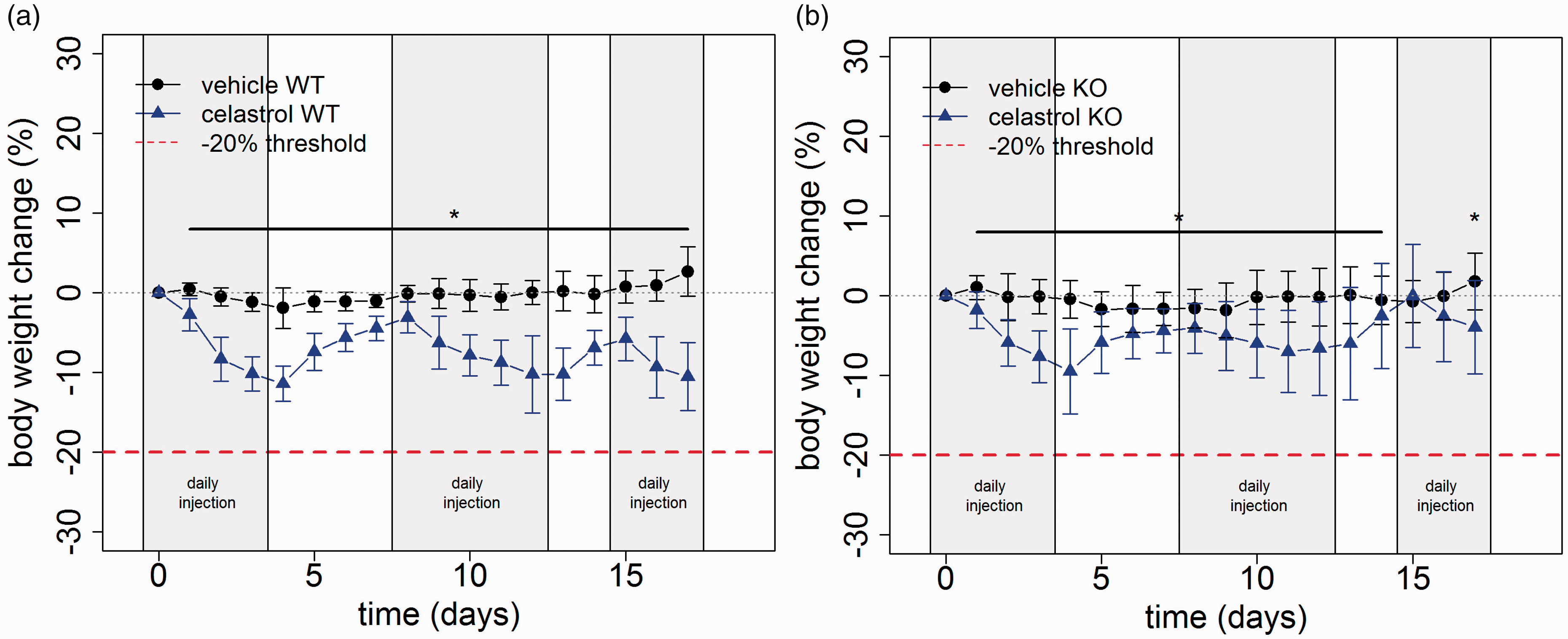
Growth curves: impact of restrictive feeding and of a genetic deficiency
Restrictive feeding
In order to assess beneficial effects on cognitive performance, CR feeding was initiated at an age of four weeks in C57BL6/J (Figure 4(a)) mice as well as ApoE-/- mice (Figure 4(b)). In comparison with mice fed AL, the body weight of mice with CR feeding proved to be reduced throughout the study by more than 20%. (Figure 4(a) and (b)). However, even mice fed with a CR diet gained body weight.
Impact of restrictive feeding on body weight. (a) Impact of restrictive feeding on C57Bl/6 J mice. Animals that were fed in a caloric restrictive (CR) manner had significantly less body weight compared to mice fed ad libitum (AL). There is a significant difference in daily body weight ranging from week 4 to 74 (Wilcoxon rank sum test, * p ≤ 0.05, nAL = 10, nCR = 10, three mice fed AL died, no mouse fed in a CR manner died, error bars are standard deviation). (b) Impact of restrictive feeding on ApoE-/- mice. A Wilcoxon rank sum test showed significant differences for weeks 4–60 (* p ≤ 0.05, nAL = 10, nCR = 10, error bars are standard deviation). (c) Dravet syndrome in mice. Animals with Dravet syndrome showed lower body weight at the time of weaning, but increased weight gain following the weaning (day 0). Dravet animals showed significantly increased weight gain over wild-type animals on indicated days (* p ≤ 0.05, nWT = 10, ndravet = 10, error bars are standard deviation).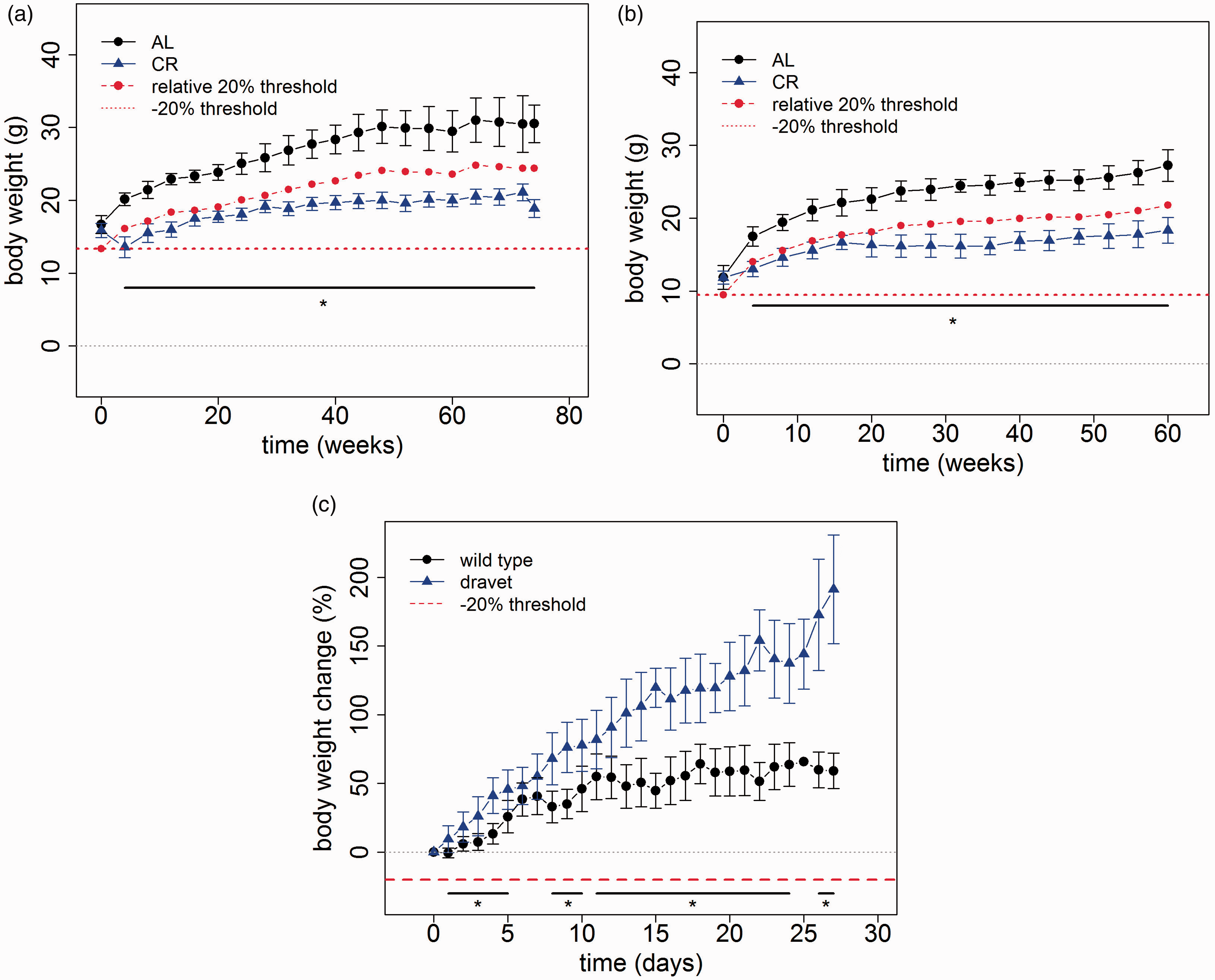
Dravet syndrome in mice
The Dravet syndrome is a rare genetic epileptic encephalopathy characterized by difficult-to-treat seizures as well as cognitive and motor impairment. 18 In a knockin Dravet mouse model, a lower body weight was evident at the time of weaning (data not shown). This is compensated over time as Dravet mice exhibit a steeper weight gain curve with a significantly increased weight gain from day 5 until day 27 post weaning (Figure 4(c)). As a consequence, the body weight of Dravet mice reaches a comparable range to wild-type mice approximately two weeks following weaning (data not shown).
Discussion
Body weight as a severity assessment parameter: simple to assess but difficult to interpret
At first glance body weight seems to be a simple parameter, which is easy to assess in an objective manner and can provide information for severity assessment scoring systems and decision about humane endpoints. However, interpretation of body weight loss in the context of severity assessment also constitutes a challenge that needs to take into account that appetite, food intake and body weight development are regulated in a very complex manner. 19 The interpretation is further complicated by the fact that weight loss can be a symptom of various conditions associated with reduced appetite, metabolic alterations, increased energy expenditure, or malabsorption.
As further discussed below there are several reasons why weight loss is considered as a parameter for severity assessment and humane endpoint. Weight loss can on one hand reflect decreased appetite as a consequence of distress, fear and pain. 20 On the other hand, it can also indicate progression of a chronic disease reflecting deterioration with an increasing burden for the animals. Moreover, weight loss related to different reasons can indicate a state of starvation, which if reaching a specific level can directly cause distress in affected animals.
Weight loss as a consequence of restrictive feeding, malabsorption, metabolic alterations, or drug effects on appetite control
The question about the level of weight loss, which should be considered as a burden itself, is for instance of relevance for experimental studies, in which a lowered body weight is a consequence of caloric restriction, changes in metabolism, or malabsorption. It needs to be taken into account that the majority of experimental animals receive food AL. Interestingly, caloric restriction can exert beneficial effects indicating overfeeding and overweight as a consequence of AL feeding.21–26 On the other hand, detrimental consequences such as an aggravation in age-related impairment of activity and anxiety-associated behaviour are also possible. 21
With the CR feeding protocols applied in the present studies, none of the animals lost more than 20% of their starting weight. Since the animals are still in the growth phase when initiating CR feeding, it is, however, better to compare the body weight of these mice to the body weight of mice fed AL. However, a decision about humane endpoint should also not be based exclusively on a 20% difference in body weight. More research is needed for an evidence-based definition of limits of tolerable weight differences while considering benefits of caloric restriction and other readout parameters measuring distress.
As mentioned above, the same question applies for models in which weight loss is related to metabolic alterations or malabsorption. In the DSS colitis model weight loss was observed, which proved to be slight and transient in response to 1% DSS but exceeded 20% in some of the animals responding to 1.5% DSS. In affected animals, we did not observe an association with a severe deterioration of the clinical state with bloody diarrhea being the only other symptom. Thus, despite the weight loss the impairment of the clinical state of the affected animals remained on a mild level, whereas wheel running performance decreased to nearly 0%, consistent with weight loss. A very similar observation has been made after induction of diabetes with streptozocin. Several mice lost more than 20% of their body weight within two months without a deterioration of the clinical state or signs of distress or pain (for parameters see online Supplementary file 3). In addition, after the first two months of diabetes the body weight increased again. In respective situations a strict application of 20% weight loss as a criterion for euthanasia might result in unnecessary loss of animals with the consequence of higher animal numbers necessary for the experiment.
Another example, which raises the question about the relevance of weight loss for severity, are drug effects on appetite regulation or energy expenditure. 27 A respective effect has been observed with Celastrol, which was administered to modulate neuroinflammatory responses in a chronic epilepsy model. Previous studies reported that Celastrol causes increased energy expenditure. 28 In our experiments, Celastrol-treated mice lost weight without persistent additional clinical symptoms except from acute adverse effects in the hours following administration. Thus, the data indicate that the effect was rather related to drug-induced metabolic alterations. Considering strict regulations applying for this experiment, we implemented treatment-free intervals to avoid weight loss exceeding 15%. The experience during this study again raises the question, which level of weight loss should be considered as a burden for the animals?
Considerations for interventions with transient weight loss
Specific considerations also seem to be necessary for interventions, which are associated with a transient weight loss. This has been observed in animals following partial liver or pancreas resection during the early phase of restraint stress as well as following SE induction. While the excess of 20% weight loss was an exception in our experiments, it is nevertheless debatable whether animals should be euthanized related to a drop in body weight, which is expected to be transient with a high level of certainty. As discussed above, losing animals as a consequence would imply the need for higher animal numbers. In this context, a transient 20% weight loss in the absence of other clinical symptoms should be considered a questionable humane endpoint considering the overarching 3Rs objectives.
Weight loss as a parameter reflecting distress
It is well known that pathophysiological factors related to distress, fear and pain can exert a strong impact on appetite thereby causing weight loss.20,29 Our findings further confirmed the impact of distress on body-weight development with restraint stress causing a transient drop in body weight during the early exposure phase. Thus, the present data further support the relevance of thorough body-weight monitoring for severity assessment in animals, which are exposed to stressful procedures.
Weight loss as an early marker for discontinuation
In models with progression of the disease, there is an urgent need for early humane endpoints markers for discontinuation decisions.30,31 In this context, body-weight reduction has been confirmed as a helpful parameter in different experimental scenarios.9,32,33 Following the same concept, our analysis in an intracranial glioma model revealed that body weight drop within 1–2 days, but not exceeding 20% is a reliable predictor of clinical deterioration. Please note that more specific markers for endpoint determination will be discussed in detail in a separate publication.
Considerations for growth curves in young animals
The assessment of growth curves during development of young animals also requires specific considerations. These are of particular relevance for severity assessment in genetically modified animals. The weight development of Dravet mice constitutes an interesting example with a reduced body weight evident at the time point of weaning, but more pronounced weight gain following weaning. The reduced weight gain at the time of weaning might be related to maternal neglect of affected animals. The strong weight gain following weaning indicates that the lower body weight is in this case not a result of inappetence or illness-associated weight reduction. Our data, therefore, suggest that a transiently lowered body weight during a specific developmental phase should always be evaluated in the context of other clinical signs, since loss of animals due to euthanasia can imply the need for higher animal numbers.
Conclusions and future recommendations
In conclusion, the fact that weight loss can be related to different reasons and that weight loss can take a different course, strongly suggests that experiment and model specific considerations are necessary for the rational integration of the parameter ‘weight loss’ in severity assessment schemes and humane endpoint criteria. For example, a weight loss of 20% as sole criterion for euthanasia would lead to the premature death of diabetic animals. In the intracranial glioma model, however, a body weight loss less than 20% is already a reliable predictor of clinical deterioration. Thus, each animal model is unique and requires tailor-made humane end-points.
In this context, it is important to consider that a less pronounced reduction of weight loss as a consequence of different interventions indicates that weight loss of less than 20% should be considered in clinical scoring sheets with consequences depending on the animal model.
This study, therefore, suggests that the decision for euthanasia should not be based solely on an arbitrary percentage of body-weight change, but should always consider other parameters indicating pain or distress and also animal model specific considerations.
Supplemental Material
LAN883319 Supplemetal Material1 - Supplemental material for Defining body-weight reduction as a humane endpoint: a critical appraisal
Supplemental material, LAN883319 Supplemetal Material1 for Defining body-weight reduction as a humane endpoint: a critical appraisal by Steven R Talbot, Svenja Biernot, Andre Bleich, Roelof Maarten van Dijk, Lisa Ernst, Christine Häger, Simeon Oscar Arnulfo Helgers, Babette Koegel, Ines Koska, Angela Kuhla, Nina Miljanovic, Franz-Tassilo Müller-Graff, Kerstin Schwabe, Rene Tolba, Brigitte Vollmar, Nora Weegh, Tjark Wölk, Fabio Wolf, Andreas Wree, Leonie Zieglowski, Heidrun Potschka and Dietmar Zechner in Laboratory Animals
Supplemental Material
LAN883319 Supplemetal Material2 - Supplemental material for Defining body-weight reduction as a humane endpoint: a critical appraisal
Supplemental material, LAN883319 Supplemetal Material2 for Defining body-weight reduction as a humane endpoint: a critical appraisal by Steven R Talbot, Svenja Biernot, Andre Bleich, Roelof Maarten van Dijk, Lisa Ernst, Christine Häger, Simeon Oscar Arnulfo Helgers, Babette Koegel, Ines Koska, Angela Kuhla, Nina Miljanovic, Franz-Tassilo Müller-Graff, Kerstin Schwabe, Rene Tolba, Brigitte Vollmar, Nora Weegh, Tjark Wölk, Fabio Wolf, Andreas Wree, Leonie Zieglowski, Heidrun Potschka and Dietmar Zechner in Laboratory Animals
Footnotes
Acknowledgements
We thank Ilona Klamfuß (Rudolf-Zenker-Institute of Experimental Surgery, Rostock University Medical Center) and Zhiqun Wu (Department of Neurosurgery, Hannover Medical School) for excellent technical assistance. We (Institute of Pharmacology, Toxicology, and Pharmacy, Ludwig-Maximilians-University, Munich) thank Uwe Birett, Sabine Vican, Katharina Gabriel, Katharina Schönhoff, Sarah Driebusch, Sieglinde Fischlein, Tamara Lindemann, Carmen Meyer, Sabine Saß, and Claudia Siegl for their excellent technical assistance. The study is properly described and the appropriate reporting guidelines have been followed. The methods used were appropriate and are described fully, the results are presented clearly and the conclusions are supported by the results. Also, any relevant ethical approval and consents have been obtained and included in the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Funding information on each study is given in online Supplementary file 1.
Supplemental Material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
