Abstract
Traumatic brain injury (TBI) is a clinical emergency with a very high incidence, disability, and fatality rate. Minocycline, a widely used semisynthetic second-generation tetracycline antibiotic, has anti-inflammatory and bactericidal effects. However, minocycline has not been explored as a therapeutic drug in TBI and if effective, the related molecular mechanism is also unclear. In this study, we examined the neuroprotective effect and possible mechanism of minocycline, in mice TBI model by studying the trauma-related functional and morphological changes. Also, in vitro cell studies were carried out to verify the animal model data. We found that minocycline significantly improved the neurobehavioral score, inhibited apoptosis, repaired the blood-brain barrier, and reduced the levels of inflammatory factors Interleukin-6 and tumor necrosis factor-α in TBI mice. In vitro, upon oxygen and glucose deprivation, minocycline reduced the levels of cellular inflammatory factors and increased the levels of tight junction and adherens junction proteins, thereby significantly improving the cell viability. Moreover, Mino treatment prevented the loss of tight junction and adherens junction proteins which were markedly reversed by an ER stress activator (tunicamycin) both in vivo and in vitro. Our findings set an effective basis for the clinical use of Mino to treat Traumatic brain injury-induced neurological deficits.
Introduction
Traumatic brain injury (TBI) denotes damage to brain parenchyma caused by trauma, in which extreme external force elicits a series of functional and morphological changes in the brain tissue. 1 It has been suggested that diseases including post-traumatic stress disorder, Parkinson’s disease, epilepsy, and chronic traumatic encephalopathy are related to neuroinflammatory reactions. 2 In TBI, external forces cause damage to the nervous system, which then triggers a cascade of secondary reactions causing further damage. Also, within a few minutes of TBI, activation of molecular pathways in the damaged area and surrounding normal tissues leads to neuroinflammation, mitochondrial dysfunction, and destruction of the blood-brain barrier (BBB). Among them, neuroinflammation is a vital factor for sustained neuronal damage. Under normal circumstances, moderate inflammation participates in countering invading pathogens, but persistent inflammation aggravates tissue damage. Presently, the early prognosis of TBI is challenging which hinders the targeted inhibition of the inflammatory factors, a potential treatment method in TBI. 3
The BBB, mainly composed of capillaries walls and glial cells, is a barrier between the plasma and brain cells or plasma and cerebrospinal fluid. 4 A compromised BBB prevents the entrance of harmful substances to the brain tissue and carry out precise regulation for material exchange and molecular transport between the cerebral microvessels and brain tissues. 5 Thereby, the BBB plays an essential role in maintaining the microenvironment of the central nervous system (CNS). After TBI, a compromised BBB leads to an altered brain microenvironment and activates secondary pathophysiological reactions such as brain edema and neuroinflammation. 6 Besides, the long-term micro-opening of the BBB is closely linked to sustained complications in TBI. Previously, it was shown that post-TBI inflammatory responses are initiated by peripheral immune mediators that enter the CNS through the porous BBB. 7 Therefore, in trauma, inhibiting the expression of related immune mediators and repairing the integrity of the BBB can stabilize the brain microenvironment, reduce the pathological response, and improve the long-term disease prognosis. Accumulation of the misfolded proteins in the endoplasmic reticulum (ER), causing dysfunction of ER, is known as ER stress. Previous studies suggested that ER stress plays an important role in several neurological diseases, including neurodegeneration disorders, 8 vasculopa, 9 and TBI. 10 The alleviation of ER stress markedly promotes the recovery of nerve function after TBI; however, the role of ER stress in post-TBI BBB disruption is poorly understood.
Minocycline (Mino), a widely used semisynthetic second-generation tetracycline antibiotic, has anti-inflammatory and bactericidal effects. 11 Notably, Mino can pass the BBB. This property makes it a fascinating drug candidate to treat various neurological diseases, such as cerebral hemorrhage and TBI. 12 Experiments showed that Mino, an inhibitor of caspase-3, stabilizes mitochondrial membranes by reducing their permeability, reduce Bid protein release, inhibits microglial activation, and reduces neuron and oligodendrocyte apoptosis. 13 However, Mino has not been explored as a therapeutic drug in TBI and if effective, the related molecular mechanism is also unclear. Given this, by studying the trauma-related functional and morphological changes, we examined the neuroprotective effect and possible mechanism of Mino in mice TBI model. Also, in vitro cell studies were carried out to verify the animal model data.
Materials and methods
Animals
C57BL/6 male mice (20–25 g) were purchased from the Animal Center of the Chinese Academy of Sciences (Shanghai, China). These were maintained at 21°C–23°C and a regular light/dark cycle with food and water ad libitum. All animal experiments were approved by the Ethics Committee of Wenzhou Medical University. According to the random number table method, 81 mice were divided into four groups (n = 6/n = 5). The mice were intraperitoneally (i.p.) injected with Mino (20 mg/kg at 30 min post-TBI, daily for 3 days). This dose of Mino was selected based on previous studies which investigated effects of Mino in iron metabolism regulation in TBI, 12 and attenuation of brain injury after intracerebral hemorrhage in rats. 14 The ER stress activator tunicamycin (TM) (10 mg/kg) was i.p. injected immediately after TBI.
Animal model construction
As described previously, the controlled cortical impact (CCI) method was used to establish TBI models 15). Briefly, after the anesthesia (pentobarbital, 40 mg/kg, i.p.), the mouse scalp was cut to expose the skull. Then, the bone flap was removed and a pneumatic cylinder (Impact One TM Stereotaxic Impactor, Leica) at 4 m/s speed, ~1 mm depth, and 150-ms impact duration was used to control the right cerebral cortical impact. After the procedure, the incision was sutured and mice were allowed to recover using a warming device. The animals in the sham group underwent the surgical procedure without cortical impact.
Cell culture and oxygen-glucose deprivation (OGD) experiment
HUVECs were cultured in endothelial cell medium (ECM, ScienCell) supplemented with 5% FBS (Gibco, Invitrogen), 1% ECGS (Gibco, Invitrogen), penicillin (final concentration, 100 U/ml) and streptomycin (final concentration, 0.1 mg/ml). These cells were maintained in a humidified atmosphere of 5% CO2 and 95% air at 37°C. For the oxygen-glucose deprivation (OGD) experiment, cells were incubated in an anaerobic chamber for 6 h with a glucose-free DMEM with TM (3 mM) and the oxygen level was maintained below 0.5%. After this, cells were further incubated for another 6 h under normal culture conditions (5% CO2 and 95% air at 37°C). Mino was added 2 h before the OGD stimulus.
Garcia neurobehavioral score
The recovery of neurological function in mice was scored for spontaneous activity, symmetrical movement of limbs, forelimb extension, climbing, body trunk reaction, tentacles reaction, and lateral rotation reaction. The scoring for each item was 0 (worst performance) to 3 (best performance), and the total score was calculated as the sum of all the items (maximum score: 21).
Brain water content
Twenty-four hours after the treatment, the mice were sacrificed to obtain the brain tissue which was divided into two parts, right and left hemispheres. To determine the wet weight, the left cerebral cortex was weighed immediately. Then, the tissue samples were dried at 72°C for 72 h and the dry weight was noted. Brain water content was calculated as follows: [(wet weight–dry weight)/wet weight] × 100%.
Cell counting kit-8 (CCK-8) assay
Five thousand cells/well were seeded into 96-well plates and incubated for 24 h at 37°C and 5% CO2. As indicated, treatment with different concentrations of Mino and OGD was performed 12 and 6 h before the CCK-8 treatment respectively. Subsequently, a total of 10 µl CCK-8 (Dojindo Molecular Technologies, Inc.) was added to each well and incubated for an additional 2 h. Lastly, the optical density (OD450) was measured using a plate reader.
Western blotting (WB)
Equal amounts of protein samples were separated by SDS-PAGE and transferred onto PVDF membranes. After that, membranes were blocked with 5% skimmed-milk-TBST for about 90 min at room temperature (RT). This was followed by an overnight incubation at 4°C with corresponding primary antibodies rabbit anti-c-caspase-3 (1:1000), rabbit anti-Bcl-2 (1:300), rabbit anti-Bax (1:300), rabbit anti-GAPDH (1:1000) from Santa Cruz Biotechnology; rabbit anti-occludin (1:1000), rabbit anti-β-catenin (1:1000), rabbit anti-GRP78 (1:1000), and rabbit anti-CHOP (1:1000) from Abcam. After that, membranes were washed four times in TBST and then incubated with secondary antibodies for an hour at RT. The signal intensities were quantified using the Image Lab 3.0 software (Bio-Rad).
TUNEL staining
The apoptotic cells were detected using the TUNEL analysis kit (40307ES20, Yeason). Firstly, the deparaffinized brain tissue was rehydrated and then washed with PBS. Then, completing the treatment with sodium citrate buffer, the tissue was stained with Alexa Fluor Labeling Mix or DAPI as indicated. Lastly, the images were obtained using a fluorescence microscope (Olympus Inc., Japan)
Immunofluorescence (IF) staining
Samples were fixed with 4% PFA (paraformaldehyde) for 30 min. Then, the prepared cells and tissue were blocked with 5% BSA for about 30 min, followed by overnight incubation at 4°C with the corresponding primary antibodies: c-caspase-3 (1:1000), Zo-1(1:1000), IL-6 (1:1000), TNF-α (1:1000), and β-catenin (1:1000). After that, samples were washed at RT and incubated with secondary antibodies for an hour. The cell nucleus was stained with DAPI. Finally, images were captured. The fluorescence intensity was analyzed using the Integrated Performance Primitives v9.0 software (Intel Corporation).
ELISA
The levels of IL-6 and TNF-α were estimated in mice brain and cell lines samples using the ELISA kits (BosterBio) as per the manufacturer’s instructions. The optical density (OD) of the samples was recorded by the SpectraMax microplate reader (Molecular Devices) and fitted to a standard curve to obtain the corresponding concentration values.
Real-time quantitative PCR
For detailed PCR operation steps, please refer to previous literature. 15 The primers used for mouse TNF-α and IL-6 are as follows: TNF-α forward: 5′-TGATCCGCGACGTGGAA-3′, reverse: 5′-ACCGCCTGGAGTTCTGGAA-3′; IL-6 forward: 5′-CCAAGAGGTGAGTGCTTC CC-3′, reverse: 5′-CTGTTGTTCAGACTCTCTCCCT-3′. GAPDH forward: 5′-TTTGTGATGGGTGTGAACCA-3′, reverse:5′-GTCTTCTGGGTGGCAGTGAT-3′. The relative gene expression levels were calculated and normalized to GAPDH.
Statistical Analysis
Statistical analysis of data is expressed as mean ± standard error of the mean (SEM). The difference between the two groups was determined by Student’s t-test and between multiple groups was performed using the One-way analysis of variance (ANOVA), respectively using GraphPad Pro 5.0 (San Diego, CA, USA). p values less than 0.05 were considered statistically significant.
Results
Mino promotes nerve function repair in TBI-mice model
Firstly, in the three experimental groups of mice, we used the Garcia neurobehavioral scoring system to explore a possible Mino repair effect on the nerve function. We found that compared to the sham group, the scores of the TBI group of mice were significantly lower, indicating a significant damage to the nervous system function. Interestingly, Mino treatment significantly restored the neurobehavioral activities (Figure 1(a)). Next, to test nerve cell apoptosis mediated nerve function damage, we performed immunofluorescence (IF) and TUNEL assays as shown in Figure 1(b) and (c). We found that the c-caspase3 level was significantly increased in the TBI group of mice. However, Mino treatment did not significantly reduce the level of c-caspase3. Consistent with the IF results, the TUNEL assay showed that Mino treatment did not significantly reduce the nerve cell apoptosis (Figure 1(d) and (e)). Besides, WB was used to detect the levels of apoptosis-related proteins (Figure 1(f) and (g)). The quantitative results showed that compared to the sham group, the levels of c-caspase3 were significantly increased in the TBI group. Here too, after Mino treatment, the levels of the protein did not decrease significantly (Figure 1(f) and (g)).
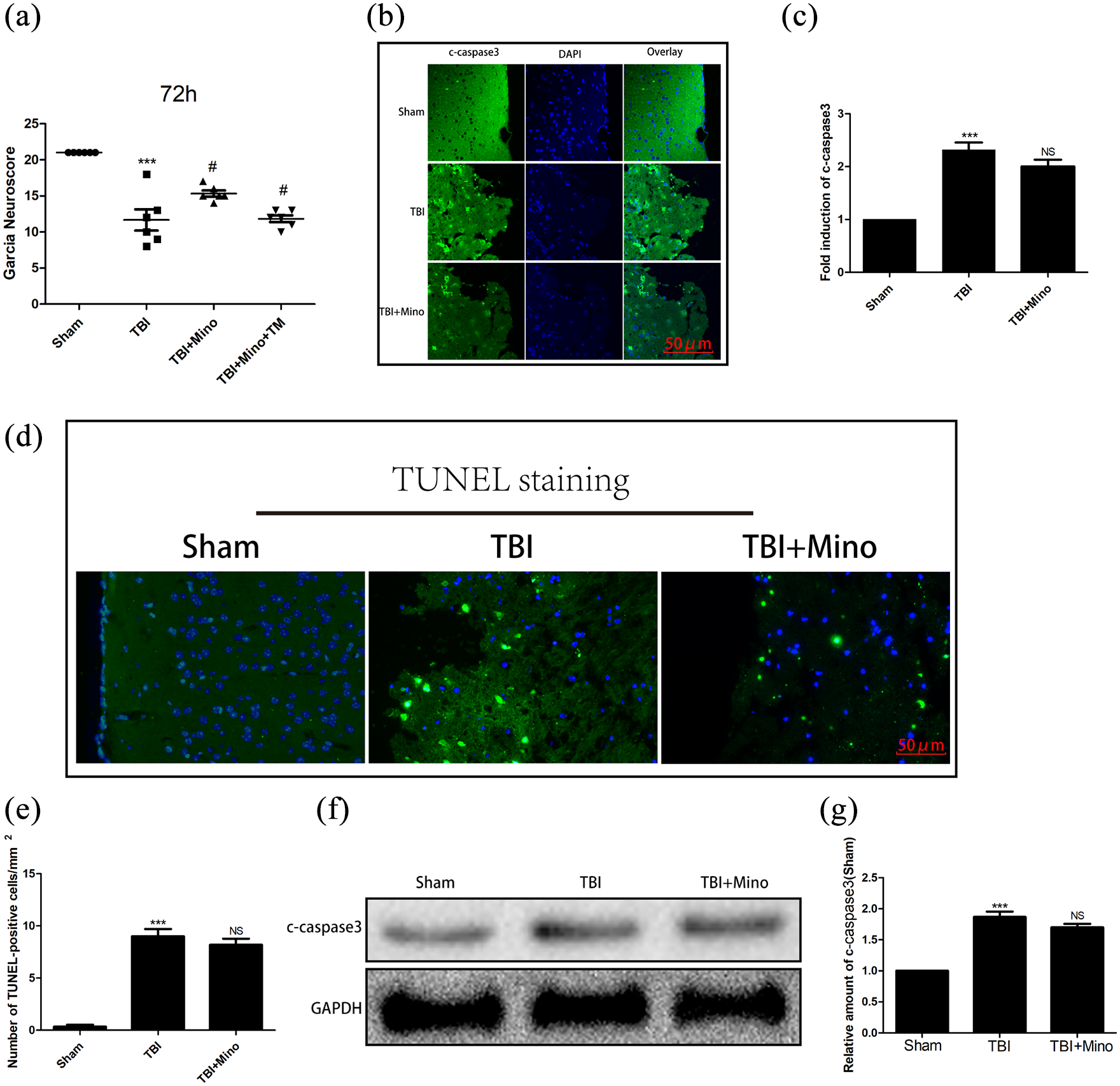
Mino promotes nerve function recovery: (a) Garcia test of three groups of mice at 72 h after TBI (n = 6 per group), (b and c) the expression level and Statistical graph of c-caspase-3 (green) in the brain tissues of the three groups of mice at 3 days post-injury detected by IF (scale bar = 50 μm. n = 5 per group), (d and e) TUNEL (green) staining and statistical graph of the apoptosis level of nerve cells at 3 days post-injury in the brain tissues of the three groups of mice (scale bar = 50 μm. n = 5per group), (f) Western Blotting (WB) was used to detect the expression levels of the three apoptosis-related proteins, representative images are shown (n = 5 per group), and (g) statistical graph of apoptosis-related proteins in the different groups.
Mino promotes the repair of BBB integrity and attenuates TBI-Induced ER Stress
Next, we explored the effect of Mino on the formation of brain edema after TBI. We found that compared to the sham group, the brain water content in the TBI group was significantly increased. However, Mino treatment reduced the water content in the brain tissue (Figure 2(a)). The BBB is composed of tight and direct junctions, maintained by the connexin family of proteins. Therefore, we examined the expression of tight junction (TJ) proteins in the brain tissue. Zonula occludens-1(ZO-1), Occludin, and β-catenin are the key tight junction proteins that maintain the mechanical barrier and permeability of cells. As shown in Figure 2(b), compared to the TBI group, in the Mino group, the fluorescence intensity corresponding to ZO-1 protein was elevated in the brain tissue. Moreover, the WB results were consistent with the IF results showing the increased levels of Occludin and β-catenin after Mino treatment. Glucose-regulated protein 78 (GRP78) and C/EBP-homologous protein (CHOP) are the markers of activated ER stress. We observed that the levels of GRP78 and CHOP were increased after TBI, and were alleviated after Mino treatment (Figure 2(c)–(g)). Notably, TM treatment significantly reversed the protective effect of Mino against TBI and reversed to loss of TJ and AJ proteins.

Mino promotes blood-brain barrier repair: (a) quantification of brain water content of the three groups of mice at 24 h after TBI (n = 6 per group), (b) the expression and statistical graph of Zo-1 (green) in the brain tissue of the three groups was detected by IF at 3 days after injury (scale bar = 50 μm. n = 5per group), (c) WB was used to detect the expression levels of two connexins in the brain tissues of the three groups, representative images are shown (n = 5per group). (d and e) statistical graph of Occludin and β-catenin proteins in the different groups.
Mino reduces the secretion of inflammatory factors
Next, we explored the possible mechanism for the neuroprotective effect of Mino. For this, we examined the expression of two inflammatory factors IL-6 and TNF-α. As shown in Figure 3(a) and (b) after brain injury, the fluorescence signal corresponding to IL-6 and TNF-α increased but Mino treatment significantly weakened the fluorescence signal, thereby lowering the levels of IL-6 and TNF-α. Next, qRT-PCR was used to estimate the mRNA expression of the two inflammatory factors in the brain tissue. Here too, the results showed that compared to the sham group, the mRNA expression levels of the two inflammatory factors in the TBI group increased significantly but Mino treatment reduced the corresponding mRNA levels (Figure 3(c) and (d)). Consistent with the above results, the ELISA results too showed that after trauma, the levels of IL-6 and TNF-α in the brain tissue increased significantly which were recovered by Mino treatment (Figure 3(e) and (f)). Overall, the above results indicate that in TBI mice, the brain tissue produce inflammation, which thereby decreases the nerve function and increases blood-brain barrier permeability. Notably, Mino can improve the recovery in brain trauma by reducing the expression of inflammatory factors.
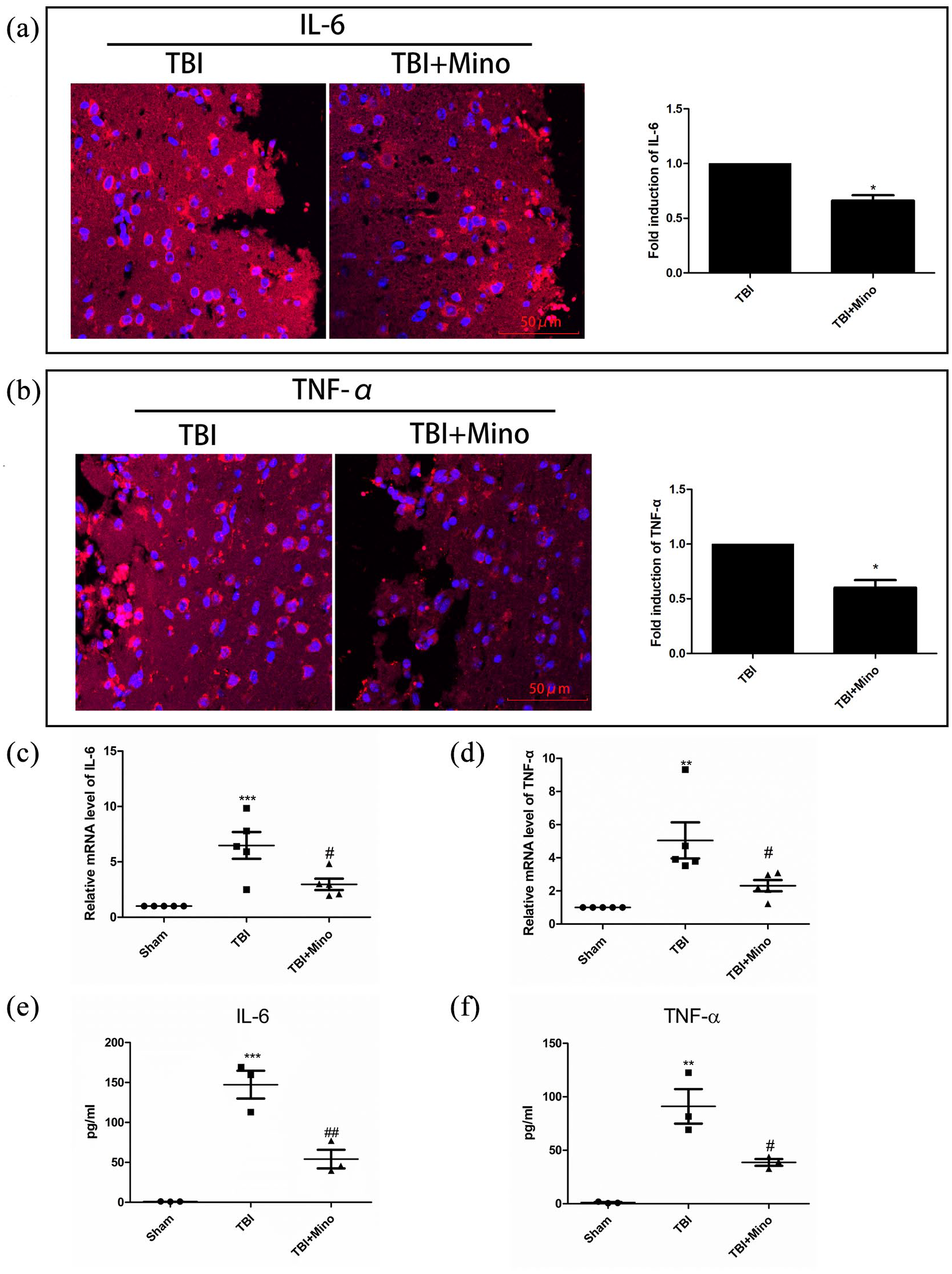
Mino inhibits the secretion of inflammatory factors: (a and b) IF staining and statistical graph of IL-6 (red) and TNF-α (red) expression levels in the brain tissues of different groups of mice (scale bar = 50 μm.n = 5per group), (c and d) relative mRNA expression levels of IL-6 and TNF-α in the brain tissues of three groups of mice (n = 5per group), (e and f) detecting the expression levels of IL-6 and TNF-α in the brain tissues in different groups of mice by ELISA (n = 5per group).
Mino improves cell survival, inhibits inflammatory factors, and promotes connexin expression in vitro
Finally, we tested the above hypothesis at the cellular level. We first pretreated the cells with different concentrations (5, 10, and 20 μm) of Mino for 2 h. Then, the cells were deprived of oxygen and glucose for 6 h and tested for cell viability. As presented in Figure 4(a), compared to the OGD group, Mino treatment prevented cell death in a dose-dependent manner. Importantly, the cell viability increased significantly after the administration of 10 μm Mino which was reversed by TM. Moreover, in the different cell groups, the expression of IL-6 and TNF-α were also detected. We found that oxygen and glucose deprivation upregulated inflammatory factors which was reduced by Mino treatment (Figure 4(b) and (c)). Notably, ELISA results were also in agreement with the aforementioned results (Figure 4(d) and (e)). Next, we performed the IF assay to estimate the level of connexin proteins. The results showed that Mino treatment alleviated the OGD-induced reduction of connexin (Figure 4(g)). Also, GRP78, CHOP, β-catenin, and Occludin were downregulated in the OGD group. Interestingly, Mino treatment upregulated these proteins. However, this effect was weakened by TM (Figure 4(f)–(k)). Overall, in vitro results indicate that Mino has a certain protective effect on the HUVEC cells. This could be the result of Mino mediated inhibition of inflammatory factors and ER stress that improved the BBB.

Mino improves the viability, reduces the expression of inflammatory factors, and increases the expression of connexin in vitro. (a) The cells were pretreated with different concentrations of mino for 2 h and then were deprived of glucose and oxygen for 6 h to estimate the cell viability, (b and c) the expression levels of IL-6 and TNF-α were detected by qPCR in the three groups of cells, (d and e) ELISA was used to verify the expression levels of inflammatory factors in the three groups of cells, (g) Western Blots (WB) and (f, h–j) statistical graph of GRP78, CHOP, Occludin, and β-catenin proteins in the different groups, and (k) detection of β-catenin protein expression in three groups of cells with IF (scale bar = 100 μm).
Discussion
Acute brain injury, a common clinical disease has a very high morbidity and mortality rate. 15 For decades, studies are trying to find targets for post-trauma interventions. 16 Mino, with low toxicity and fast metabolism in the human body, has evolved as one such potential drug for the treatment of acute brain injury. 17 Notably, Mino has been used clinically for more than 30 years. 18 Presently, it is mainly used to treat skin diseases, sexually transmitted diseases, rheumatoid arthritis, and so on. 19 It has the characteristics of easily passing through the BBB. 20 Therefore, many studies have focused on Mino for its neuroprotective effects. In 1998, for the first time, using a mouse model, Professor Yrjanheikki showed that Mino has a protective effect in cerebral ischemia via inhibiting the activity of pro-inflammatory factors. 21 Mino was also found to have an attenuated microglial activation effect on mice after TBI.22,23 Furthermore, the neuroprotective effects of Mino have been studied in multiple neurological disease models. For instance, in multiple sclerosis, matrix metalloproteinases (MMPs) are up-regulated. Under the influence of inflammatory factors, the spinal cord and brainstem produce demyelinating lesions which lead to extracellular matrix deposition. 24 This excessive extracellular matrix cross-linking then triggers the proliferation of glial fibers and the formation of calcification spots, causing multiple sclerosis. Interestingly, Mino can alleviate multiple sclerosis by downregulating inflammatory factors and inhibiting the production of extracellular matrix. 25 In Parkinson’s disease, the substantia nigra striatum pathway is disrupted, and dopamine receptor agonists are one of the popular therapeutic drugs. Notably, a study in the zebrafish model of Parkinson’s disease reported that Mino can produce effects like dopamine. 26 Therefore, we speculated that Mino may have a neuroprotective effect in TBI too. As we expected, in our experiment, Mino improved the neurological behavior score of TBI mice indicating its potential therapeutic role in TBI.
Tight junctions (TJs), a closed structure formed by the fusion of the outer layers of adjacent cell membranes with specific transmembrane proteins, are the main regulators of the BBB. 27 Occludin, one of the key TJ proteins, is directly involved in BBB fence function. 28 ZO-1, another important TJ protein, is uniformly distributed along the cell membrane, and its levels determine the permeability of BBB. 28 β-catenin, an adhesive protein is responsible for BBB stability. 29 After TBI, the immediate damage to BBB increases its permeability for neurotoxic substances and immunological cells, causing serious damage to the nervous system. ER stress is a kind of cellular stress caused by the accumulation of misfolded proteins in the ER. 30 GRP78, a binding protein of ER involved in protein folding and transport, is considered the marker of ER stress. 30 Excessive ER stress activates CHOP, a downstream protein in ER stress-induced apoptosis pathway which triggers apoptosis. 30 A study has shown that excessive ER stress is one of the key features of TBI which can be reduced by Mino. 10 Apart from that, Mino is shown to have a stabilizing effect on the mitochondrial membrane, 31 thereby inhibits the inflammatory factors that activate caspase-dependent and independent apoptotic pathways. Our data, both in vivo and in vitro, show that Mino downregulates apoptosis-related proteins to inhibit apoptosis in nerve cells.
After the brain injury, inflammation is a key event that activates the secondary injury processes. 32 Also, the phenotypic and functional changes in Microglia, a key inflammatory cell in the CNS, complicate the inflammatory response. The Type M1 microglia express inflammatory cytokines such as TNF-α, IFN-γ, etc., while the type M2 microglia express different molecules such as IL-4, IL-10, etc., producing distinct neuroprotective effects. A study, using the in vivo and in vitro models, showed that Mino can inhibit the expression of M1 microglia surface markers and thereby reduces the numbers of inflammatory cells. Furthermore, a study showed that Mino nanoparticles can effectively regulate microglia, reduce the M1 phenotype, increase the M2 phenotype, and reduce the pro-inflammatory response. 33 Besides, Mino has also been shown to affect lymphocytes and inhibit the generation of free radicals. 34 In general, numerous regulatory factors inter-regulate each other to protect the nervous system. Here too, both animal and cell-based studies show that Mino reduced the expression of inflammatory factors, which is also consistent with the afore-discussed scientific reports. Maintaining the BBB integrity promotes central nervous system (CNS) recovery after TBI. Recent studies have been demonstrated that Minocycline Preserves the Integrity of BBB in ICH Model. 33 Thus, we investigated the effect of Mino on the integrity of BBB after TBI. We found that Mino treatment eased the decreases of TJ (β-catenin) and AJ (Occludin) proteins to reverse the increased permeability of the BBB during TBI. Recent studies indicated that restraining ER stress alleviates LPS-induced BBB disruption. 35 Notably, minocycline treatment induces apoptotic effects via regulation of ER stress pathways in photoreceptor cells. 36 Based on these, we speculated that Mino could promote restoration of BBB after TBI or OGD. Our data show that Mino treatment inhibited the activation of ER stress which was reversed by TM. Nonetheless, this study has some limitations. For example, Mino treatment including various duration (2–4 weeks) and doses must be tried to confirm its effects on autophagy and immunomodulatory properties. That may strengthen Mino protective effects against TBI.
Conclusions
In conclusion, through a series of in vivo and in vitro studies, we show that Mino can improve neurological function by protecting the integrity of the BBB and downregulating inflammatory factors such as IL-6 and TNF-α. Moreover, our data demonstrate that Mino treatment rescue the BBB disruption from TBI via inhibiting ER stress, our findings provide the basis to use Mino as a potential therapy to treat TBI-induced brain injury or other similar nervous system diseases.
Footnotes
Acknowledgements
The authors are thankful to Hui ya Huang (Wenzhou Medical University) for technical assistance on the Real-time quantitative PCR.
Animal welfare
The present study followed Institutional Animal Care and Use Committee (IACUC) of Wenzhou Medical University for humane animal treatment and complied with NIH guidelines.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding from the program of Wenzhou Municipal Science and Technology Bureau (No. 2016Y0394).
Ethical approval
The present study followed Institutional Animal Care and Use Committee (IACUC) of Wenzhou Medical University (approval no: wydw2016-0079) for humane animal treatment.
Ethical approval
Ethical approval for this study was obtained from the Institutional Animal Care and Use Committee (IACUC) of Wenzhou Medical University (approval no: wydw2016-0079).
