Abstract
Thermal threshold testing is commonly used for pain research. The stimulus may cause burning and merits prevention. Thermal probe modifications hypothesized to reduce burning were evaluated for practicality and effect. Studies were conducted on two humans and eight cats. Unmodified probe 0 was tested on two humans and promising modifications were also evaluated on cats. Probe 1 incorporated rapid cooling after threshold was reached: probe 1a used a Peltier system and probe 1b used water cooling. Probe 2 released skin contact immediately after threshold. Probe 3 (developed in the light of evidence of ‘hot spots’ in probe 0) incorporated reduced thermal mass and even heating across the skin contact area. Human skin was heated to 48℃ (6℃ above threshold) and the resulting burn was evaluated using area of injury and a simple descriptive scale (SDS). Probe 1a cooled the skin but required further heat dissipation, excessive power, was not ‘fail-safe’ and was inappropriate for animal mounting. Probe 1b caused less damage than no cooling (27 ± 13 and 38 ± 11 mm2 respectively, P = 0.0266; median SDS 1.5 and 4 respectively, P = 0.0317) but was cumbersome. Probe 2 was unwieldy and was not evaluated further. Probe 3 produced even heating without blistering in humans. With probe 3 in cats, after opioid treatment, thermal threshold reached cut-out (55℃) on 24 occasions, exceeded 50℃ in a further 32 tests and exceeded 48℃ in the remainder. No skin damage was evident immediately after testing and mild hyperaemia in three cats at 2–3 days resolved rapidly. Probe 3 appeared to be suitable for thermal threshold testing.
Pain relief is an essential component of treatment for injury and disease; study of the applied pharmacology of analgesic drugs is required so that optimal treatments can be devised. It is particularly important that appropriate pain relief is provided for laboratory animals undergoing painful intervention. All analgesic drugs have side-effects and cannot be used indiscriminately. Those most appropriate for the prevailing conditions should be documented; and knowledge of their efficacy, safety and duration of effect can optimize pain relief in each species and circumstance.
Nociceptive threshold testing (NTT) is widely used in studies investigating pain and its treatment. This applies both when a laboratory species is used as a model for humans and when a potential analgesic is under investigation for therapy in the target species. Pain is a complex phenomenon involving both sensory and affective components, so investigation into its mechanisms requires that pain is inflicted during the study, and NTT commonly fulfils this requirement. Analgesic treatment generally increases NT, 1 thereby allowing assessment of the drug's behaviour under controlled laboratory conditions without confounding from the inevitable variability encountered in clinical studies.
A variety of stimuli have been used for NTT. Of necessity they must cause pain, but, as described by Beecher, 2 the intensity of the stimulus should be related to the perceived pain intensity and should not result in lasting tissue damage or harm to the animal. The stimulus must also be repeatable, reliable, and easy to apply, and must result in a clear endpoint.
Thermal stimuli are commonly used in NTT. Such stimuli may, however, cause local damage, which if serious, makes the technique unacceptable. In some studies3,4 attempts to reduce burn injury have led to the use of a low maximum ‘cut-out’ temperature if there is no response. Lowering the maximum temperature reduces the ability to detect an increased threshold, and may make the investigation worthless. It is essential that animals used in research are not wasted; any refinement which maximizes data robustness is worthwhile.
This paper reports refinements made to thermal NTT probes to allow sufficient ‘headroom’ above the baseline thermal threshold to reduce burning without compromising data collection. The original probes have been widely used in several species, and are well tolerated.5–8 However, the high thresholds resulting from treatment with potent analgesics have led to skin burns, even with the cut-out reduced to 55℃.9,10
We hypothesized that reducing heat transfer to the skin should reduce burning and aimed to evaluate two methods of reducing skin damage: either by cooling the thermal probe after threshold is reached, or by decreasing the subsequent heat transfer by reducing probe skin contact. We hypothesize that either of these systems should reduce skin damage from prolonged contact between the probe and skin at threshold temperature (TT). A third probe design was also evaluated in the light of information collected during studies of the first two. The investigation was conducted after approval by the local institutional review board, and the investigations in cats were conducted under the Animals (Scientific Procedures) Act 1986. The human studies were conducted on two of the authors.
Materials and methods
Original equipment
A heater probe (probe 0) used for thermal NTT in cats
9
was evaluated prior to and after a number of modifications. Probe 0 had been developed from the 9 × 8 × 5 mm probe (heating contact area 72 mm2) originally designed for thermal NTT in cats
11
and used in numerous feline studies12–16 (Figure 1). Probe 0 was smaller, and therefore had lower thermal mass than the original design. It contained the resistor heating element embedded in a minimal volume of thermally conductive epoxy with an adjacent temperature sensor, resulting in a 39 mm2 skin heating contact area (Figure 2). Probe 0 weighed less than 5 g and heated at 0.6℃/s. The probe was connected to the control unit by a five-way ribbon cable, and incorporated a safety cut-out at 60℃
11
which was subsequently reduced to 55℃.
14
For thermal threshold testing in animals the probe was attached to a modified blood pressure bladder and held in place on the shaved thorax by an elasticated surcingle. The bladder was inflated manually to 100 mmHg to provide reproducible contact between the probe and the skin. A handheld switch activated the probe heater, and TT was recorded and held on the display when the animal reacted visibly to the stimulus. Simultaneously, the heater was switched off.
Probe 0 incorporating heater and sensor embedded in a minimal quantity of thermally conductive epoxy reducing the heating contact area to 39 mm2.

Pilot studies
Pilot studies were conducted on the forearms of two of the authors (MJD and PMT) using probe 0. For human testing the probe was mounted on a handheld pneumatic mechanical actuator (ProdPlus; Topcat Metrology Ltd, Ely, UK) in order to maintain constant contact with the skin. The heater was held against the hairless skin of the ventral antebrachium at 1.5N. Thermal nociceptive threshold (the first point at which the sensation was painful) was measured in both individuals as the mean of three tests taken at 5 min intervals at adjacent sites on the antebrachium. To examine each test probe's characteristics heating was continued 6℃ beyond threshold to a reading of 48℃ and was then switched off. Skin damage was evaluated by visual inspection 20 h after all the tests.
Test probes
Three fundamental probe modifications were investigated. All systems were first evaluated on the bench to examine heating and cooling characteristics and assess potential for use in vivo. The internal probe sensor was used to track temperature and recorded using a data logger sampling at 10 Hz.
(1) Cooling of the probe immediately after each test
Probe 1a
Rapid cooling of the probe was achieved by mounting a series of two Peltier heat pumps (9 × 9 × 3.4 mm, cat no 189–1576; 30 × 30 × 3.5 mm, cat no 693–7040; RS Components Ltd, Corby, UK) immediately behind the thermally conductive epoxy layer that surrounded the heating element. The pump was switched on at TT to conduct heat away to a heat sink. The heat sink, a 50 × 25 × 1 mm copper plate, was attached behind the Peltier pump.
Probe 1b
An alternative water-cooled probe was developed in which the heating element was placed inside a 9 mm long brass tube (ID 2.4 mm and OD 3.2 mm). A second brass tube of the same dimensions was mounted in parallel (soldered) and carried water from a 2 L reservoir containing ice and water mounted 60 cm above the assembly. Gravity-driven water flow from the reservoir was controlled by a roller constrictor (Vygon infusion set, model 027421; Vygon Vet, Swindon, UK) at 170 mL/min. Flow rate was calibrated by measuring the volume delivered over one minute. When heating was stopped, a tap was immediately opened manually allowing iced water to flow through the tube parallel to the heater and cool the probe (Figure 3).
Water-cooled probe 1b. The heater was placed inside a brass tube and a second brass tube was mounted alongside, carrying iced water flowing at 170 mL/min to cool the probe after heating.
(2) Release of the probe pressure on the skin immediately after the endpoint
Probe 2
A vent valve (Bio-Chem part no P/N075T2NC12/32M; Biochem-Fluidics, Cambridge, UK) was incorporated into the air line supplying the pressure bladder holding the thermal probe against the skin. The valve was activated automatically when the heating was stopped at TT, thereby reducing probe–skin contact. Once the probe had cooled to room temperature, the bladder was re-inflated for the next test.
(3) Reduced thermal mass with even heating
Probe 3
Probe 3 was developed in response to the pilot studies conducted on the authors' forearms which suggested that heating from probe 0 was uneven (Figure 4). This was further supported by evidence of even heating in the non-cooled tests with probe 1b. For probe 3, the heating element was encased in an 8 mm long brass tube (ID 2.4 mm and OD 3.2 mm) to produce even heating without any hot spots (Figure 5). The thermal mass of the heating components was less than in probe 0. The brass tube was positioned behind low heat conducting plastic material with 0.4 mm depth exposed, giving a heating contact area of about 20 mm2 when the plastic was held on the skin surface with the contour of the exposed brass tubing depressing the skin.
Blister (approximately 1 mm diameter) produced on human skin within the heating contact area of probe 0 heated to threshold (42℃). This provided evidence of uneven heating and ‘hot spots’ at temperatures higher than recorded by the sensor. Black square indicates heating contact area. Even heating, low thermal mass probe 3. The heater is placed inside a brass tube which protrudes 0.4 mm above the low thermal conductivity plastic shielding. The plastic is flat against the skin maintaining consistent contact between the heating bar and skin. Heating contact area 20 mm2.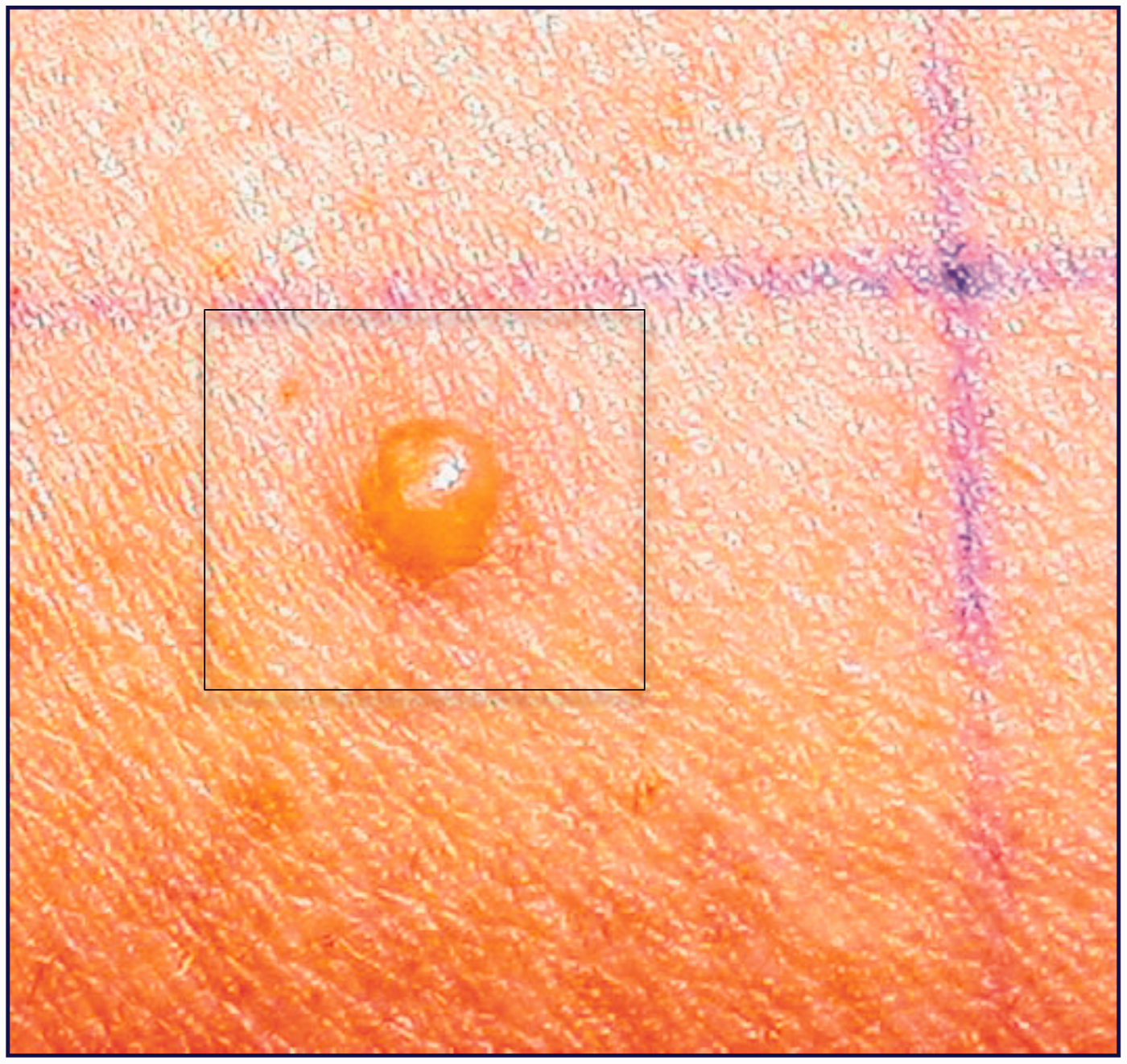

Human testing
Numerical Rating Scale scoring system for skin damage.

Probe 1a
The Peltier pump-cooled probe, although effective on the bench, proved inappropriate and was not tested further.
Probe 1b
The water-cooled probe was tested in 24 randomized experiments (2 subjects, 2 limbs, 6 tests per limb): on each individual, on adjacent sites on both forearm and leg, three cooled to 9–10℃ immediately after heating to 48℃, and three not cooled, where the probe was held on the skin without water flow for a further 2 min after heating was stopped.
This cooling method was considered to be potentially effective for large animals but was not tested further on animals in this investigation because, with the water reservoir and substantial tubing for sufficient water flow, it appeared too cumbersome for use in cats.
Probe 2
The pressure relieving method was also effective on the bench but was cumbersome and bulky, again unsuitable for mounting on an animal. Hence no further pressure experiments were conducted.
Probe 3
The low thermal mass probe was compared with the original in 24 randomized experiments as above: three at each site on each individual with each probe. Each probe was heated to 48℃ (6℃ above threshold) and the test subject was blinded to the identity of the probe. Skin damage was described by the SDS scoring system (Table 1) and the area was measured 20 h later.
Cats
Eight purpose-bred neutered adult domestic shorthair cats (2 male and 6 female) were studied. They were housed in groups of four in accordance with Home Office (UK) regulations. Testing was carried out on two cats at a time, in a separate room where they were housed individually within sight of each other in large cages. Cats were taken to this room the day before the study to become accustomed to the new environment, and were returned to their long-term housing after testing was complete. A minimum two weeks' rest period was allowed between any repeat testing on the same animal.
Only the most promising probe modification was tested on animals. Hence the low thermal mass, uncooled tubular probe 3 (Figure 5) was studied in eight cats. The probe was used in a wireless threshold testing system operated by infrared control which allowed the animal free movement during testing and facilitated detection of a natural behavioural endpoint when TT was reached 17 (WTT1, Topcat Metrology Ltd, Ely, UK).
The probe was placed on the shaved skin of the thorax as previously described11,12,18 and heated at 0.8℃/s until threshold was reached, detected by the cat's response, generally turning to the site, jumping forward, twitching the skin or occasionally vocalizing. Three pretreatment tests were performed at 10 min intervals to confirm normal responses. Buprenorphine (20 µg/kg) or fentanyl (20 µg/kg) was then injected intravenously and threshold was measured eight times at 30 min intervals. The probe was not moved between tests and the skin was allowed to return to pretesting temperature without active cooling or removing the thoracic band. The skin was examined for any evidence of heat damage immediately, at 2, 15 and 24 h, and then daily for three days after the last test.
Historical data (unpublished) from four of the same eight cats, tested six months previously after similar opioid treatment using probe 0, were examined for descriptive comparison of skin damage to confirm published experience of the extent of post threshold testing skin burns in cats.
Data analysis
Human data from cooled and not cooled burn sites were compared using Student's t-test for normally distributed data (area) and Mann–Whitney U-test for SDS. P < 0.05 was considered significant. No formal analysis was applied to the cat data.
Results
Pilot studies
Probe 0
Probe 0 produced blisters of 1–3 mm diameter at TT (42℃) indicating that the heater produced ‘hot spots’ where the skin reached temperatures considerably higher than those recorded by the sensor (Figure 4).
Human testing
Probe 1a
The heat was removed from the probe itself, but still needed dissipation from the site. This required a considerable bulk of equipment that was unsuitable for mounting on an animal. In addition, since Peltier heat exchangers are inefficient and consume a great deal of power, this proved too much for battery-driven equipment, further preventing the mounting of the system on the animal, or the use of light, flexible cables. A third disadvantage was that the system is ‘fail-dangerous’: if power is lost the extracted heat returns very quickly, leading to a rapid increase in temperature and a significant risk of serious burns. This was inappropriate for further testing.
Probe 1b
The ice water-cooled probe resulted in a significantly smaller area of damaged skin than when not cooled (27 ± 13 and 38 ± 11 mm2 respectively, P = 0.0266) (Figure 6). The SDS scores were lower with the cooled probe than the not cooled (median 1.5 and 4 respectively, P = 0.0317).
Effect of ice water cooling: burns on human skin after heating to 48℃ (6℃ above thermal threshold). Sites 2, 4, 6 were cooled with iced water flowing through a brass tube adjacent to the heating surface immediately after threshold was reached. Sites 1, 3, 5 were not cooled.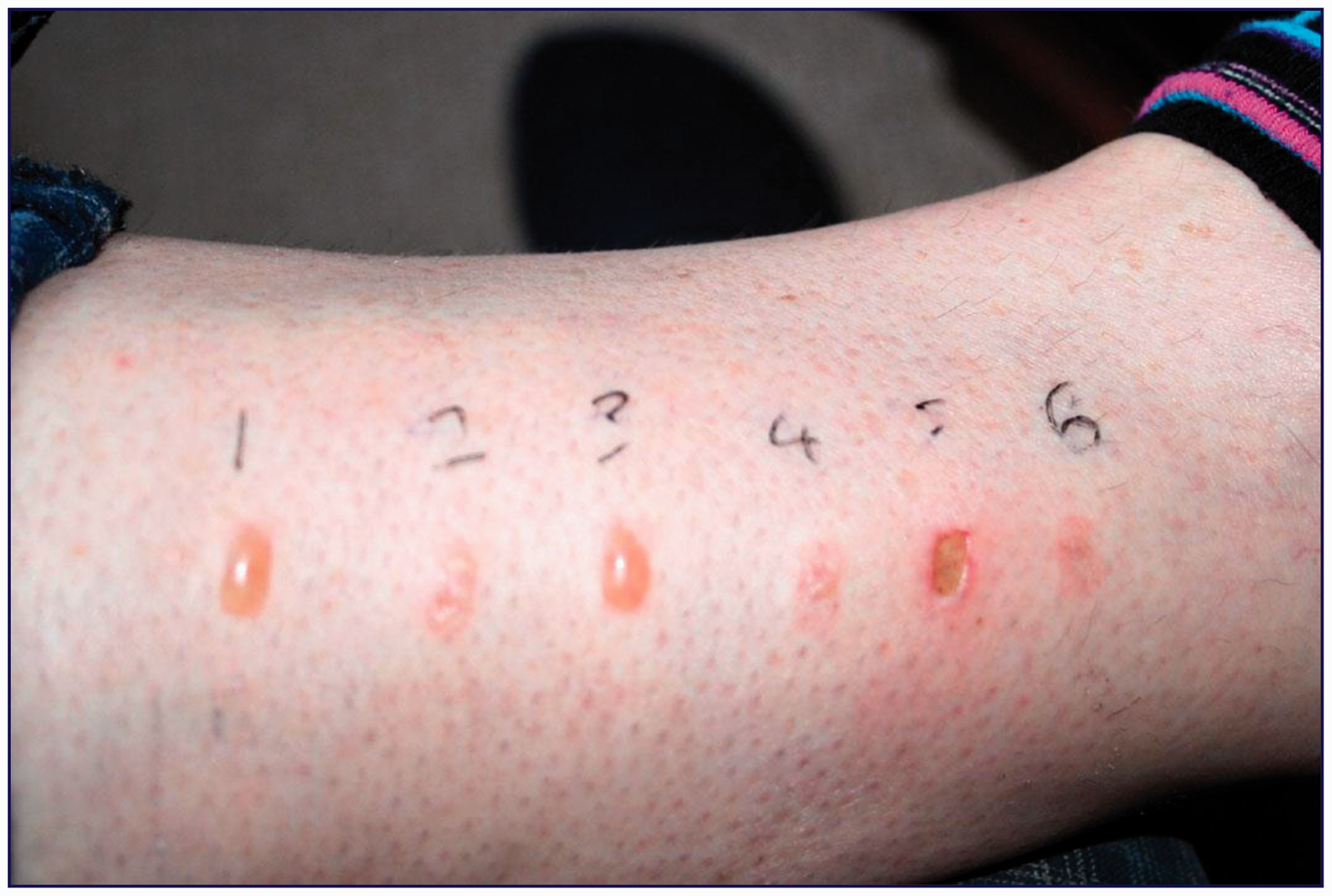
Probe 2
Probe 2 proved too cumbersome for mounting on animals and was not evaluated further.
Probe 3
The low thermal mass/even heating probe (Figure 5) produced skin reddening after heating to 48℃ over an area of the same shape and size as the probe footprint and with no blistering, indicating even heating across the sites with no evidence of ‘hot spots’.
Cats
Probe 3
In cats the mean ± SD skin temperature was 37 ± 0.4℃ before testing. Mean ± SD pretreatment thermal threshold was 43.5 ± 2.0℃. Post opioid administration, all eight tests went to cut-out (55℃) in three cats and all eight tests were above 50℃ in six cats. In the remaining two cats, one and four tests respectively exceeded 50℃, and the rest exceeded 48℃. Immediately after testing the imprint of the probe was evident, confirming the testing site, but there was no skin damage (Figure 7). There was no sign of damage at 2, 15 and 24 h, but three days after testing one cat had a mildly reddened area at the site of the brass tube heating element (Figure 8). Also at 24 h, two other cats had similar reddening in an oval shape that did not clearly correspond with the probe site. These marks were entirely ignored by the individual cats and did not elicit any signs of pain or discomfort on palpation. They resolved completely within a few days.
Immediately after testing, the imprint of the probe was evident, confirming the testing site, but there was no skin damage. Lesion on cat skin three days after eight threshold tests reached cut-out (55℃). The probe outline (black marker pen at testing, black arrows) is faint, the red mark (white arrow) is the heating surface contact area. The white marks are old scars present before testing.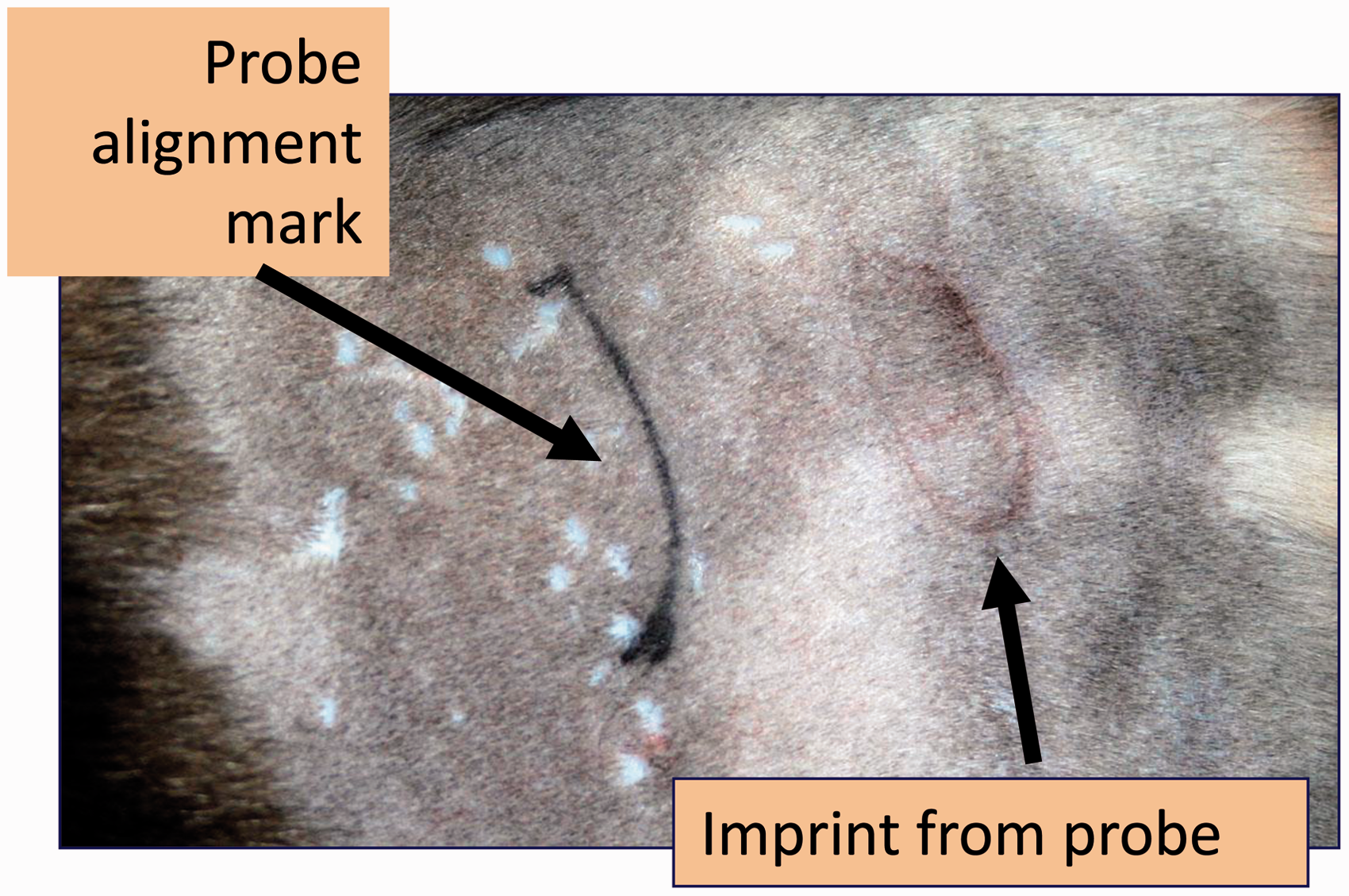
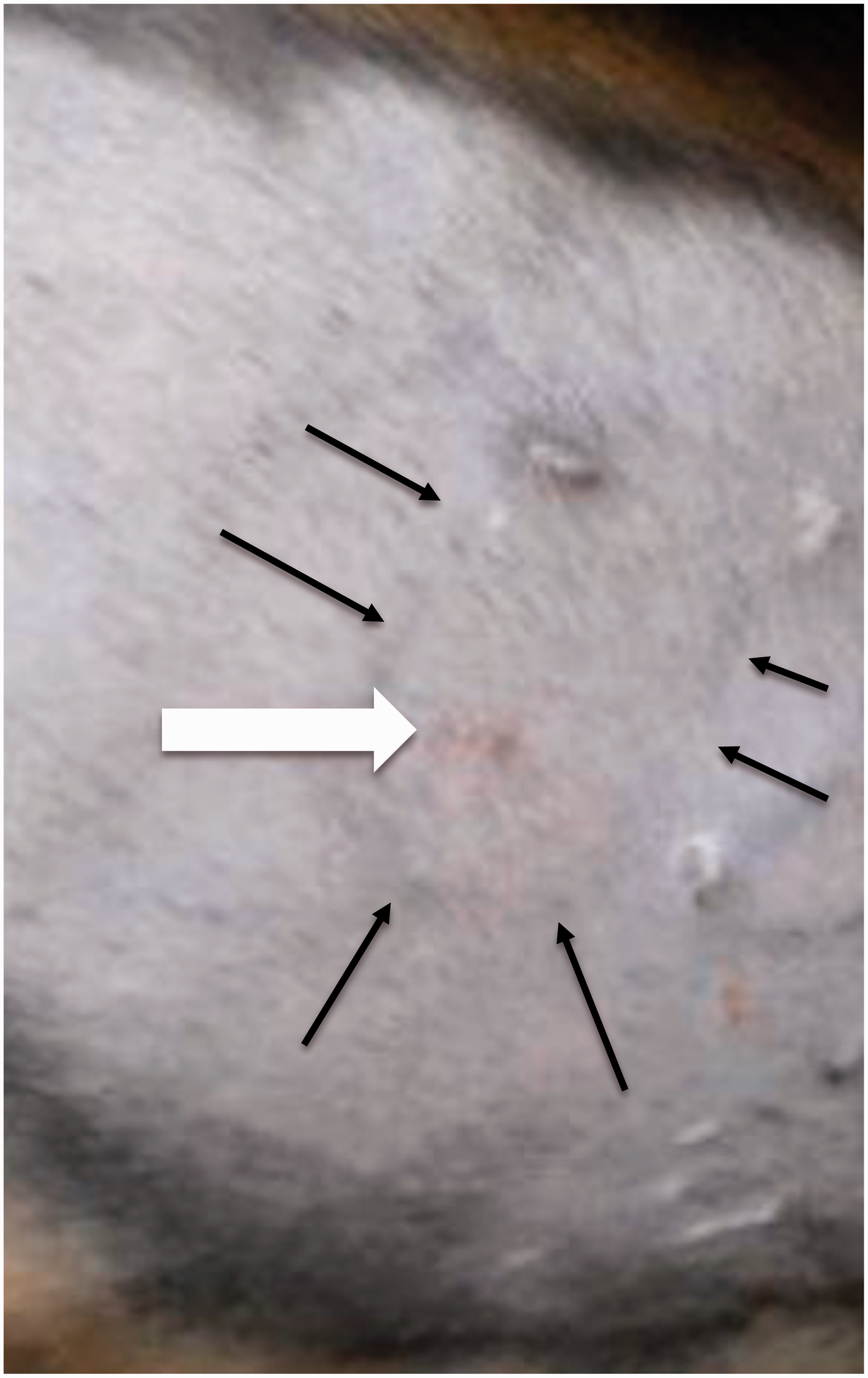
Historical data using probe 0 in four of the eight cats recorded that, after opioid treatment, cut-out (55℃) was reached in 2/8 tests in only two cats. In one cat threshold never exceeded 44℃, and overall, 8/32 tests exceeded 50℃ and a further seven exceeded 48℃. No skin damage was detected on the cat with thresholds <44℃, but 2–3 circular damaged areas were detected at the probe site in the other three cats. These resolved over the following few days and did not appear to cause discomfort.
Discussion
Thermal NTT has become a well accepted laboratory method of assessing the likely therapeutic efficacy of an analgesic. 1 Studies in cats have also demonstrated that increased TT appears to correlate with clinical perception of the efficacy and duration of opioid analgesics. This method of analgesiometry is well validated5,9,13,14,16,19–21 and is now accepted for assessment of efficacy prior to analgesic drug registration. 22 Early laboratory studies of buprenorphine in cats contributed data used in the successful application for buprenorphine UK Market Authorization (MA) for feline use in 2005 (Vetergesic®; Alstoe Animal Health, York, UK) and this analgesic has had considerable impact on feline clinical pain management.23–25 Since evaluation of analgesics is a major application for NTT it is inevitable that the stimulus will frequently reach temperatures higher than the normal threshold and may burn the skin. Any improvements made to this methodology which reduce the heat transfer and subsequent injury has the potential for refinement affecting numerous laboratory animals.
We have demonstrated that adaptations can be made to the heating probe which reduce skin damage. This was effective under conditions occurring during laboratory testing of the most potent class of analgesics, the opioids, when TT may be well above normal for several hours.5,12,20
The Peltier heat pump was effective in rapidly cooling the probe after heating. However it proved to be impractical for use with a probe designed to be attached to the subject. The process depends on a relatively inefficient use of power (http://en.wikipedia.org/wiki/Thermoelectric_cooling), and it proved to be impossible to run on batteries small enough to be mounted on the animal. In addition, removing the heat rapidly from the site requires a very large heat sink which itself needed cooling. All this was too bulky for small animal use. Commercially available devices which use Peltier heat pumps to provide hot and cold stimuli such as the handheld Physitemp (http://www.physitemp.com) and the Medoc TSAII Neurosensory Analyser (http://www.medoc-web.com) incorporate a substantial probe connected to a large and bulky mains-powered control box via a thick, stiff cable which is required for power and water cooling. These are impractical for attachment to an animal and are unsuitable for use on unrestrained subjects. The final deciding feature which made this method unsuitable for further testing was its ‘fail-dangerous’ nature. It is essential that a stimulus probe fixed on the subject ‘fails-safe’ if power is lost for any reason. After a Peltier pump has withdrawn heat, and if power is lost the heat returns very rapidly, causing significant burning; which is clearly unacceptable for a remote-controlled probe attached to an animal.
A notable result from this investigation was confirmation that rapid cooling after threshold is reached is of considerable benefit in preventing skin injury. The iced water system was effective in cooling the skin and reducing heat injury; and although this is cumbersome for small animals such as cats and dogs, it has potential in larger species. Horses are often tested in stocks 26 using equipment incorporating cables connected to bench top control units. Under these conditions the addition of an ice water reservoir and tubing would be quite feasible.
Heat transfer is influenced by probe pressure and its effect on skin contact. Reducing the contact between the probe and skin at threshold, so that no further heat is transferred to the skin should therefore reduce the transfer of remaining probe heat to the skin. Although the prototype system was effective in releasing the pressure at threshold, the valve and its connections were too heavy and bulky for use on a small animal such as the cat. Even with a larger animal the overall benefit of the additional complexity was questionable. However, this approach still merits further evaluation.
The tubular probe 3 allowed testing to threshold during opioid treatment in cats with virtually no subsequent skin damage. Although the burns encountered with the original 9 × 8 × 5 mm probe first described 11 were not severe or extensive, and did not appear to cause discomfort,9,10 entirely preventing any burning would clearly be worthwhile. The heating contact area and the thermal mass of probe 0 were less than those of the original probe, but minor skin lesions were still reported. 9 In probe 3 the thermal mass and heating contact area were further reduced. Reduction of the thermal mass in contact with the skin could be expected to reduce heat transfer to the skin; the maximum temperature reached is unchanged, but the probe cools faster after the stimulus is switched off. The value of reducing the thermal mass was not formally evaluated in the present investigation, but fundamental physical principles suggest that it should have some benefit. Burn injury relates to the amount of heat energy transferred to the skin. Thermal mass is an indication of the amount of heat the probe can store and is therefore available for transfer. Thermal mass depends on the size of the object and its specific heat capacity. Specific heat capacity is a property of the material, so a larger probe (of the same material) has greater thermal mass than a smaller one and therefore also provides more heat energy available for transfer.
It is likely that even heating across the whole contact area contributes substantially to preventing skin burns by eliminating ‘hot spots’of higher temperature than the sensor's readout. Although the temperature reached at the hot spots was not precisely known, it must have been higher than the displayed temperature as less than one minute at 42℃ is very unlikely to cause damage. 27 The lesions seen with probe 0 both on human skin (Figure 4) and in cats used in previous studies were small spots in the centre of the probe contact area where the maximum heat is generated by the resistor. Although embedding the sensor and resistor in thermally conductive epoxy facilitates heat transfer, the temperature across the probe surface is still not uniform. Skin in contact with the centre of the resistor will reach a higher temperature than the sensor separated from the resistor by epoxy. Placing the resistor inside brass tubing ensures consistent heat transfer to both skin and sensor, as thermal conductivity of brass is much greater than the epoxy. Hence the temperature across the probe 3 contact area was consistent and was more accurately recorded by the sensor. A recent report of thermal NTT in cats may support this view: 28 two cats treated with tramadol and morphine developed minor skin lesions a few days after testing even though the peak mean threshold was only 45℃. It is plausible that the probe may have produced hotter areas than detected by the sensor. The historical data from probe 0 presented here may be an additional reflection of this. Most of the cats experienced some burns with probe 0, in spite of fewer post opioid tests reaching cut-out (55℃) than with probe 3. This may be a result of small areas of skin reaching temperatures higher than those detected by the sensor, and triggering a response. In addition, the baseline feline threshold recorded with probe 3 (43.5 ± 2.0℃) was higher than that in older published studies using probe 0 or earlier designs (40–42℃).15,16,20,29 These observations support the concept that uneven heating leads to ‘hot spots’ above the displayed temperature. This may not only cause burns but may also lead to TT recorded at lower values than reality.
A higher heating rate was used with probe 3 than that employed previously with both the original probe and probe 0. The effect of heating rate on burning was not examined in this investigation. From fundamental principles, as the area under the temperature–time curve (AUC) is smaller, a higher heating rate is more likely to decrease heat transfer and the likelihood of burning. This suggests that the AUC should be kept as low as possible. Even relatively low temperatures (44℃) for a long period of time (5 h) caused burns; 27 a slower rise in temperature will tend to increase the risk of burning as the AUC is larger. This is difficult to study on the bench as the effective heating rate can only be measured when the probe is heated in situ on the subject: heat transfer from the probe and hence its actual temperature during heating are affected by the thermal conductivity of its surroundings. Heating rate is faster in air than on tissue, as air is a relatively effective insulator. Various adjustments in heating rate and probe design have been made in previous investigations, particularly when adapting the system to new species; horses appeared to burn easily. 30 Reducing the heating rate appeared to reduce skin injury in horses, but this was already minimal in the study reported. 30 It is also recognized that fast nociceptive stimulus ramps lead to higher thresholds primarily because the stimulus overshoots further after the control is switched off. 31 The effect of heating rate on tissue injury merits further investigation.
The fundamental approaches to reducing skin damage were first evaluated on ourselves, thereby including subjective sensation as well as avoiding unnecessary use of animals. We studied cats to test the most promising probe design as there is a considerable body of information from feline thermal NTT. Cats are generally regarded as a difficult species for NTT, due to their size and temperament, so we hypothesize that equipment they tolerated is likely also to be suitable for other species. However this approach prevented further evaluation of the water-cooled system.
In conclusion, the low thermal mass, even-heating probe appears to offer the best features of those evaluated, being the most practical for mounting on an unrestrained small animal. Water cooling was effective in cooling the probe and reducing the potential for skin injury, but is only suitable for larger animals. The even-heating non-cooled design is now used in thermal threshold testing equipment supplied by Topcat Metrology Ltd.
Footnotes
Funding
This investigation was supported by a grant from the National Centre for Replacement, Refinement and Reduction of Animals in Research (Grant/Award Number: ‘Taylor.P.22102008’).
Conflict of interest statement
PM Taylor and MJ Dixon are directors of Topcat Metrology Ltd.
