Abstract
Reported analgesic use following experimental surgery is low in rodents and there has been little published information on the frequency of analgesic use in other laboratory species. A structured literature review was conducted to examine analgesic administration in larger laboratory animals. The Scirus search engine was used to identify studies published in peer-reviewed journals that reported carrying out experimental surgery on ‘large’ laboratory animals, specifically rabbits, pigs, sheep, dogs and non-human primates. Seventy-four studies between 2000 and 2001 and 75 studies between 2005 and 2006 were included in the review. There was an increase in the reported administration of systemic analgesics to these species from 50% in 2000–2001 to 63% in 2005–2006. When all agents with analgesic properties were considered (systemic analgesics, local anaesthetics and anaesthetics with analgesic components), the proportion of papers that reported some form of analgesic administration to ‘large’ laboratory animals increased from 86% in 2000–2001 to 89% in 2005–2006. Overall rabbits, pigs, sheep, dogs and non-human primates were more likely to receive analgesics following potentially painful experimental procedures than has been reported in laboratory rodents but analgesic administration to ‘large’ laboratory species is still not optimal.
Administration of systemic analgesics to laboratory rodents is likely to be low. Stokes et al. 1 reported that only 20% of studies describing surgical procedures on rodents, published between 2005 and 2006, specify the administration of a systemic analgesic agent. Little is known about the frequency of analgesic administration in other laboratory species but there is some evidence that larger laboratory animals may be more likely to receive postoperative analgesia than laboratory rodents. In a survey of UK scientific establishments, it was noted that there was variation in policies on postoperative pain relief to laboratory animals, particularly with respect to the provision of analgesia for different species. 2 In some establishments, brief records of clinical signs of pain were kept for rodents, whereas detailed descriptions were kept for the larger animals. It was hypothesized that this may be due to ‘speciesism’ possibly because larger laboratory animals such as dogs and non-human primates may be favoured by animal care staff because they tended to be housed in animal units for longer periods of time. 2 Certainly, the general public is more concerned about the use of some laboratory species than others and acceptance of the use of animals in scientific procedures depends largely on the species being used. There is considerably more support for the use of mice compared with monkeys (e.g. MORI 3 ). Public opinion regarding the use of animals in research is also influenced by whether the procedures cause pain or distress. 4
In companion animal veterinary practice, some species are more likely to receive analgesia than others, specifically dogs seem to be more likely to receive analgesics than cats. 5–7 Small mammals, including rabbits, ferrets and rodents are even less likely to receive analgesics in companion animal veterinary practice than cats. Only 20% of small animal veterinary practitioners routinely administered analgesics to small mammals and this may be due to difficulty in recognizing pain in small mammals and lack of knowledge concerning suitable drugs. 6
To investigate analgesic administration to dogs, pigs, non-human primates, rabbits and sheep following experimental surgery, and to determine whether species differences in analgesic administration to animals used in scientific procedures exist, a literature review of studies published between 2000 and 2001 and from 2005 to 2006 was carried out. The aim of the review was to assess the frequency of use of analgesics, the agents used and to determine if the use of analgesics was related to the severity of the experimental procedure.
Materials and methods
Search strategy
The Scirus search engine (
Inclusion criteria
A paper was eligible for inclusion if it involved the use of a ‘large’ laboratory animal in an experimental surgical procedure under general anaesthesia with a postoperative recovery period of at least 24 h. All papers included in this review had methods that were described in detail. Papers that: (i) described multiple studies on different species of animals, (ii) described fetal surgery, (iii) described neuropathic models of pain and (iv) stated the efficacy of analgesia following surgery as the purpose of the study was excluded. Review articles, abstracts, letters and meta-analyses were also excluded. When multiple papers from a single group of authors described a similar series of procedures, only the first paper listed by Scirus was included in the study.
Classification
The classification of the experimental procedure was based on criteria used in our earlier literature review. 8 Each paper was classified into one of six categories: skin incision, craniotomy, laparotomy, burn study, thoracotomy or orthopaedic study. Thoracotomies and orthopaedic procedures were considered to be the most potentially painful procedures, laparotomies and burn studies were considered to be slightly less potentially painful, and skin incisions and craniotomies were considered to be the least potentially painful procedures. If multiple procedures were described in one paper (such as a blood vessel cannulation followed by a thoracotomy), the paper was classified on the basis of the more potentially painful procedure.
Anaesthetic and analgesic regimens used were also noted. Classification of systemic analgesics was based on Lamont and Mathews, 9 where opioids and non-steroidal anti-inflammatory drugs (NSAIDS) were classified as systemic analgesic agents. The dose rate, duration, frequency and time of analgesic administration were noted when specified. The use of local anaesthetic agents (when specified) was also noted. Animals anaesthetized with dissociative anaesthetic agents (ketamine or tiletamine) and/or alpha2 agonists (xylazine or medetomidine) were classified as having been anaesthetized with an ‘anaesthetic agent with analgesic properties’. Preanaesthetic medications (for example sedatives) were considered as anaesthetics for this classification. The number of animals that underwent surgery (‘study size’) was also recorded.
Statistical analysis
All statistical analyses were conducted using SPSS software (SPSS 14.0 statistical package for Windows; SPSS Inc, Chicago, IL, USA). Chi-squared analyses were used for all comparisons except to analyse ‘study size’. A Mann-Whitney test was used to compare ‘study size’ between time periods. Within time periods, Kruskal-Wallis tests were used to compare ‘study size’ between species followed by post hoc analyses using a Bonferroni correction. A value of P < 0.05 was considered statistically significant.
Results
One hundred and fifty papers, 75 from each time period (2000–2001 and 2005–2006) were selected from 61 journals for inclusion in this review. One of the papers originally selected was later excluded as the study size of one was considered to be inappropriate for inclusion in this study. The journals included in the review were: Alcohol, Annals of Thoracic Surgery, Atherosclerosis, Autonomic Neuroscience, Biological Psychiatry, Biomaterials, Biomedicine and Pharmacotherapy, Biosensors and Bioelectronics, BMC Musculoskeletal Disorders, BMC Physiology, Bone, Brain Research, Brain Research Bulletin, Brain Research Protocols, British Journal of Oral and Maxillofacial Surgery, Burns, Cardiovascular Research, Cardiovascular Surgery, Diabetes Research and Clinical Practice, Drug and Alcohol Dependence, European Journal of Cardio-Thoracic Surgery, European Journal of Pharmacology, European Journal of Vascular and Endovascular Surgery, Experimental Neurology, Gastrointestinal Endoscopy, Head and Face Medicine, International Journal of Oral and Maxillofacial Surgery, Journal of Cardiothoracic Surgery, Journal of Cranio-Maxillofacial Surgery, Journal of Gastrointestinal Surgery, The Journal of Hand Surgery, Journal of Hepatology, Journal of Neuroimmunology, Journal of Neuroscience, Journal of Neuroscience Methods, Journal of Neuroscience Research, Journal of Oral and Maxillofacial Surgery, Journal of Orthopaedic Research, Journal of Pharmacological and Toxicological Methods, Journal of Plastic, Reconstructive and Aesthetic Surgery, Journal of Shoulder and Elbow Surgery, Journal of Surgical Research, Journal of the Neurological Sciences, Journal of the Society for Gynecologic Investigation, Journal of Vascular Surgery, Methods, Neuroscience, Neuroscience Research, Oral Surgery, Oral Medicine, Oral Pathology, Radiology and Endoscopy, Osteoarthritis and Cartilage, Otolaryngology- Head and Neck Surgery, Peptides, Physiology and Behaviour, Physiological Measurement, Regulatory Peptides, Resuscitation, The Spine Journal, Transplantation Proceedings and Urology.
Due to diversity of the papers selected (papers describing 6 different categories of procedures, in 5 different species, over two time periods were included) the number of papers in each group (and therefore the statistical power of the study) was generally small and statistical analyses were non-significant unless otherwise specified.
Number (‘study size’) and species of animals
The median number of animals that underwent surgery in each study (‘study size’) for ‘large laboratory animals’ was 15 with a minimum and maximum ‘study size’ of 2 and 135. There was a small decrease in the median study size from 15 in 2000–2001 to 14 in 2005–2006. Median and range of group sizes are given in Table 1. Overall, the number of animals undergoing surgery in each time period differed significantly between species (P < 0.001) and the median ‘study size’ for rabbits and pigs was significantly larger than that for primates in both time periods (P ≤ 0.005). In 2000–2001, the median ‘study size’ for rabbits and pigs was also significantly larger than that for dogs (P < 0.01).
Median number of animals used in study (minimum, maximum ‘study size’)
Within each time period, values with different superscripts are significantly different (P < 0.05)
Of the 30 papers describing experimental procedures in non-human primates included from both time periods in this review, 26% of the papers described experimental procedures on rhesus macaques, 23% on cynomolgus macaques, 16% on squirrel monkeys, 16% on common marmosets, 13% on Japanese monkeys, 3% on Bonnet monkeys and 3% on stumptail monkeys.
Classification of experimental procedures
Table 2 shows the number of papers included in the survey for each species classified by procedure and time period. In 2005–2006 there were also three burn papers included in the survey. The distribution of papers by category of procedure differs between the two time periods (P < 0.05); there is an increase in the number of papers describing orthopaedic studies and a decrease in the number of papers describing thoracotomies. Despite the difference in the distribution of categories of procedures however, the papers included in the survey do not differ significantly between the two time periods when classified according to their potential to be painful (e.g. skin incisions are classed as the ‘least potentially painful’ whereas thoracotomies are classed as the ‘most potentially painful’).
Number of papers included in the review for each species classified by category of procedure and by year
Although a statistical analysis was not carried out due to small sample sizes, categories of procedures included in the survey varied with species (Table 2). Specifically there was a high proportion of papers describing: (i) sheep undergoing thoracotomies and orthopaedic procedures, (ii) rabbits undergoing orthopaedic procedures, (iii) pigs undergoing laparotomies and (iv) non-human primates undergoing craniotomies (Table 2). The procedures did vary significantly between species according to their potential to be painful (P < 0.01).
Systemic analgesic administration
The reported administration of systemic analgesics to dogs, pigs, non-human primates, rabbits and sheep increased from 50% in 2000–2001 to 63% in 2005–2006. There was an increase in the reported analgesic administration for all of the species examined between the two time periods with the exception of pigs, where the reported systemic analgesic administration was the same for both time periods, 67% (Figure 1). The reported systemic analgesic administration increased by 9% between time periods for sheep (64–73%) and 13% for rabbits and dogs (40–53%). The largest increase in analgesic use was seen for non-human primates where there was an increase in reported analgesic administration from 40% to 67%.
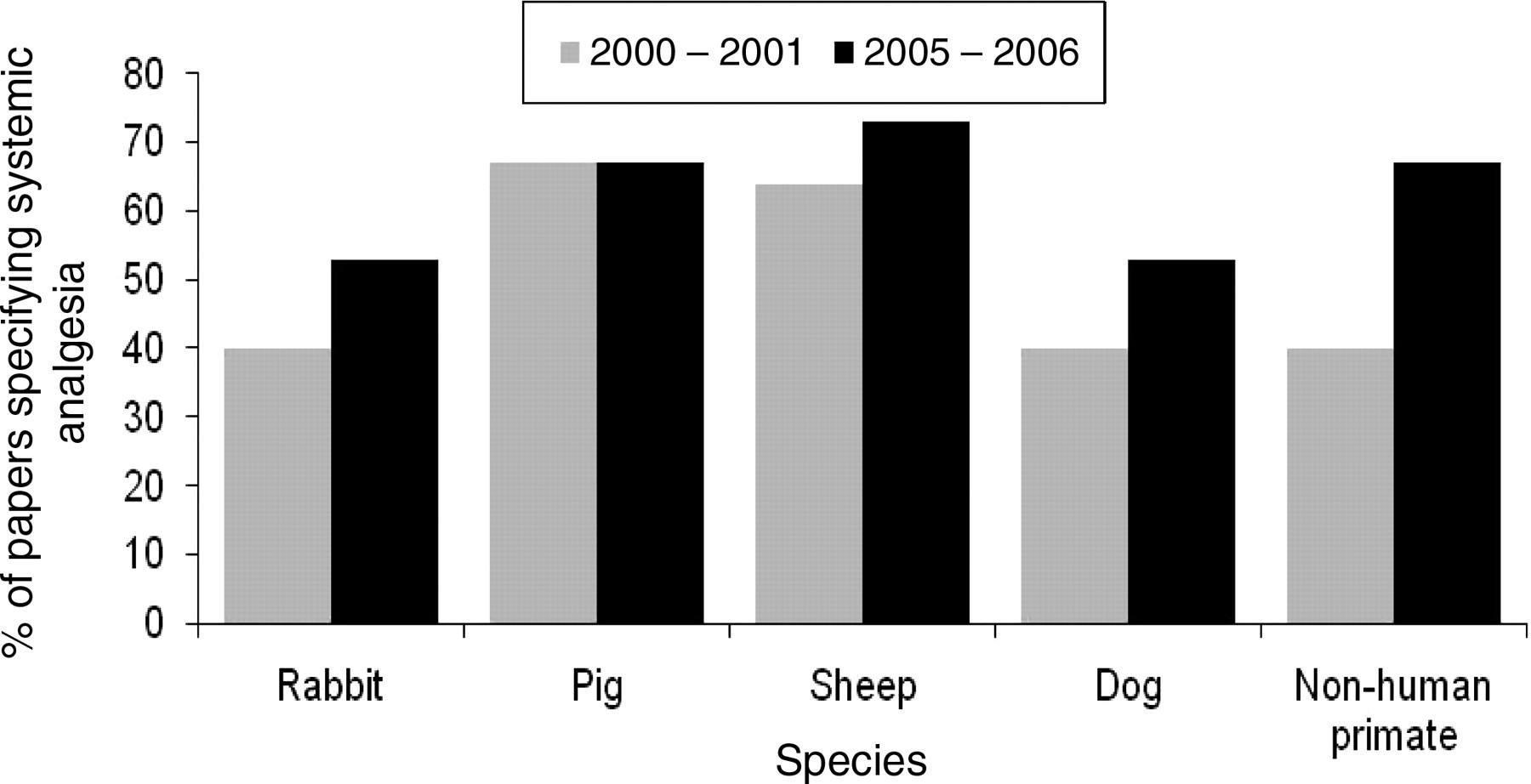
Reported systemic analgesic administration classified by species
Reported systemic analgesic administration increased between the time periods examined for papers describing craniotomies, skin incisions, laparotomies and thoracotomies; however, there was a small decrease in reported systemic analgesic for papers describing orthopaedic procedures (Figure 2). In the 2000–2001 papers, animals that underwent the most potentially painful procedures were more likely to receive systemic analgesics than animals that underwent less potentially painful procedures (P < 0.02); this was not observed in the 2005–2006 papers. Buprenorphine was the most commonly reported systemic analgesic both in 2000–2001 (44%) and in 2005–2006 (40%) (Table 3). The dose range of buprenorphine administered to all ‘large’ laboratory species was 0.001–0.3 mg/kg in both time periods and there was also a large reported dose range for buprenorphine in many of the species examined (Table 4). The reported use of NSAIDs increased between the two time periods from 24% in 2000–2001 to 37% in 2005–2006 primarily due to an increase in the use of carprofen and flunixin meglumine (Table 3). Dose ranges for carprofen and flunixin (when specified) are given in Table 4. In most cases when the administration of a systemic analgesic was specified, the quantity of analgesic administered was given (92% of cases in 2000–2001, 89% of cases in 2005–2006), however dose was often given in mL/animal or mg/animal rather than mg/kg.
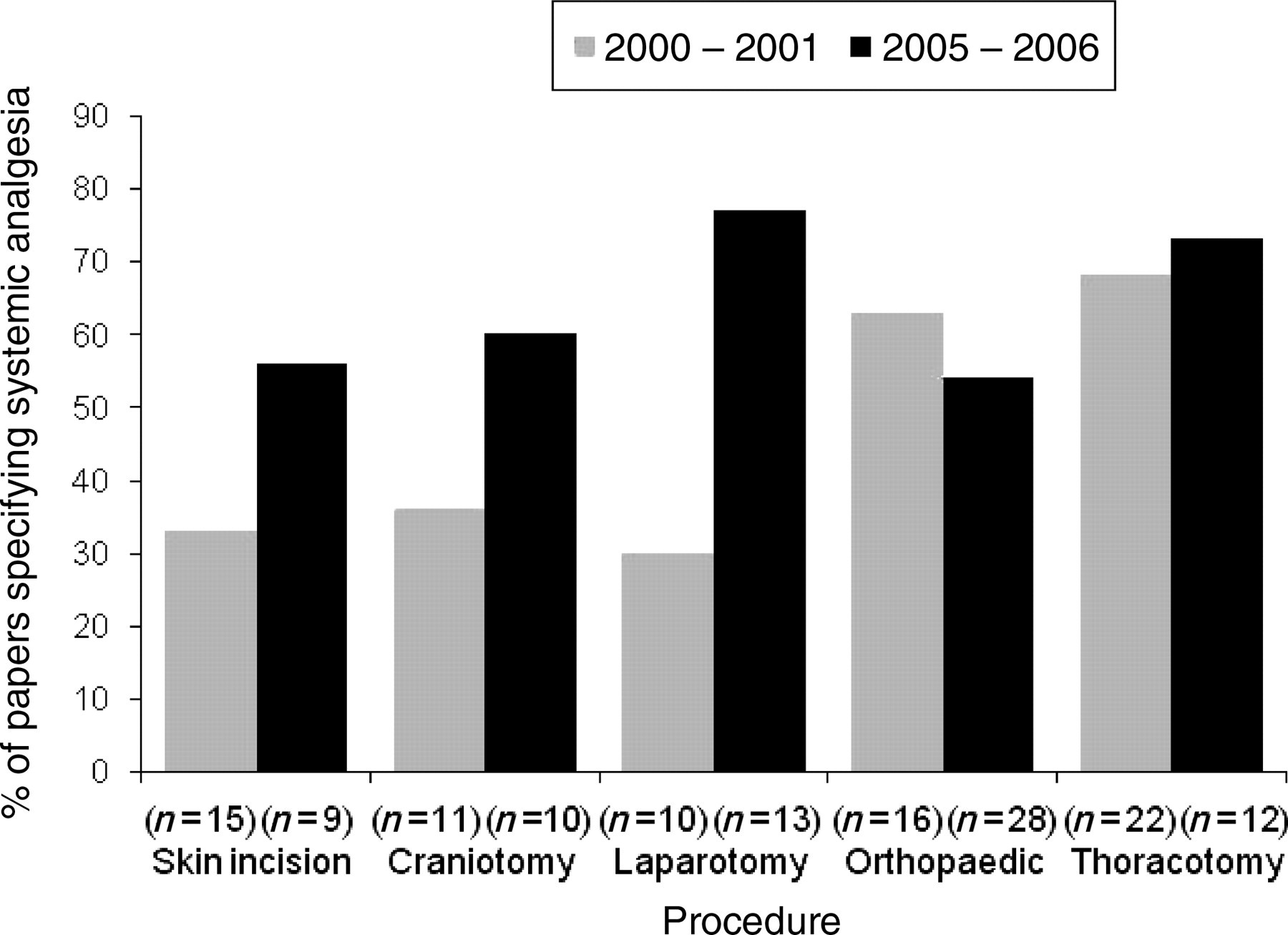
Reported systemic analgesic administration classified by category of procedure
Number of papers reporting systemic analgesic administration classified by agent
Superscript numbers denote the number of papers that reported administration of an analgesic to a given species. Superscript letters denote the species that underwent the experimental procedure: aRabbit; bPig; cSheep; dDog; eNon-human primate. NSAIDs = non-steroidal anti-inflammatory drugs
Range of reported doses of systemic analgesic administered when specified (mg/kg)
n denotes number of papers in which dose was specified
*One paper also reported the use of a non-steroidal anti-inflammatory drug (either acetaminophen, flunixin or carprofen)
†One paper also reported the use of an additional opioid (either methadone or morphine)
‡One paper also reported the use of buprenorphine
§Two papers also reported the use of fentanyl patches
Duration of systemic analgesic administration is given in Table 5. In both time periods, duration of systemic analgesic administration was not specified in 46% of papers that reported the use of a systemic analgesic. Sixteen percent of papers from 2000 to 2001 specifying the use of a systemic analgesic reported one dose of systemic analgesic; in these papers the analgesic was most commonly given intraoperatively. In contrast, none of the papers from 2005 to 2006 specifically reported the administration of only one dose of a systemic analgesic. When repeated doses of systemic analgesic were given, the most common duration of administration was 48 h in 2000–2001 and 72 h in 2005–2006. The duration of systemic analgesic administration did not vary greatly with species (Table 5a), however administration for 48 h or more was more commonly reported in papers describing the most potentially painful procedures (thoracotomies and orthopaedic procedures) compared with papers describing less potentially painful procedures (skin incisions and craniotomies) (Table 5b).
Number of papers specifying the duration of systemic analgesic administration classified by (a) species, (b) severity of procedure
*Three papers (describing pigs undergoing laparotomies) also reported administration of butorphanol or flunixin for an unspecified duration
†One paper also reported administration of buprenorphine for two days
When the administration of a systemic analgesic was specified, the frequency of systemic analgesic administration was not specified in the majority of cases (68% of papers in both time periods). If the frequency of systemic analgesic administration was specified, analgesics were most commonly administered every 12 h.
Administration of all agents with analgesic properties
The proportion of papers specifying the administration of some form of analgesic agent increased from 86% in 2000–2001 to 89% in 2005–2006. The use of ‘agents with analgesic properties’ including systemic analgesics, anaesthetics with analgesic components and local anaesthetic agents is summarized in Table 6. The largest group of papers describing potentially painful procedures without the administration of any analgesic agent, are papers involving dogs published between 2000 and 2001; seven papers (47% of papers involving dogs from that period) did not specify the use of any analgesic agent (Table 6a). Of these seven papers, one skin incision, two laparotomies, two orthopaedic procedures and two thoracotomies were described.
Number of papers included in review classified by analgesic administered and (a) species, (b) severity of procedure
Papers are double counted if they specify the use of a local anaesthetic and the use of a systemic analgesic or an anaesthetic with an analgesic component. Superscripts denote papers that were also classified in another analgesic category. The superscript number denotes the number of papers and the superscript letter denotes the analgesic category
aLocal anaesthetic infiltration and analgesic component in anaesthetic
bLocal anaesthetic infiltration and systemic analgesic
cLocal anaesthetic nerve block or epidural with systemic analgesic
The use of local anaesthetics (through local infiltration, nerve blocks and the epidural route) increased between the two time periods. Nerve blocks were only reported in papers involving animals undergoing thoracotomies (2 papers involving sheep and 1 paper involving dogs). The administration of an analgesic through the epidural route was reported for animals undergoing laparotomies (1 paper each on pigs and dogs) and orthopaedic procedures (1 paper on dogs). The application of fentanyl patches was only reported in papers published between 2005 and 2006.
Multimodal analgesia
The administration of an opioid analgesic with a NSAID was reported in three of the papers published in 2000–2001 and 10 papers published in 2005–2006 (5 of the papers in the latter time period also used an anaesthetic regimen with an analgesic component). When local anaesthetic agents were used, they were frequently combined with systemic analgesic agents and/or anaesthetic with analgesic components (Table 6). Systemic analgesics were also frequently used in conjunction with an anaesthetic regimen with an analgesic component (57% of papers from 2000 to 2001 and 51% of papers from 2005 to 2006 that specified the administration of a systemic analgesic also used an anaesthetic regimen with an analgesic component).
Discussion
The administration of systemic analgesic agents to ‘large’ laboratory species is much higher than that has been reported for rodents. In 2000–2002, systemic analgesic administration to rodents was reported in 10–14% 1,8 of papers, whereas 50% of papers describing surgical procedures on ‘large’ laboratory species reported systemic analgesic administration. The lower reporting of systemic analgesic administration in rodents compared with ‘large’ laboratory species is likely to be attributable to some of the unique challenges associated with assessing pain in laboratory rodents. One of the most common reasons for not administering analgesics to animals is not observing signs of pain. 6,8,10 This is likely to be a particular problem when attempting to assess rodents postoperatively as there may be an evolutionary advantage for rodents to hide overt signs of pain allowing them to avoid predation. 11 Rodents that are group-housed may also try to hide pain from their cage mates in an attempt to maintain their social status and avoid disturbance. 12 Group sizes for rodent studies tend to be larger than studies involving use of larger laboratory species, and rodents are likely to be housed in groups, adding to the challenge of assessing individual animals; the median ‘study’ size for ‘large’ laboratory species was 15 compared to 40 for laboratory rodents in papers published during the same period. 1 Similarly, some of the cages used for rodents (including opaque and individually ventilated cages) and cage furnishings (including shelters and nesting materials) may provide barriers to observing the animals. 12 Finally, since rats and mice are nocturnal, pain-associated behaviours may be more obvious in the hours when staffing levels are lowest. 2
Although compared with laboratory rodents, the level of analgesic administration to ‘large’ laboratory species is high (particularly when administration of local anaesthetic agents and anaesthetics with analgesic components are also considered), the findings of this review indicate that analgesic administration to these larger species is still not optimal. Given the sensitivity surrounding the use of animals in experiments, particularly when they involve non-human primates and companion species such as dogs, it is surprising that some peer-reviewed papers still do not report the administration of any form of analgesic. Although it is possible that in some studies analgesics were administered but not reported, this is unlikely as the level of under-reporting of analgesic administration is thought to be low. 8
Even when papers specified administration of an analgesic agent, it is possible that effective pain relief was not provided in many cases. To relieve pain one must administer an effective analgesic, at the correct dose and frequency, for an appropriate duration of time. There was a wide reported dose range of analgesics for many species, particularly for the most commonly used systemic analgesic, buprenorphine (Table 4). In rabbits, e.g. the dose range for buprenorphine varied by 30-fold from 0.01 mg/kg to 0.3 mg/kg. Although one might expect some variation in dose based on the severity of the procedure and whether other analgesics were administered, such a large variation suggests uncertainty about dose. Similarly, although there was some evidence that the duration of analgesic administration increased with the severity of the procedure (Table 5b), this was not always the case, particularly for the papers published in 2000 and 2001, which suggests that duration of analgesic administration may often be inappropriate.
It is possible that, as with rodents, analgesics may be withheld when signs of pain are not recognized. Although eight papers described the administration of analgesics ‘as necessary’, indicating that animals were evaluated for signs of pain, none of the papers included in the survey described how pain was assessed postoperatively and none of the papers described signs that were considered to be indicative of pain. It is therefore likely that in most cases, analgesic administration was based on subjective impressions of the animal or that analgesics were administered blindly without the use of objective pain assessment techniques. This is perhaps not surprising given the lack of pain assessment schemes available. Ideally, the level of pain should be assessed in each animal to ensure that sufficient pain relief is provided but that the animals are not overdosed since this may result in undesirable side-effects. 13
The current lack of pain assessment techniques available for many species increases the likelihood that many individuals carrying out potentially painful experimental procedures will refer to published literature to determine an analgesic regimen for the specific procedure they intend to carry out. The findings of this review suggest that this may often be problematic given that information about analgesic administration is often insufficient. When systemic analgesics were administered, 68% of papers did not specify the dosing interval or frequency and 46% of papers did not specify the duration of analgesic administration. Similarly, when administration of a systemic analgesic was specified it was often given in inappropriate units (mg/mL, mg/animal, mL, mL/animal, ‘1 ampoule’) making replication of the analgesic regimen difficult (dose rate should always be given in mass of agent per body weight of animal typically mg/kg). Hawkins 2 recommended that analgesic administration should always be included in the methods section of mainstream life science journals however reporting of analgesic administration is still not sufficient in many journals.
Although analgesic administration to ‘large’ laboratory species is still not optimal, several findings of this review suggest that analgesic administration is improving. More papers from 2005 to 2006 reported the use of multimodal analgesia. Combining analgesics from different classes (multimodal analgesia) is a widely used technique in man 14 and in veterinary clinical practice. 15 The administration of an opioid and a NSAID is thought to provide more effective pain relief than when high dose rates of a single analgesic are used. 16,17 Side-effects from analgesic drugs may also be minimized because lower doses of each drug can be administered, than if the drugs were administered individually. Increased reporting of non-steroidal anti-inflammatory agents and fentanyl patches also suggest an improvement in overall analgesic administration to ‘large’ species used in scientific procedures.
Footnotes
ACKNOWLEDGEMENTS
CC was supported by the Fund for Animal-Free Research. CR was supported by a 3Rs Liaison Group Research Studentship from UFAW.
