Abstract
Each different gas that is used to induce a pneumoperitoneum (PP) exhibits individual effects within the peritoneal cavity. This might include adverse effects such as pain and/or inflammatory reactions. The acute effects of ozonized oxygen (O3/O2), a highly oxidative gas mixture, after being insufflated into the peritoneal cavity are analysed in this study. Using the abdominal constriction response (‘writhing’) assay of chemical nociception in C57BL6/N mice, O3/O2-PP was found not to be associated with visible pain responses and did not alter the c-fos expression in the spinal cord. In addition, mRNA expression levels of the pro-inflammatory cytokines, interleukin (IL)-1β and IL-6, were found unaltered in the spleen 2 h after insufflation. In conclusion, O3/O2-PP is free of adverse pain and does not trigger inflammatory immune responses.
Induction of an artificial pneumoperitoneum (PP) by medical insufflation of gas into the peritoneal cavity is used to extend the abdominal cavity to enable adequate visualization in laparoscopic surgery. Besides the harmless mechanical distension of the peritoneal cavity, insufflated gases have been found to harm the structure of the peritoneal surface,1–3 to induce metabolic changes within the peritoneal cavity4,5 and to influence local and systemic immunological parameters.6–8 The immunological parameters encompass the activation and function of various leukocytes, e.g. neutrophils, monocytes/macrophages, lymphocytes, and the amount of several inflammatory mediators such as interleukin (IL)-1, IL-6 and tumor necrosis factor (TNF)-α synthesized by laparoscopic interventions.6,7,9 Interestingly, each type of gas used for the induction of a PP varies in its effect on mechanical, physiological and immunological parameters.10–12 The individual effect of each type of gas on immune parameters might be responsible for the different side-effects and outcomes of laparoscopic surgery.
The gases most commonly used for PP are carbon dioxide (CO2), helium (He), and nitric oxide (NO). Besides these conventional pure gases, an ozonized oxygen (O3/O2) gas mixture is also applicable for inducing a PP. Moreover, insufflation of O3/O2 into the peritoneal cavity has been found to have advantageous effects in several clinical conditions. In a rat model for experimental surgery, O3/O2-PP reduced the formation of postsurgical peritoneal adhesions, 13 and in the rat model of polymicrobial-induced peritonitis, O3/O2-PP reduced the death rate.14–16 These data suggest that the targeted use of O3/O2 gas mixtures in intestine surgery might be useful. Additionally, the immune modulatory effect of O3/O2-PP has proved to be an effective oncolytic approach in an experimental tumour model. 17
With these beneficial therapeutic properties O3/O2-PP is recommend for use in further animal studies. Therefore, with animal welfare in mind it is essential to know whether O3/O2-PP is associated with pain when used in animal experiments.
To investigate the nociceptive property of O3/O2-PP, we used the established abdominal constriction response (‘writhing’) assay of chemical nociception in mice,18–20 and analysed the pain-associated activation of immediate early gene c-fos expression in corresponding spinal cord segments. With regard to the effect of O3/O2 on immune functions,21,22 and considering that cytokines can modulate pain sensitivity,23,24 the gene expressions of the pro-inflammatory cytokines IL-1β and IL-6 in the spleen were quantified 2 h after gas insufflation.
Animals
Six to 8 week-old-male and female C57BL6/N mice, derived from Charles River, Sulzfeld, Germany were housed in groups of six animals per type III Makrolon cage (Tecniplast, Buguggiate, Italy) in a temperature-controlled environment, maintained on a 12 h artificial day/night rhythm. Each cage was enriched with nesting material consisting of scraps of soft paper and one paper roll. Animals were acclimatized in the institute’s own animal facility for at least 14 days and had free access to water and food. Prior to the experiment, animals were acclimatized in pairs of two animals derived from the same grouped cage for at least 24 h in a separate testing room to the Plexiglas observation chamber in which the abdominal constriction response (‘writhing’) test was performed. During this acclimatization phase, the Plexiglas observation chamber contained the usual softwood bedding and enrichment as used in the Makrolon cages and animals had free access to water and food. Immediately before the 2 h long writhing test the enrichment was removed from the Plexiglas observation chamber and the two animals were separated by a non-transparent wall to avoid social modulation of pain by visible contact as described in dyads. 25 All writhing tests were performed near mid-photophase between 12:00 and 16:00 h to minimize circadian variability. 26
Material and methods
Abdominal constriction (‘writhing’) test
The animal experiment was approved by the regional board in Giessen, Germany (V54-19c20-15(1) MR 2011, Nr 47/2008) according to the German Animal Protection Law. The experimental design is shown in (Figure 1a). For sample size calculation, the determination of c-fos mRNA expression levels in the spinal cord as well as possible inflammatory effects of the O3/O2 gas mixture on cytokine mRNA expression levels within the spleen were defined as the critical factor. Due to the lack of any information about the effect size in molecular changes, the standardized effect size (Cohen’s d) was set at 1.4 (large effect under standardized conditions) according to the references given by the 3R (refinement, replacement and reduction) platform,
27
which resulted in a sample size of n = 12 required for detecting possible differences at the 5% probability level.
Experimental design. (a) Time sequence of observation periods and tissue sampling. (b and c) The lengthwise stretches of the torso with a concomitant concave arching of the back and a flexure of the head to the tail sagittal line above an angle of 30° were defined as criteria for positive abdominal constriction (writhing).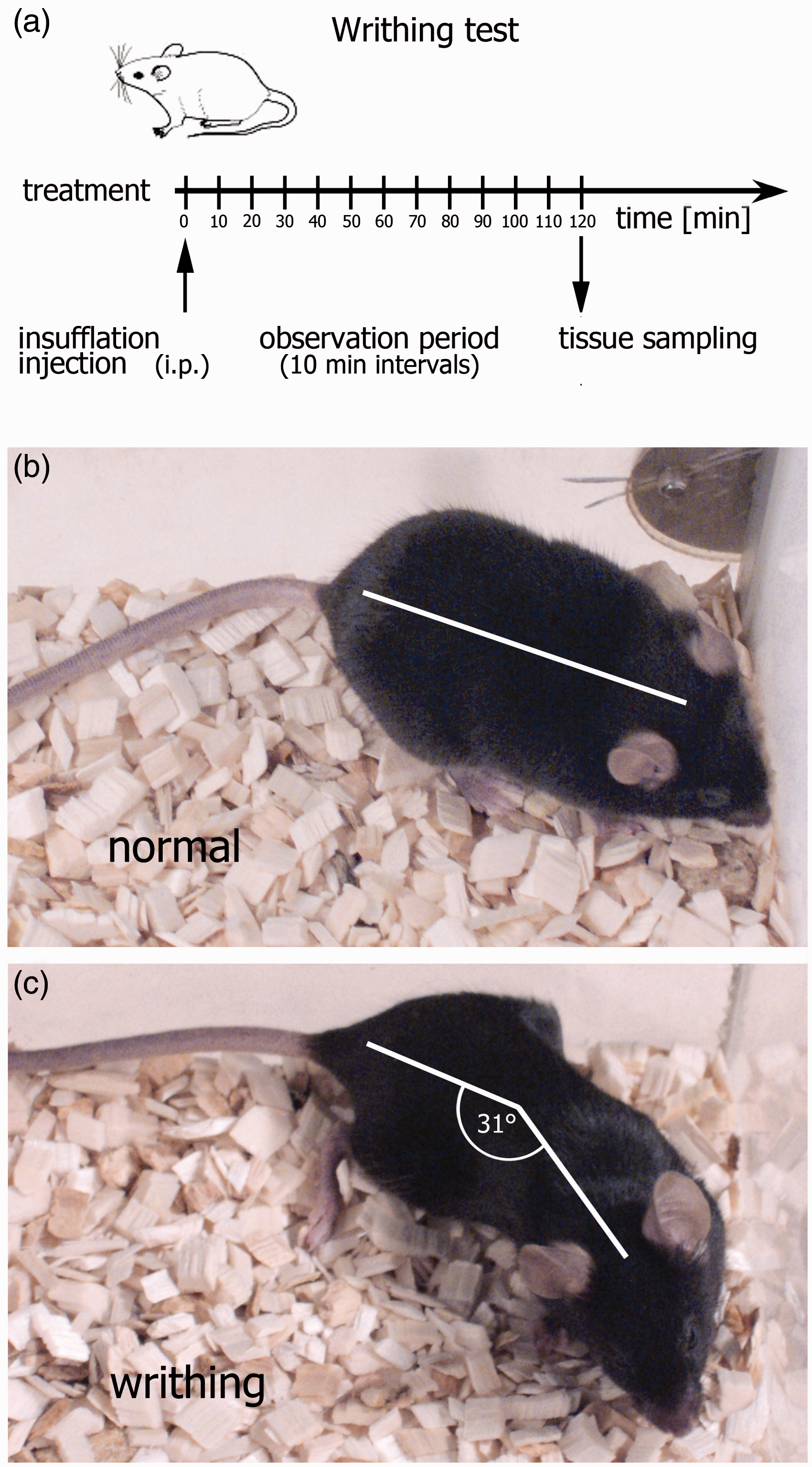
The animals were randomly divided into one of the four experimental groups. Two cages with six animals each were assigned by a blinded experimenter to groups I to IV. The animals within each given group received an intraperitoneal injection of either: (i) 0.2 mL of a 0.7% glacial acetic acid in physiological sodium chloride (NaCl) solution, used as a positive control for the induction of writhing responses; 28 (ii) 0.2 mL of a physiological NaCl solution used only as a negative sham control; (iii) 1 mL of pure O2 gas; and (iv) 1 mL of a medical O3/O2 gas mixture containing 80 µg O3 per 1 mL of gas synthesized using an ozone gas processor MedozonIP® (Herrmann Apparatebau GmbH, Kleinwallstadt, Germany) as described previously. 29 This volume and O3 concentration were chosen because 80 µg ozone/mL is the maximal O3 amount which can be synthesized by the MedozonIP® processor. Moreover it also equates to the amount of the total amount of O3 per kg body weight used in other animal studies. A 19 G needle was used for the injection of the liquid solutions as well as for the insufflation of the gaseous substances. The one-millilitre volume of the insufflated gaseous substances was applied within 15 s and led to a slightly strained abdominal wall.
The observation period started immediately after application of the substance and consisted of 12 intervals of 10 min each in which the numbers of writhing actions were noted. During the observation period the experimenter left the testing room and the behaviour of the animals was recorded using a webcam positioned straight above the Plexiglas observation chamber. All recorded files were assigned a number and analysed later by an experimenter blinded to the treatment. A lengthwise stretch of the animal with a concomitant concave arching of the torso and a flexure of the head to the tail sagittal line above an angle of 30° was defined as a positive abdominal constriction writhing (Figures 1b and c). At the end of the observation period animals were euthanized by CO2 inhalation, the abdomen was examined for injuries due to the intraperitoneal injection post-mortem, and the spleen and thoracolumbal spinal cord segments were removed for molecular biological analysis and stored in RNAlater® (Sigma, Steinheim, Germany). Animals with any pathological signs of a puncture of the gut were excluded (one animal in total).
Quantitative reverse transcription PCR
Messenger RNA expression levels of c-fos in thoracolumbal segments of the spinal cord and IL-1β and IL-6 in the spleen were measured by quantitative real-time PCR (qPCR) using the ABI PRISM® 7900HT system (Applied Biosystems, Foster City, CA, USA). Total RNA was extracted from tissue samples using the RNeasy® mini kit (Qiagen, Hilden, Germany), its degree of purity and integrity was analysed by the Experion™ automated gel electrophoresis system (Bio-Rad Laboratories GmbH, Munich, Germany) and quantity was determined using a Nanodrop 2000c spectrophotometer (Peqlab, Erlangen, Germany). Finally one microgram of the total RNA was reverse transcribed to cDNA with the Transcriptor First Strand cDNA Synthesis Kit® (Roche Diagnostics GmbH, Penzberg, Germany) according to the manufacturer’s protocols. The following oligonucleotide primers were purchased from Invitrogen GmbH (Darmstadt, Germany): β-actin (forward: 5′-AGC TTC TTT GCA GCT CCT TCG-3′; reverse: 5′-AGG GTC AGG ATA CCT CTC TTG CT-3′, designed according to NM_007393.3), c-fos (forward: 5′-CCG CGA ACG AGC AGT GAC CG-3′; reverse: 5′-GGA AAG CCC GGC AAG GGG TC-3′, NM_010234.2), IL-1β (forward: 5′-TGC AGC TGG AGA GTG TGG ATC CC3′; reverse: 5′-TGT GCT CTG CTT GTG AGG TGC TG-3′, NM_008361.3), IL-6 (forward: 5′-CCT CTC TGC AAG AGA CTT CCA TCC A-3′; reverse: 5′-AGC CTC CGA CTT GTG AAG TGG T-3′, NM_031168.1). Amplicons of all primer pairs were generated by PCR using 2× Kapa fast ready mix (Peqlab), purified from agarose gels using the NucleoSpin® gel and PCR clean-up (Macherey-Nagel, Dueren, Germany) and quantified using the Nanodrop 2000c spectrophotometer. To generate standard curves for absolute quantification assays serial dilutions of known copy numbers were used for qPCR amplification. For absolute quantifications, amplicons or cDNA probes derived from tissue samples were amplified in triplicates with the Power SYBR Green PCR Master Mix (Applied Biosystems, Darmstadt, Germany) according to the manufacturer’s protocol. For calibration of variances between different qPCR plates quantifying the mRNA expression of the same gene, a dilution containing 10exp5 copies of amplicons of the corresponding gene was used as an interpolate calibrator. The calculated copy number from each sample was normalized to 10exp3 copies of β-actin.
Statistics
The GraphPad Prism Software 6.0 (GraphPad Software Inc, San Diego, CA, USA) was used. In all analysis a P value of < 0.05 was considered to be a significant difference between the four groups. For calculation of statistic differences in the number of writhing actions between all experimental groups within each 10 min observation interval, a one-tailed analysis of variance (ANOVA) test with Bonferroni’s multiple comparison test was performed. For statistical analysis of group differences in the copy number of c-fos, IL-1β, or IL-6 an unpaired, two-tailed t-test with Welch’s correction was used.
Results
Insufflation of the highly oxidative O3/O2 gas mixture as well as pure O2 into the peritoneal cavity did not lead to visible pain reactions and was comparable with the control (NaCl) (Figure 2a). Injection of the 0.7% glacial acetic acid solution induced a significant number of writhing actions, which was highest within the first 10 min (15.6 ± 3.4), decreased until 30 min (5.5 ± 1.0), and remained constant over the remaining observation period of 120 min. The molecular genetic analysis of c-fos mRNA in thoracolumbal segments of the spinal cord revealed a significantly enhanced c-fos gene expression in the acetic acid-treated animals when compared with the NaCl-treated control animals (Figure 2b). No significantly increased expression of c-fos mRNA was measured in the O3/O2-treated group when compared with the sham animals, although a slight increase was present.
Pain response. (a) Abdominal constriction (writhing) assay after intraperitoneal injection of 0.2 mL of a 0.7% glacial acetic acid solution or insufflation of a 1 mL of an ozonized oxygen (O3/O2) gas mixture into the peritoneal cavity. Physiological saline solution and pure O2 gas were used as controls for the acetic acid solution or the highly oxidative O3, respectively. Mean number of writhing actions within a 10 min interval ± SEM are shown. (b) Absolute quantification of c-fos mRNA templates in the spinal cord by real-time quantitative polymerase chain reaction (qPCR) analysis. The numbers of c-fos templates were calibrated to 10exp3 β-actin mRNA templates. Statistical differences were calculated: (a) by one-way analysis of variance (ANOVA) test followed by Bonferroni’s multiple comparison test; (b) by an unpaired t-test with Welch’s correction. Statistical differences were marked with: *P < 0.05, **P < 0.001, ***P < 0.0001.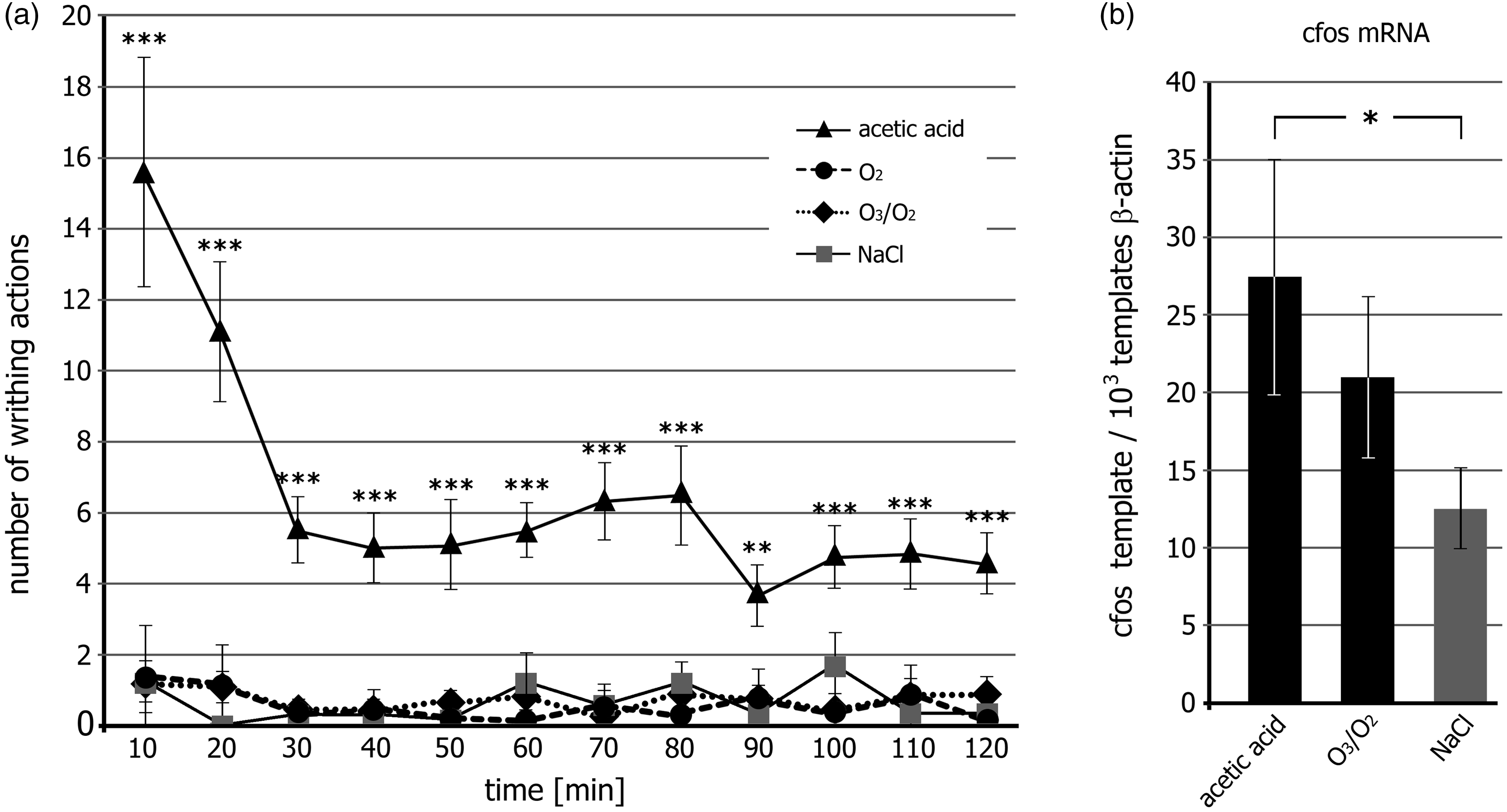
The impact of O3/O2-insufflation on the immune status was examined by gene expression analysis of the pro-inflammatory cytokines IL-1β and IL-6 in spleen homogenates and compared with the effect of acetic acid injection. Absolute quantification of IL-1β mRNA (Figure 3a) and IL-6 mRNA (Figure 3b) revealed a basal expression of both cytokines in the spleen of the NaCl-treated sham animals, and no significant enhancement by intraperitoneal O3/O2-insufflation or acetic acid injection.
Immune response. (a) Number of interleukin (IL)-1β mRNA and (b) IL-6 mRNA templates in the spleen of mice 120 min after injection of the respective test substance. The numbers of IL-1β and IL-6 templates were calibrated to 10exp3 β-actin mRNA templates. Statistical differences were calculated by an unpaired t-test with Welch’s correction. No statistical differences were observed.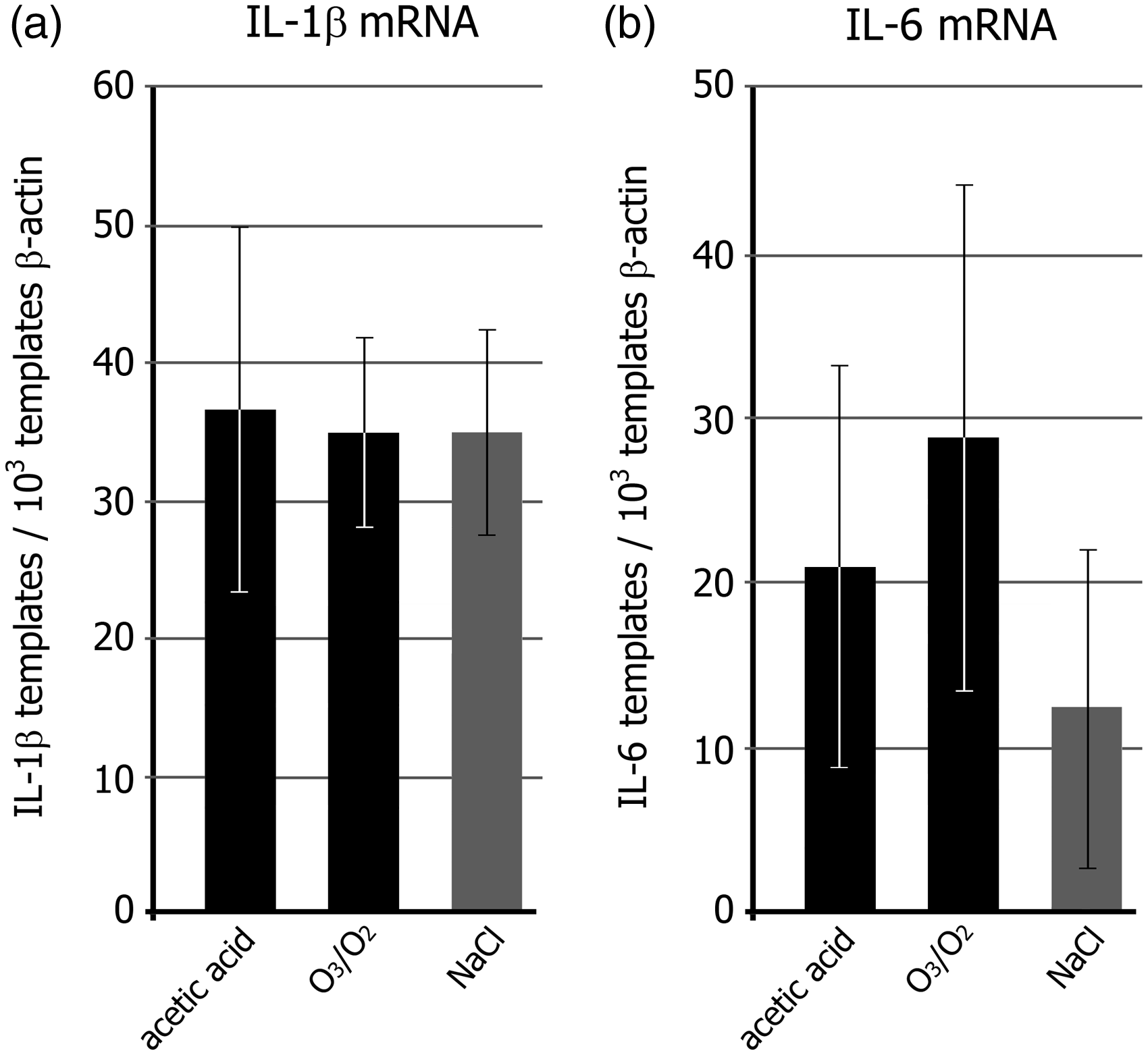
Discussion
The insufflation of the highly reactive gas mixture of O3/O2 or the lower reactive O2 into the peritoneal cavity did not lead to early or later pain responses as observed by the writhing test. This shows that the abundant presence of free O2 radicals did not generate a chemical nociception. Furthermore, due to the lack of pain reactions, a mechanically-induced nociception generated by the rapid insufflation of one millilitre of the gaseous substances and the associated increase in intra-abdominal pressure can be ruled out.
The intraperitoneal injection of acetic acid used as a positive control led to visible writhing actions of the C57BL6/N mice with most writhing actions occurring within the first 30 min, but persisted over the whole observation period of 120 min at lower frequencies.18,30 The number and early time course of the writhing actions are in accordance with the published data which analysed the nociceptive sensitivity in 11 different commercially available inbred mouse strains, including the C57BL6 strain. 28 The C57BL6 was found to be a mouse strain which exhibited a mean sensitivity to acetic acid-induced chemical nociception when compared with all the other strains. This characterizes the C57BL6 strain as suitable for use in detecting not only the lower but also the higher pain sensitivity for insufflated O3/O2 compared with acetic acid.
Increased neuronal expression of the immediate-early gene c-fos in the spinal cord has been associated with pain-induced neuronal activation in response to noxious somatic, articular, and visceral stimulation.31,32 In this study a significant increase in c-fos mRNA expression within the spinal cord 2 h after administration of acetic acid was measured, but not after O3/O2-PP administration, which is in line with our results of the writhing test which showed pain reactions exclusively in the acetic acid-treated animals. Therefore, we conclude that O3/O2-insufflation into the peritoneal cavity of awake mice did not induce measurable visceral or somatic pain. Additionally, the animals behaved normally within the 2 h observation period after insufflation, indicating that they were not distressed. Thus anaesthesia and pre- or post-analgesic care do not seem to be essential for this treatment paradigm. However, unrecognized adverse effects on the well-being of the animals cannot be excluded completely. Animals should always be observed thoroughly within the first few hours after treatment and pre- or post-analgesic care must be used whenever it is necessary.
Besides a direct mechanical or chemical effect of an insufflated gas on viscero- and/or somatosensory afferent neurons, the individual capacity of any gas to influence the intraperitoneal immunity6–8,33 might lead to pain as a result of direct cell cytotoxicity or the generation of an inflammatory process.34,35 Inhaled ozone was found to be cytotoxic to lung tissue and could cause acute inflammation by the recruitment and activation of leukocytes as well as the release of pro-inflammatory cytokines such as TNF-α, IL-1β and IL-6.36–42 In the case of the lung, insufflated ozone might lead to airway inflammation by direct cytotoxicity or by immune cell activation triggered by oxidation products such as lipid ozonation products (LOPs) 43 or reactive oxygen species (ROS). 44
The O3/O2-PP did not induce IL-1β or IL-6 gene expression in the spleen, suggesting that the abundant presence of free O2 radicals per se did not trigger inflammatory processes. This is in line with observations in rats showing unaffected gene expression levels of IL-1β and TNF-α in the liver, spleen, Peyer’s patches and mesenteric lymph nodes one hour after O3/O2-PP. Therefore, an acute immune stimulatory effect of ozonized oxygen on the abdominal immune system seems to be unlikely. 14
In summary, our experimental study shows that the usage of a O3/O2 gas mixture for laparoscopic interventions or as a novel therapeutic agent does not lead to acute discomfort and immune stimulatory processes in mice. However, it cannot be ruled out that other unknown negative side-effects may occur in mice and that pain and/or adverse immune reactions may occur when O3/O2 gas mixtures are used in other animal species.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded in part by the Wirtschafts- und Infrastrukturbank Hessen (TM MBF_04).
