Abstract
Air pressure is commonly used to drive a mechanical stimulus for nociceptive threshold testing. This may be bulky, noisy, non-linear and suffer from friction, hence development of a better system is described. A novel, light (14 g) rolling diaphragm actuator was constructed, which supplied 20 N force via a constant actuation area irrespective of the pressure and position in the stroke. Three round-ended pins, 2.5 mm diameter, mounted in a triangle on the piston, provided the stimulus. Pressure was increased manually using a syringe with the rate of rise of force controlled at 0.8 N/s by warning lights. The pressure/force relationship was calibrated using a static force transducer and mercury column. Data were collected with the actuator attached to the anteromedial radius of 12 cats and four dogs. Mechanical threshold was recorded when the animal withdrew the limb and/or turned towards the actuator. Safety cut-off was 20 N. The pressure/force relationship was linear and independent of the start point in the actuator stroke. Baseline feline thresholds were 10.0 ± 2.5 N (mean ± SD), which increased significantly 30 min after butorphanol administration. Baseline canine thresholds were 5.5 ± 1.4 N and increased significantly between 15 and 45 min after administration of fentanyl or butorphanol. The system overcame the problems of earlier devices and detected an opioid-induced increase in threshold. It has considerable advantages over previous systems for research in analgesia.
Research in pain and analgesia requires laboratory investigation as well as clinical studies to establish appropriate analgesic dosing protocols suited to the individual species. This necessitates the use of objective analgesiometry: a repeatable noxious stimulus, without injury to the subject, used to measure nociception and drug-induced antinociceptive effects. A variety of mechanical stimuli have been widely used in this way, alongside other modalities such as thermal, electrical and chemical. 1
A mechanical stimulus, particularly for larger animals, is commonly generated using a pneumatic actuator to drive a pin into tissue. Air pressure from a compressed gas container, or generated by an electric motor and pump, is often used to pressurize the actuator.2-4 Such systems have a number of drawbacks, and those developed for animals such as sheep, cattle and horses are too heavy and cumbersome for use in the smaller animals such as cats and dogs, particularly where a behavioural endpoint is sought. 5 In addition to the bulk of the actuator itself, the pump and motor noise is distracting and the subject may learn to recognize it. A further problem inherent in the larger sliding seal actuators is the potential for high static and dynamic friction, leading to a proportion of the applied pressure being ‘shunted’ away into overcoming this force. Both the larger sliding seal systems 2 as well as the smaller bladder-type actuator 5 may demonstrate a pressure/force relationship which is non-linear across the full stroke, and this may change unpredictably as parts become worn and alter the compression area.
Even if compressed gas is not used to generate pressure, some or all of the above problems apply in mechanical threshold (MT) testing in larger animals; we describe the development of a small, light actuator, suitable for a range of species, which overcomes the drawbacks of the older systems.
This project was carried out under UK Home Office licence (Animal [Scientific Procedures] Act) and was approved by the ethical review committee for animal experimentation of Lower Saxony according to §8 of the German Animal Welfare Act (Tierschutzgesetz).
Materials and Methods
Actuator
A novel actuator was constructed using a 18 mm wide Penrose drain (Bard Ltd, Crawley, Sussex, UK) as a rolling diaphragm to form a seal between a piston and cylinder (Figure 1). The assembly is compact (25 mm diameter and 27 mm high) weighing 14 g with a force range up to 20 N. Mechanical stimulus was provided by three pins, 2.5 mm diameter, 7 mm long, with a 2.5 mm diameter ball bearing glued onto the stimulating end. The pins were mounted in a 10 mm triangle on the piston in the formation previously described for use in cats. 5
Rolling diaphragm pressure actuator. The three pins are driven into the tissue by pressurizing the chamber behind the diaphragm
Pressure within the actuator was increased by manual inflation using a 20 mL syringe with a vent hole sealed by the operator's thumb. Pressure was released at threshold by lifting the thumb, thereby removing the stimulus. Pressure in the actuator was measured by a 0–15 psi pressure transducer with integral signal conditioning (Model 2938339, Radio Spares Ltd, Corby, Northants, UK). At threshold pressure, the reading was held on the display until reset for the next test.
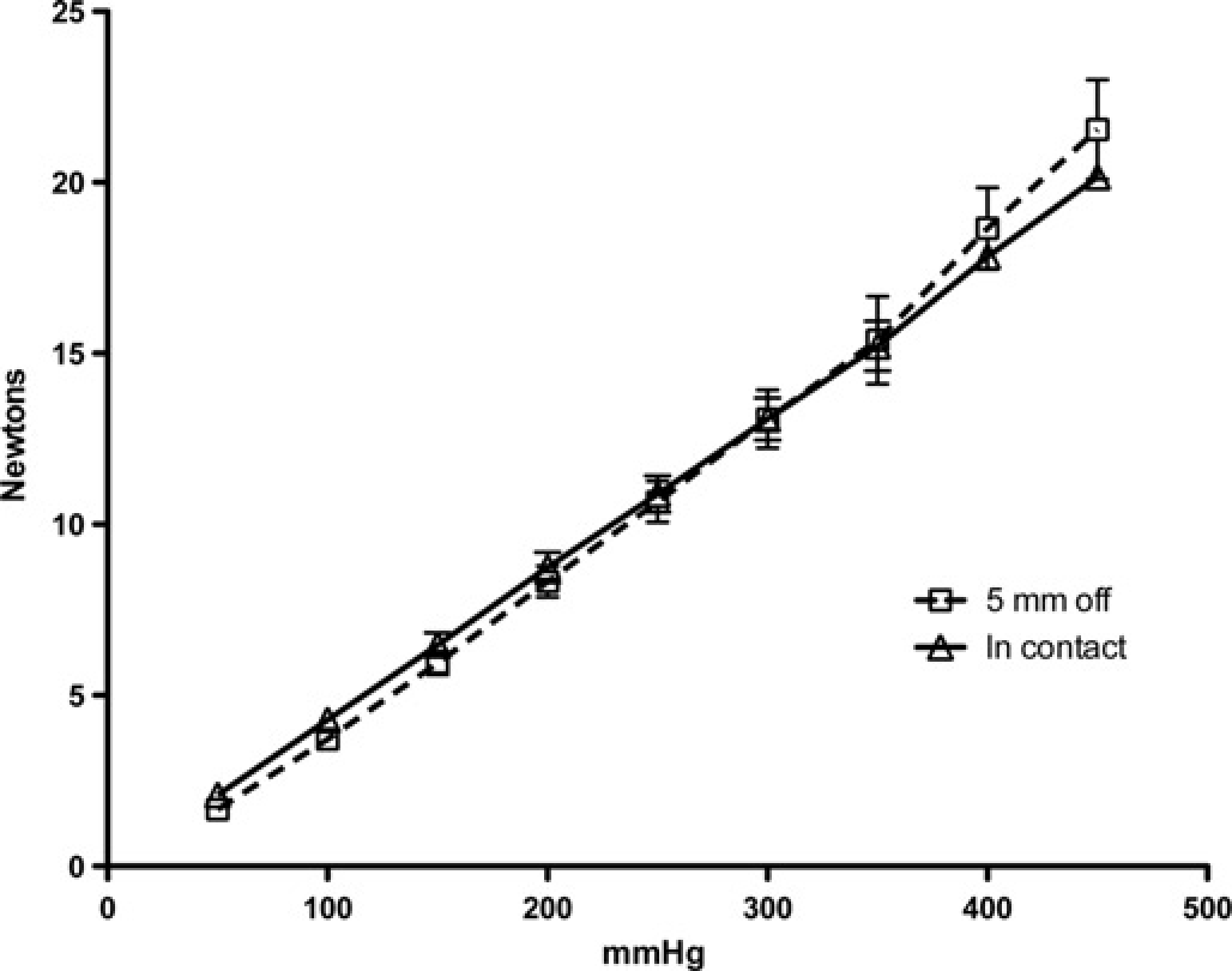
The pressure–force relationship was calibrated using a static force transducer (Kenwood, UK) and mercury column and the display adjusted to indicate N. The pressure–force relationship was plotted four to five times for each of three actuators made to the same design, over a period of five months (n = 13). Tests were performed both with the pins in contact with the force transducer at the start of the stroke and with the pins 5 mm away from the transducer at the start.
The pressure–force relationship from the rolling diaphragm actuator was compared with that generated from the original bladder system previously described. 5 Tests were performed both with the pins in contact with the force transducer at the start of the stroke and with the pins 5 mm away from the transducer at the start.
The manual increase in pressure was controlled in order to give an increase in force of 0.8 N/s using warning lights that allowed a window of ± 1 N at any point during application of the stimulus. A green light indicated that the rate was too low (the expected pressure for the time taken had not been reached), and a red light that the rate was too high (the expected instantaneous pressure had been exceeded). A safety stimulus cut-off was set at 20 N to prevent tissue damage if the animal did not respond before this force was reached.
Initially the actuator was fixed onto a limb using only a buckle and non-elastic tape (Figure 2a). More recently a soft boot with a cylindrical locator has been used, which allows predictable and repeatable location of the actuator on the limb (Figure 2b).
(a) Actuator secured to a dog's forelimb with non-elastic tape. (b) Actuator held on a cat's leg using the boot and cylindrical placer
Animals
Cats
Twelve in-house neutered laboratory-bred domestic cats (8 female, 4 male), aged 6–8 months, weighing 2.8–6.0 kg were studied. They were housed in groups, according to the regulations outlined in the Animal Scientific Procedures Act 1986, and, a few hours before the first test, two cats were placed in adjacent individual cages (1.2 m x 0.7 m x 0.6 m) for the testing procedure. They were fed commercial moist whole diet (Whiskas®) and dry cat food (Hills Feline Maintenance, Watford, Herts, UK) supplied in small amounts during the day. Bowls of drinking water were always available.
A 5 cm ring of hair was clipped from both front limbs between the carpus and elbow. The actuator was secured on the anterolateral aspect of the clipped ring midway between the carpus and elbow, using non-elastic tape to position the actuator so the pins were just in contact with the limb, but exerting no pressure. A dummy actuator was similarly secured to the contralateral limb. The cat was then allowed a minimum of 15 min acclimatization before any testing.
Nine baseline readings were taken at 15 min intervals before any treatment was given. The first four were for training both cats and operator, and the data were discarded. At time 0, butorphanol 0.2 mg/kg was injected intramuscularly and further readings were made at 15 min intervals until 150 min.
Dogs
Four laboratory-bred beagle dogs (1 female, 3 castrated male), aged 8–10 years, weighing 14–16 kg were studied. They were were housed in groups, according to regulations of the German Animal Welfare Act 1998 and fed a commercial moist whole diet (Hills Canine P/D Prescription Diet™) and dry dog food (Hills Canine P/D Prescription Diet™) twice a day. A few hours before the first test, at least two dogs were placed in a cordoned area of the laboratory for the testing procedure. Bowls of drinking water were available at all times.
The dogs’ limbs were clipped in a similar way to the cats, the actuator attached and the dogs allowed time to acclimatize. A dummy actuator was placed on the contralateral limb.
Testing was performed 3–5 times at 15 min intervals on each dog prior to treatment with either butorphanol 0.25 mg/kg intramuscularly (2 dogs) or fentanyl 5–10 μ/kg intravenously (2 dogs). Testing was continued at 15–60 min intervals after dosing until thresholds returned to pretreatment values.
Mechanical threshold testing
For each test, the actuator was connected via 150 cm non-expandable polyethylene tubing ID 1.5 mm (Vygon, France) to a 20 mL syringe filled with air. The syringe plunger was depressed manually, applying force increasing at 0.8 N/s. MT was recorded when the animal withdrew or raised its limb, jumped forwards or turned towards or tried to chew the actuator. The pressure was released immediately the animal reacted and the force recorded as MT.
Statistical analysis
Statistical analysis was carried out using GraphPad Prism®. Mean ± SD are reported unless otherwise stated. Descriptive statistics were applied to the actuator data. Cat and dog data were checked for normality using the Kolmogorov–Smirnov test. Cat baseline data were averaged to give a mean ± SD baseline value. Taking this value as time 0, repeated-measures ANOVA followed by Dunnett's test was used to assess changes with time in response to butorphanol treatment. Canine baseline MTs were compared with peak MTs using Student's unpaired t-test and the Welch correction for data that were not normally distributed. Cat baseline data were compared with baselines taken from the same cats six months later 6 using Student's paired t-test.
Results
Actuator
The pressure–force relationship of the rolling diaphragm actuator was linear over the full 0–20 N range, with a pressure of 450 mmHg producing 20 N force (Figure 3). The coefficient of variation for 13 tests over five months ranged from 3.1% at 400 mmHg to 14.6% at 50 mmHg. Extrapolation of the pressure–force relationship towards the origin shows intersection with the X-axis at less than 10 mmHg, indicating minimal internal resistance. Put another way, less than 10 mmHg pressure was needed to overcome the internal resistance of the actuator. The gain of the actuator (its amplification factor), here defined as the ratio of the input pressure to the output force, and its internal resistance were also unaffected by the position of the piston at the start of the stroke (Figure 4).
Pressure/force relationship of the rolling diaphragm actuator measured against a static force transducer. Mean ± SD of 13 tests taken from three actuators over five months. Extrapolation of the line back to the X-axis (at <10 mmHg) is an indication of the negligible pressure required to overcome the internal resistance of the actuator (analogous to friction in a system with sliding seals) Effect of pin position at the start of the stroke: pressure/force relationship of the rolling diaphragm actuator measured against a static force transducer. Mean ± SD of nine tests with pins starting in contact with skin (open triangles, solid line) and four tests starting with pins 5 mm above skin (open squares, dotted line). Extrapolation of the lines back to the X-axis is similar to Figure 3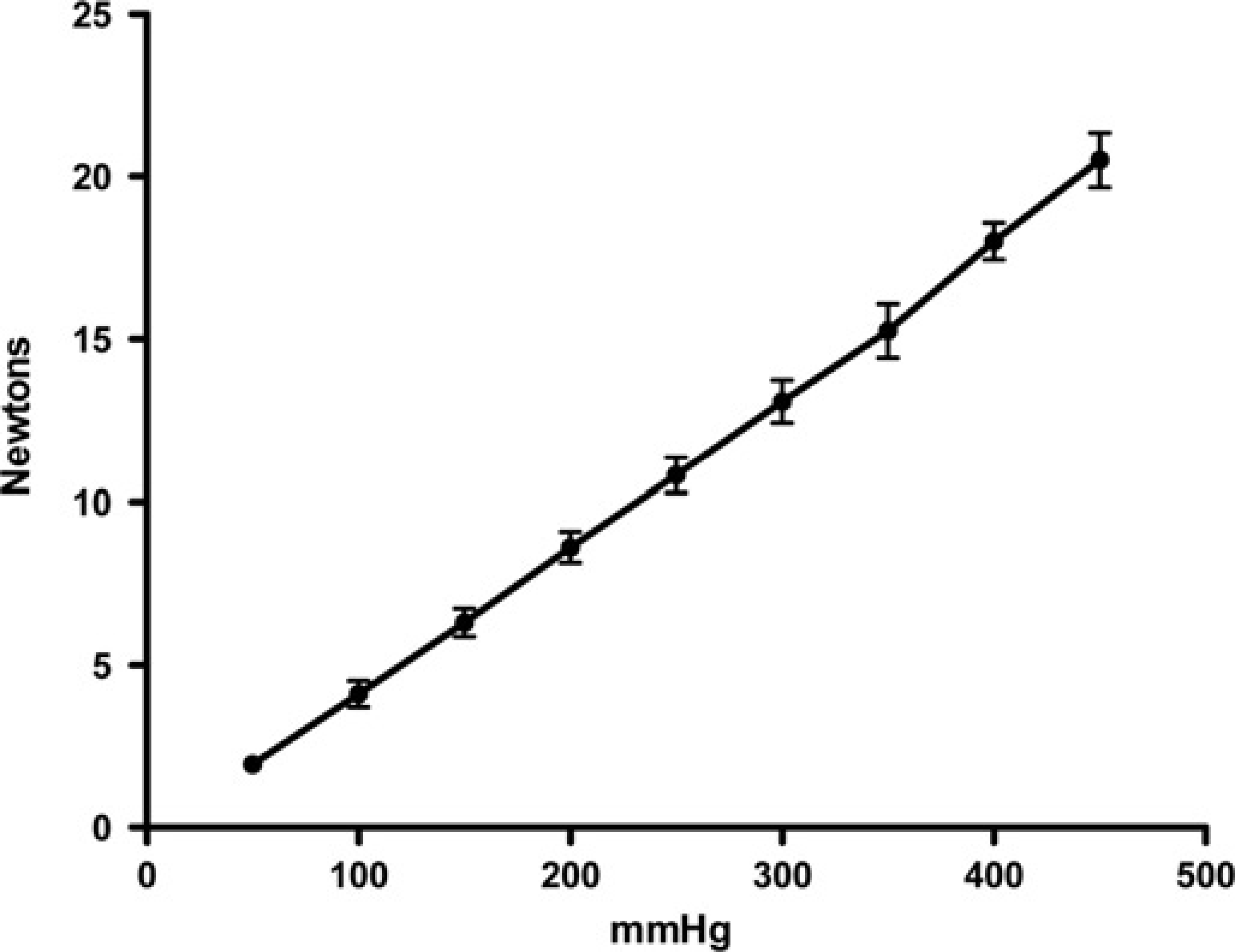
The gain of the rolling diaphragm actuator was similar to that of the bladder actuator, which was linear over the range 0–20 N and had a similar degree of internal resistance (from extrapolation), but only when the calibration was started with the pins in contact with the force transducer (Figure 5). The internal friction for the bladder became significant when the calibration was performed with the pins starting 5 mm away from the force transducer. Extrapolation back to the X-axis then intersected at approximately 40 mmHg, indicating that more of the pressure generated was then used to deform the bladder initially (analogous to starting friction in a sliding seal actuator). The gain of this actuator was also lower when the pins were not in contact with the force transducer at the start of the stroke (Figure 5), indicating that the effective area of the bladder changed with extension.
Comparison of the pressure/force relationship of the rolling diaphragm actuator with the bladder actuator measured against a static force transducer. Rolling diaphragm actuator (open symbols, dotted lines), bladder actuator (closed symbols, solid lines), pins starting in contact with skin (triangles) and pins starting 5 mm above skin (squares). The rolling diaphragm line extrapolated back to zero would cross the X-axis at less than 10 mmHg. The extrapolated bladder line starting in contact with skin would also cross the X-axis at less than 10 mmHg, but the bladder line for 5 mm above skin has >40 mmHg offset when extrapolated back to zero, indicating that the internal resistance is four times higher, hence four times the force is required to inflate the bellows to contact position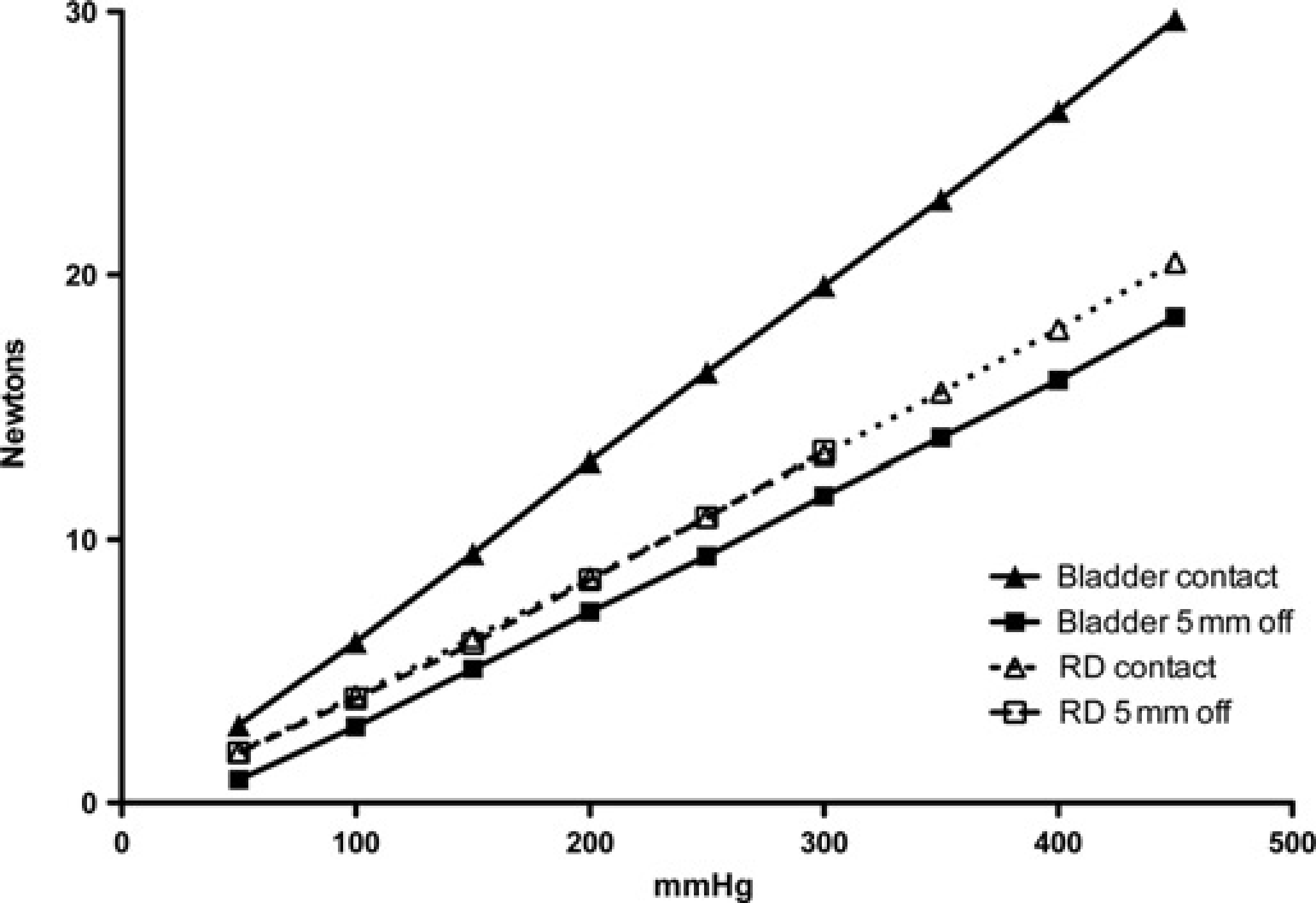
Use of equipment
Consistent positioning of the actuator on the limb was greatly facilitated by the use of the boot and cylindrical guard. The guard also ensured that the pins were consistently positioned just in contact with the skin. The warning lights enabled all testers to control the rate of rise of force within the set limits without difficulty; all individuals had good control after 4–5 test runs. None of the testing caused any skin damage.
Cats
Data were normally distributed. Mean pretreatment baseline MT were 10.0 ± 2.5. Individual cat data are shown in Table 1. MT 30 min after butorphanol treatment (15.0 ± 5.1 N) was significantly higher than baseline (P = 0.0005) (Figure 6). There were no significant differences between these MT baselines and those recorded from the same cats six months later (8.8 ± 1.5 N) (P = 0.4134), 6 and the variances were not significantly different.
Individual cat and dog mechanical thresholds (MTs)
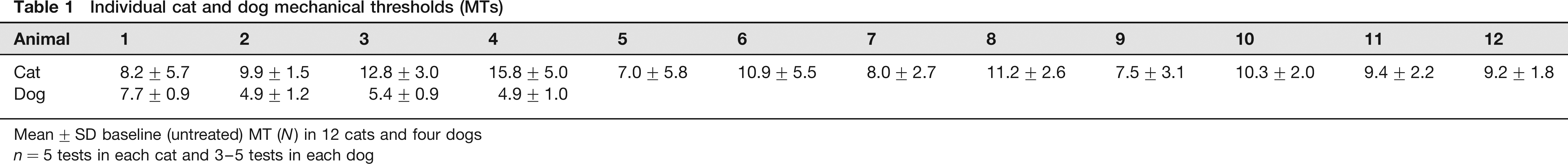
Mean ± SD baseline (untreated) MT (N) in 12 cats and four dogs n = 5 tests in each cat and 3–5 tests in each dog
Mean ± SEM mechanical threshold in 12 cats before (0 = mean of 5 pretreatment tests) and after treatment (arrow) with butorphanol 0.2 mg/kg intramuscularly. *Denotes significant increase above pretreatment baseline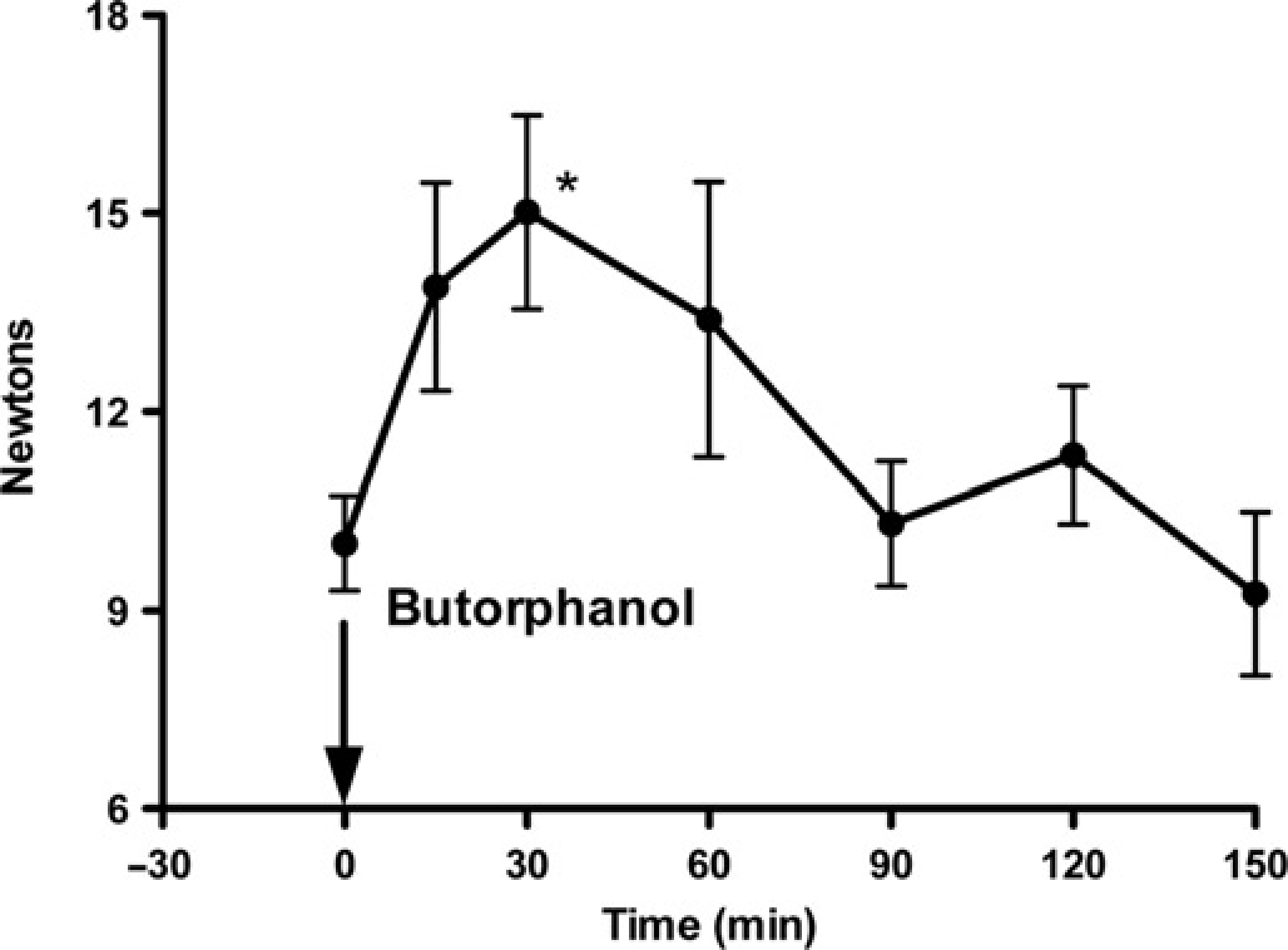
Dogs
Mean pretreatment baseline thresholds were 5.5 ± 1.4 N. Individual untreated dog data are shown in Table 1. Peak MT after opioid treatment (14.9 ± 2.1 N) was significantly higher than baseline (P = 0.0232).
Discussion
The rolling diaphragm actuator was light and easy to apply to the limb of a small animal; as a result, it was well tolerated, even by cats. Use of a small, manually driven syringe to generate the driving pressure made the system completely silent and did not disturb the animals.
A rolling diaphragm actuator has two inherent advantages over a sliding seal actuator, both of which were used to advantage in the current system. Firstly, a rolling diaphragm has low internal resistance (similar to friction), so that nearly all of the generated pressure contributes to the force at the output, thus preventing a significant zero offset in the pressure/force relationship. Secondly, the effective piston area of the rolling diaphragm actuator does not change with the position in the stroke, again leading to the same gain under all working conditions.
In contrast, the gain of the bladder actuator was seen, under calibration, to vary with the distance that the pins had to travel before touching the surface of the calibration transducer (0 or 5 mm). It is also of note that, in use, the pins drive into soft tissue rather than a stiff surface (the calibration transducer). The total travel, especially to generate higher forces, will therefore be greater than the 5 mm simulated during calibration and the resultant gain (see Figure 5) even lower than that from the 5 mm starting point.
The starting point, travel and therefore repeatability will inevitably vary depending on how tightly the actuator is fixed to the animal's limb; in addition to the inherent improvements from the rolling diaphragm device, this variability was also reduced in the current investigation by the use of a boot and cylinder, which gave a stiffer (less compliant) reaction path to the applied load and greater repeatability of positioning.
Consistency of data from the bladder actuator is, therefore, heavily dependent on how it is positioned and on the proximity of the pins to the skin before the start of the test. The consistency of data reported by Steagall and co-workers7–11 suggest that, with experience, this may be reasonably repeatable within one laboratory and one group of experimenters.
Another feature of note in the current system was the use of warning lights to control the rate of force increase. It has been previously indicated that the threshold may be affected by the rate of application of the stimulus,1,12 hence it is important to ensure a consistent rate of application of force to generate valid data.
The present actuator incorporated the three-pin stimulus previously reported in developing a system suitable for cats. 5 This is intended to ensure that the stimulus remains perpendicular to the main axis of the limb, where a single pin may tilt, reducing the pressure on the limb for any given force in the actuator. However, this does not prevent the use of another profile with the rolling diaphragm, as others can be fixed into the present system in place of the three pins.
The aim of this investigation was to evaluate a new actuator, not the pharmacology of the drugs administered. However, in order to demonstrate the potential benefit of the new system, and to understand how it fits into the existing knowledge base, the effect of recognized opioid analgesics was examined in two species. In both cases MT was increased, demonstrating that this system was able to detect antinociception in the same way as described for other mechanical systems.2,5,13
When new techniques are developed it is important to evaluate their place within the existing knowledge base. MT testing is notoriously susceptible to difficulties in comparison of data between investigations conducted by different groups. This is a result of the relationship between pressure and force and the fact that often only the force in the actuator is reported without details of the stimulus area and pin profile. As pressure = force/area, it is evident that the pressure in the tissue in contact with the stimulus can only be estimated if the stimulus area, and preferably the profile, are known as well as the applied force. It is notoriously difficult to measure the area of contact, as this changes when the probes deform the surface of the skin, hence only limited comparison can be made between two different reports unless they use identical equipment.
Nevertheless, we have attempted to evaluate how the data generated in this report compare with previously published MTs. We have used the same pin profile as previously described, 5 and report data directly comparing the bladder and rolling diaphragm systems calibrated against a static force transducer (Figure 5). Steagall and co-workers7–11 reported untreated baseline MT ranging from 200 to 240 mmHg using the bladder system developed for cats 5 and peak values of 450 to 500 mmHg after treatment with a number of opioids. With reference to Figure 5, describing the gain (force/pressure relationship) of both bladder and rolling diaphragm, it can be seen that baseline values were closely comparable when the ‘5 mm off skin contact’ gain line is used: 200–240 mmHg is equivalent to 8.5–10 N, the range reported in the present investigation for untreated baselines. Peak values of 450–500 mmHg correspond to forces of 17.5–20 N if the 5 mm gain line is still used. However, as noted above, the greater extension from driving into soft tissue rather than into a stiff calibration plate will lower the gain of the bladder actuator even further, leading to a still lower gain line than shown in Figure 5. Reasonable extrapolation from the 0 and 5 mm lines to a further gain line with a total travel of about 8 mm would then indicate a force of about 13–15 N at 450–500 mmHg, which compares well with the peak postopioid value of 15 N reported here.
The data presented in this study from the cats suggest that the MT recorded in the same animals is relatively stable. The MT from the same group of cats after several months appeared slightly lower, although the differences were not significant. This may, however, reflect some degree of learning, emphasizing the importance of using crossover studies when evaluating different drug treatments. The cats were relatively young at the first testing period, so the slight decrease in baseline MT may also reflect increasing maturity. It was notable that the cats had considerably higher baseline MT than the dogs. This may simply reflect species differences, but until data have been collected from a number of species using the same stimulus it is impossible to draw any further conclusion on this aspect.
In conclusion, this rolling diaphragm actuator proved straightforward to use, well tolerated by cats and dogs, produced linear data and detected opioid-induced increases in MT. It has considerable advantages over previous mechanical systems for research in analgesia.
