Abstract
The induction of acute hepatic damage by acetaminophen (N-acetyl-p-aminophenol [APAP]), also termed paracetamol, is one of the most commonly used experimental models of acute liver injury in mice. The specific values of this model are the highly reproducible, dose-dependent hepatotoxicity of APAP and its outstanding translational importance, because acetaminophen overdose is one of the most frequent reasons for acute liver failure (ALF) in humans. However, preparation of concentrated APAP working solutions, application routes, fasting period and variability due to sex, genetic background or barrier environment represent important considerations to be taken into account before implementing this model. This standard operating procedure (SOP) provides a detailed protocol for APAP preparation and application in mice, aimed at facilitating comparability between research groups as well as minimizing animal numbers and distress. The mouse model of acetaminophen poisoning therefore helps to unravel the pathogenesis of APAP-induced toxicity or subsequent immune responses in order to explore new therapeutic interventions for improving the prognosis of ALF in patients.
Historic background of the model
Acetaminophen (N-acetyl-p-aminophenol [APAP]) is the most widely used antipyretic and analgesic drug, which is considered to be safe at recommended therapeutic concentrations. An overdose of APAP can cause severe liver damage resulting in acute liver failure (ALF). 1 Indeed, APAP overdose is the most common cause of liver failure in the United States. 2 As with humans, an overdose of APAP in mice also results in ALF. Therefore, in contrast to most other animal models of acute liver injury, the model of APAP-induced liver injury (AILI) in mice is directly transferable to the pathogenesis of an important and life-threatening human disease.
While mice are very suitable as experimental animals for studying metabolic functions or immune reactions following APAP overdose, large animal models like dogs or pigs give the opportunity to measure additional physiological parameters (e.g. intracranial pressure, cardiac output, systemic vascular resistance and levels of methaemoglobinaemia). 3 For instance, specific interventions addressing haemodynamics in APAP-induced ALF can be sufficiently studied in such large animal models. 4 By contrast, using mice as experimental animals offers unique potential for understanding the pathogenic mechanisms of liver injury and the subsequent immune response in detail. Due to the high amount of immune markers and antibodies available for mice, the easy handling of large numbers of animals, and the wide availability of knockout mice, this species is highly suitable for understanding the mechanisms in AILI and for exploring novel treatment options in a preclinical setting. Therefore, our protocol focuses on the AILI model for mice.
Pathogenic mechanisms of liver damage
The initial underlying mechanism of AILI is the massive necrosis of hepatocytes. In humans, APAP is taken up in the intestine within the first 2 h after oral consumption. The main proportion of acetaminophen is metabolized in the liver by glucuronidation and sulphation and excreted via urine. A small fraction (10–15%) is metabolized in hepatocytes by cytochrome P450 isoforms into the alkylating and highly toxic metabolite N-acetyl-p-benzoquinone imine (NAPQI). The antioxidant glutathione (GSH) converts NAPQI into a harmless reduced form, which can then be excreted via bile (Figure 1). Due to the restricted capacity for glucuronidation and sulphation, the amount of metabolized APAP increases in the case of an overdose. When GSH is depleted, this increasing amount of NAPQI binds to mitochondrial proteins, where they form cytotoxic protein adducts, leading to necrosis of hepatocytes.1,5
Degradation of N-acetyl-p-aminophenol (APAP) and formation of N-acetyl-p-benzoquinone imine (NAPQI) protein adducts. The metabolism of acetaminophen (APAP) by different isoforms of cytochrome P450 (mainly CYP2E1) leads to formation of the toxic metabolite NAPQI in hepatocytes. Low levels of NAPQI can be fully converted into the inactive reduced form by the binding of glutathione. Due to an overdose of APAP, existing glutathione is depleted, and NAPQI starts binding to mitochondrial proteins. The NAPQI protein adducts lead to necrosis of hepatocytes. N-acetylcysteine as a precursor of glutathione is a treatment option after APAP overdose in humans.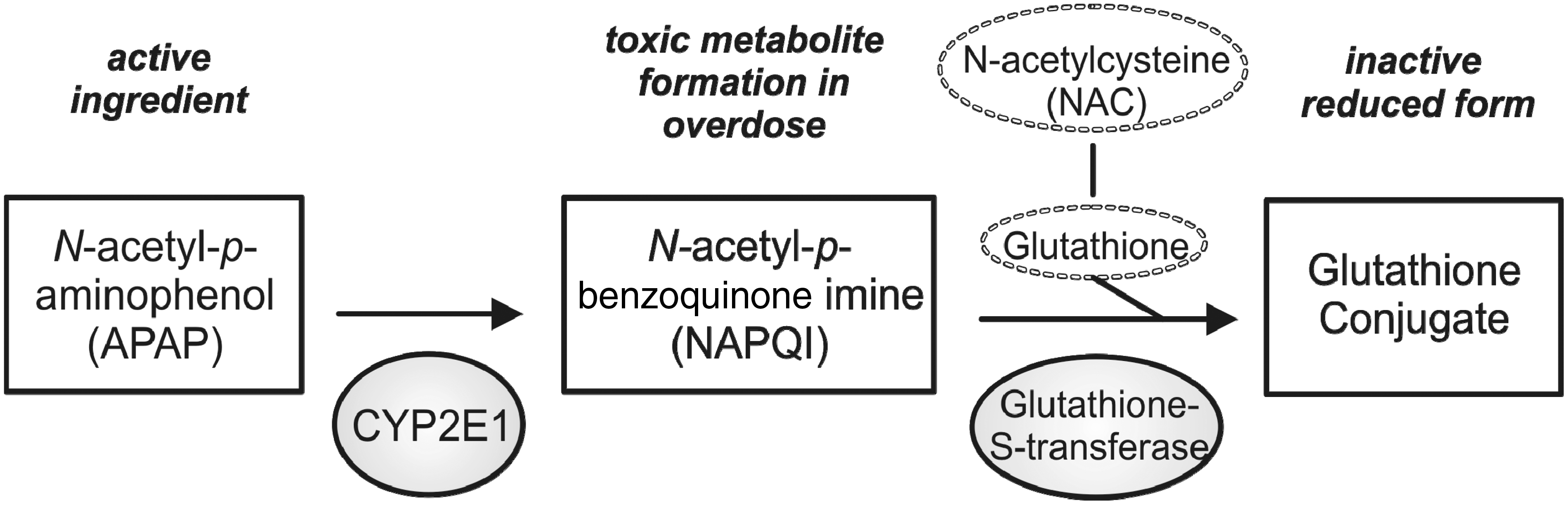
The second step in AILI is a sterile inflammation as a response to the necrosis of hepatocytes, which leads to the massive release of various danger-associated molecular patterns (DAMPs) and the formation of the inflammasome complex in various cells such as Kupffer cells (liver macrophages). 6 Different non-parenchymal cell types, cytokines and chemokines contribute to the inflammatory response following acute liver injury. The activation of the innate immune system leads to the recruitment of immune cells to the site of inflammation and perpetuates hepatocyte necrosis. All of these steps, including NAPQI accumulation, hepatocyte necrosis, and strong inflammatory response, are well recapitulated in the AILI model in mice.
Experimental procedure
General considerations
Sex and genetic background
The differences between female and male mice in AILI are well established.7,8 Female mice usually develop less necrosis in the liver than male mice, independent of their genetic background. Therefore most studies used either solely female or solely male mice to obtain comparable results.
Principally, mice from different genetic backgrounds can be used in this model, as the mechanism initiating liver damage is a direct toxic effect of APAP within the hepatocytes. However, it is known that different mouse strains from different genetic backgrounds require either dose adjustments of APAP or develop significantly less or more injury. This is thought to be a consequence of different immune cell activation following the injury. As such, C57BL/6 mice show enhanced liver injury and increased expression of tumour necrosis factor-α (TNF-α) compared with BALB/c mice. This might be due to a C57BL/6 specific Th1-dominant response of T-helper cells in AILI or to other strain-dependent differences. 9 The overwhelming body of experiments published to date with this model have employed male C57BL/6 mice. Therefore, our procedure is focused on this strain (male C57BL/6 mice), and other genetic backgrounds or female mice might require dose optimization prior to experimentation.
Fasting period
Before APAP is applied, mice should be fasted to create comparable conditions for APAP metabolization by depleting hepatocytic glutathione levels. 10 The duration of this fasting period described in the literature alternates between 12 and 16 h, with free access to water during the whole time. We recommend a fasting period of 12 h for male, 10 to 12 weeks old C57BL/6 mice, which receive an APAP dose of 250–300 mg/kg body weight. In different conditions (mouse strain, sex, age of mice, dose of APAP), the fasting period might be adjusted.
Duration of treatment and appropriate endpoints
Immediately after the fasting period, the mice should be weighed and APAP applied at a concentration of 250 mg/kg body weight in cases of intravenous application, or 300 mg/kg body weight in cases of intraperitoneal application. After APAP treatment, free access to food (standard chow diet) should be allowed. The necrosis of the hepatocytes starts (depending on the application route) about one hour after APAP administration. Different papers have shown elevated liver aminotransferase activities (aspartate aminotransferase [AST], alanine aminotransferase [ALT]) in the serum within 2 h after intraperitoneal application.7,8 In cases of intravenous application, liver damage can be expected earlier, and in cases of gavage at later time points.
Choosing the appropriate endpoint depends on the research objective of the experiments. During the first 5 h after application, APAP metabolism and the initial wave of hepatocyte necrosis are the central ongoing processes. Starting at around 6 h after APAP treatment, different immune processes are activated by the hepatocyte necrosis. Typical endpoints are 2 h (for metabolism), 4 h (for intrinsic cell death pathways), as well as 6, 12 and 24 h (for inflammation and immune-related responses).
Application routes
APAP can be administered through intraperitoneal or intravenous injection, or by gavage. The majority of investigators use intraperitoneal application of APAP, mainly due to its high reproducibility and ease of handling. Intravenous injection via the lateral tail vein can be done using already dissolved APAP (e.g. APAP commercially available for human patients such as Paracetamol Actavis® 10 mg/mL) and has even higher reproducibility. We do not recommend using the enteral application route via gavage, because the induction of liver injury has much higher variability due to individual differences in enteral absorption, which then requires a considerably higher number of animals per experiment.
Biometric calculation of required cohorts
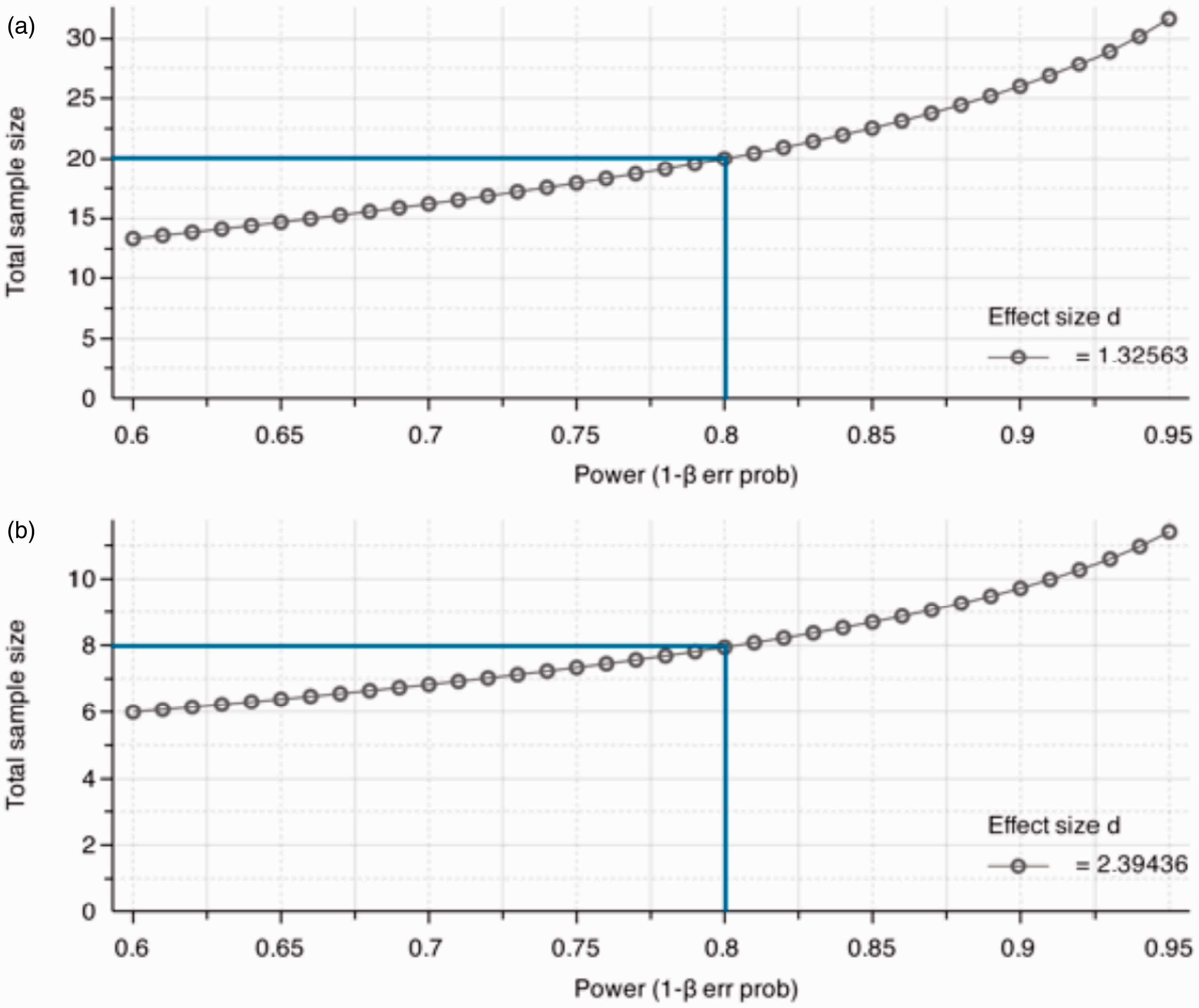
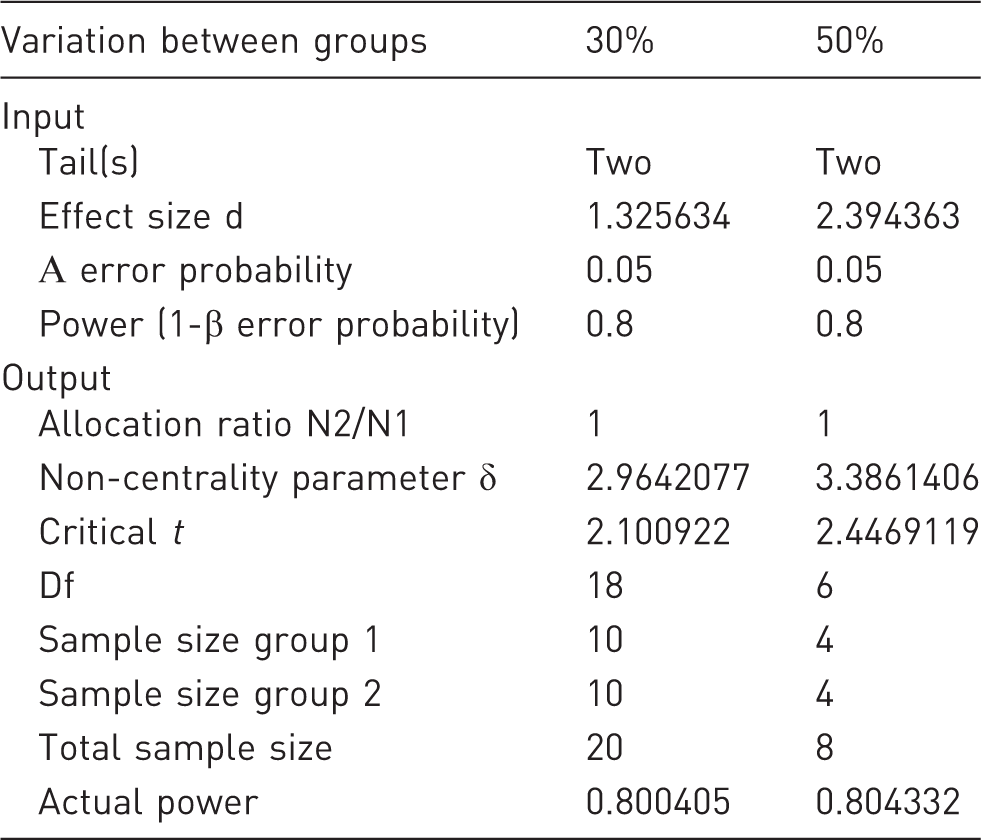
In a typical AILI experiment we compare the level of hepatic damage between wild-type (wt) mice and pharmacologically-treated or genetically-modified mice. A useful laboratory value for quantifying the liver damage is the activity of transaminases (AST and ALT) in the serum (Figure 2). From our own experience a variance in transaminases of around 25% in wt mice 12 h after APAP application can be expected (Figure 2). With respect to the clinical outcome and for preclinical studies with translational impact, we consider a difference in transaminase activities between groups (e.g. wt versus transgenic, wt placebo versus protective drug) of at least 30% to be clinically relevant. From these parameters, the calculated effect size d is 1.33, as determined using G Power V.3.1.9 software.
11
To obtain a statistical power of 80% and a specific α error probability of 0.05, the minimum required sample size in each group should be n = 10 mice. Comparing two groups with a 50% difference in transaminases, the calculated effect size d is 2.39, and in each group n = 4 mice are needed to reach a statistical power of 80% and a specific α error probability of 0.05 (Table 1 and Figure 3).
Liver injury after N-acetyl-p-aminophenol (APAP) overdose. Male C57BL/6 mice (10 to 11 weeks old) received one intraperitoneal injection of 300 mg APAP/kg body weight after a fasting period of 12 h. (a) Livers from control mice (ctr, treated with NaCl 0.9%) and APAP-treated mice stained with haematoxylin and eosin (H&E) show progressing necrotic areas (lighter), 12 h and 24 h after APAP overdose. Scale bar: 500 µm. (b) The liver damage is also reflected by the levels of transaminase activities (aspartate aminotransferase [AST] and alanine aminotransferase [ALT]) in the serum. ctr: n = 7; APAP: 12 h, n = 7; 24 h, n = 10. ***P < 0.001 (Student’s unpaired t-test). Relationship between total sample size and statistical power for the 12 h time-point in the N-acetyl-p-aminophenol (APAP) injury model. (a) Twenty animals (10 per group) are needed under the described conditions and in case of intraperitoneal injection of acetaminophen, if the difference in transaminases between pharmacologically treated and untreated mice (or different genetic backgrounds) is 30%. In cases of intravenous injection, the standard deviation in transaminases might be lower and fewer animals would then be needed. (b) Eight animals (4 per group) are necessary, if the transaminases have a 50% deviation between two groups. Biometric calculation of sample size for 12 h after acetyl-p-aminophenol (APAP) application.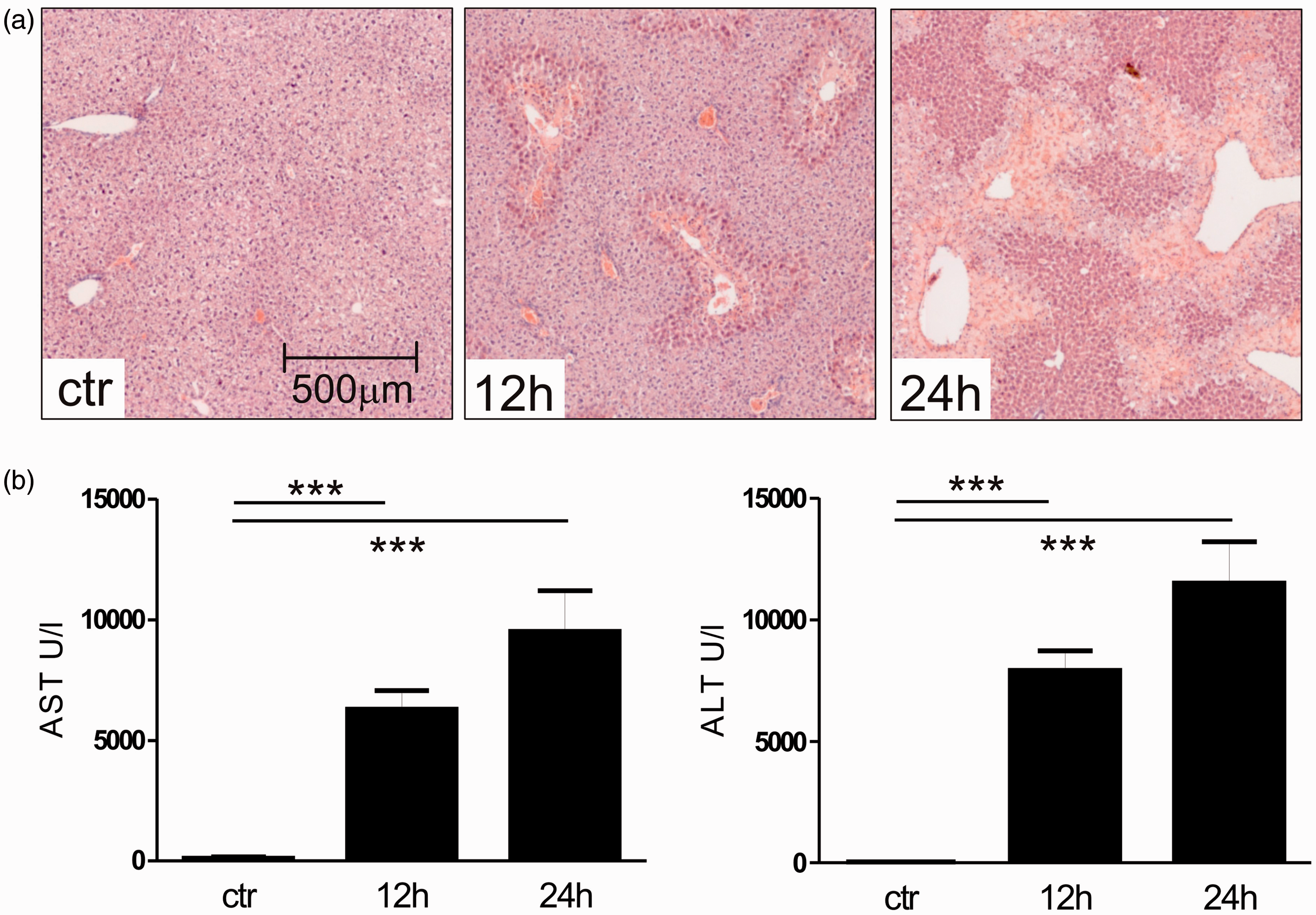
Practical implementation
Fasting of mice should be done in a clean cage, without food, but with free access to water. Care must be taken that no deposit of chow is left in the cage. After a fasting period of 12 h, mice should be weighed and examined for signs of distress (i.e. changes in respiration, rough hair coat, unusual behaviour, hunched posture). Animals showing these abnormalities should be excluded from the study and euthanized according to humane endpoints. Acetaminophen (for example, Sigma Aldrich Cat. No. A3035) should be dissolved in 0.9% sodium chloride (NaCl 0.9%) at a concentration of 15 mg/mL at 30℃ in a water bath. The volume required for the single administration of APAP depends on the concentration of the working solution, body weight of the mice and route of injection. Intraperitoneal injection: APAP is injected at doses of 250 mg/kg body weight up to 500 mg/kg body weight via intraperitoneal injection into the lower side of the abdomen. As the highest possible concentration of APAP in sodium chloride solution is limited to about 15 mg/mL (at 30℃), the appropriate injection volume will vary according to the body weight of the animals.
We recommend a dose of 300 mg/kg body weight after a fasting period of 12 h. As an example, a mouse with a total body weight of 20 g needs to be injected intraperitoneally with 400 µL APAP solution (15 mg/mL).
This volume is slightly higher than the maximal injection volume recommended according to the guiding principles of the Society for Laboratory Animal Science (GV SOLAS).
12
Due to the limited solubility of acetaminophen, it is not possible to work reproducibly with higher concentrated solutions.
13
It is also not advisable to split the APAP dose into two injections, as this would cause additional stress to the mice and might increase variability due to diverse absorption rates from different injection sites.
Intravenous injection: APAP can also be injected intravenously via the lateral tail vein. We recommend a dose of 250 mg/kg body weight after a fasting period of 12 h. For intravenous application, it is important to ensure that the compound is completely dissolved to avoid complications like embolisms. Alternatively, commercially available solutions of dissolved APAP (e.g. Paracetamol Actavis® 10 mg/mL) can be used. APAP solution should be injected in mice using disposable, sterile syringes. For intraperitoneal injection the mouse should be fixed behind the neck region between the thumb and forefinger. After turning over, the animal should rest on the palm against the base of the thumb using a third finger to stabilize the pelvic region with the head down. APAP should be injected into the lower side of the abdomen avoiding injury to the bladder, diaphragm or intestine using a 27 gauge needle (e.g. 27 G Microlance; Becton Dickinson, Franklin Lakes, NJ, USA). Intraperitoneal injection can lead to local irritation of the skin. For intravenous injection, mice should be placed in a restrainer. To expand the veins, the tail should be dipped in warm water (35℃) or should be warmed up under a heat lamp. During this process, the breath frequency and behaviour of the animal needs to be continuously monitored. APAP should be injected into the lateral tail vein using a 27 gauge needle (see above). After APAP administration, mice should be allowed free access to food. Mice should be re-inspected regularly, for instance, after one hour and every 12 h after injection. At the end of the experiment (e.g. after 24 h), animals should be sacrificed by blood withdrawal from the heart using deep isoflurane anaesthesia.
Animal burden/side-effects
In general, handling mice (i.e. taking them out of their cage, fixation, and exposure to a foreign surrounding) is already stressful for them. To reduce the burden of the animals only trained laboratory personnel should be allowed to perform the application of APAP ensuring short and precise animal handling times. Mice treated with an overdose of APAP develop an acute liver injury resulting in dose-dependent liver failure. Acute liver injury is not associated with pain in humans, and mice subjected to this model also do not show signs of pain. However, the animals show signs of fatigue and move more slowly within the first 12 h after injection of APAP. We assume that the treatment with APAP itself reduces possible slight abdominal irritations because of its action as a pain killer.
When the model is established for the first time in a research unit, the animals have to be observed every hour. Due to difficulties in dissolving APAP and due to differences depending on the mouse strain, sex, genetic background and duration of the fasting period, it is possible that the mice may die due to the liver damage. If the mice have low body temperature (hypothermia <35℃), show changes in respiration and are not easy to wake, they need to be euthanized. In case of high (lethal) APAP concentrations, the animals may die due to the typical complications of ALF such as cerebral oedema and encephalopathy, coagulopathy, renal, haemodynamic and cardiorespiratory failure. 2 Very likely, the decreased hepatic metabolizing capacity for APAP in high doses or longer observational studies may render secondary target organs, such as the kidneys, susceptible to the harmful side-effects of APAP, allowing the study of both hepatotoxicity and nephrotoxicity of APAP in these scenarios. 14
Classification of severity of procedure
Based on our experiences and according to Article 15 of the EU Directive 2010/63 (http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri = OJ:L:2010:276:0033:0079:en:PDF) the estimated degree of pain, suffering, distress or lasting harm of the animals subjected to APAP application should be classified.
Details about the classification criteria underlying this assessment have been established by the Expert Working Group on severity classification of scientific procedures performed on animals. These can be found at: http://ec.europa.eu/environment/chemicals/lab_animals/pdf/report_ewg.pdf.
The APAP application to induce liver failure with no major impairment of liver function is classified as a moderate procedure according to Article 15 of the above-mentioned EU Directive 2010/63. The induction of liver failure and death due to the typical complications of ALF such as cerebral oedema and encephalopathy, coagulopathy, renal, haemodynamic and cardiorespiratory failure is classified as a severe procedure. In line with the 3R principle, the procedure should undergo a refinement, and humane endpoints must be implemented with frequent observation points to restrict pain, suffering, distress or lasting harm to the animals. Then the procedure can be reclassified as moderate.
Concluding remarks
Administration of acetaminophen (overdose) is a key model in experimental liver research, because this is the only model for acute liver injury in mice, which directly corresponds to the cause of a leading entity of ALF in humans. The model is robust and leads to highly reproducible results. In our view, the best established route is the application via intraperitoneal injection, but intravenous injection might be an interesting option for gaining further comparable results and for reducing the number of experimental animals. This SOP gives detailed recommendations for technical and biometric standards, facilitating reproducible animal experimentation according to the new European Animal Welfare Regulations.
Ethical statement
The experiments were performed in accordance with the German legislation governing animal studies following the ‘Guide for the care and use of laboratory animals’ (NIH publication, 8th edition, 2011) and the Directive 2010/63/EU on the protection of animals used for scientific purposes (Official Journal of the European Union, 2010). Official permission was granted from the governmental animal care and use office (LANUV Nordrhein-Westfalen, Recklinghausen, Germany).
Footnotes
Acknowledgements
The authors thank all members of the Tacke Lab and the Department of Medicine III for helpful discussions.
Funding
This work was supported by the German Research Foundation (DFG SFB/TRR57). The authors disclose no conflict of interests.
