Abstract
The ovine model is generally considered to be the best for testing bioprosthetic heart valve durability. Although echocardiography is the method of choice for the interim evaluation of the valve, literature on sheep echocardiography is scarce. Within the context of a study on treatment of pericardial heart valve prostheses, 19 adolescent sheep underwent transthoracic echocardiography six days after mitral implantation of bioprosthetic valves. Echocardiographic examination was performed under mild anesthesia and animals were put in a right lateral decubitus position. Four images were obtained: right parasternal long axis four and five chamber views, right parasternal long axis view with left ventricular outflow, and right parasternal short axis view through the mitral valve. We measured aortic annulus and velocity time integral over the aortic valve to determine stroke volume, cardiac output and cardiac index. The mitral valve was evaluated through color Doppler imaging for valvular and paravalvular leakages. Pulsed wave spectral Doppler was used for the measurement of velocities, pressures and velocity time integrals. For the evaluation of valve stenosis deceleration time and pressure half-time were determined. Effective orifice area of the mitral valve was derived. And, although not measured, other structures could clearly be visualized: right and left ventricle and atrium, wall thicknesses, tricuspid valve. This study shows that echocardiography in sheep is feasible, and that right parasternal images, obtained in animals in a right lateral decubitus position, are well qualified for the interim evaluation of bioprosthetic valves implanted in the mitral position. Besides the implanted valve, other cardiac structures like atria and ventricles can be visualized and evaluated.
Valve replacement surgery has dramatically improved outcome of patients with valvular heart disease. Approximately half of cardiac valve replacements are performed using bioprosthetic valves made of bovine pericardium or porcine valve tissue. Besides in vitro testing of new bioprosthetic heart valves, in vivo testing of these devices in an animal model is crucial. Implantation in a living animal gives valuable information on parameters not ascertainable by in vitro tests. Not only can technical and surgical particularities of the valve be assessed but valve characteristics and influence of different treatment regimens on hemolysis, thrombosis and valve degeneration and calcification can also be evaluated.
The ovine model is generally considered to be the model of choice for testing cardiac valves. Although the species is not specified in the ISO guidelines for cardiovascular implants and cardiac valve prostheses (ISO 5840:2005), Gallegos et al. have shown that almost 80% of large animals used for testing heart valves are sheep. 1 This species offers different advantages over other large animals like pigs or calves, especially regarding their limited somatic growth which avoids important adverse effects like paravalvular leakage and increase in cardiac output. Besides, sheep are widely available, easy to keep, and their cardiovascular anatomy and physiology closely mimic humans.
Although echocardiography is the method of choice for the interim evaluation of implanted heart valves, literature on sheep echocardiography, especially after bioprosthetic mitral valve implantation, is limited. Several studies have been conducted to determine echocardiographic values in normal, healthy sheep.2–5 And although echocardiographic evaluation of mitral valves is mentioned in different studies, no details have been provided on methodology or parameters and characteristics of the implanted mitral valve. The aim of the present study is, therefore, to describe the methodology of echocardiographic evaluation of a biological heart valve prosthesis implanted in the ovine mitral position and to report echocardiographic features of such a valve one week post-operatively.
Materials and methods
Study protocol
Within the context of a study on tissue treatment of biological heart valves, 19 sheep underwent echocardiographic evaluation of the mitral valve one week after mitral valve replacement surgery. For study purposes two types of bioprosthetic valves were implanted in the mitral position: nine sheep received Pericarbon More valves (Sorin Group, Saluggia, Italy) and 10 animals received Mitroflow valves (Sorin Group), all 23 mm in size. Since the objective of this paper is to describe the methodology of post-operative echocardiographic evaluation of a biological valve implanted in the mitral position, treatment regimens of the valves and further evaluation of the valves are not discussed. The study protocol was approved by the ethical committee of our institution (Ethische Commissie Dierproeven, KU Leuven ECD P097/2012) and all animals received humane care in compliance with the Guide for the care and use of laboratory animals published by the National Institutes of Health (8th edition, 2011).
Animals
All the sheep were female and were less than 10 months old at the time of surgery. They were Swifter sheep, except for three which were a mixed breed of Swifter × Charolais. All animals were obtained from Zootechnisch Centrum KU Leuven, Lovenjoel, Belgium. Prior to surgery all animals were examined by a veterinarian to exclude existing cardiac problems and signs of infection.
Surgery
After premedication with ketamine 15 mg/kg intramuscularly (Anesketin; Eurovet, Heusden-Zolder, Belgium) the animals were induced with isoflurane (Iso-Vet, Eurovet) through a mask. Isoflurane was also used for maintenance. Surgery was performed as previously described and included cardiopulmonary bypass. 6 Analgesia was performed with 0.5 mg/kg meloxicam intravenously (Metacam; Boehringer Ingelheim Vetmedica, Ingelheim am Rhein, Germany).
The first days after surgery, the animals were thoroughly examined by a veterinarian and antibiotics (15 mg/kg amoxicillin intramuscularly, Clamoxyl LA; Pfizer Animal Health, Louvain-la-Neuve, Belgium), anticoagulants (20 mg enoxaparine subcutaneously, Clexane; Sanofi Aventis, Paris, France), diuretics (20 mg furosemide intramuscularly, Lasix; Sanofi Aventis) and analgesia (0.5 mg/kg meloxicam subcutaneously) were given. The animals were allowed to recover properly from prior surgery and anesthesia, and post-operative echocardiography was consequently performed one week after surgery.
Echocardiography
After premedication with 15 mg/kg ketamine intramuscularly, echocardiographic examination was performed under isoflurane anesthesia delivered through a mask. Animals were positioned in a right lateral decubitus position on a custom-made table containing a cut-out at the level of the chest (Figure 1). Therefore, images could be obtained from the right side of the animal, reducing the distance between the heart and the ultrasound probe.
Position of the sheep for echocardiographic examination on a scanning table with a cut-out at the level of the thorax. Right parasternal images are obtained from below the animal.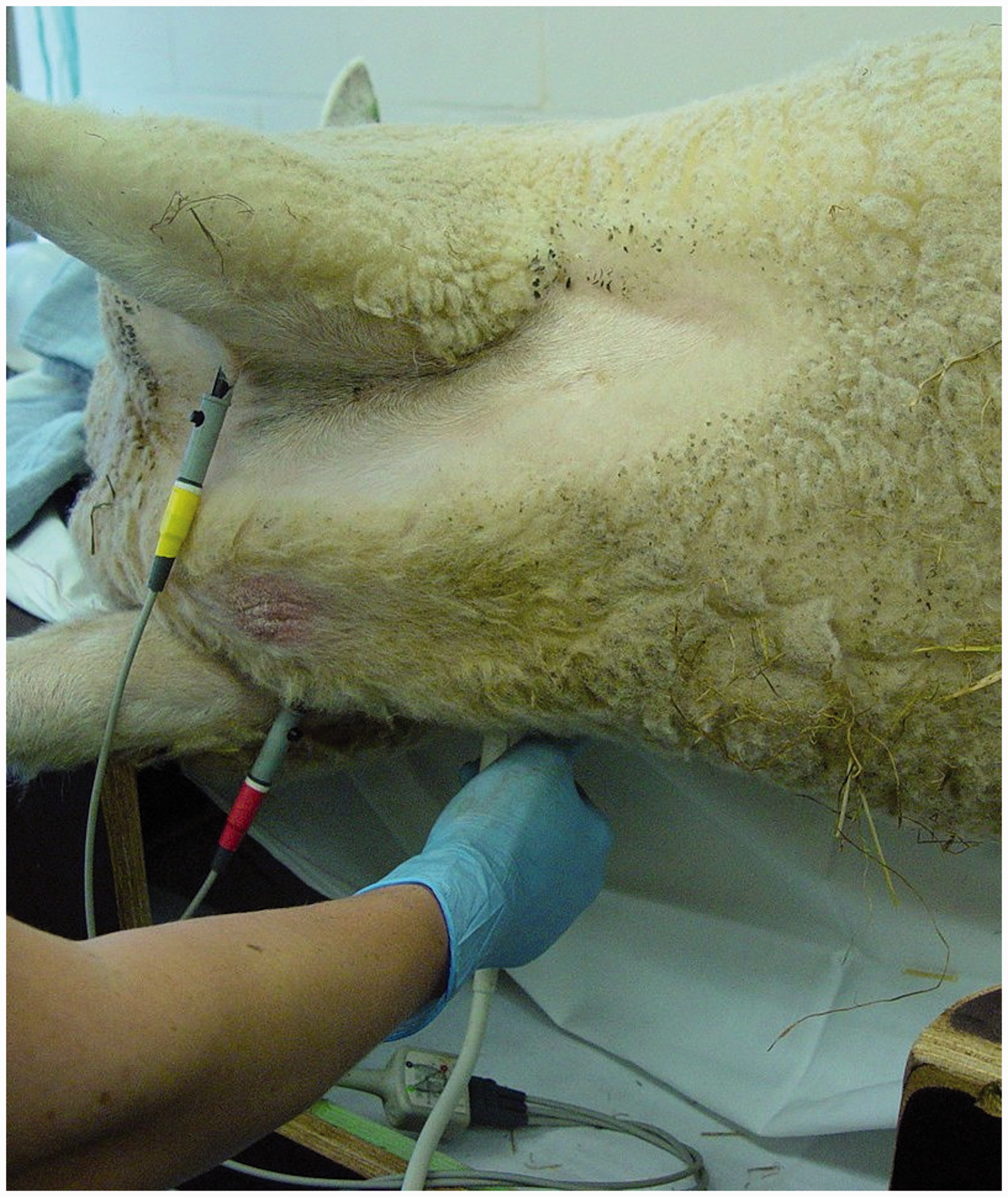
All images were obtained by the same person (SDV) using a Vivid 7 (GE Medical Systems, Chalfont St Giles, UK) and a 5.0 MHz phased array probe. Electrocardiography was monitored throughout the examination and echocardiographic examination started when the heart rate stabilized. Each measurement was repeated three times and the results were averaged afterwards.
For the evaluation of the valve, four images were obtained: right parasternal long axis four and five chamber views, right parasternal long axis view with left ventricular outflow, and right parasternal short axis view through the mitral valve. These images were obtained in the third to fifth intercostal space, in the alopecic area of the axilla, making clipping unnecessary. Usually only one window could be used to obtain good qualitative images. Obtaining apical views in sheep is difficult due to the very wide ovine sternum and the position of the heart in the chest.
Right parasternal long axis four chamber view
In this view (Figure 2), the mitral valve was evaluated in both two dimensional (2D) and color Doppler modes. In the 2D mode, the general appearance of the valve was evaluated, and leaflet thickness, movement and motility were assessed. During color Doppler evaluation, valvular and paravalvular leakages were looked for. Flow through the mitral valve was evaluated by pulsed wave Doppler placing the sample gate at the tips of the open leaflets. Velocities (Vmax and mean), pressure gradients (Pmax and mean), velocity time integral, deceleration time and pressure half-time were recorded. Effective orifice area was calculated using the continuity equation: effective orifice area = stroke volume/mitral velocity time integral.
7
Right parasternal long axis four chamber view. (a) 2D echocardiographic image showing the left atrium (LA), right atrium (RA), left ventricle (LV) and right ventricle (RV). The implanted mitral valve is clearly visualized. (b) color Doppler through the mitral valve to evaluate valvular and paravalvular leakages.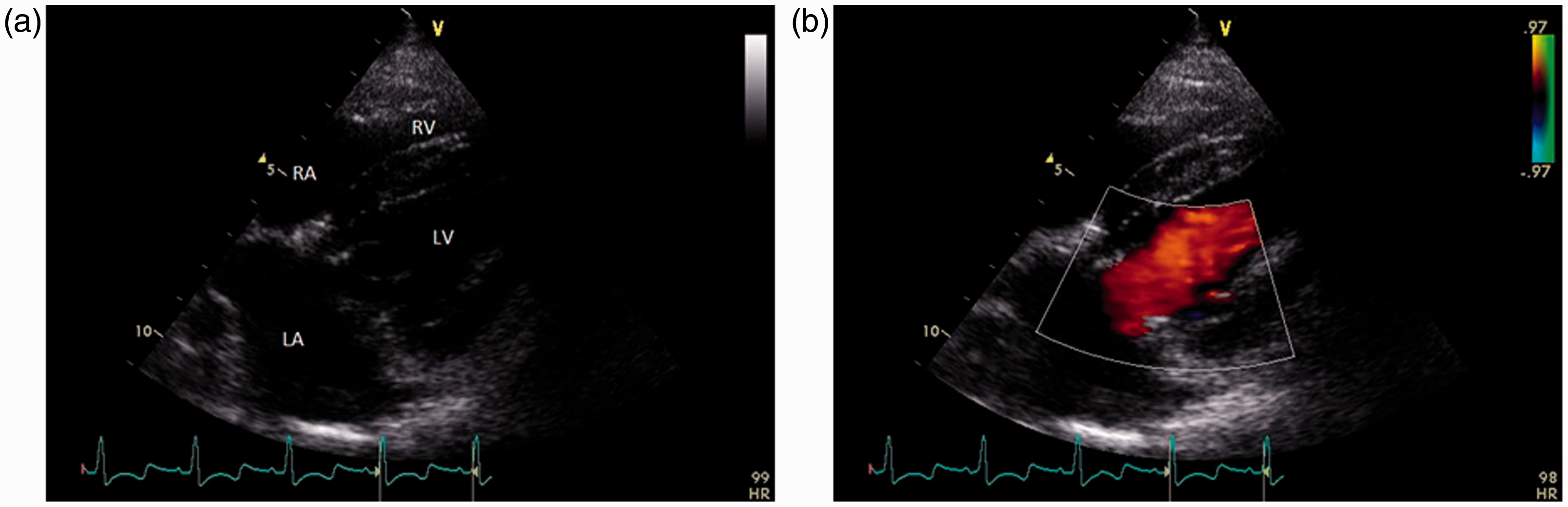
Right parasternal long axis five chamber view
In this view (Figure 3), the evaluation of the mitral valve was repeated by 2D and color Doppler imaging. Aortic velocity time integral was recorded through pulsed wave Doppler for evaluation of stroke volume, cardiac output and cardiac index. The pulsed wave Doppler gate was positioned in the left ventricular outflow tract. Cardiac output was measured using the following equation: heart rate × stroke volume with stroke volume = πr (annulus/2)2 × aortic velocity time integral. The cardiac index is the cardiac output divided by the body surface area. Body surface area was calculated using the formula generally accepted for animals: body surface area = 0.101 × 0.67 body weight.
8
Right parasternal long axis five chamber view. LA: left atrium; RA: right atrium; LV: left ventricle; RV: right ventricle; Ao: aorta.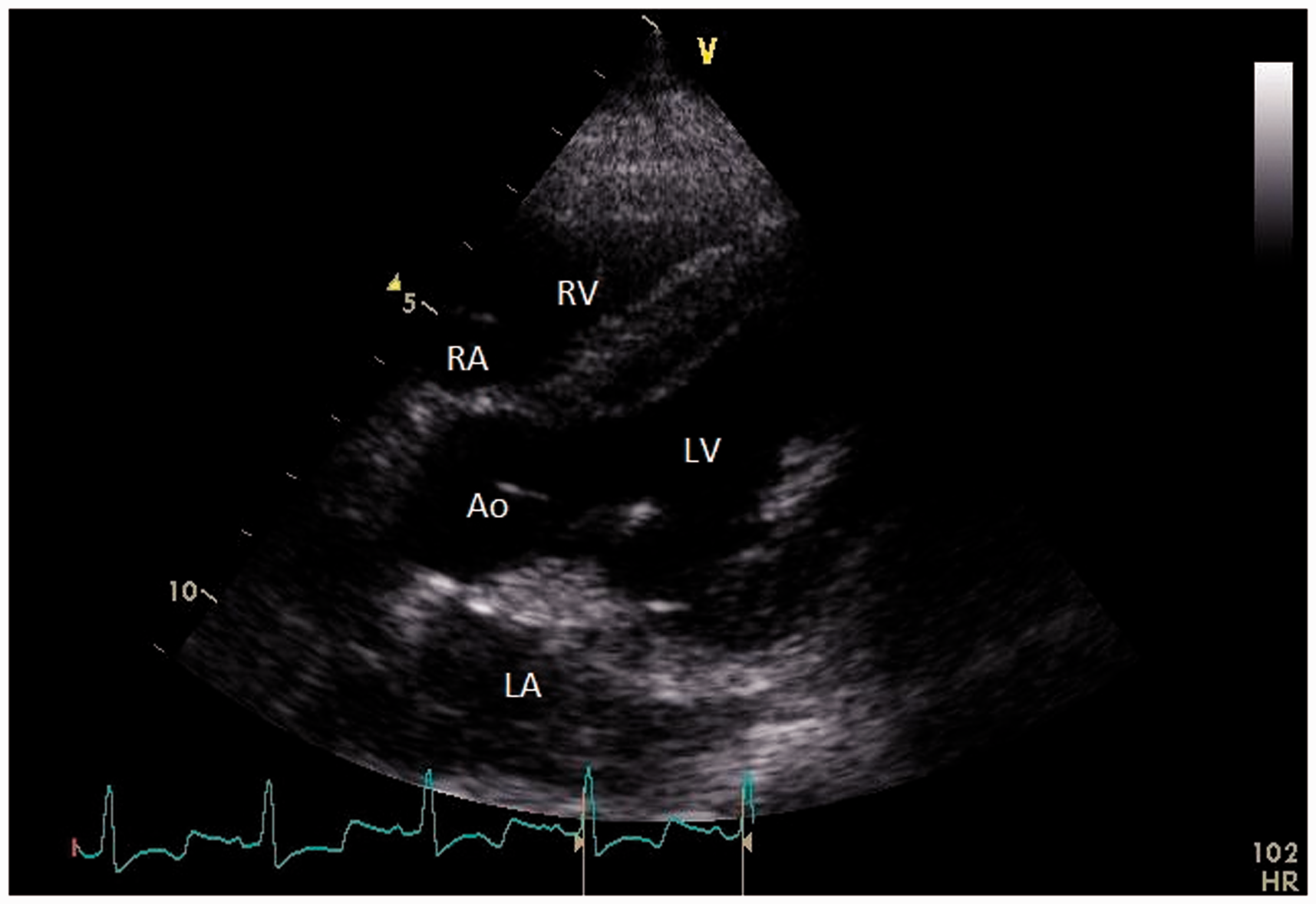
Right parasternal long axis with left ventricular outflow view
In this view (Figure 4), the size of the aortic annulus (Figure 4b) was measured to evaluate cardiac output and cardiac index. In this view, as well as in the other views, the general appearance of the heart was also evaluated (wall thickness of left and right ventricles, tricuspid valve, size of atria and ventricles, presence of pericardial fluid) but were not recorded unless visually abnormal.
Right parasternal long axis with left ventricular outflow view (a), in this image the annulus of the aorta can be measured (b). LA: left atrium; RA: right atrium; LV: left ventricle; RV: right ventricle; Ao: aorta.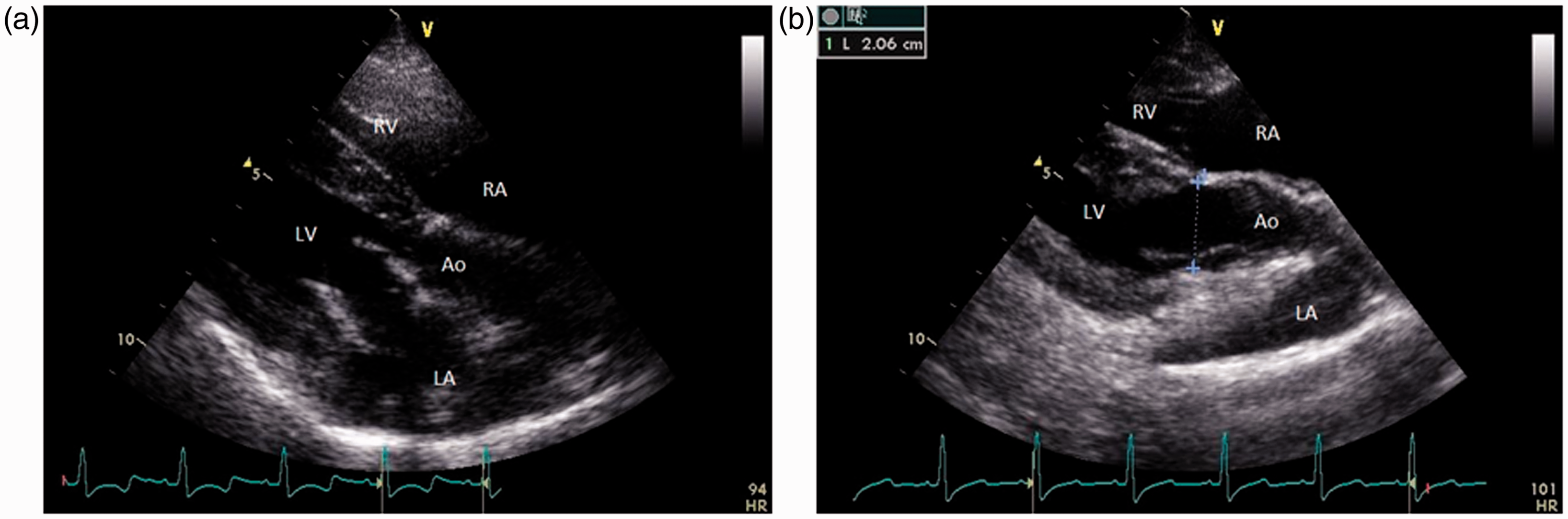
Short axis view
The short axis view (Figure 5) was taken through the mitral valve. Leaflet thickness, movement and motility as well as opening and closure of the orifice were evaluated with the 2D method. Color Doppler is also applied in this view to evaluate valvular and paravalvular leakages.
Right parasternal, short axis view through the mitral valve. RV: right ventricle; MV: mitral valve.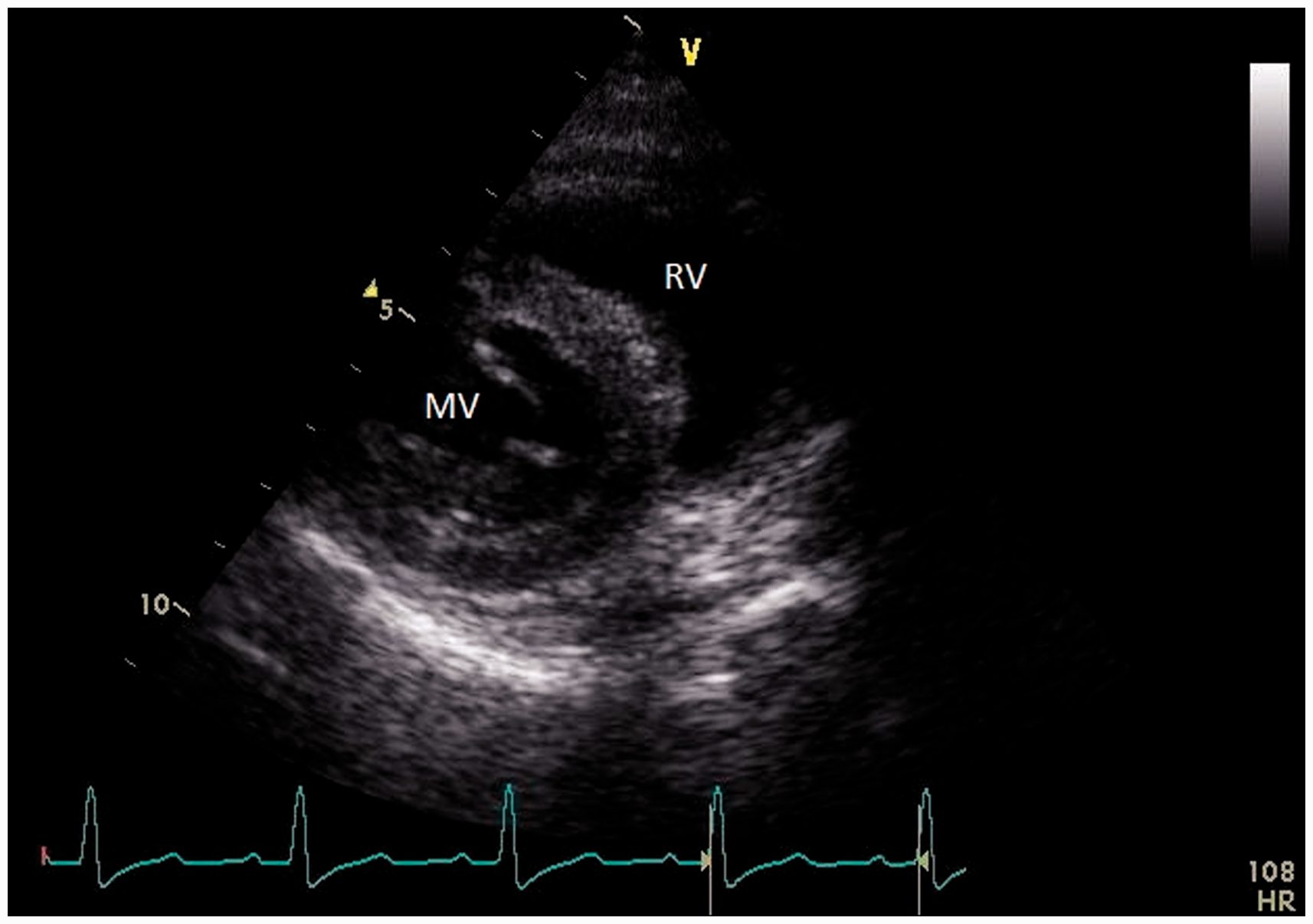
Overview of the different echocardiographic views including structures which can be visualized, and which structures and parameters were assessed for this study.

Statistical analysis
Statistical analysis was performed using Graphpad Prism 6 (Graphpad Software Inc, San Diego, CA, USA). Differences between the two groups were assessed by unpaired t-tests. Results are reported as median values (interquartile range) and P values <0.05 were considered to be statistically significant.
Results
General
Echocardiographic evaluation was performed at a median of six days post-operatively, and good echocardiographic images could be obtained in all animals although some animals were less echogenic. Although not measured, all echocardiographic exams could be performed within 15–20 min, limiting the duration of anesthesia and allowing the quick recovery of the sheep.
Animals had a median age of 229 (197–273) days at the time of surgery with no difference in age between the groups receiving the Pericarbon More and the Mitroflow valves.
Median weight of the animals at the time of echocardiography was 26.5 (24.1–28.5) kg with no difference between both groups. Body surface area was 0.91 (0.85–0.95) m2 for the whole group. Median heart rate of the animals, reported here as the heart rate assessed during aortic velocity time integral measurements, was 110 (104–126) bpm.
Echocardiographic data of sheep one week after bioprosthetic valve implantation in a mitral position.
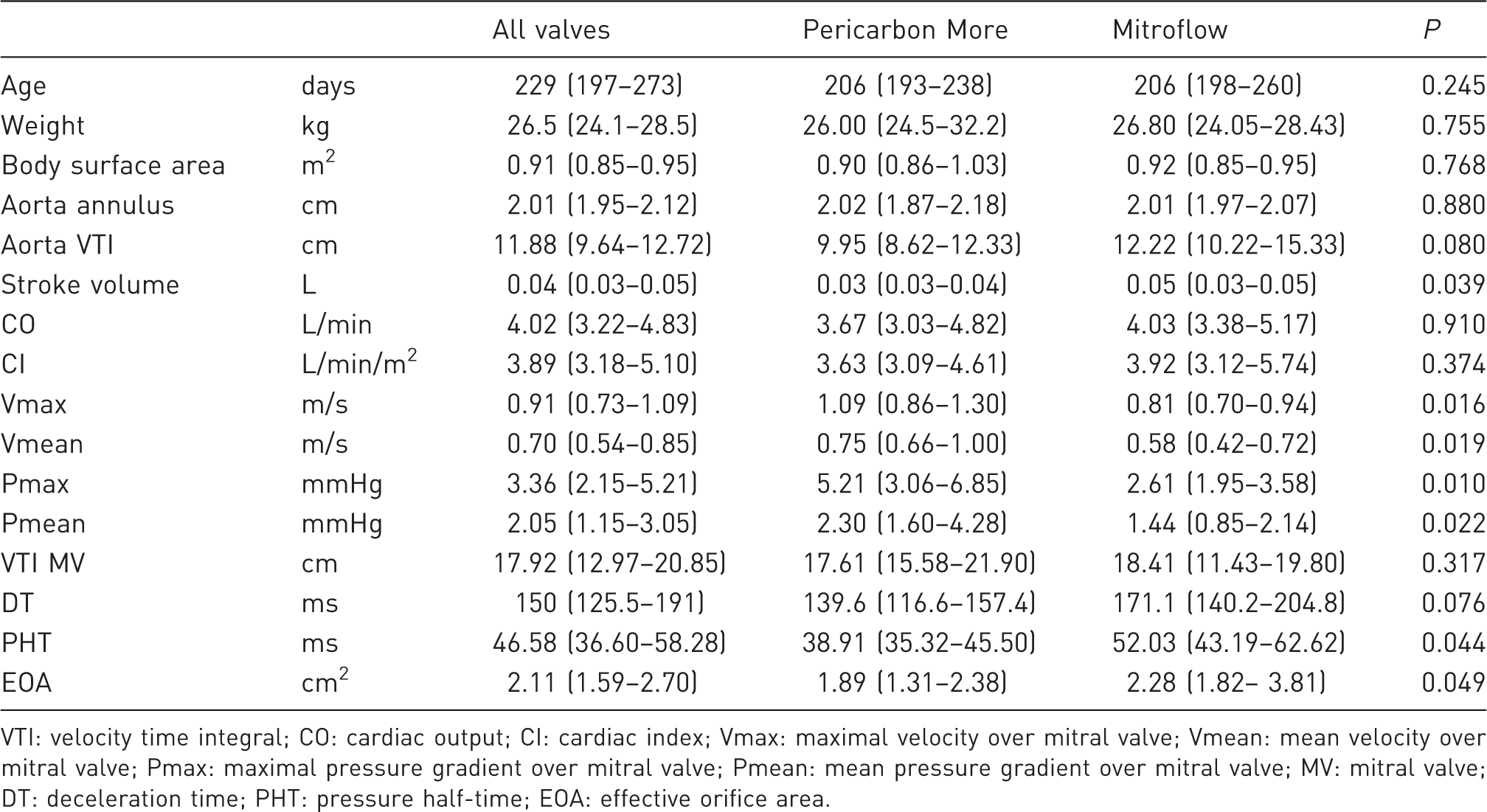
VTI: velocity time integral; CO: cardiac output; CI: cardiac index; Vmax: maximal velocity over mitral valve; Vmean: mean velocity over mitral valve; Pmax: maximal pressure gradient over mitral valve; Pmean: mean pressure gradient over mitral valve; MV: mitral valve; DT: deceleration time; PHT: pressure half-time; EOA: effective orifice area.
Stroke volume, cardiac output and cardiac index
The aortic annulus was 2.01 (1.95–2.12) cm for the whole group, 2.02 (1.87–2.18) cm in the Pericarbon More implanted sheep and 2.01 (1.97–2.07) cm in the Mitroflow implanted sheep (P = 0.880). Median velocity time integral of the aorta was 11.88 (9.64–12.72) cm for the whole group, 9.95 (8.62–12.33) cm for the Pericarbon More valves and 12.22 (10.22–15.33) cm for the Mitroflow valves. This difference was not statistically significant (P = 0.080).
Median stroke volume was 0.04 (0.03–0.05) L. In the Pericarbon More group, stroke volume was 0.03 (0.03–0.04) L and this was significantly lower than in the Mitroflow group in which the stroke volume was 0.05 (0.03–0.05) L (P = 0.039). Median cardiac output was 4.02 (3.22–4.83) L/min for the whole group, 3.67 (3.03–4.82) L/min for the Pericarbon More valves and 4.03 (3.38–5.17) L/min for the Mitroflow valves (P = 0.910). Overall cardiac index was 3.89 (3.18–5.10) L/min/m2 for the whole group, 3.63 (3.09–4.61) L/min/m2 for the Pericarbon More implanted sheep versus 3.92 (3.12–5.74) L/min/m2 for the Mitroflow valves. Again this difference was not statistically significant (P = 0.374).
Evaluation of the bioprosthesis implanted in mitral position
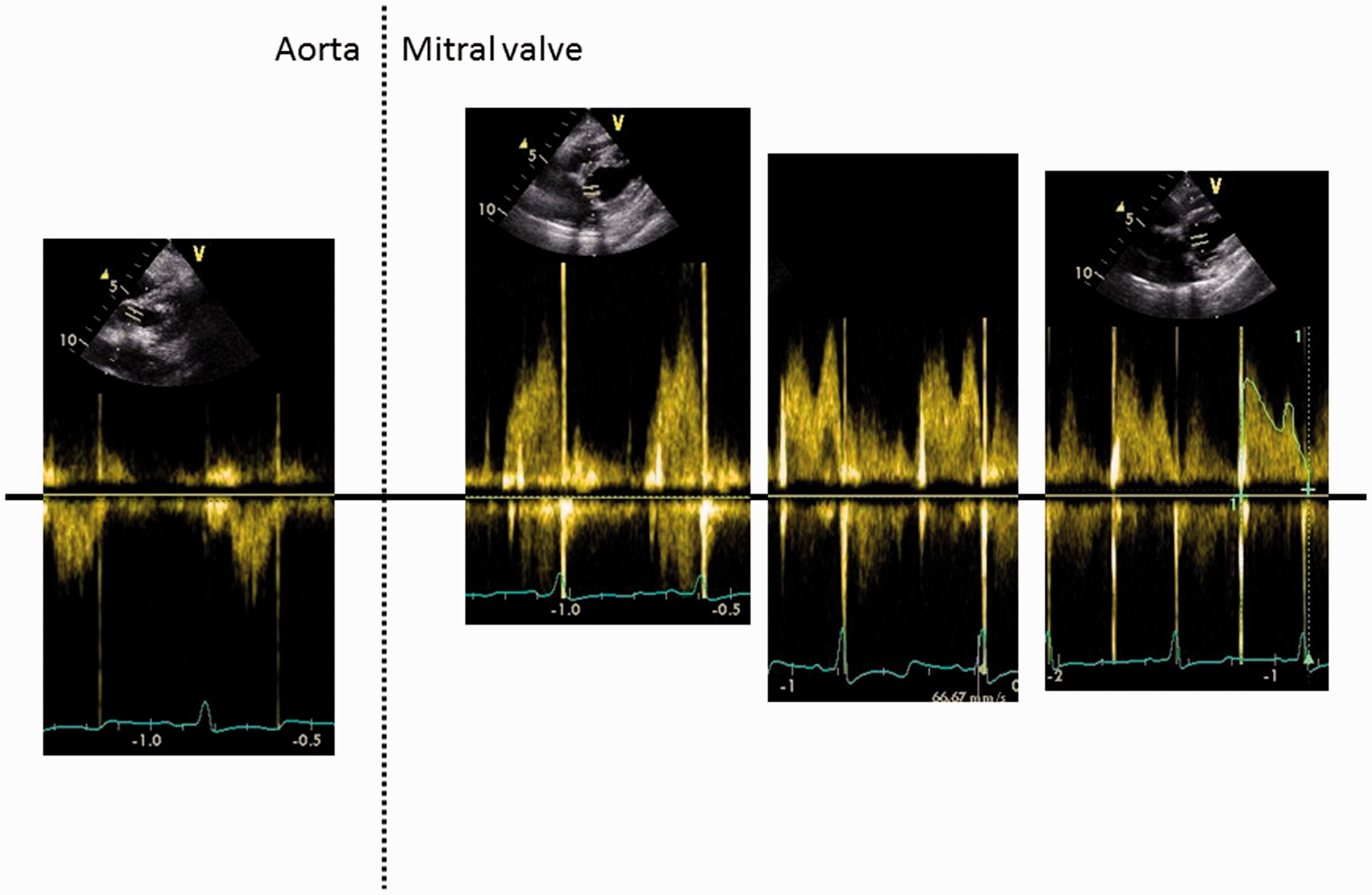
None of the implanted valves demonstrated structural valve deterioration. An echocardiographic image of both types of valves, which are both open and obtained in right parasternal four chamber view is given in Figure 6. One of the sheep in the Mitroflow group showed a mild valvular regurgitation, graded ¼. Transmitral flow patterns differed within and between animals depending on the heart rate, going from a clear distinction between E and A peaks to total summation of these peaks. When the E and A peaks were distinguishable, E could be larger, much larger or smaller than the A peak (Figure 7).
Right parasternal long axis four chamber view. (a) pericarbon More valve. (b) mitroflow valve. Flow patterns through the aortic and mitral valves. The left panel shows an aortic flow pattern; the right side shows three different patterns of mitral flow with early and late diastolic velocities which could have different shapes.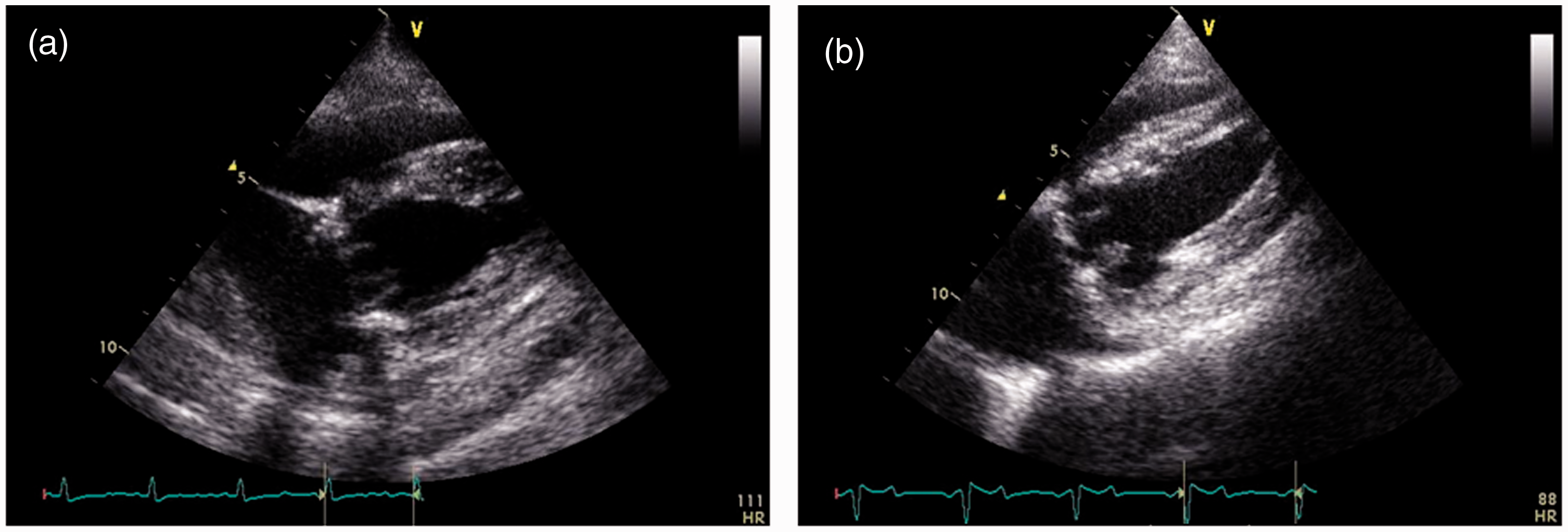
Median maximal velocity over the implanted valve was 0.91 (0.73–1.09) m/s for the whole group. There was a significant difference between both groups regarding maximal velocity, with higher peak velocities in the Pericarbon More valves than in the Mitroflow valves: 1.09 (0.86–1.30) m/s versus 0.81 (0.70–0.94) m/s, respectively (P = 0.016). The same was true for mean velocities, with mean velocities of 0.70 (0.54–0.85) m/s for the whole group, 0.75 (0.66–1.00) m/s for the Pericarbon More valves and 0.58 (0.42–0.72) m/s for the Mitroflow valves (P = 0.019).
The peak pressure gradient over the bioprosthesis was 3.36 (2.15–5.21) mmHg for the whole group, with higher peak pressure gradients in the Pericarbon More valves (5.21 [3.06–6.85] mmHg) than in the Mitroflow valves (2.61 [1.95–3.58] mmHg; P = 0.0099). Mean transvalvular pressure gradients were 2.05 (1.15–3.05) mmHg in the whole group, 2.30 (1.60–4.28) mmHg in the Pericarbon More valves and 1.44 (0.85–2.14) mmHg in the Mitroflow valves (P = 0.022).
Velocity time integral of the implanted valve was 17.92 (12.97–20.85) cm for the whole group, and there were no differences between the Pericarbon More valves and the Mitroflow valves with velocity time integral values of 17.61 (15.58–21.90) cm and 18.41 (11.43–19.79) cm, respectively (P = 0.317).
Deceleration time was 150 (126–191) ms for the whole group, 140 (117–157) ms for the Pericarbon More group and 171 (140–205) ms for the Mitroflow group (P = 0.076).
Median pressure half-times were 46.6 (36.6–58.3) ms in the whole group. Pressure half-times were significantly lower in the Pericarbon More group: 38.9 (35.3–45.5) ms versus 52.0 (43.2–62.6) ms in the Mitroflow group (P = 0.044).
Overall the effective orifice area was 2.11 (1.59–2.7) cm2. In the Pericarbon More group the effective orifice area was 1.89 (1.31–2.38) cm2 and in the Mitroflow group the effective orifice area was significantly bigger: 2.28 (1.82–3.81) cm2 (P = 0.049).
Discussion
The aim of this study was to describe the methodology of echocardiographic evaluation of mitral valve implants in sheep and to determine the echocardiographic parameters of biological mitral valve prostheses implanted in the mitral position one week after surgery.
Finding an optimal tissue treatment for improving device durability of bioprosthetic heart valves is still an important challenge, and testing these different treatment regimens in an animal model is indispensable. According to the ISO guidelines for cardiovascular implants and cardiac valve prostheses (ISO 5840:2005), these animals should survive for a minimum of 20 weeks. Besides clinical examination of the animal, echocardiography is the method of choice for the interim evaluation of the implanted valve. Echocardiography allows us to obtain information on the implanted valve in an animal under mild anesthesia. This can be done quickly and non-invasively with minimal side-effects and a short recovery time. Although sheep are most often used for testing heart valves, the literature on sheep echocardiography is scarce.
Besides some studies conducted to assess echocardiographic values in normal sheep2–5, echocardiography is described in several papers which evaluate mitral valves. However the present study identified some important differences. The first studies describing echocardiographic evaluation of implanted, often stentless, mitral valves in the 1990s used either transesophageal echocardiography9,10 or epicardial echocardiography10,11 or the valves were evaluated in vitro. 12 Leaflet motion, vegetation, calcification, leakage through color flow Doppler, velocities, pressure gradients and effective orifice areas were evaluated. Epicardial echocardiography was reported immediately before and after surgery in acute experiments11,13 or at the time of autopsy about five months after implantation. 10 Epicardial echocardiography is, however, for obvious reasons, not a good tool for interim evaluation. Transesophageal echocardiography was also reported immediately after surgery 10 or as means of follow-up after mitral valve implantation. 9 However the latter reported some technical difficulties with the temperature sensor. Due to the physiologically higher body temperature of sheep, and consequently the small difference with the safety circuit, the system automatically shut down quickly. Besides these technical difficulties with transesophageal echocardiography, transthoracic echocardiography has the advantage of being less invasive and it can be performed on animals under mild anesthesia or even without anesthesia.
To obtain good qualitative echocardiographic images, movement of the animals should be minimized. This is especially true for the measurements/evaluations done requiring moving images like the assessment of valvular leakage using color Doppler wherein a stable position of the gate is necessary. Echocardiographic examination of sheep has been described in awake, unsedated, standing animals2,3. Although (frequent) anesthesia could be avoided, performing these examinations could require more training and more personnel to restrain the animal if images are to be taken in a right decubitus position. Another option is to restrain the sheep in a hoof trimming device, but although this immobilizes the animals, stress and tachycardia will most likely occur. Tachycardia will inevitably affect different echocardiographic measurements (e.g. showing no distinction between E and A peaks). So far, no comparison has been made between the quality of images obtained in standing, unsedated animals and images obtained in a right decubitus position in sedated animals. We are currently also exploring the possibility of performing echocardiography in awake animals, but several questions need to be answered first by means of some pilot experiments like position and restraining method of the sheep, quality of images obtained, etc.
Vetter et al. 12 studied echocardiographic characteristics of a stentless mitral valve in an in vitro set-up wherein heart rate and cardiac output could be adjusted. More recently, echocardiography has also been described in studies evaluating mitral annuloplasty by different groups.14–16
The only study describing transthoracic echocardiographic evaluation of the mitral valve was published previously by our group in 2007. 17 Recently, our group adopted another technique for performing transthoracic echocardiography by placing the ultrasound probe underneath the animal and obtaining right parasternal images instead of left parasternal images obtained from sheep in a right lateral decubitus position, with the latter increasing the distance between the heart and ultrasound probe. This technique, for post-operative mitral valve evaluation, has never been described in detail before and was therefore the aim of the present study.
In contrast to what has been described previously, 4 early diastolic (E peak) and late diastolic (A peak) flow in the transmitral flow spectrum did not show the typical M shape in all animals. E was not always bigger than A, and both were not always separated from each other. These differences in transmitral flow shape were seen both within and between sheep (Figure 7). Since transmitral flow patterns in this study were used to assess the mitral valve and not the diastolic function of the heart, the shape of this pattern and the relationship between early and late diastolic function are less important.
Although the aim of this study was not to compare Mitroflow and Pericarbon More bioprostheses, we could see some significant differences between both types of valves. The Pericarbon More valves showed higher peak and mean velocities and higher peak and mean pressures. Effective orifice area was also smaller in these valves, which could explain the higher transprosthetic velocities and pressure gradients. All these differences can be explained by a difference in design between both types of valves. While in Mitroflow valves the pericardial sheet is mounted externally on the stent, in the Pericarbon More valves the pericardium is sutured inside the ring, explaining the smaller effective orifice area.
In 2009, the American Society of Echocardiography published some recommendations for the evaluation of prosthetic valves by using echocardiography and Doppler ultrasound. 18 According to these, mitral valves should be evaluated by 2D echocardiography in both parasternal and apical images, and Doppler images should include assessment of peak velocities, mean pressure gradients and pressure half-times. Although parasternal images in humans are taken on the left side, qualitatively good parasternal images can be obtained in animals from the right side by putting them in a right lateral decubitus position. 19 This is due to the anatomical features of the animal thorax which is not dorsoventrally flattened as in humans. Obtaining true apical images is, however, difficult in sheep due to the anatomical relation between the heart and the sternum, making it very difficult to align the ultrasound probe with the heart to obtain these images. Color Doppler and Doppler flow parameters were, therefore, obtained from parasternal images instead of from apical images. The Doppler beam was aligned with the flow as parallel as possible, which resulted in transmitral flows of a sufficient quality. Some underestimation and variation of velocities and consequently of pressure gradients were however likely. Nevertheless, these measurements will most often be repeated in follow-up studies, and valuable information on valve insufficiency and stenosis can be obtained by comparing data taken at different time-points.
In conclusion, we can say that it is feasible to perform transthoracic echocardiography in sheep and that this is a good tool for evaluating bioprosthetic valves in a mitral position. Besides evaluation of the mitral valve implant, other cardiac structures and features like atria and ventricles, tricuspid valve and pericardial fluid, can also be assessed in sheep in right parasternal images obtained in sheep in a right lateral decubitus position.
Footnotes
Funding and Acknowledgements
All bioprosthetic valves were provided by the Sorin Group. The Sorin Group designed the study regarding the treatment of the valves. They had, however, no role in data collection, analysis and interpretation or in the writing and submission of this paper.
Conflict of interest statement
The authors did not receive any benefit, including financial, by reporting the data of this paper.
