Abstract
We have investigated by immunochemistry the distribution of H Type 3/4 chains of the ABO histo-blood group system in human submandibular gland using a monoclonal anti-H MBr1 antibody specific for H Type 3/4 chains, and have found the expression of H Type 3/4 chains was mainly in the serous cells. Serous cells from secretors were stained by MBr1 but not by anti-A and anti-B antibodies, whereas serous cells from nonsecretors exhibited a negative reaction with MBr1. Mucous cells were not stained by MBr1. Only a few striated duct cells showed a weak reaction with anti-H MBr1. These results suggested that the H Type 3/4 chains were distributed predominantly in the serous cells of the human submandibular gland and that secretor Type α (1,2) fucosyltransferase (Se enzyme) controlled the synthesis of H Type 3/4 chains in vivo. Saliva also contained H Type 3/4 chains, which were controlled by the secretor gene (FUT2). The differences in the distributions of H Type 1, H Type 2, and H Type 3/4 chains of the ABO histo blood group system in the submandibular gland are discussed.
B
In this study, we used MAb anti-H MBr1, which is defined as a breast cancer-associated antigen and is specific for H Type 3/4 antigens (Menard et al. 1983; Bremer et al. 1984; Kannagi et al. 1984; Clausen et al. 1986a,b; Adobati et al. 1997), to examine the distribution of H Type 3/4 chains in cell types of the human submandibular gland through an immunohistochemical method, and have demonstrated that the H Type 3/4 chains were expressed mainly in the se rous cells, with their expression dependent on the secretor status.
Materials and Methods
The ABO histo-blood group phenotypes of tissue donors were determined on red blood cells using the conventional hemagglutination method with mouse MAbs (Kokusai; Kobe, Japan) against histo-blood group antigens A and B (Liu et al. 1996). In addition, the Se genotypes of tissue donors were determined by PCR after extraction of DNA from frozen salivary glands, as described previously (Koda et al. 1996).
Two anti-H MAbs, MBr1 and 1E3, were used in the present study. MAb MBr1, specific for H Type 3/4 structures (Fucot1-2Galβ1-3GalNAc-) (Bremer et al. 1984; Clausen et al. 1986a,b; Adobati et al. 1997), was a kind gift from Dr. Maria I. Colnaghi (Division of Experimental Oncology E, National Institute for Cancer Research; Milan, Italy). Anti-H MAb 1E3 was a kind gift from Drs. Ken Furukawa and Shin Yazawa (Department of Legal Medicine, Gunma University School of Medicine). The anti-H 1E3 has been demonstrated to be reactive with H Type 1-H Type 4 oligosaccharides and to be reactive with Fuca1-2Galβ disaccharide, using a series of synthetic oligosaccharides (Nakajima et al. 1993), but to be reactive predominantly with the H Type 1 and H Type 2 antigens in situ in the submandibular gland, as shown in our previous study (Liu et al. 1998).
Submandibular glands from individuals with different ABO histo-blood groups were obtained from autopsy cases (five secretors and four nonsecretors from O blood group individuals and four secretors from A and four secretors from B blood group individuals). Deparaffinized sections of 10% formalin-fixed, paraffin-embedded submandibular glands were immunostained using the streptavidin-biotin complex immunoperoxidase method as described previously (Liu et al. 1998). The primary MAbs, anti-H MBr1, anti-H 1E3, anti-A, or anti-B, diluted 500 times in PBST (0.01 M phosphate buffer, pH 7.4, 0.15 M NaCl, and 0.5% Tween 20), were reacted at 4C overnight in a moist chamber.
A dot ELISA was carried out as described previously (Liu et al. 1996, 1998). Briefly, boiled human saliva samples were diluted in twofold serial dilution, and 1 μl of each of the diluted samples was dotted onto a nitrocellulose membrane. After quenching intrinsic peroxidase activity with 3% H2O2, the nitrocellulose membrane was blocked with 0.05% Tween 20 in 0.9% NaCl. Then the antigenicity was detected using MAb MBr1 or 1E3 (both diluted 200 times), followed by peroxidase-conjugated goat anti-mouse IgM (Cappel; West Chester, PA) and 4-chloro-1-naphthol for color development.
To examine whether the H Type 3/4 antigens were synthesized by the H enzyme or the Se enzyme, the wild-type allele of the FUT1 for H enzyme or the FUT2 for Se enzyme was subcloned into a mammalian expression vector pRc/CMV (Invitrogen; San Diego, CA) (pRc/CMV-H or pRc/CMV-Se), as described previously (Koda et al. 1996, 1997). The plasmid (pRc/CMV-H or pRc/CMV-Se) was transfected into COS7 cells by a DEAE-dextran method (Gonzalez and Joly 1995). After 48 hr of culture, the expression of H antigens on cells was examined using anti-H MBr1 or anti-H 1E3 and fluorescein-labeled anti-mouse IgM (Koda et al. 1993).
Results
The murine MAb MBr1, raised against the human breast cancer cell line MCF-7, recognized a saccharide epitope on human breast, ovary, and lung carcinomas (Mariani-Costantini et al. 1984a,b; Martignone et al. 1993; Perrone et al. 1993). This antigen was originally identified as a globo series glycosphingolipid with an H-like determinant at its terminus (Fuca1-2Galβ1-3GalNAcβ1-3Galα1-4Galβ1-4Glcβ1-Cer) in MCF-7 cells (Bremer et al. 1984). Later, it was shown that MBr1 recognized the trisaccharide sequence (Fucα1-2Galβ1-3GalNAc, H Type 3/4) on glycolipids and on glycoproteins, irrespective of the anomeric structure of the internal GalNAc residue (Clausen et al. 1986a,b). Recently, Adobati et al. (1997) demonstrated that MBr1 was more specific for Fuco 1-2Galβ1-3GalNAcβ1 (H Type 4 chain).
Previous studies have revealed the presence of Type 3 chain-based ABH antigens in saliva and ovarian cyst fluids (Donald 1981) and in seminal plasma (Hanischi et al. 1986) after isolation and chemical characterization. In dot ELISA, anti-H MBr1 reacted with human saliva from secretor individuals but not with saliva from nonsecretors (Figure 1). This finding indicated that human saliva contained H Type 3/4 chains and that their secretion was dependent on the secretor status.
Next, we immunohistochemically investigated the human submandibular gland using anti-H MBr1 and anti-H 1E3 (Figure 2). Serous cells and some serous demilune cells from secretor donors, but not from nonsecretor donors, were stained intensely by anti-H MBr1, irrespective of ABO phenotypes (Figures 2A and 3A). These results suggested that the production of H Type 3/4 antigens in serous cells of human submandibular glands was under the control of the Se gene. In contrast, mucous acini, vascular endothelia, and erythrocytes showed no reaction with anti-H MBr1, although these cells showed a good reaction with anti-H 1E3 (Figure 2), anti-A (Figure 3), or anti-B (not shown) in tissues from corresponding ABO group individuals. Positive staining by MBr1 was observed in the luminal surface and luminal contents of many striated ducts, but only a few epithelial cells (less than 1%) of striated ducts showed a weak reaction with MBr1. Staining by 1E3 appeared to localize on basolateral plasma membranes of striated duct cells, and this mode of distribution obviously differed from that in mucous cells (Figures 2B and 2D). Epithelial cells of interlobular ducts demonstrated a negative reaction with MBr1 (not shown). In the submandibular gland of A secretors, we also observed a strong reaction with anti-A in mucous acini (Figure 3) and a weak reaction in some ducts, but no reaction in serous acini despite the strong staining by anti-H MBr1 in serous cells, suggesting the expression of the ABO gene in mucous cells and duct cells but not in serous cells. It appeared that all serous cells from the A secretors showed a positive reaction with MBr1 (Figure 3A, whereas only a portion of serous cells from the O secretors reacted with the antibody (Figure 2A. The difference in staining of serous cells between A and O secretors was due to individual variation. All results are summarized in Table 1.

Reaction of saliva with anti-H MBr1 and anti-H 1E3 on dot ELISA. The saliva from secretor individuals showed a positive reaction with anti-H MBr1 (
COS7 cells transfected by the H gene (FUT1) or the Se gene (FUT2) showed strong fluorescence with both anti-H MBr1 and 1E3 (not shown), suggesting that both α (1,2) fucosyltransferases (H enzyme and Se enzyme) can fucosylate a precursor of Type 3 or Type 4 to form H Type 3 chain or H Type 4 chain in vitro, consistent with a previous report (Henry et al. 1996).
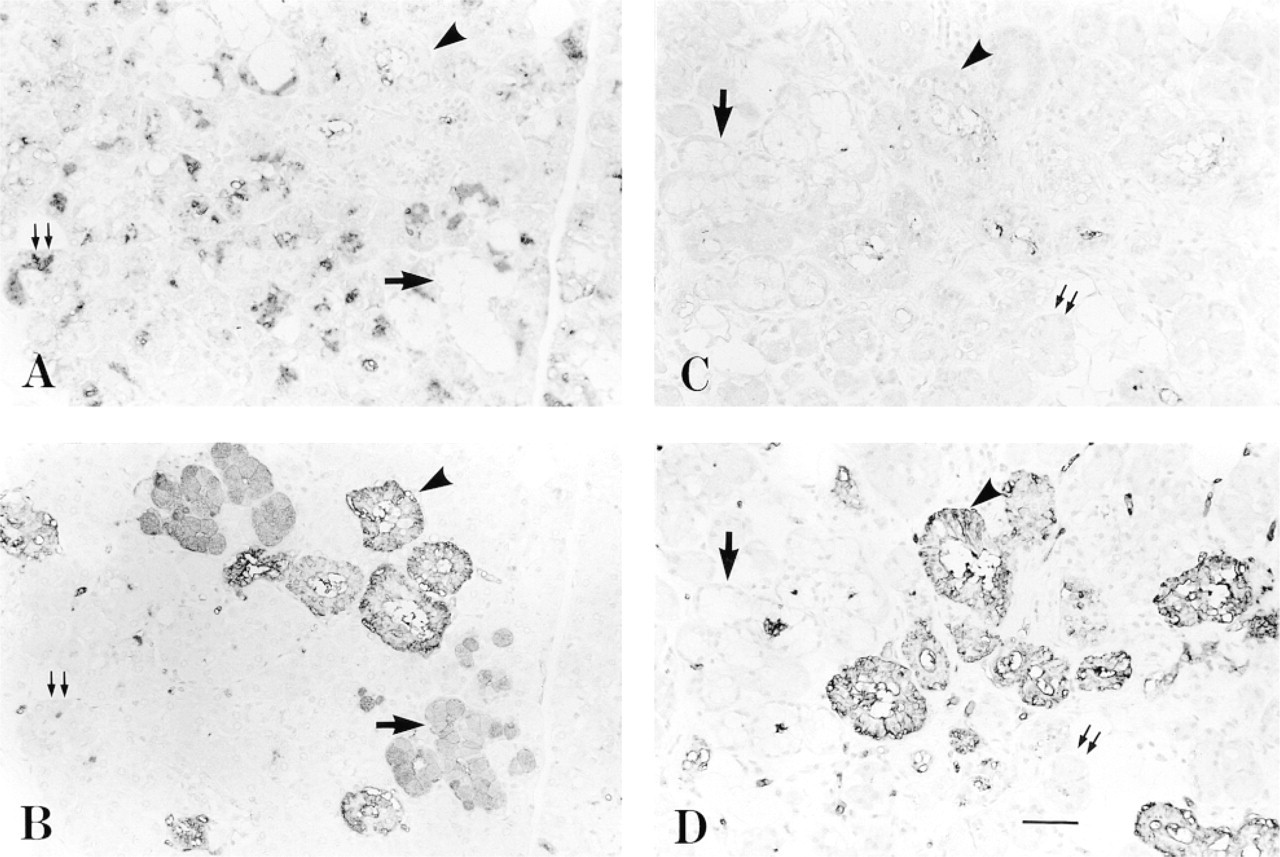
Serial sections of submandibular gland from an O secretor stained by anti-H MBr1 (
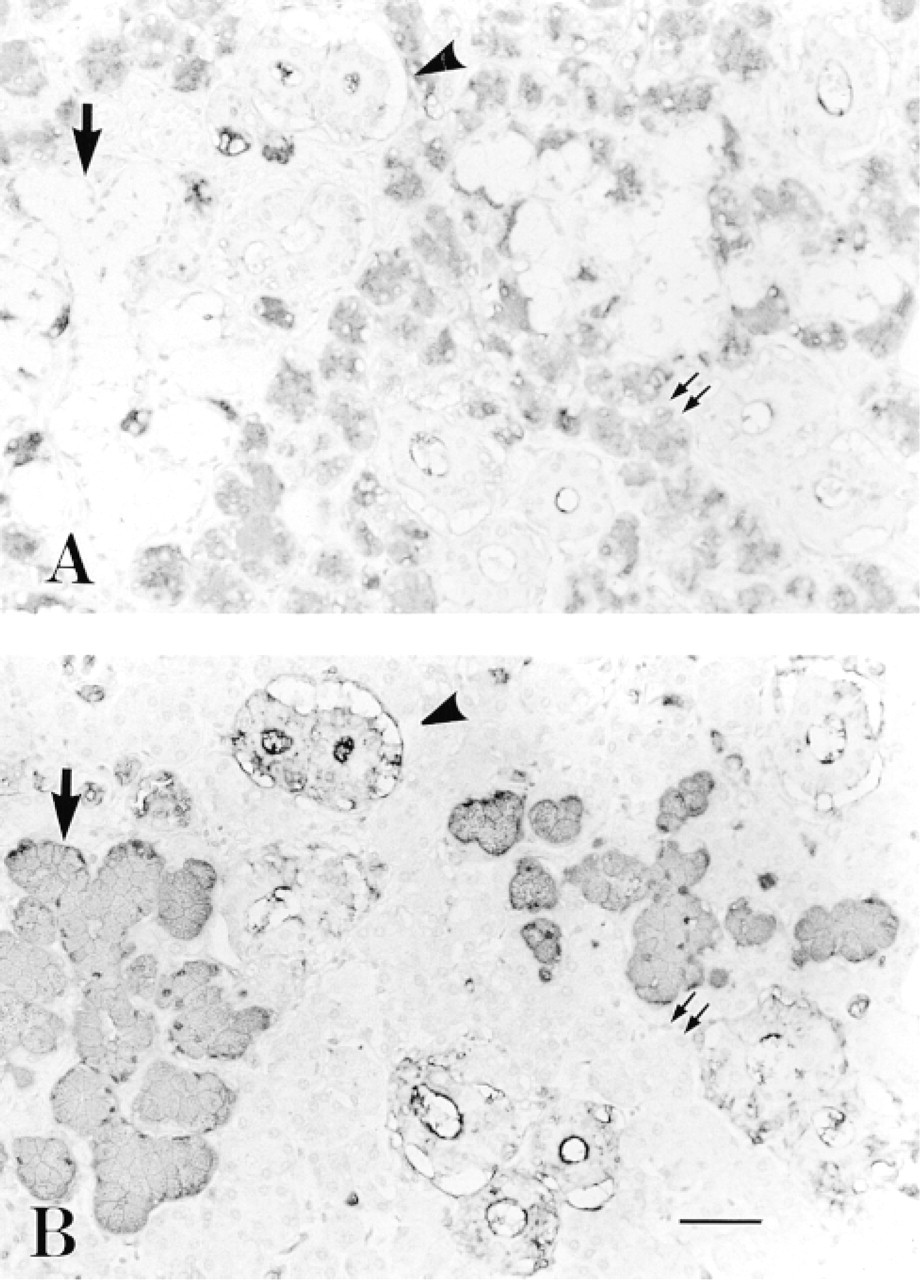
Immunohistochemical staining of the submandibular gland from an A secretor by anti-H MBr1 (
Discussion
In this study we examined immunohistochemically the location of H Type 3/4 antigens in cell types in the human submandibular gland using anti-H MBr1, and we identified cell types expressing H Type 3/4 chains as serous cells. Our results extended previous observations (Ito et al. 1989; Cossu et al. 1990; Cossu and Lantini 1996), which implied the presence of the H antigen in serous cells of human submandibular gland by lectin histochemical and immunoelectron microscopic methods. From our previous and present studies, we have now clearly identified cell types expressing H Type 1, H Type 2, and H Type 3/4: H Type 1 in mucous cells and duct cells, H Type 2 in duct cells, and H Type 3/4 in the serous cells of the submandibular gland (Table 2) (Liu et al. 1998). The expression of H Type 1 in mucous cells and of H Type 3/4 in serous cells was regulated by the Se enzyme in vivo. However, the H enzyme also can form H Type 3/4 antigens, as demonstrated by the positive reaction with MBr1 on COS7 cells after transfection of the H gene (FUT1).
The H Type 3 has been found in ovarian cyst fluid and saliva (Donald 1981) and in erythrocytes (Clausen et al. 1986a) from individuals with blood group A. The H Type 3 glycosphingolipid (a precursor of repetitive A) (Fucα-2Galβ1-3GalNAcα1-3[Fucα1-2]Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAc-Cer) was constructed by adding a Gal residue and then a Fuc residue sequentially to the terminal GalNAc of the A Type 2 structure. Alternatively, the formation of the H Type 3 chain needs both the H Type 2 chain as a precursor and the activity of A glycosyltransferase. The other Type 3 chain-based H antigen is a fucosylated T antigen (Fucα 1-2Galβ1-3GalNAcc 1-O-Ser/Thr), which has been found in the mucosa of the stomach and colon (Macartney 1986; Nakayama et al. 1987; Okada et al. 1994). Using peanut agglutinin (PNA) and glycosidase digestion, Ito et al. (1989) concluded that O-linked Type 3 chain-based H antigen (Fucα1-2Galβ1-3GalNAcα1-O-Ser/Thr) was produced in serous cells and that its fucosylation was under the control of the Se gene in human submandibular glands. Okada et al. (1994) also demonstrated the expression of the Thomsen-Friedenreich antigen (T antigen) (Galβl-3GalNAcα-O-Ser/Thr) and fucosylated T antigen (H Type 3) in human gastric surface epithelial cells and the dependency of its fucosylation on the Se gene, using anti-H MBr1 and PNA as probes. However, the specificity of PNA has been questioned because of different distributions of PNA-positive and anti-T-positive substances before and after neuraminidase treatment and different reactivities of PNA and anti-T antibody after galactose oxidation (Macartney 1986; Nakayama et al. 1987). Ito et al. (1989) reported that PNA-reactive substances were not accumulated in serous cells of the submandibular gland from nonsecretors.
Immunohistochemical staining of human submandibular gland by monoclonal anti-H and anti-A antibodies a
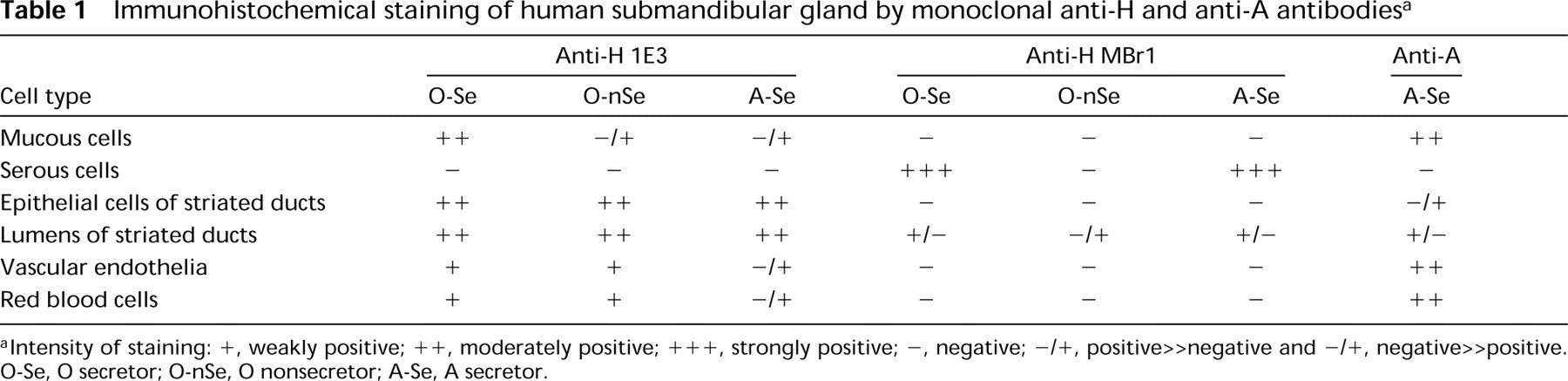
aIntensity of staining: +, weakly positive; ++, moderately positive; +++, strongly positive; -, negative; -/+, positive<negative and -/+, negative>>positive. O-Se, O secretor; O-nSe, O nonsecretor; A-Se, A secretor.
Distribution of H type 1, H type 2 and H type 3/4 chains in human submandibular gland a
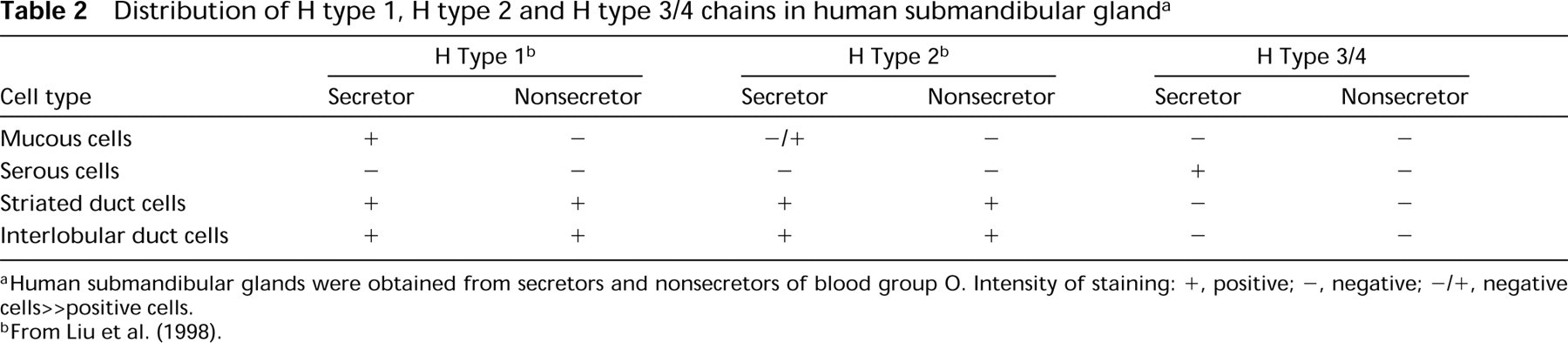
aHuman submandibular glands were obtained from secretors and nonsecretors of blood group O. Intensity of staining: +, positive; -, negative; -/+, negative cells>positive cells.
bFrom Liu et al. (1998).
In this study we have demonstrated that reactivity of anti-H MBr1 was found in the serous cells of the submandibular gland from O individuals, and that anti-A- and anti-H MBr1-positive substances were present in different cell types of the submandibular gland from A individuals, suggesting that an MBr1-reactive substance appeared to be not a precursor of repetitive A (H Type 3 glycosphingolipid). Moreover, no A, B, or H Type 2 antigens were detected in serous acini (Figure 3) (Laden et al. 1984; Ito et al. 1989; Liu et al. 1998). The H Type 4 chain isolated from the human O erythrocytes (Kannagi et al. 1984) and from the human mammary carcinoma cell line MCF-7 (Bremer et al. 1984) was originally identified as the antigen defined by anti-H MBr1. Recently, Adobati et al. (1997) examined the specificity of MBr1 and demonstrated that the terminal tetrasaccharide of globo-H (Fucα1-2Galβ1-3GalNAcβ1-3Galα1-R) is the MBr1-defined epitope and that the terminal fucose is essential for MBr1 recognition. However, because anti-H MBr1 cannot discriminate H Type 3 from H Type 4, it is not known whether MBr1-reactive material in serous cells is H Type 3, H Type 4, or both. Because the A type 4 antigen was isolated from human kidney (Breimer and Jovall 1985; Holgersson et al. 1992), it would be of interest to examine the kidney immunohistochemically using anti-H MBr1.
It is known that the UEA-I-positive substances, which are regulated by the Se gene, are present in the serous cells of human submandibular gland (Ito et al. 1989; Liu et al. 1998). Here, we have demonstrated the presence of H Type 3/4 chains in the serous cells and the dependency of their expression on the Se gene. However, Mollicone et al. (1996) and Baldus et al. (1996) examined the specificity of UEA-I using synthetic oligosaccharides and demonstrated no reaction of UEA-I with H Type 3 and H Type 4 chains. Therefore, the characteristics of UEA-I-positive substance(s) in serous cells still remain unclear.
Footnotes
Acknowledgements
Supported by grants-in-aid for Scientific Research from the Ministry of Education, Science, Culture and Sports of Japan.
We are grateful to Drs Ken Furukawa and Shin Yazawa (Department of Legal Medicine, Gunma University School of Medicine) and to Dr Maria I. Colnaghi (Division of Experimental Oncology E, National Institute for Cancer Research, Milan) for the kind gifts of MAb anti-H 1E3 and anti-H MBr1, respectively. We thank Mr Shigeo Kamimura and Ms Yasuko Noguchi for making serial thin sections.
