Abstract
We have examined the immunohistochemical distribution of H Type 1 and of H Type 2 substances of the ABO blood group system in human submandibular gland using either of the two anti-H monoclonal antibodies MAb 1E3 and MAb 3A5. MAb 3A5 was specific for H Type 2, and MAb 1E3 reacted with each of H Type 1-H Type 4 artificial antigens. We have developed a competitive inhibition method against H Type 2 and have obtained MAb 1E3, which is fairly specific for H Type 1 under certain conditions. Mucous cells from secretors were strongly stained by 1E3 and weakly by 3A5, whereas those from nonsecretors showed no reaction with 1E3 and 3A5. Serous cells from both secretors and nonsecretors were stained neither by 1E3 nor by 3A5. Striated and interlobular duct cells were strongly stained by 1E3 and by 3A5, regardless of the secretor status. These results indicated that the expressions of the H Type 1 and H Type 2 in different cell types of the submandibular gland were controlled by different genes. In addition, we have determined the acceptor specificity of two α(1,2)fucosyltransferases (H and Se enzymes) after transient expressions of the FUT1 and FUT2 in COS7 cells, and found that the H enzyme activity was similar for both Type 1 and Type 2 precursors, and that Se enzyme activity with the Type 1 precursor was higher than that with the Type 2 precursor. Expression of the H Type 1 antigen in mucous cells was found to be dependent on the Se gene, whereas expressions of the H Type 1 and H Type 2 antigens in striated and interlobular duct cells were dependent on the H gene.
Keywords
T
In the present study, we compared the stainings of the H antigens in cell types of human submandibular gland by two anti-H MAbs (MAb 3A5 and MAb 1E3, which are specific for H Type 2 and for H Types 1–4, respectively) (Nakajima et al. 1993; Liu et al. 1996a). We developed a competitive inhibition method against H Type 2 by absorption of MAb 1E3 with synthetic H Type 2-albumin complex or red blood cell ghosts and we obtained MAb 1E3 fairly specific for H Type 1 antigen. In addition, we have examined the acceptor specificities of two α(1,2)fucosyltransferases, H enzyme and Se enzyme, for interpreting our immunohistochemical findings.
Materials and Methods
Blood Group Phenotypes and FUT2 Genotypes
The ABO and Lewis blood groups of tissue donors were determined on red blood cells by conventional hemagglutination techniques using mouse MAbs against histo-blood group antigens, A, B (Kokusai; Kobe, Japan), Lea, and Leb (Ortho Diagnostic; Tokyo, Japan), respectively. Because the secretor status in saliva of ABO substances is closely related to the Lewis phenotypes on red blood cells, an individual with Le(a-b+) was recognized to be a secretor, with Le(a+b+) to be a nonsecretor, or with Le(a+b+) to be a weak secretor (Henry et al. 1995). In addition, the Se and Lewis genotypes of tissue donors were determined by PCR after extraction of DNA from frozen salivary glands, as described previously (Koda et al. 1996; Liu et al. 1996b).
Antibodies and Lectin
Two anti-H MAbs, 3A5 and 1E3, were used in the present study. The specificity of MAb 3A5 has been demonstrated to be specific for the H Type 2 structure using a series of synthetic oligosaccharides (Liu et al. 1996a). Anti-H MAb 1E3 was a kind gift from Drs. Ken Furukawa and Shin Yazawa (Department of Legal Medicine, Gunma University School of Medicine). The anti-H MAb 1E3 has been demonstrated to be reactive with Fucα1-2Gal disaccharide and H Type 1-H Type 4 oligosaccharides (Nakajima et al. 1993). UEA-I agglutinin conjugated with horseradish peroxidase (HRP) was purchased from EY Laboratories (San Mateo, CA). Working concentrations were as follows: MAb 3A5 and MAb 1E3 (ascites) were diluted 500 times in PBST (0.01 M phosphate buffer, pH 7.4, 0.15 M NaCl, and 0.5% Tween 20), and HRP-UEA-I was 10 μg/ml in PBS containing 0.1% bovine serum albumin (BSA).
Absorption of MAb 1E3 with Sonicated Red Blood Cell Ghosts
Saliva and sonicated erythrocyte membranes (ghosts) from O blood group individuals were prepared as described previously (Liu et al. 1996a). A total of 100 μl of MAb 1E3 (ascites) diluted 500-fold was mixed with sonicated ghosts of O red blood cells (about 9 μg of membrane protein) as H Type 2 substance and kept at 4C for 1 hr. The ghost solution with birefringence became completely clear after sonication. After addition of a small quantity of a clear ghost solution to the diluted ascites containing MAb 1E3, the solution was still clear. In some experiments, MAb 1E3 was absorbed with synthetic H Type 2 structure coupled to BSA (Syntagen H Type 2) (Chembiomed; Edmonton, Canada), synthetic H Type 2 coupled to silica beads (Synsorb H Type 2) (Chembiomed), or boiled saliva from an O blood group individual, and the absorbed MAb 1E3 was used for an enzyme-linked immunosorbent assay (ELISA) and a dot ELISA, as described previously (Liu et al. 1996a). Peroxidase-conjugated goat anti-mouse IgM (Cappel; West Chester, PA) and Syntagen H Type 1 (Chembiomed) were used for ELISA.
Immunohistochemistry and Lectin Histochemistry
Submandibular glands from individuals with blood group O were obtained from autopsy cases (five secretors and four nonsecretors). The glands were fixed in 10% formalin at room temperature (RT), embedded in paraffin, and sectioned serially at 3–4 μm. Paraffin sections were mounted on gelatin-dichromate-coated glass slides. Sections were depar-affinized, rehydrated, and pretreated with 3% hydrogen peroxide for 30 min at RT to quench endogenous peroxidase activity. Then the pretreated sections were used for immunohistochemistry or lectin histochemistry.
Sections were incubated with 10% normal rabbit serum for immunohistochemistry or with 1% BSA for lectin histochemistry for 30 min at RT to block nonspecific sites, then incubated with the primary MAb (anti-H 3A5, anti-H 1E3, or anti-Leb) or HRP-conjugated UEA-I at 4C overnight in a moist chamber. After washing three times in PBST, the sections for immunohistochemistry were incubated with biotinylated rabbit anti-mouse immunoglobulins (IgG + IgM + IgA) (Nichirei; Tokyo, Japan) for 30 min at RT, followed by rinsing in PBST, and were then incubated with peroxidase-conjugated streptavidin (Nichirei). Positive sites were developed with 200 μg/ml of 3,3′-diaminobenzidine containing 0.04% hydrogen peroxide in PBS, and finally the sections were counterstained with hematoxylin. Control sections, incubated as above but without the primary antibody or UEA-I, showed no staining.
Transient Expression of FUT1 (H gene) and FUT2 (Se gene) in COS7 Cells and Assay of α(1,2)fucosyltransferase Activity
The wild-type allele of the FUT1 or the FUT2 was subcloned into a mammalian expression vector pRc/CMV (Invitrogen; San Diego, CA) (pRc/CMV-H and pRc/CMV-Se, respectively), as described previously (Koda et al. 1996; Wang et al. 1997). The plasmid with DNA inserted in the sense direction of the FUT1 or of the FUT2 was transfected into COS7 cells by a DEAE-dextran method (Gonzalez et al. 1995). After 48 hr of culture, the expression of H antigen on the cells was examined using monoclonal anti-H antibody 1E3 or 3A5 and then fluorescein-labeled anti-mouse IgM antibody (Koda et al. 1993). At the same time, the α(1,2)fucosyltrans-ferase activity in cell extracts was also determined using lacto-N-biose I as a Type 1 acceptor or N-acetyl-lactosamine as a Type 2 acceptor (Masutani and Kimura 1995).
Results
The FUT2 genotypes of all four individuals with Le(a+b+) were identified to be homozygous for se2 (A385T), which is one of the Se enzyme-deficient alleles (Koda et al. 1996) and is also believed to be a weak secretor allele (Yu et al. 1995). All nine individuals had at least one functional Le allele in the FUT3.
Although MAb 1E3 has been shown to be reactive with the H Type 1-H Type 4 synthetic oligosaccharides (Nakajima et al. 1993), different reactions with the various tissues have been observed. MAb 1E3 reacted weakly with O red blood cells compared with MAb 3A5, but strongly with saliva from O secretors in a dot ELISA method (Liu et al. 1996a). We have demonstrated that the reactivity of MAb 1E3 with H Type 2 was about 50% compared to that with H Type 1 (Wang et al. 1994; see also Figure 2). These previous results suggested a lower reactivity of MAb 1E3 to H Type 2. Therefore, we designed a competitive inhibition method against H Type 2 in which we could determine whether or not the reactivity of MAb 1E3 to H Type 2 would change after absorption with H Type 2 substance. As shown in Figure 1, we found that MAb 1E3 still reacted with O saliva but not with O red blood cells in a dot ELISA after being absorbed with a particular quantity of ghosts (Figure 1B), whereas MAb 1E3 before absorption with ghosts strongly stained O secretor saliva and weakly reacted with O red blood cells (Figure 1A). When MAb 1E3 was absorbed with synthetic H Type 2-BSA complex, similar results as with ghosts were obtained (not shown). However, when MAb 1E3 was absorbed onto Synsorb H Type 2 silica beads, MAb 1E3 did not react with either saliva or red blood cells (Figure 1C). We also confirmed the effect of absorption of MAb 1E3 with a small quantity of ghosts by an ELISA method using synthetic H Type 1- and H Type 2-BSA complexes (Figure 2). The reactivity of MAb 1E3 to H Type 1-BSA was decreased by only about 25% after absorption with O erythrocyte membranes, and that to H Type 2-BSA was remarkably lowered, which led to increase in the ratio of the reactivity of MAb 1E3 to H Type 1 and to H Type 2 from 2 to 10 (Figure 2). This result was also confirmed in the present immunohistochemical study. MAb 1E3 before absorption by ghosts stained not only mucous and duct cells but also red blood cells and endothelial cells of vessels in the submandibular gland from O secretors. However, after absorption with a limited quantity of ghosts, MAb 1E3 stained only mucous cells and duct cells and did not stain red blood cells and endothelial cells of vessels whose major component was H Type 2 (Breimer et al. 1988; Clausen and Hakomori 1989) (Figure 3). When MAb 1E3 was absorbed with diluted boiled saliva from an O secretor whose major component was H Type 1 (Wang et al. 1994), staining was not observed at all in tissue sections (not shown), suggesting that positive material by 1E3 after absorption with ghosts was H Type 1 chain. Although it is difficult to determine the affinity constant for each of H Type 1-H Type 4 of MAb 1E3, our results suggest that MAb 1E3 appears to not react with the H Type 2 under some conditions. Therefore, MAb 1E3 after absorption with sonicated ghosts was used as a putative anti-H specific for H Type 1 throughout the present study.
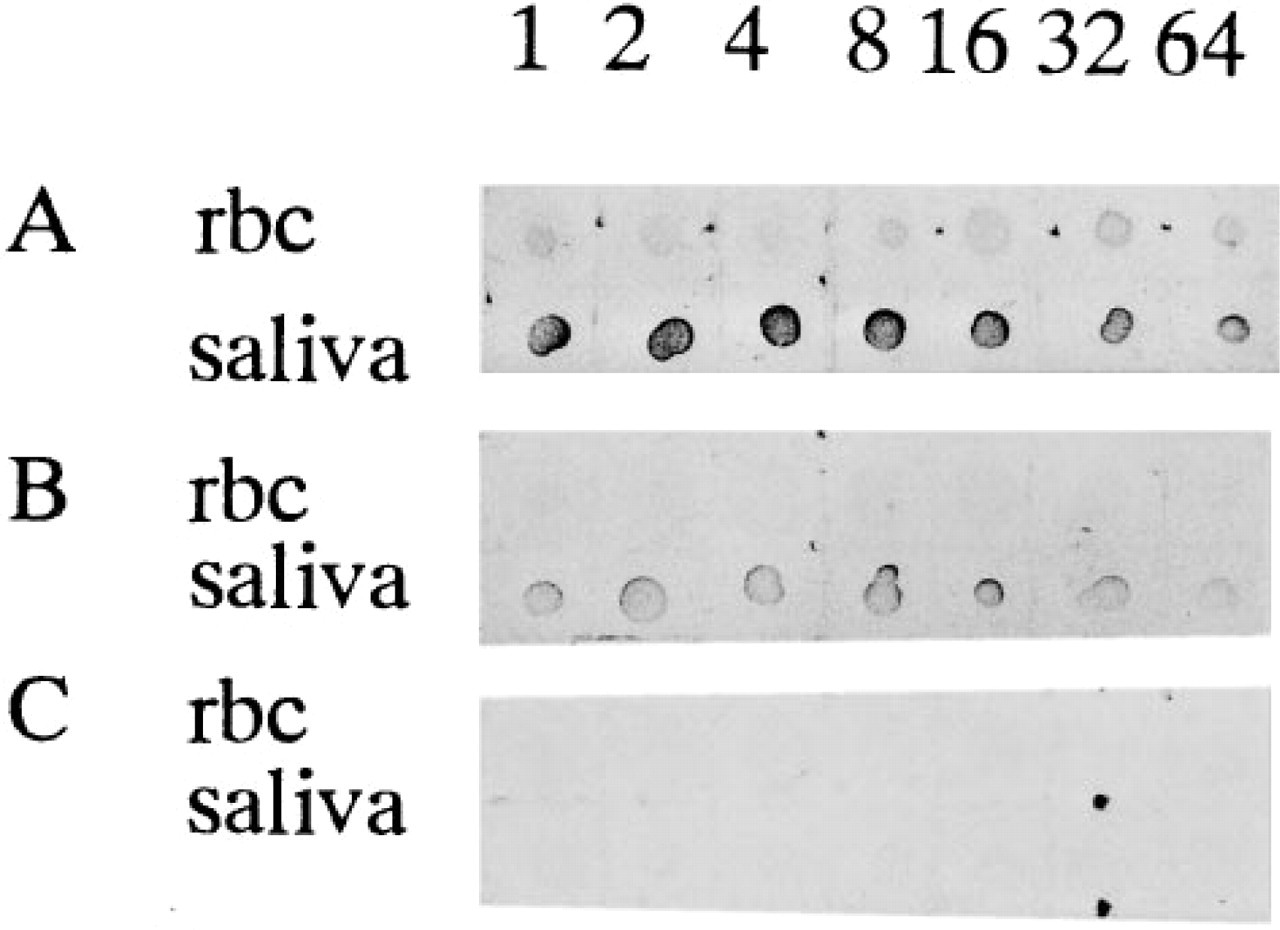
The dot ELISA of saliva and red blood cells from O secretors with MAb 1E3 (
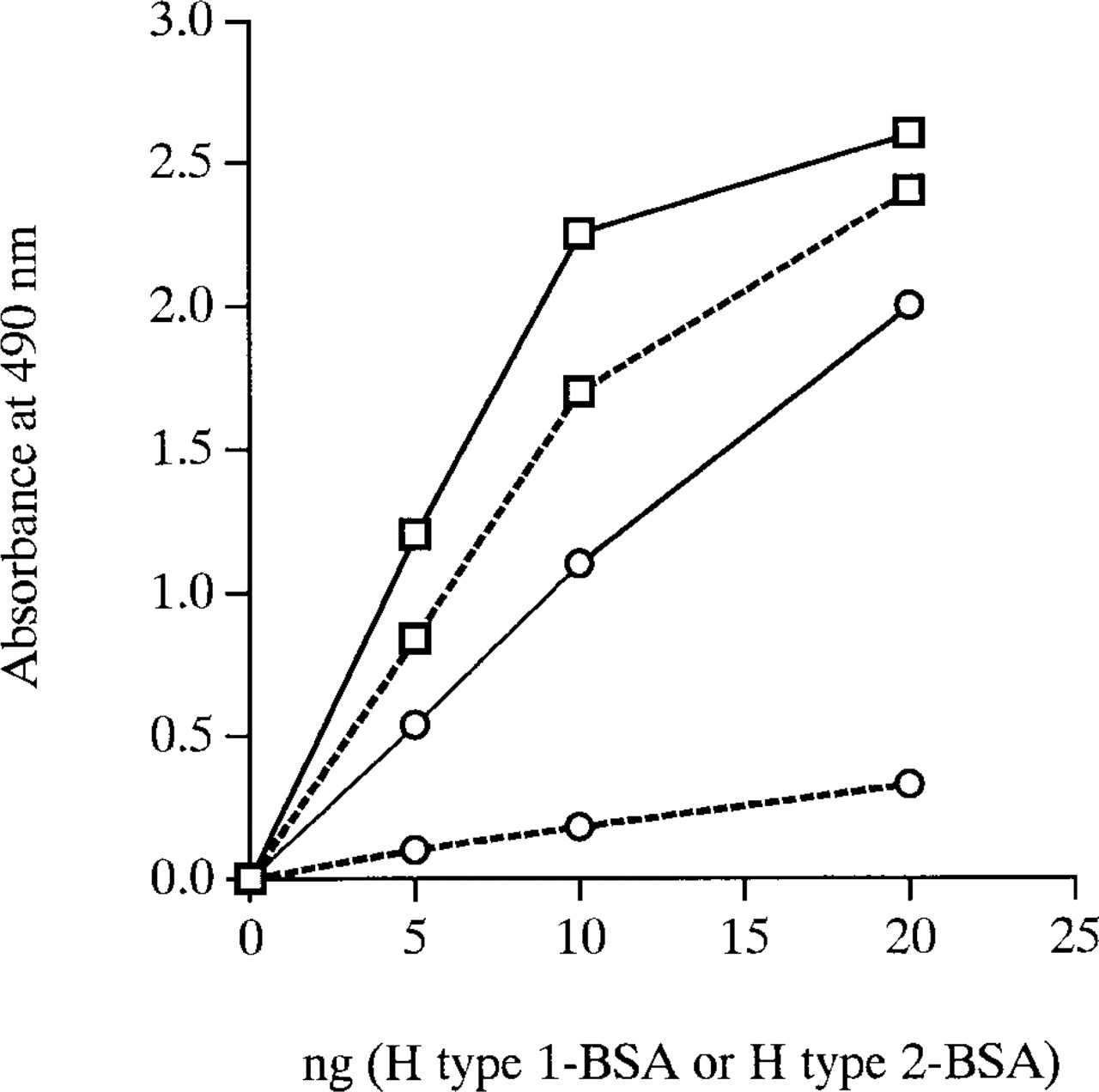
Reactivity to H Type 1-BSA and to H Type 2-BSA complexes of MAb 1E3 before and after absorption with sonicated ghosts determined by an ELISA method. Squares represent reaction with H Type 1 and circles with H Type 2. Solid lines represent before and broken lines after absorption with ghosts.
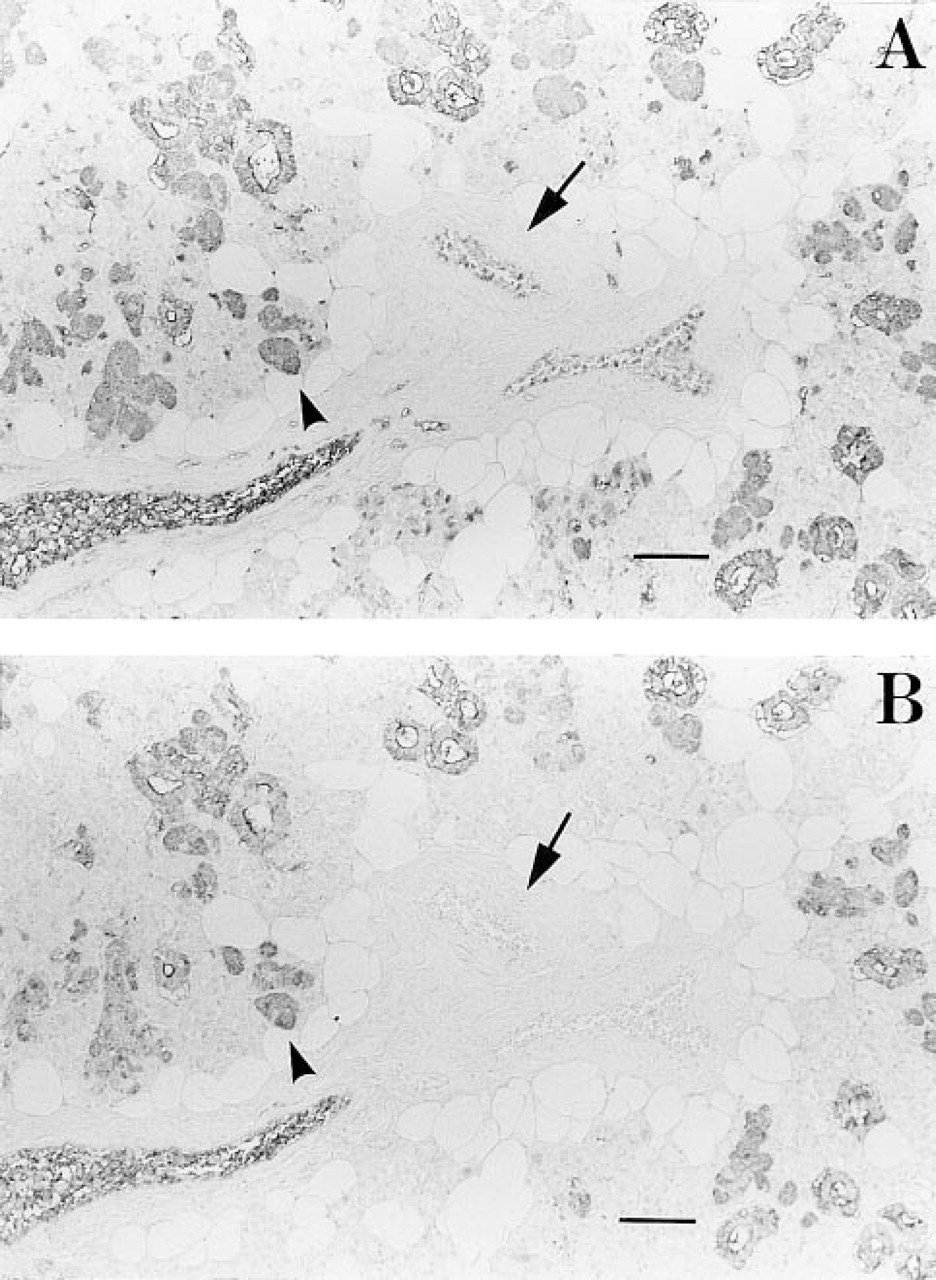
Immunohistochemical staining of secretor submandibular gland by MAb 1E3 before (
The majority of mucous cells of acini from secretors were stained intensely by MAb 1E3 (Figure 4A) and weakly by MAb 3A5 (Figure 4B). Most mucous cells from nonsecretors were negative by 1E3 and 3A5 (Figures 4D and 4E), but some mucous cells from non-secretors still displayed a weak positive reaction with MAb 1E3, probably due to the weak secretor phenotype. However, mucous acini from nonsecretors were stained by MAb anti-Leb (not shown) despite the lack of staining of H Type 1 on these cells. These results suggested that the major H substance in the mucous cells was H Type 1 rather than H Type 2 and that its expression was closely related to the secretor status. Serous acini from both secretor and nonsecretor individuals were not labeled by MAbs anti-H, 1E3, and 3A5. The staining of some intercalated cells by anti-H was the same as that of mucous cells, dependent on the secretor status. Interestingly, striated and interlobular duct cells were stained by both 3A5 and 1E3, which was independent of the secretor status of the donors, indicating the presence of both H Type 1 and H Type 2 antigens.
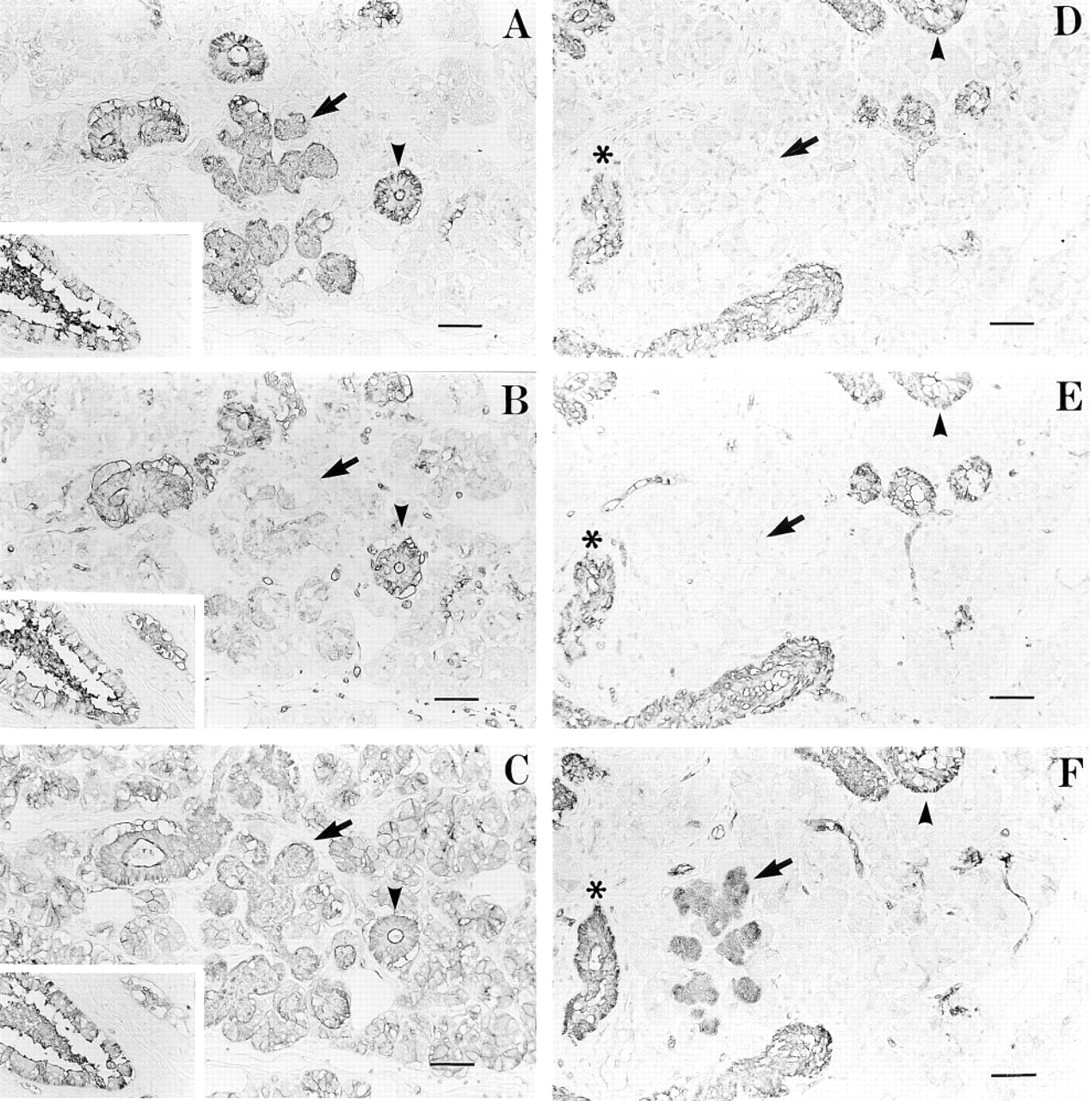
Serial sections of the submandibular gland from an O secretor stained with MAb 1E3 (A), MAb 3A5 (B), and UEA-I (C), and those from a nonsecretor stained with MAb 1E3 (D), MAb 3A5 (E), and UEA-I (F). MAb 1E3 was used after absorption with ghosts. The mucous cells (arrow) from the secretor were stained strongly by 1E3 (A) and weakly by 3A5 (B), but those from the nonsecretor showed no reaction with MAbs 1E3 and 3A5 (D,E). The serous cells showed no reaction with 1E3 or 3A5. Striated ducts (arrowhead) and interlobular ducts (asterisk in D-F) showed a positive reaction with 1E3 and 3A5, independent of the secretor status of the tissue donor. Insets in A-C show an interlobular duct (at left) and a vessel containing red blood cells (at right). UEA-I stained all cells except for the serous cells from nonsecretors. Bars = 50 μm.
As shown in Figures 4C and 4F, almost all cells, including mucous cells, serous cells, and duct cells in tissue sections, except for the serous cells from nonsecretors, were uniformly stained by UEA-I lectin. The staining of the serous cells by UEA-I was dependent on the secretor status of donors, whereas that of the mucous and duct cells was independent of the secretor status. All results are summarized in Table 1.
Immunohistochemical staining of different cells with monoclonal anti-H antibodies 1E3 and 3A5 and with UEA-I lectin in human submandibular gland a
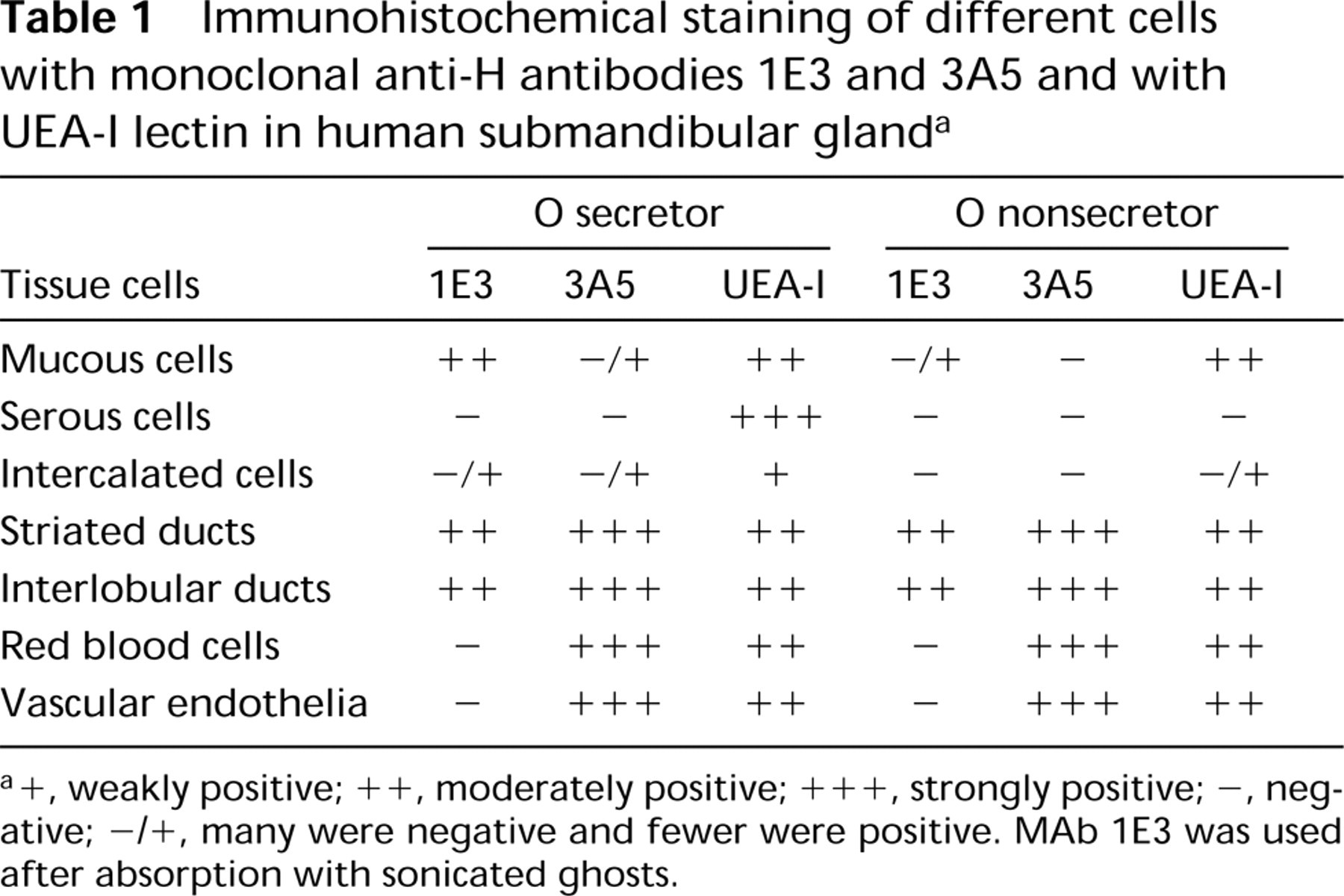
+, weakly positive; ++, moderately positive; +++, strongly positive; -, negative; -/+, many were negative and fewer were positive. MAb 1E3 was used after absorption with sonicated ghosts.
The H enzyme activity in pRc/CMV-H-transfected cell extracts was similar with lacto-N-biose I (Type 1 chain) and with N-acetyl-lactosamine (Type 2 chain) as acceptors, whereas the Se enzyme activity in pRc/CMV-Se-transfected cell extracts showed about six-fold higher activity with lacto-N-biose I than with N-acetyl-lactosamine (Table 2). In addition, COS7 cells transfected by the Se gene (FUT2) displayed strong fluorescence with 1E3 but very weak fluorescence with 3A5, whereas cells transfected by the H gene (FUT1) showed strong fluorescence with both monoclonal anti-H antibodies (not shown), suggesting similar reactivities of the H enzyme with both Type 1 and Type 2 acceptors and higher reactivity of the Se enzyme with the Type 1 acceptor than with the Type 2 acceptor in cells.
Activity of α(1,2)fucosyltransferases in extracts of COS7 cells transfected with plasmid DNA inserted with FUT1 or FUT2 a
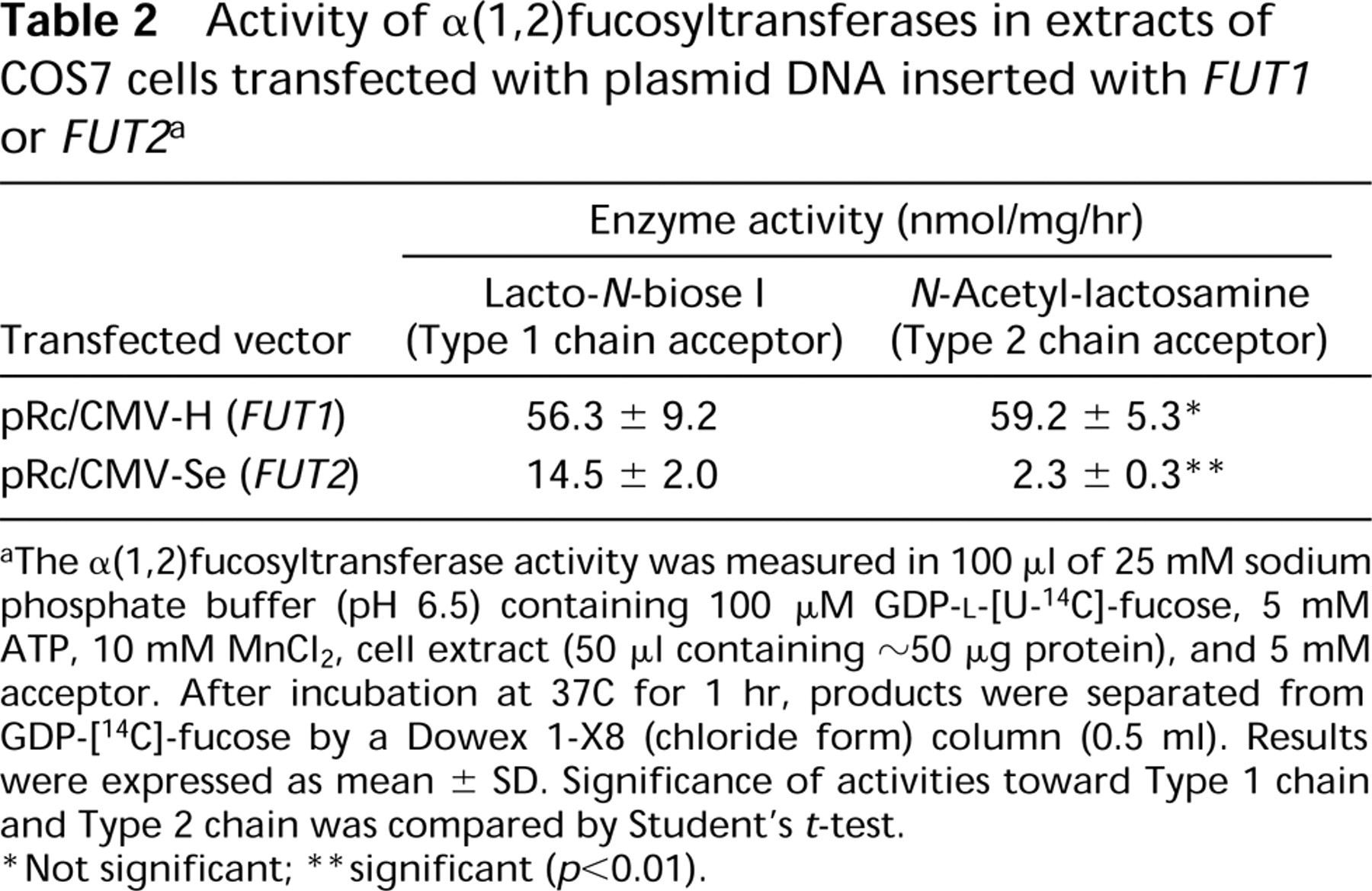
The α(1,2)fucosyltransferase activity was measured in 100 μl of 25 mM sodium phosphate buffer (pH 6.5) containing 100 μM GDP-
Not significant;
significant (p<0.01).
Discussion
Many immunohistochemical studies of the ABO substances in the salivary gland have been reported (Laden et al. 1984; Ito et al. 1985,1989; Nakajima et al. 1988; Cossu et al. 1990), and these reports have identified the cell types producing blood group antigens. These cell types are the mucous cells, serous cells, and duct cells. To date, however, direct differential staining of H Type 1 and H Type 2 antigens has not been examined because of the limited availability of a specific antibody. In the present study we examined the location of H Type 1 and H Type 2 antigens in cell types of the human submandibular glands using two MAbs and UEA-I lectin by an immunohistochemical technique. The competitive inhibition method against H Type 2, involving the use of ghosts and synthetic H Type 2, reduced the ability of MAb 1E3 to bind H Type 2 and enhanced its specificity for H Type 1. The mechanisms can be explained as follows. Because the antigen-antibody reaction is reversible, the formation of a stable complex is dependent on the affinity of antibody to antigen (Owen and Steward 1993). In general, an antibody with a high affinity requires a low antigen concentration to achieve binding of the antigen to half its combining sites, whereas an antibody with a low affinity requires a much higher concentration of antigen to achieve this. In other words, if more than two antigens are recognized by a single antibody molecule, the majority of binding sites of an antibody molecule are occupied by the antigen with the higher affinity under the same concentrations. In our competitive inhibition method described here, after 1E3 was absorbed with ghosts (H Type 2) in advance, the ability of 1E3 to bind H Type 2 in tissue sections was strikingly inhibited, whereas the ability to bind H Type 1 was only slightly affected. It was assumed that the binding sites on the 1E3 molecule occupied by ghosts would be replaced gradually by H Type 1 in tissue sections during the process of equilibration, in that the higher affinity of 1E3 to H Type 1 had the stronger tendency to form a stable complex with it. As a result, MAb 1E3 was discovered to be specific for anti-H Type 1 in this limited condition. MAb 1E3 before absorption stained not only mucous and duct cells but also stained red blood cells and vascular endothelium in the submandibular gland from secretors. On the contrary, MAb 1E3 stained only mucous and duct cells but did not stain red blood cells and vascular endothelium at all after absorption with a small quantity of ghosts (Figure 3). Therefore, even though it was a monoclonal antibody, the competitive inhibition method was confirmed as a successful immunohistochemical technique.
In the light of the present study, we concluded that the expression of H Type 1 in the mucous cells of submandibular glands was a consequence of the activity of the Se enzyme encoded by the Se gene (FUT2), whereas the expressions of the H Type 1 and the H Type 2 in striated and interlobular duct cells were a consequence of the H enzyme encoded by the H gene (FUT1). However, the possibility of Se enzyme expression in duct cells has not been completely eliminated, in that the stainings of duct cells from nonsecretors by 1E3 absorbed with ghosts were weaker than those from secretors. Previously, the Se enzyme was believed to use both the Type 1 and the Type 2 structures as acceptors but preferred the Type 1, and the H enzyme used only the Type 2 precursor as an acceptor (Oriol et al. 1986). This thinking had been based on comparing the apparent Kms for Type 1 and Type 2 precursors of the H and Se enzymes. However, Kyprianou et al. (1990) failed to discover any preference for either the Type 1 or the Type 2 precursors by highly purified plasma H Type α(1,2)fucosyltransferase, and obtained similar activities against the Type 1 and Type 2 precursors. Our results were similar to that of Kyprianou et al. (1990). When enzyme activities toward Type 1 and Type 2 precursors were determined after transient expression of FUT1 or FUT2 in COS7 cells, the H enzyme activities were similar for both Type 1 and Type 2 chain precursors as substrates to form H Type 1 and H Type 2 substances, whereas the formation of the H Type 1 catalyzed by the Se enzyme was about six times higher than that of the H Type 2 (Table 2). The results of the H and Se enzyme activities were consistent with our immunohistochemical findings. We have recently demonstrated that bone marrow cells and erythroleukemic HEL cells expressed FUT1 but did not express FUT2, whereas peripheral leukocytes and several leukemic cell lines expressed neither of them by a reverse transcription-polymerase chain reaction method (Koda et al. 1997). A correct conclusion of the distributions of the H and Se enzymes in the various cell types of tissues awaits direct immunohistochemical staining by a specific antibody against the H enzyme or the Se enzyme.
UEA-I has been known to be and is still used as a lectin specific for H Type 2 (Pereira et al. 1978; Holtöfer et al. 1982; Laden et al. 1984; Ito et al. 1989, 1990). Comparing the immunohistochemical and lectin histochemical results, we have found a significant difference in the binding properties between the monoclonal anti-H antibodies and UEA-I. MAb anti-H 3A5, specific for H Type 2 chain, reacted weakly with the mucous cells from secretor and failed to react with the serous cells regardless of the secretor status of the tissue donors. In contrast to MAb 3A5, the UEA-I stained the mucous cells, duct cells, erythrocytes, and the vascular endothelia, irrespective of the secretor status, and stained serous cells from secretors but not those from nonsecretors. Ito et al. (1989,1990) also obtained positive substances in serous cells from secretors but not from nonsecretors using UEA-I lectin his-tochemistry. Therefore, Ito et al. (1990) concluded that expression of the H antigen in serous cells was controlled by the Se gene. However, our present study demonstrated that no H antigen was present in serous acini of the submandibular gland, because there was no staining of the H antigens by the anti-H MAbs and no A and B antigens demonstrated previously (Ito et al. 1989; Laden et al. 1984). Therefore, the substance(s) in mucous cells from nonsecretors and in serous cells from secretors stained by UEA-I was not H Type 2 antigen (Figure 4). Despite being used as a blood group H Type 2-specific lectin for a long time (Pereira et al. 1978; Laden et al. 1984), UEA-I shows heterogeneous specificity. Recently, Mollicone et al. (1996) and Baldus et al. (1996) have systematically investigated the characterization of the binding specificity of UEA-I using an ELISA method. They found that UEA-I reacted mainly with H Type 2 and had a crossreaction with H Type 6, Ley, H Type 5, Lex, and A Type 2. The material(s) stained by UEA-I in serous cells, which appeared to be dependent on the secretor status of tissue donors, is not known at present. In view of the complexity of the distribution of blood group substances in tissues and the as yet unknown characterization of lectin, we suggest that the monoclonal antibody instead of lectin should be used for immunohistochemical localization of blood group substances.
Footnotes
Acknowledgements
Supported by a grant-in-aid for Scientific Research from the Ministry of Education, Science and Culture of Japan.
We are grateful to Drs K. Furukawa and S. Yazawa, Department of Legal Medicine, Gunma University School of Medicine, for the kind gift of the MAb anti-H 1E3, and to Mr Shigeo Kamimura for making serial thin sections.
