Abstract
Homeobox-containing (Hox) genes play important roles in development, particularly in the development of neurons and sensory organs, and in specification of body plan. The Hmx gene family is a new class of homeobox-containing genes defined by a conserved homeobox region and a characteristic pattern of expression in the central nervous system that is more rostral than that of the Hox genes. To date, three closely related members of the Hmx family, Hmx1, Hmx2, and Hmx3, have been described. All three Hmx genes are expressed in the craniofacial region of developing embryos. Here we show, for the first time, the expression of the transcription factor Hmx3 in postnatally developing salivary glands. Hmx3 protein is expressed in a cell type-specific manner in rat salivary glands. Hmx3 is present in both the nuclei and cytoplasm of specific groups of duct cells of the submandibular, parotid, and sublingual glands. Hmx3 expression increases during postnatal development of the submandibular gland. The duct cells show increasing concentrations of Hmx3 protein with progressive development of the submandibular gland. In contrast, the acinar cells of the three salivary glands do not exhibit detectable levels of Hmx3 protein.
H
The major salivary glands (submandibular, parotid, and sublingual) of mammals exist as three pairs of organs that cooperate functionally to produce saliva for the oral cavity. They all share a common developmental pattern in rodents, in that they fully develop post-natally (Denny et al. 1997). The submandibular gland (SMG) develops through the process of branching morphogenesis, undergoing a complex process of growth and differentiation during the prenatal and postnatal periods, which is regulated by hormones, elements in the extracellular matrix (ECM), and the autonomic nervous system. The acini are connected to the oral cavity by a rather complicated duct system. At birth (in rodents), the submandibular gland consists of a ramifying duct system ending in terminal tubules consisting of terminal tubule cells and proacinar cells. By day 2 the terminal tubules are mostly cell buds consisting of proacinar cells, which give rise to acinar cells during the first week of development. At the end of the first week, the cell buds are composed predominantly of acinar cells (Chang 1973). The terminal tubules are also the site of future intercalated ducts (IDs). The postnatal development of rat salivary glands can be divided into two phases. In the first phase, the secretory units of the adult type, the acini, are formed from the rudimentary secretory units known as terminal tubules. In the second phase, the striated ducts give rise to the granular convoluted tubules (Jacoby and Leeson 1959). The parenchyma of the mature submandibular gland of the rat consists of acini, IDs, granular convoluted tubules (GCTs), and striated ducts (SDs). The SDs are continuous with the interlobular ducts that form the common excretory duct (ED) leading to the oral cavity (Gresik 1980; Gresik et al. 1981).
The study presented here represents the first examination of the expression and distribution of a transcription factor, Hmx3, in postnatally developing SMG. We find that Hmx3 expression in SMG is increased during postnatal development. We have also examined the expression of the protein in all three salivary glands of adult rats. Hmx3 protein is present in all three salivary glands, (submandibular, parotid, and sublingual). In addition, Hmx3 expression is cell type-specific: The duct cells show increasing concentrations of Hmx3 with progressive SMG development. Hmx3 is present in both the nuclei and cytoplasm of specific sets of duct cells in the submandibular, sublingual, and parotid glands, but not all duct cells show nuclear localization of Hmx3. In contrast, acinar cells do not exhibit detectable levels of Hmx3 protein in any of the salivary glands.
Materials and Methods
Materials
The peroxidase-labeled streptavidin-biotin kit was from DAKO LSAB (Carpinteria, CA). The proteinase inhibitor cocktail was from Roche (Mannheim, Germany). The Bradford protein reageant and horseradish peroxidase-coupled goat anti-rabbit antibody were from Bio-Rad (Hercules, CA). Hybond ECL membranes and enhanced chemiluminescence (ECL) substrate were from Amersham Pharmacia Biotech (Arlington Heights, IL). The protein standards were the BenchMark Prestained Ladder from Gibco (Gaithersburg, MD). All other materials were purchased from commercial sources (Sigma; St. Louis, MO and Fisher, Pittsburgh, PA) and were of the highest purity available.
Animals
Female Sprague-Dawley rats (Charles River; Wilmington, DE) were kept in a temperature- and humidity-controlled environment (12 hr light/12 hr dark cycle) and had free access to water and standard laboratory chow. All experimental protocols were reviewed by the Mount Sinai Institutional Animal Care and Use committee and conducted in accordance with the NIH guidelines for the care and use of laboratory animals. Female adult rats and pups of 2, 3, 4, 5, 6, 8, 10, 15, 20, 25, 28, and 33 days of age were analyzed. The animals were sacrificed by CO2 and the salivary glands were removed. For morphological studies, the glands were fixed in 4% paraformaldehyde in 1 × PBS (0.01 M phosphate buffer, 2.7 mM potassium chloride, and 0.137 M sodium chloride, pH 7.4) for 2 hr to several days at 4C and embedded in paraffin. The glands for extraction of total proteins were frozen in liquid nitrogen and stored at −80C until used.
Staining of Submandibular Glands
Five-μm sections were stained with hematoxylin and eosin (H&E) for morphological examination.
Hmx Protein Localization in Salivary Glands
Five-μm paraffin sections were deparaffinized with two changes of xylene and rinsed with two changes of 100% ethanol and double distilled water. For immunostaining, a peroxidase-labeled streptavidin-biotin procedure (DAKO) was used. Sections were incubated with 3% H2O2 for 5 min and washed in water. They were rinsed with 1 × PBS, drained, and then the sections were covered with blocking solution (DAKO LSAB Kit) for 20 min. The primary antibody, rabbit anti-mouse Hmx3 [raised to an MAP-conjugated peptide in the COOH-terminal region of Hmx3 (IVRVPILYHENSAA EGAAAA)] was used at 1:100 dilution (in PBS, 0.1% BSA) for 60 min at room temperature. The specificity of the antibody is shown in Figure 6. As a negative control, BSA was applied in place of the antibody. The slides were washed in PBS, and biotinylated anti-rabbit immunoglobulin (linking solution in the kit) was applied to the sections for 20 min. After washing, freshly made streptavidin-peroxide solution was applied to the slides for 20 min. The sections were then washed in PBS. Diaminobenzidine (DAB) was added for approximately 6 min, the slides were washed in water, and then were counterstained with 0.2% Light Green (Fisher). The sections were then dehydrated and coverslipped.
Extraction of Total Proteins
Total proteins were prepared from the submandibular (SMG), sublingual (SL), and parotid (PA) glands of adult Sprague-Dawley rats. The tissues were removed, cut into small pieces, and homogenized in 1 ml of buffer (per 160 mg tissue) containing 1 × PBS, 0.25% Triton X-100, and a complete protease inhibitor cocktail (1 tablet per 50 ml buffer; Roche). The homogenates were centrifuged at 12,000 rpm at 4C. Protein concentrations of the supernatants were determined using the Bradford protein assay reagent (Bio-Rad). Extracts containing total proteins were aliquotted and stored at −80C until use.
Specificity of the Hmx3 Antibody
Western blotting analyses were performed using the homeodomain-containing proteins GST-Hmx3, GST-Hmx1, PreScission cleaved (P-cleaved) Hmx1 pure protein, P-cleaved Nkx2.5 pure protein, P-cleaved PITX2C pure protein, P-cleaved PITX2A pure protein, and P-cleaved Msx2 pure protein. These proteins were purified and subjected to PreScission cleavage as previously described (Amendt et al. 1999, Cox et al. 2002). Various concentrations of these proteins were electrophoresed on a 10% SDS-polyacrylamide gel and transferred to PDF filters (Millipore; Bedford, MA). The membranes were then treated with the polyclonal Hmx3 antibody at a 1:2000 dilution and the secondary antibody, horseradish peroxidase-coupled goat anti-rabbit (anti-rabbit HRP Ab), at a 1:5000 dilution. Immunodetection was performed using the ECL substrate (Amersham Pharmacia Biotech). As controls, GST-Hmx1 and GST only were analyzed by Western blots to demonstrate that the Hmx3 antibody did not bind to GST. The protein markers are from BRL and range from 200 to 29 kD.
Western Blotting Analyses of Hmx3 in Salivary Glands
Six micrograms of total protein were loaded into each well of a 12% SDS-polyacrylamide gel, electrophoresed at 100 V for 50–60 min at 4C, and electrotransferred (Bio-Rad Mini PROTEAN II Cell) to Hybond ECL membranes (Amersham Pharmacia). As a positive control, 36 and 360 ng of pGST-Hmx 3, which includes amino acids 309–458 containing the homeodomain 20-amino-terminal flanking residues and the entire COOH-terminal region, were electrophoresed in the same gel. The membranes were probed with the polyclonal antibody to Hmx3 at a 1:2000 dilution as described above. The blots were then developed using the anti-rabbit HRP antibody at a 1:4000 dilution (Bio-Rad). Immunodetection was performed using the ECL substrate from Amersham according to the manufacturer's instructions. The protein markers were the BenchMark Prestained Ladder and ranged from 9.3 to 172.6 kD (Gibco).
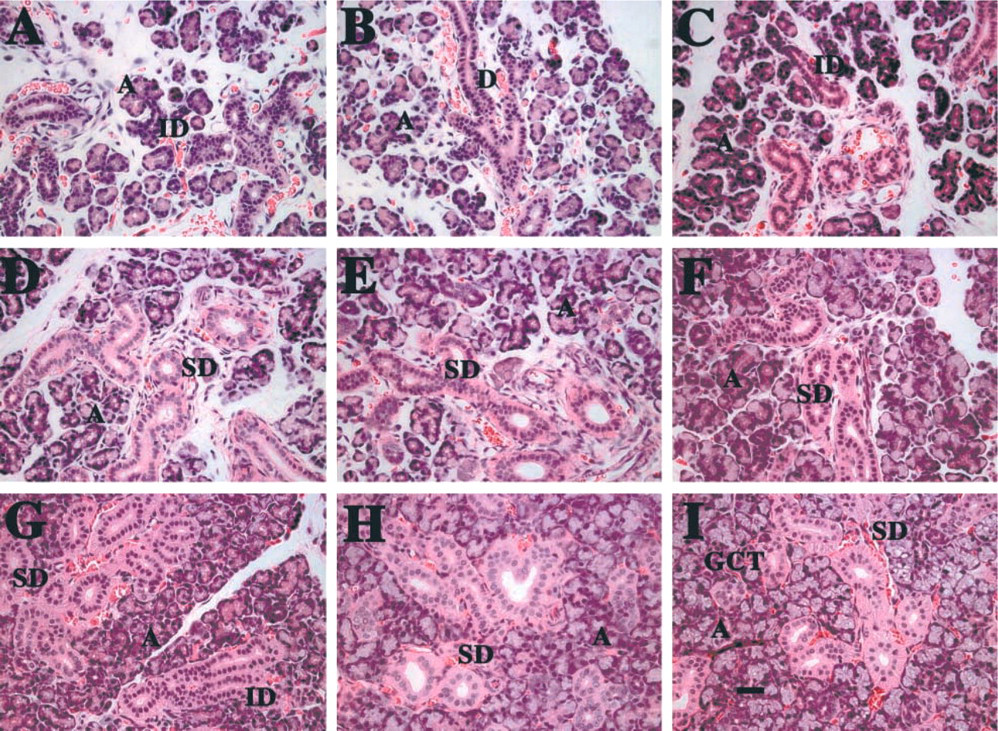
Submandibular glands of developing rats, stained with H&E. (
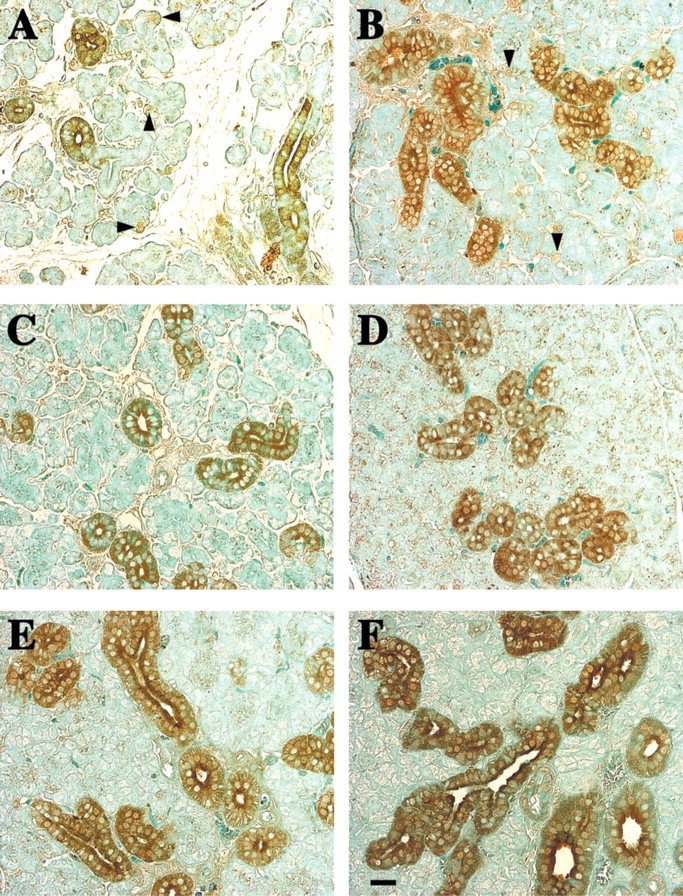
Increased Hmx protein in the ducts of submandibular glands of developing rats. (
Results
The well-documented postnatal development of rat SMGs is depicted in Figure 1. Particular emphasis on the development of the duct components is described. During days 2, 4, and 6, progressive differentiation of the acinar cell (A) compartment is noted, with increasing elongation of the ductal components of the gland (Figures 1A-1C). In addition, there is a large amount of loose connective tissue connecting the newly formed lobes, and an extensive blood supply. Short intercalated ducts (IDs) are observed at day 2 (Figure 1A), but by days 4 and 6, long and branched ducts, whose epithelial cells are more cuboidal, are evident (Figure 1C). A few striated ducts (SDs) are detected at 8 days, and the acinar (A) components are much more numerous (Figure 1D). Many striated ducts are present by day 15, as well as a few granular convoluted tubules (Figure 1F). The granular convoluted tubule components, as well as the excretory ducts, are greatly increased during days 20, 25, and 33 (Figures 1G-1I).
Immunocytochemical analyses of Hmx3 protein distribution indicate that, as the SMG develops with progressive increase in the duct components of the gland, increased Hmx3 protein is evident in the ducts (Figure 2). It is also interesting to note that, at 4 and 10 days of development of the SMG, Hmx3 protein is present in specific cells of the cell buds (Figures 2A and 2B, arrowheads). These cells could be those that give rise to or are the precursors of duct cells. When the sections are visualized by Nomarski optics, Hmx3 is observed in specific nuclei and the cytoplasm of some duct cells as early as 4 days of development (Figure 3B). By day 10, the duct epithelial cells contain Hmx3 protein in both their nuclei and cytoplasm. However, not all of the nuclei contain Hmx3 (Figure 3D). At 15 days, immunostaining for Hmx3 can be observed in both the nuclei and the cytoplasm of the SDs and the granular convoluted tubule (GCT) cells (Figure 3F). The majority of the duct components contain large amounts of Hmx3 at 20 days of development (Figure 3H). At 25 and 28 days of development of the SMG, there is a large accumulation of Hmx3 in the cytoplasm of the larger ducts, but not all of the nuclei show nuclear staining, as was also observed at the earlier time points (Figures 3J and 3L). Note that there is slight peroxidase staining in the ducts in some BSA-treated SMGs (Figures 3A, 3C, 3E, 3G, 3I, and 3K). Higher magnification using Nomarski imaging of Hmx3 antibody staining of adult female SMGs demonstrates that the nuclei and cytoplasm of only certain duct cells contain Hmx3 protein (Figure 4]). At lower magnification, it is obvious that not all duct cells are stained (Figure 4A). Staining of nuclei and cytoplasm of specific duct cells can be discerned at higher magnification (Figure 4B).
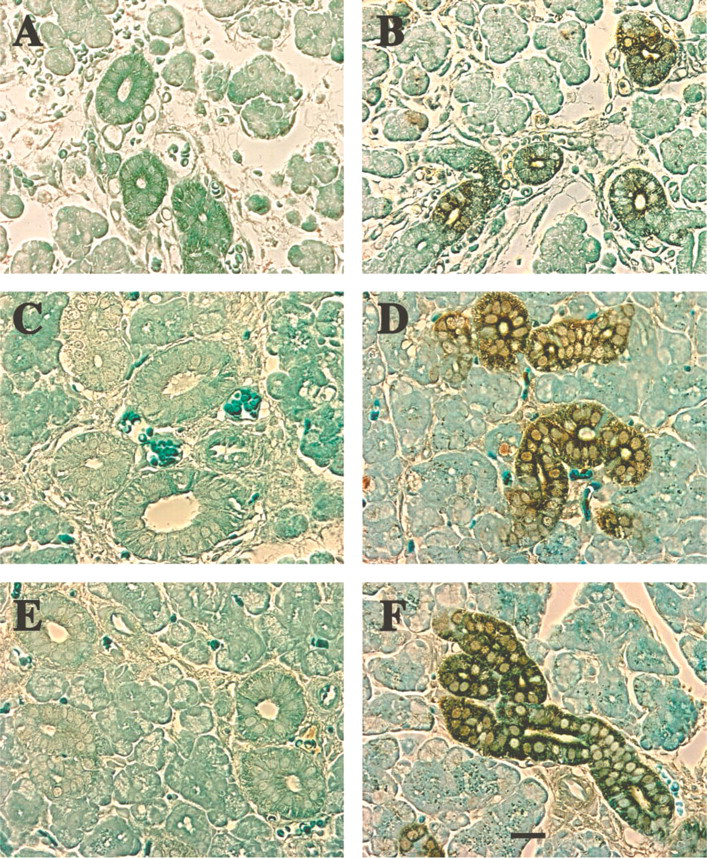
The other two salivary glands from adult rats, the parotid and the sublingual, both show Hmx3 protein in the epithelial cells of the ducts (Figure 5). In the case of the sublingual gland, whose secretory components consist entirely of mucous acini, slight antibody staining is observed in the intercalated ducts. More pronounced staining in the striated duct cells and less staining in a few mature granular duct cells are observed (Figure 5C). Not all ducts are stained, nor are all epithelial cells in the ducts stained. In addition, both the nuclei and the cytoplasm of some cells are stained, but the staining is principally cytoplasmic. Hmx3 protein is also observed in selective cells of the acini (arrow, Figure 5C). The immunocytochemistry of the parotid also shows slight staining in the intercalated duct cells and the striated duct cells (Figure 5I). In both cases, the striated ducts seem to be more heavily stained. The H&E-stained sections of the submandibular, parotid, and sublingual glands demonstrate the typical staining pattern of the acini and ducts (Figures 5A, 5D, and 5G).
The concentration and the approximate size of rat Hmx3 protein were determined by Western blotting analyses of equivalent amounts of total protein from the three salivary glands using the Hmx3-specific antibody. The difference in the concentration of Hmx3 protein in the three salivary glands is most striking. The concentration of Hmx3 protein is significantly higher in the submandibular and sublingual glands than in the parotid gland (Figure 6A). Three major proteins (approximately 77.5, 48.8, and 44 kD) are detected in the SMG protein extract by the Hmx3 antibody (Figure 6A, Lane 1). Two major proteins, approximately 77.5 kD and 72 kD in size, are observed in the sublingual gland (Figure 6A, Lane 2). The parotid shows very faint binding of Hmx3 antibody to two proteins of 77.5 and 72 kD (Figure 6A, Lane 3). The smaller molecular weight proteins are, more than likely, difficult to detect due to low concentration (they show up on much longer exposure) in the sublingual and parotid glands (data not shown). The 48.8- and 44-kD Hmx3 proteins found in the salivary glands are in the range of the deduced size for Hmx3 protein: 51 kD (Bober et al. 1994) and 43 kD (Wang et al. 2000). These data are consistent with the Hmx3 immunocytochemistry data in which the concentration of Hmx3 is much higher in the SMG.
The specificity of the Hmx3 antibody is demonstrated by Western blotting analyses of five different homeodomain proteins in addition to Hmx3: Hmx1, Nkx2.5, PITX2C, PITX2A, and Msx2. Figures 6B and 6C show two different exposures of the Western blots performed in the Amendt lab (University of Tulsa) for these homeodomain proteins. The approximate molecular weights of the homeodomain proteins analyzed are GST-Hmx1 64 kD, purified Hmx1 37 kD, purified Nkx2.5 42 kD, purified PITX2C 36 kD, purified PITX2A 32 kD, purified Msx2 30 kD. The Hmx3 antibody is specific for Hmx3 and does not bind GST-Hmx1 (Figures 6B and 6C, Lane 2), purified Hmx1 (Figures 6B and 6C, Lane 3), purified Nkx 2.5 (Figures 6B and 6C, Lane 7), purified PITX2A (Figures 6B and 6C, Lane 9), and purified Msx2 (Figures 6B and 6C, Lane 10). Furthermore, the Hmx3 antibody does not bind GST (Figures 6B and 6C, Lane 4). However, it is of note that PITX2C (Figures 6B and 6C, Lane 8) shows binding of the Hmx3 antibody represented by two bands in molecular weight regions that are not specific for PITX2C or Hmx3. We do not know at this time what these two bands represent.
Nomarski imaging of sections immunoperoxidase stained for Hmx protein in submandibular glands of developing rats. (
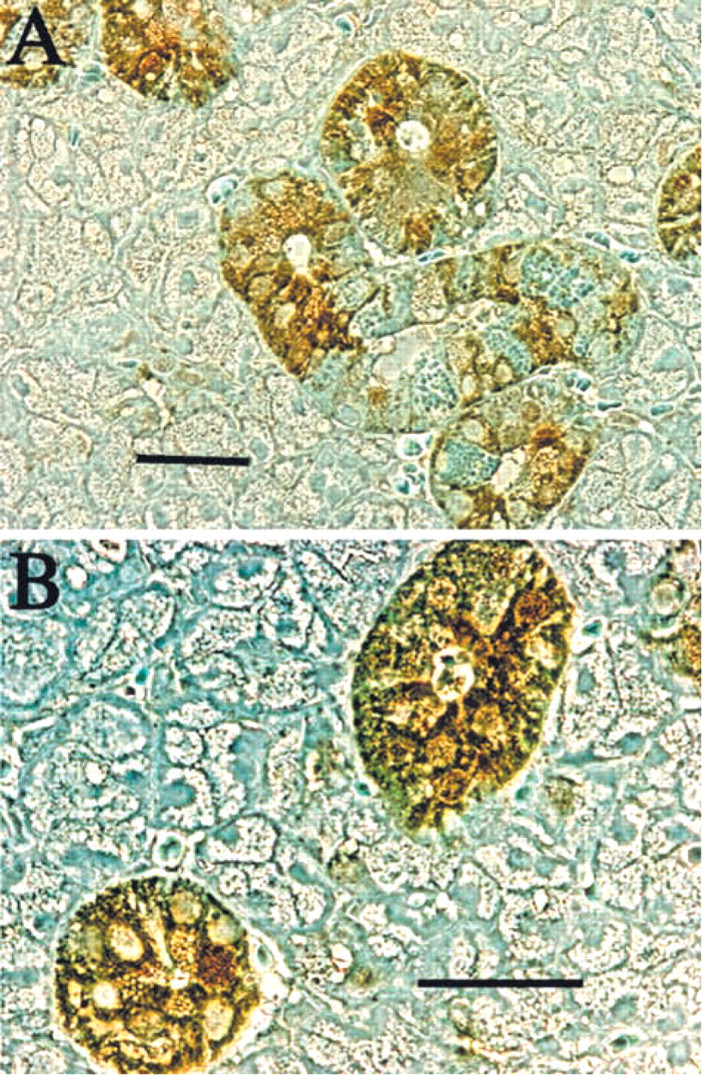
Nomarski imaging of immunoperoxidase-stained Hmx3 protein in submandibular glands of adult rats. (
Discussion
The three major rodent salivary glands develop through a similar pattern of morphogenesis that is regulated by elements in the ECM, hormones, and the autonomic nervous system. Previous studies have indicated that salivary gland development entails temporal and spatial coordination among cell proliferation, cytoarchitectural organization, and the establishment of stable cell-substratum interactions (Fernandes et al. 1999). However, the molecular mechanisms underlying these events are not known. Rodent parotid, sublingual, and submandibular glands begin to form around embryonic day 12 (E12). These glands are not readily distinguishable from each other until roughly E15, at which time the parotid gland is not only much smaller than the submandibular and sublingual gland but each gland's characteristic acinar organization of serous and mucous cells becomes apparent (Borghese 1950; Kaufman 1992). The major development of salivary glands occurs postnatally. At birth, the salivary glands consist of ramifying duct systems ending in transient tubular structures (terminal tubules), which give rise to acini and are the sites of future intercalated ducts (Jacoby and Leeson 1959). The three glands differ in the extent to which the intralobular ducts are developed. In the parotid gland, the intercalated ducts are long and abundant, whereas the striated ducts are less elaborate. The striated ducts are best developed in the submandibular gland; the intercalated ducts are not as numerous. In the sublingual gland, both intercalated and striated ducts are poorly developed (Denny et al. 1997). Little is known, however, about genetic programs regulating the development of rodent salivary glands. The study presented here represents the first examination of the distribution and expression of a transcription factor, Hmx3, in the three rat salivary glands. Because significant development occurs postnatally in salivary glands, we have examined the expression of the homeobox gene, Hmx3, in the postnatally developing submandibular gland.
In recent years, a number of homeobox-containing genes have been identified. The homeobox genes in Drosophila show restricted patterns of expression during embryonic development and are believed to encode transcription factors that recognize and bind to specific DNA sequences via their highly conserved homeodomains. Based on their expression patterns, homeobox genes are involved in the development of the sensory organs and the central nervous system (Bober et al. 1994; Rinkwitz-Brandt et al. 1995) and in the creation of craniofacial structures (Stadler et al. 1992). Studies in Drosophila have suggested a model for regulatory events that determine the sites at which salivary glands will eventually develop. The position of the salivary gland primordia, the number of cells committed to form salivary glands, and the difference between secretory and duct cells are controlled by localized expression of transcription factors and by localized cell signaling. Two homeotic genes, Sex combs reduced (Scr) and Abdominal-B (Abd-B), and a third gene encoding a zinc-finger protein, teashirt (tsh), restrict salivary glands to the anterior-posterior position in the embryo to parasegment 2 (PS2) (Panzer et al. 1992; Andrew et al. 1994). A signaling pathway within PS2, initiated by the product of the decapentaplegic (dpp) gene, limits which cells are committed to form salivary glands (Panzer et al. 1992; Henderson et al. 1999). Epidermal growth factor (EGF) signaling in the most ventral cells of the salivary gland primordia specifies a duct cell fate (Kuo et al. 1996). Formation of salivary glands also requires the function of the transcription factors extradenticle (exd) and homothorax (hth) (Henderson and Andrew 2000; Andrew et al. 2000). The positive action of the Scr gene initially dictates where Drosophila salivary glands develop (Panzer et al. 1992). Heat shock-activated Scr minus mutants do not initiate salivary gland formation. Although the homologue of Scr, hox-1.3, has been identified in mice, its direct participation in embryonic development of salivary glands is unknown (Zhao et al. 1993). However, immunocytochemistry for the hox-1.3 gene product and Northern blots show distribution in the head at late stages of embryological development, and hox-1.3 transcripts in some cranial tissues in adult mice (Odenwald et al. 1987; Tani et al. 1989). Expression of the Hox-1.3 gene in adult animals is not surprising because many mammalian homeobox genes are expressed in adult tissues. For example, every mouse homeobox gene whose expression has been examined in detail in adult tissues has been found to be expressed in both embryonic and adult tissues (Awgulewitsch et al. 1986; Colberg-Poley et al. 1985; Jackson et al. 1985; Rubin et al. 1986; Wogelmuth et al. 1986).
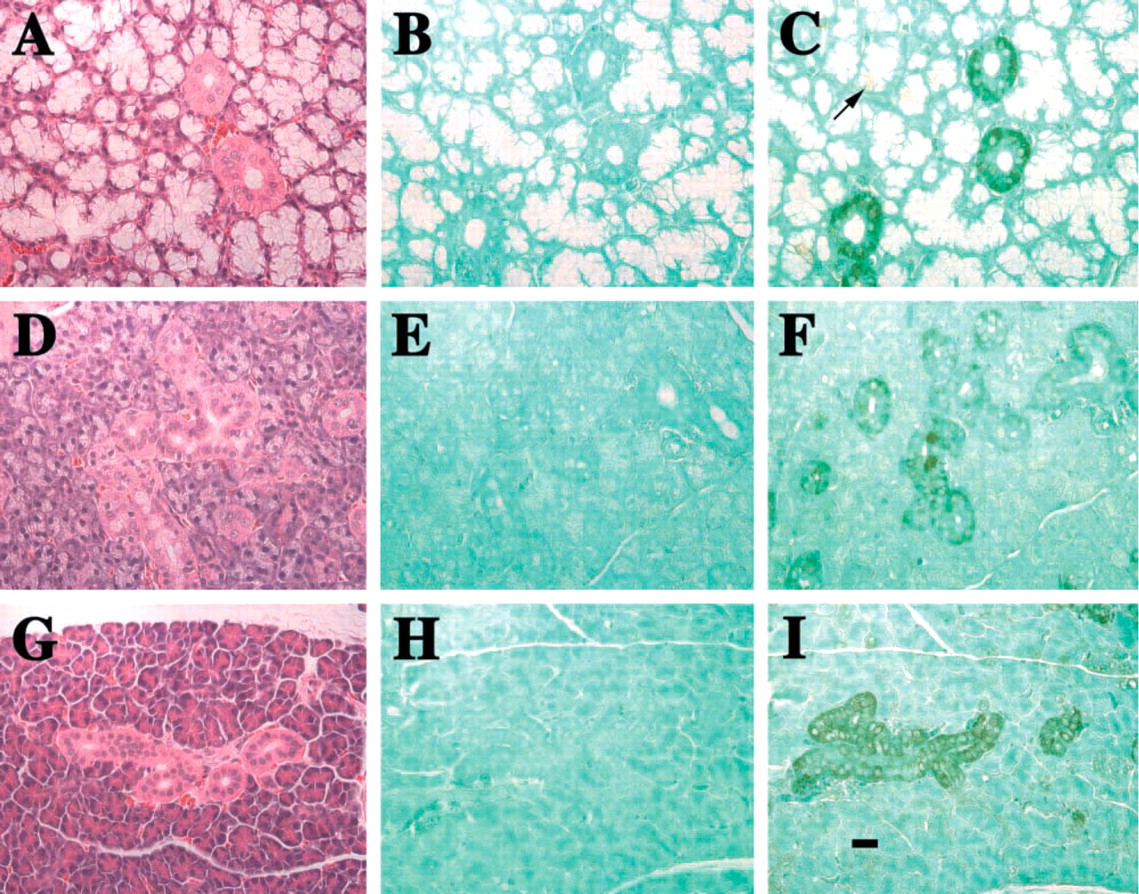
Immunoperoxidase staining for Hmx protein in sublingual, submandibular, and parotid glands of adult rats. (
To date, the only homeobox gene that had been reported to be expressed in mouse submandibular glands is Tlx-1 (Raju et al. 1993). Tlx-1 is a murine homologue of the homeobox gene Hox 11 with putative oncogenic potential. Tlx-1, originally discovered at the chromosome 10 breakpoint in T-cell acute lymphoblastic leukemias bearing chromosomal translocations (Dubé et al. 1991; Hatano et al. 1991; Kennedy et al. 1991), has a restricted pattern of expression. It is largely confined to the mouth region, including the tongue, mandible, tooth primordia, salivary glands, and specific cranial ganglia that innervate these structures. Tlx-1 expression is observed in the SMG from the earliest stages of its embryonic development, and at E15.5 is distributed evenly throughout the lobules (Raju et al. 1993). Tlx-1 expression is also observed in the facial and glossopharyngeal ganglia that receive sensory input for taste from the tongue and which innervate the salivary glands (Raju et al. 1993).
The Hmx family is a new class of homeobox-containing genes. Three new members of the Hmx gene family, Hmx 1, Hmx 2, and Hmx 3, have been identified in humans, mice, and Drosophila (Stadler et al. 1992,1995; Yoshiura et al. 1998; Wang et al. 2000, 2001). The Hmx genes are believed to play important roles in the development of sensory organs and neurons (He et al. 1989; Gruss and Walther 1992; McGinnis and Krumlauf 1992; Herr and Cleary 1995). All three murine genes are expressed in the central and peripheral nervous systems during embryogenesis (Bober et al. 1994; Rinkwitz-Brandt et al. 1995; Wang et al. 1998, 2000). Gene knockout of Hmx3 in mice leads to behavioral abnormalities such as hyperactivity and circling movements; these balance defects are correlated with anomalies of the vestibular organ in Hmx3 null mice (Hadrys et al. 1998; Wang et al. 1998). In the Hmx3 null inner ear, the gross structure of the three semicircular canals forms but the horizontal ampulla and crista are absent. Loss of Hmx3 primarily influences the development of a subset of sensory receptor cells, but not the overall morphogenesis of the inner ear (Hadrys et al. 1998; Wang et al. 1998). In addition, the majority of Hmx3 null females have a reproductive defect; there is a failure of the Hmx 3 null uterus to support normal postimplantation pregnancy (Wang et al. 1998). However, it is not known how the knockout of the Hmx3 gene affects salivary glands because these investigators did not examine salivary glands (personal communication; Thomas Lufkin, Mount Sinai School of Medicine).
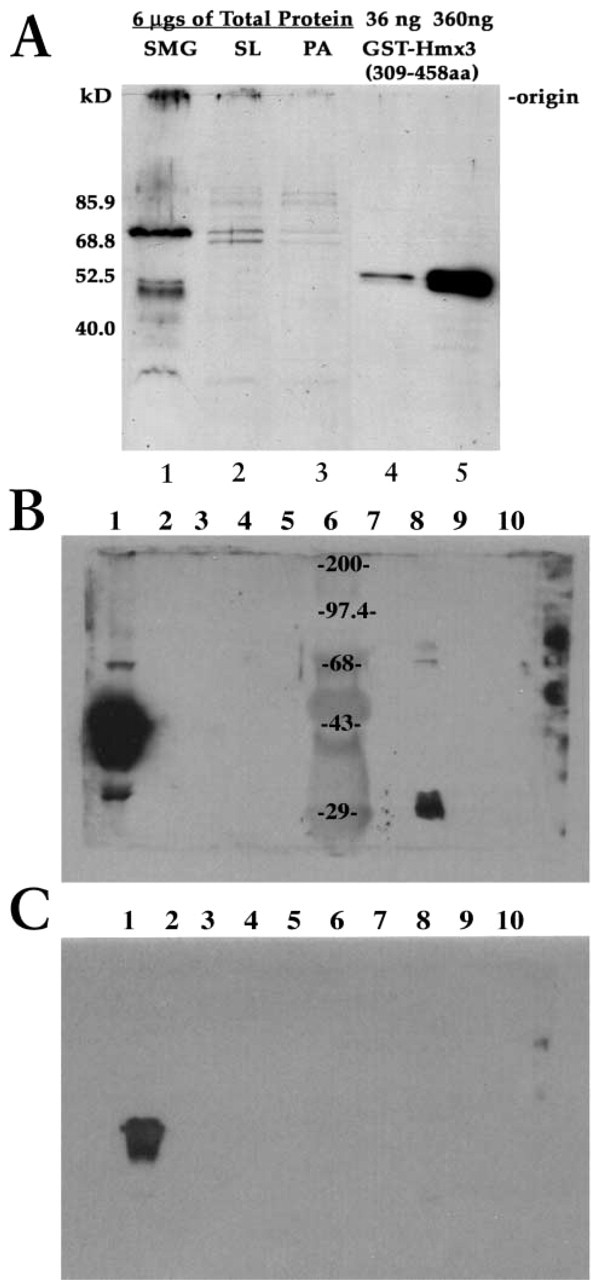
Western blotting analyses of total proteins in sublingual, submandibular, and parotid glands of adult rats using Hmx-specific antibody. (
The study presented here provides the first evidence of the presence of the homeodomain protein Hmx3 in rat salivary glands and, in particular, in the postnatally developing submandibular gland. Our findings are in agreement with previous reports of Hmx1, Hmx2, and Hmx3 expression in other sensory organ-related structures (He et al. 1989; Gruss and Walther 1992; McGinnis and Krumlauf 1992; Herr and Cleary 1995). Hmx3 protein is present in all three rat salivary glands. How Hmx3 functions in salivary glands is unknown. The only clue to date is that Hmx3 shares sequence homology with DNA binding proteins, suggesting that the Hmx3, which is localized in the nucleus of specific duct cells, functions by regulating transcription. Not only is Hmx3 localized in the nuclei, it is also found in cytoplasm of specific duct cells in the salivary glands. These findings are not without precedent because other transcription factors are also localized in both the nuclei and the cytoplasm. These include the homeodomain protein engrailed, a transcription factor that is involved in midbrain/hindbrain patterning (Maizel et al. 1999). We do not know whether the cytoplasmic location of Hmx3 indicates a regulatory aspect of its role(s) in transcription, or whether it reflects an additional cytoplasmic function. Hmx3 protein increases in amount in duct cells with progressive development of the duct compartment of submandibular glands. However, Hmx3 is expressed only in a subset of duct cells and not in the acini of the three salivary glands. Therefore, the expression of Hmx3 shows significant cell type specificity. We note, however, that although we did not detect Hmx3 in the acinar cells, we cannot rule out the possibility that this is simply due to the level of sensitivity of immunocytochemistry.
The fact that Hmx3 is expressed in the striated, granular, and intercalated ducts suggests that Hmx3 has a common function in these three diverse duct systems. Relatively little is known about the physiology of salivary duct cells. However, what is known is that the duct system of submandibular glands serves as a conduit for the fluid rich in exocrine proteins that is synthesized in the acini. As this primary fluid is carried into the oral cavity via the ducts, it is progressively modified. It is at the level of the epithelial cells of the ducts that a considerable amount of electrolyte flux occurs. Here, most of the sodium and chloride ions are reabsorbed and a small amount of potassium and bicarbonate ions is secreted. In addition, a few members of the aquaporin protein family of water channels that play a fundamental role in transmembrane water transport are expressed in salivary glands (Matsuzaki et al. 1999; Gresz et al. 2001). Available data suggest that aquaporin 5 (AQP 5) is found in both acini and duct cells (Matsuzaki et al. 1999), whereas AQP 3 is found in mucous and serous acini (Gresz et al. 2001). However, it is possible that duct-specific aquaporin(s) exist, because acinar cells are water-permeable and duct cells are water-impermeable, suggesting water channels of different protein composition in the two cell types. Although it is premature to speculate about the function of Hmx3 in salivary duct cells, it is plausible that it could participate in regulating the expression of those genes involved in electrolyte transport or possible duct-specific aquaporin genes involved in water transport. Furthermore, it remains to be determined if Hmx3 plays a role in controlling the postnatal development of certain duct components of salivary glands or is temporally expressed as a consequence of postnatal development of salivary glands. Further insight into Hmx3 function(s) in salivary glands should come from the identification of genes under its transcriptional control, as well as through generation of mutant animals with targeted mutations in the Hmx3 gene.
Footnotes
Acknowledgements
Supported by Grant DE08174 to PAS from the National Institute of Dental and Craniofacial Research, National Institutes of Health, and DE13076 to AFR. Microscopy was performed at the MSSM-Microscopy Shared Resource Facility, supported, in part, by funding from NIH-NCI shared resources grant (1 R24 CA095823–01).
We wish to thank Drs Tibor Barka and Serafín Piñol-Roma for critical reading of the manuscript. Thanks also to Konstantin Gaengel for help with the imaging and Jian Luo for technical assistance.
