Abstract
We have characterized the subcellular distribution of S100A3, a cysteine-rich calcium binding protein, in human scalp hair shaft. This was accomplished using rapid-freezing immunocytochemistry, a technique that combines rapid-freezing, freeze-substitution fixation without chemical fixatives, and subsequent electron microscopic detection of immunocytochemical labeling. This technique preserves both the antigenicity and the ultrastructural integrity of fully keratinized tissues, which are highly unmanageable when prepared for immunoelectron microscopy. In the hair shaft, S100A3 was primarily identified in the endocuticle and was also present in the intermacrofibrillar matrix surrounding macrofibril bundles of intermediate filament keratins in cortex cells. Double immunolabeling of S100A3 and hair keratins revealed the in situ spatial relationship between them. In the endocuticle, S100A3 was present on the inner portion of the endocuticle adjacent to the cell membrane complex, whereas hair keratins were present on the outer portion. These results provide the first ultrastructural evidence that an S100 protein is localized in specific subcompartments in human hair cells.
Keywords
W
S100A3 was initially described by Engelkamp et al. (1993). Its gene is tightly clustered with other S100 genes in the genome located on human chromosome 1q21, and it is distinguished by its high cysteine content (10%). Although many of its biochemical properties have been described (Föhr et al. 1995), very little information about which cells or tissues express S100A3 is currently available. Recently, however, we observed that S100A3 was highly expressed in human hair (Kizawa et al. 1996).
In this study we show the unique distribution of S100A3 in the ultrastructure of the human scalp hair shaft. The hair shaft, which projects above the surface of the skin, consists of two or three layers of highly organized keratins: the cuticle as the outermost layer, the cortex as the inner bulk, and the medulla as the central core (Figure 1). The medulla is absent in finer hairs. Localization of S100A3 was accomplished with rapid-freezing immunocytochemistry because this technique preserves both antigenicity and ultrastructural integrity of the hair shaft. Being fully keratinized, the hair shaft is one of the most unmanageable samples to prepare for immunoelectron microscopy (Takizawa et al. 1998a,b). We observed that S100A3 is located primarily within the endocuticle in cuticle cells and it is also present in the intermacrofibrillar matrix in cortex cells. These findings suggest that S100A3 may function to preserve the structural integrity of hair shafts. A preliminary account of this work has appeared in abstract form (Takizawa et al. 1996).
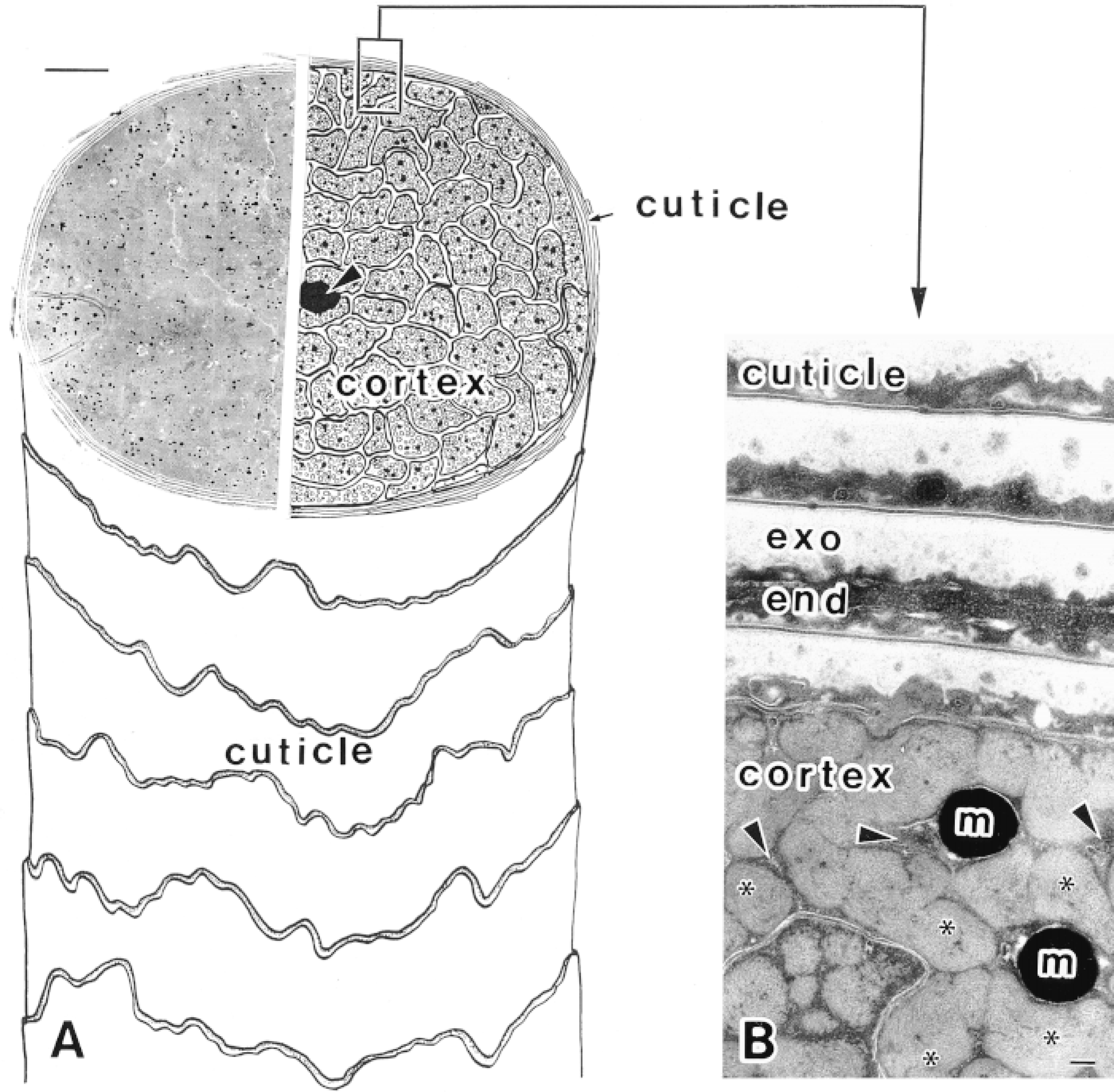
Human scalp hair shaft. (A) The structure of a hair shaft is shown schematically (right) and in a lowmagnification electron micrograph (left). The cuticle, the outer layer of the hair shaft, consists of several sheaths overlapping flattened cuticle cells, the cortex, the inner bulk of the hair shaft, contains closely packed cortex cells, and the medulla, the central core (arrowhead). Bar = 10 μm. (B) Higher-magnification electron micrograph of a hair shaft. In the cuticle, exocuticle (exo), and endocuticle (end) are evident. In the cortex, macrofibril bundles of intermediate filament keratins (∗), intermacrofibrillar matrix (arrowheads), and melanin granules (m) are also observed. Bar = 0.1 μm. The hair shafts in the electron micrographs were processed by rapid-freezing and subsequent freeze-substitution in OsO4-acetone (Takizawa et al. 1998a).
Materials and Methods
Reagents
Bovine serum albumin (BSA), dimethylformamide (DMF), and normal goat serum were purchased from Sigma (St Louis, MO). Glutaraldehyde (25% aqueous) and polyoxy-ethylenesorbitan monolaurate (Tween 20) were obtained from Nacalai Tesque (Kyoto, Japan). Medium grade LR White resin was supplied by London Resin (London, UK). All other chemicals were of the highest grade available.
An affinity-purified rabbit antibody against a synthetic peptide corresponding to residues 60–74 of human S100A3 was employed. Characterization of the antibody and its specificity was carried out as described previously (Kizawa et al. 1996). An affinity-purified mouse anti-human hair keratin antibody was prepared by immunization of the S-carboxymethyl derivative of purified human hair keratins as described previously (Uchiwa et al. 1994). Fluorescein-labeled goat anti-rabbit IgG and SlowFade-Light Antifade kit were obtained from Molecular Probes (Eugene, OR). Goat antirabbit IgG conjugated to 10-nm colloidal gold was obtained from Zymed (South San Francisco, CA). Goat anti-mouse IgG conjugated to 15-nm colloidal gold was purchased from British BioCell (Cardiff, UK). Carboxylated polystyrene beads (Immutex; 0.19 μm in diameter) were from Japan Synthetic Rubber (Tokyo, Japan). All immunological reagents were handled in accordance with the manufacturer's recommendations.
Hair Samples
Healthy, chemically untreated sections of scalp hair shaft located 15–20 cm from the surface of the skin were taken from Japanese girls 12–13 years of age.
SDS-PAGE and Western Blotting
The relative quantities of S100A3 protein in extracts of the cuticle and cortex of hair shafts were estimated by Western blot. The extracts were prepared as described previously (Kizawa et al. 1996). Briefly, the hair shafts were divided into cuticle fragments and the cylindrical core of cortex by vigorous agitation using a 7010 Waring blendor (Waring; New Hartford, CT) (Swift and Bews 1974). The cuticle fragments and the cortex were then extracted in 0.2 M Tris containing 8 M urea and 0.2 M 2-mercaptoethanol (pH 9.5) under nitrogen at 50C for 3 hr (Baden et al. 1980). The suspensions were then centrifuged at 10,000 x g for 20 min to obtain supernatant as protein extracts. The extracts were not treated with iodoacetic acid to give the S-carboxymethyl (SCM) derivative.
An aliquot of 0.1–10 μg of the cuticle extract or 1–30 μg of the cortex extract was applied to each lane. The extracts were resolved on a 15% polyacrylamide gel in the presence of 6 M urea and then electrophoretically transferred to immunoblotting membranes [Hybond-enhanced chemiluminescence (Hybond-ECL); Amersham, Little Chalfont, UK]. The membranes were blocked with 5% nonfat dry milk and probed with rabbit anti-human S100A3 (1 μg/ml); bound antibody was visualized using an ECL detection system (Amersham).
Immunofluorescence Microscopy
Recently, the immunohistochemical distribution of S100A2, S100A4, and S100A6 in the skin has been reported by Shrestha et al. (1998). In their study, these S100A proteins were present in epidermal basal cells and/or hair root sheath cells but were absent in hair cells. To confirm the cellular distribution of S100A3, normal human scalp skin was examined by immunofluorescence microscopy. The scalp tissue was fixed with 4% paraformaldehyde in 0.1 M cacodylate buffer (pH 7.4) containing 5% sucrose at 22C for 2 hr. The samples were washed in the same buffer, dehydrated in a graded series of DMF, embedded in LR White resin, and then polymerized at either 4C under UV radiation or at 50–60C in an oven (Yokota and Okada 1997).
Sections 2 μm thick were cut and mounted on silane-coated glass coverslips (13 mm in diameter; number 1 thickness). Once on the coverslips, sections were incubated in PBS containing 1% BSA to block nonspecific protein binding sites and then incubated with rabbit anti-human S100A3 (5–20 μg/ml) for 120 min at 22C. The sections were then rinsed five times in PBS over 15 min. Primary antibody binding sites were detected by incubating specimens for 60–120 min at 22C with fluorescein-conjugated goat anti-rabbit IgG (34 μg/ml). All antibody solutions were diluted with PBS containing 1% BSA and 10% normal goat serum. After PBS washing, the coverslips were mounted upside down on glass slides with SlowFade-Light Antifade reagent and then examined by epifluorescence and differential interference contrast (DIC) microscopy with an Olympus Provis AX80 equipped with a U-MCB photographic attachment and a PM-C35DX camera (Tokyo, Japan). Controls received the same treatment except that primary antibody was replaced by PBS or nonimmune rabbit serum.
Rapid-freezing Immunocytochemistry
The subcellular distribution of S100A3 in human scalp hair shafts was visualized using rapid-freezing immunocytochemistry as described previously (Takizawa et al. 1998b). Briefly, small pieces of the hair shafts (0.5 cm in length) were rapidly frozen on a liquid nitrogen-cooled copper block in a Polaron E7200 Slammer quick-freezing apparatus (Watford, UK) and then freeze-substituted in acetone at −80C for 3 days without chemical fixatives. After freeze-substitution fixation, the temperature was gradually increased, first to −20C for 1 hr and then to 4C. Acetone was substituted by graded DMF and then DMF/LR White resin mixtures. The mixtures were exchanged three times with pure resin, into which the specimens were subsequently embedded. The resin was then polymerized as described above.
Ultrathin sections were cut with a diamond knife and mounted on formvar-coated nickel grids. Once on the grids, sections were treated with BSA-PBS and then incubated with anti-S100A3 as described above. After five rinses in PBS, the sections were incubated with 10-nm gold-conjugated goat anti-rabbit IgG (A520 was 0.15) for 120 min at 22C. After immunostaining, the sections were fixed with 2% glutaraldehyde, stained with uranyl acetate and lead citrate, and observed with a Hitachi H-7000 electron microscope (Hitachinaka, Japan) operated at 75 kV. Controls received the same treatment except that primary antibody was replaced by PBS or nonimmune rabbit serum.
Some specimens were incubated with a mixture of rabbit anti-S100A3 antibody and mouse anti-hair keratin antibody (4 μg/ml). Anti-S100A3 binding sites were detected with 10-nm colloidal gold-conjugated IgG, whereas anti-hair keratin antibody binding was simultaneously visualized using 15-nm colloidal gold-conjugated IgG (A520 was 0.20).
Morphometric Analysis of the Distribution of S100A3 and Hair Keratins in the Cuticle
To confirm the electron microscopic observation of the specific distribution of S100A3 and hair keratins in cuticle cells, the relative distribution of these proteins was determined by measuring the distances from the inner-facing aspect of each cuticle cell to the respective immunogold particle. The measurements were made using electron micrographs of double-labeled human hair shafts printed at the same magnification (x 120,000).
Immunoscanning Electron Microscopy
Anti-S100A3 (1 mg/ml) was added to a 1% suspension of carboxylated polystyrene beads and stirred at 4C overnight. The resultant anti-S100A3-conjugated immunobeads were then washed twice by repeated centrifugation and resuspended in PBS containing 0.1% BSA and 0.1% Tween 20. Ultrastructural localization of the beads in the cuticle was accomplished by scanning electron microscopy of hair shafts that had been partially delaminated by mechanical extension on a Toto Dianos A2 hair tester (Tokyo, Japan). The partially delaminated hair shafts were incubated with the immunobeads for 2 hr at 22C. After washing with PBS containing 0.1% Tween 20, the samples were dried, ion-sputtered with Pt-Pd in a Hitachi E102 ion sputterer (Hitachinaka, Japan), and visualized using a Hitachi S-800 scanning electron microscope at an accelerating voltage of 30 kV.
Results
Immunoblot Analysis
The presence of S100A3 in both cuticle and cortex was initially determined by Western blot analysis of extracts from human scalp hair shafts. Figure 2 shows that anti-S100A3 recognized a 7-kD protein in extracts of both cuticle and cortex. Anti-S100A3 identified a strong 7-kD band in the cuticle extract, but only small amounts of S100A3 were detected in cortex; note that cortex samples were 30 times larger than cuticle samples (Figure 2, Lanes 2 and 4). In the cortex extract, a smear band observed at the upper position of a 7-kD band might be a dimer of S100A3 (Figure 2, Lane 4). Anti-S100A3 exhibited no crossreactivity with other S100 proteins (i.e., S100A1, S100B) but was strongly reactive with purified human recombinant S100A3 (not shown).
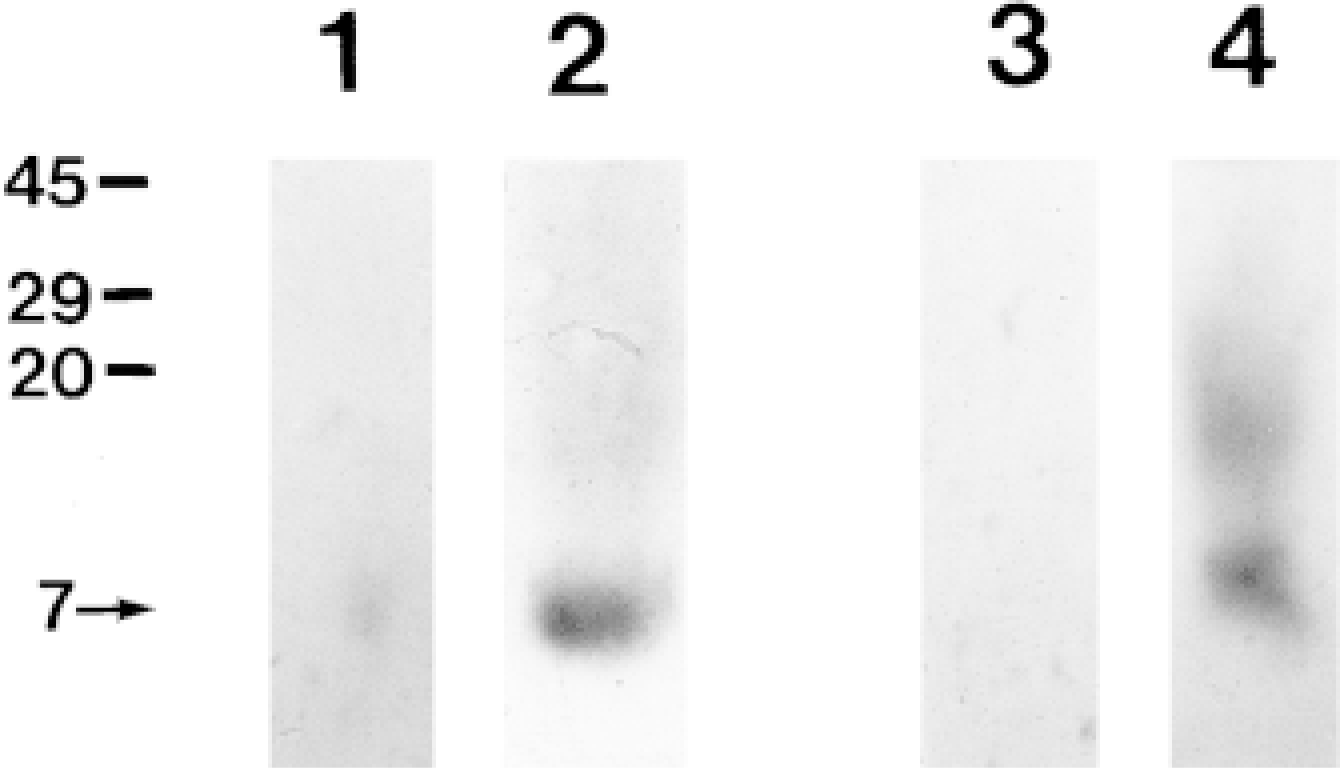
Western blot analysis of S100A3 in human scalp hair shafts. Cuticle extracts (Lane 1, 0.1 μg; Lane 2, 1 μg) and cortex extracts (Lane 3, 3 μg; Lane 4, 30 μg) were probed with an anti-S100A3 antibody. In both the cuticle and cortex extract, anti-S100A3 recognizes a 7-kD band. Note that small amounts of S100A3 are detected only after application of relatively large amounts of cortex extract (Lane 4). Molecular weight markers in kD are indicated at left.
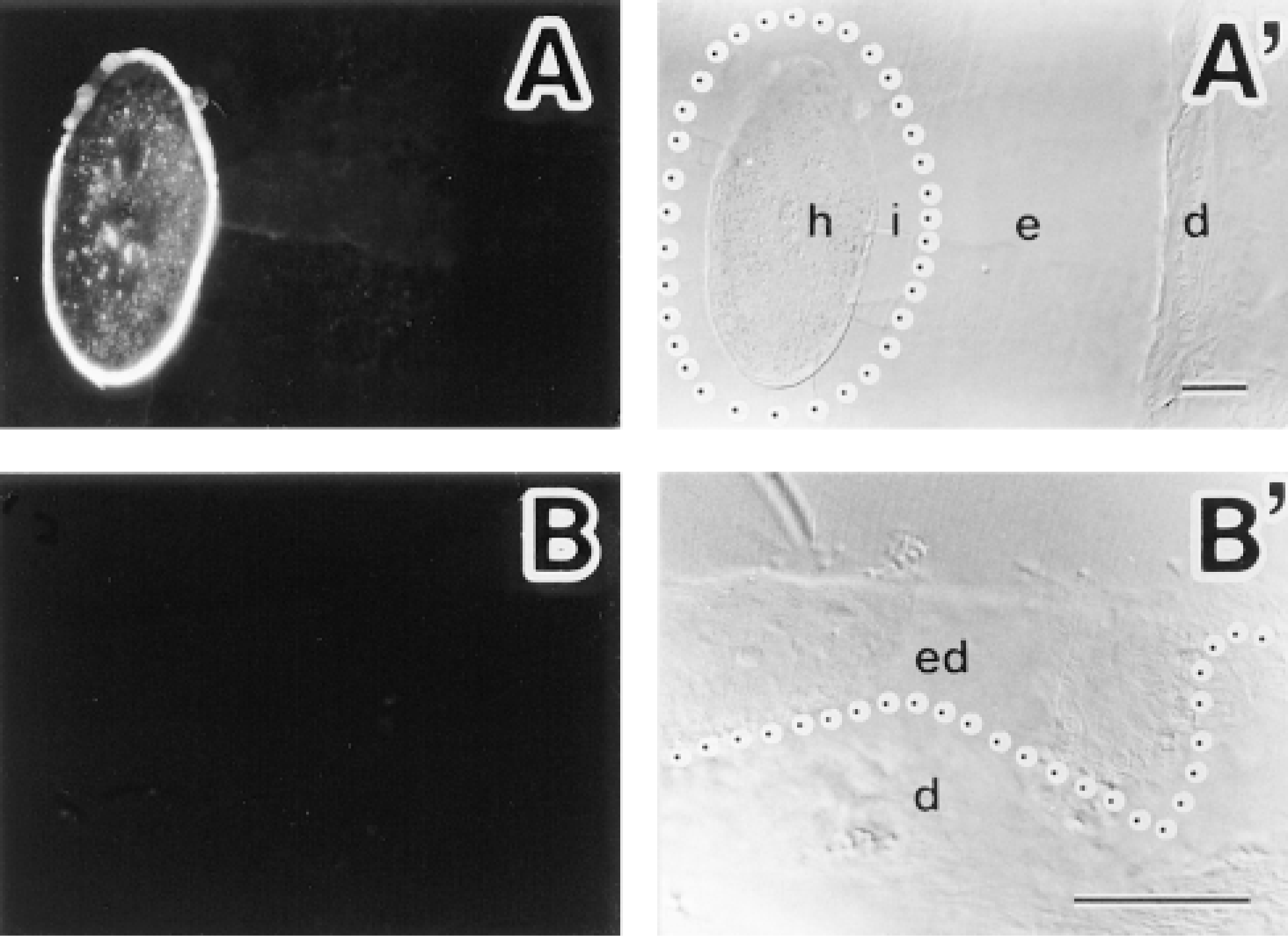
Immunofluorescence localization of S100A3 in normal human scalp skin. (A) Localization of S100A3 by immunofluorescence in hair follicle. (A′) Differential interference contrast (DIC) image of the same tissue seen in A. S100A3 is present only on hair cells (h). Note absence of immunofluorescence labeling in internal root sheath (i) and external root sheath (e). Dermal sheath is evident (d). A boundary between internal and external root sheaths is marked with a dotted line. (B) Localization of S100A3 by immunofluorescence in epidermis. (B′) DIC image of the same tissue seen in B. None of the epidermal cells show S100A3 immunostaining (ed). Dermis is evident (d). The epidermal-dermal junction is marked with a dotted line. Bars = 100 μm.
Immunofluorescence Microscopy
Immunofluorescence analysis with anti-S100A3 showed that, in normal human scalp skin, S100A3 was expressed only on hair cells (Figure 3). In hair cells, the cuticle showed strong positive signal for S100A3, whereas the cortex showed weak but definite staining (Figure 3A). Neither epidermis, internal root sheaths, nor external root sheaths exhibited S100A3 immunostaining (Figure 3). Control specimens in which anti-S100A3 was replaced by PBS or nonimmune rabbit serum lacked immunofluorescence labeling (not shown).
Rapid-freezing Immunocytochemistry
Although hair shafts were freeze-substituted in acetone without chemical fixatives, preservation of the hair shaft ultrastructure was generally good. Exocuticle, endocuticle, microfibrils in macrofibril bundles, and melanin granules were readily identified. In addition, rapid-freezing followed by freeze-substitution without fixatives preserved the antigenicity of S100A3 for immunocytochemistry (Figure 4). Consistent with the Western blots, the intensity of labeling was much lower in the cortex than in the cuticle covering the hair shaft. Immunogold particles identifying S100A3 were predominantly localized in the endocuticle (Figure 4A). In the endocuticle, most of immunogold particles were present in the nonstructural matrix, although some were associated with granule-like structures. In the cortex, the immunogold particles were found mainly in the region of the intermacrofibrillar matrix between macrofibril bundles (Figure 4B). No particles were found in melanin granules. Control specimens in which anti-S100A3 was replaced by PBS or nonimmune rabbit serum lacked colloidal gold labeling (Figure 4C).
When hair shafts were double labeled with anti-S100A3 (10-nm colloidal gold particles) and anti-hair keratin (15-nm colloidal gold particles) on the same ultrathin sections, both antigens were localized primarily within the endocuticle (Figure 5). The inner portion of the endocuticle was heavily labeled with 10-nm immunogold particles (S100A3), whereas 15-nm immunogold particles (hair keratins) were most likely to be present on the outer portion of the endocuticle (Figure 5A). Few immunogold particles were observed in the exocuticle. In the cortex, large numbers of 15-nm immunogold particles identifying hair keratins were present on microfibrils in the macrofibril bundles, but very few 15-nm gold particles were associated with the intermacrofibrillar matrix, which was labeled with 10-nm immunogold particles showing S100A3 (Figure 5B).
Morphometric Analysis of the Distribution of S100A3 and Hair Keratins in the Cuticle
Although S100A3 and hair keratins were both largely present within the endocuticle, there were striking differences in their respective distributions (Figure 6). The majority of S100A3 was present on the inner portion of the endocuticle. On the other hand, hair keratins were primarily located on the outer portion of the endocuticle.
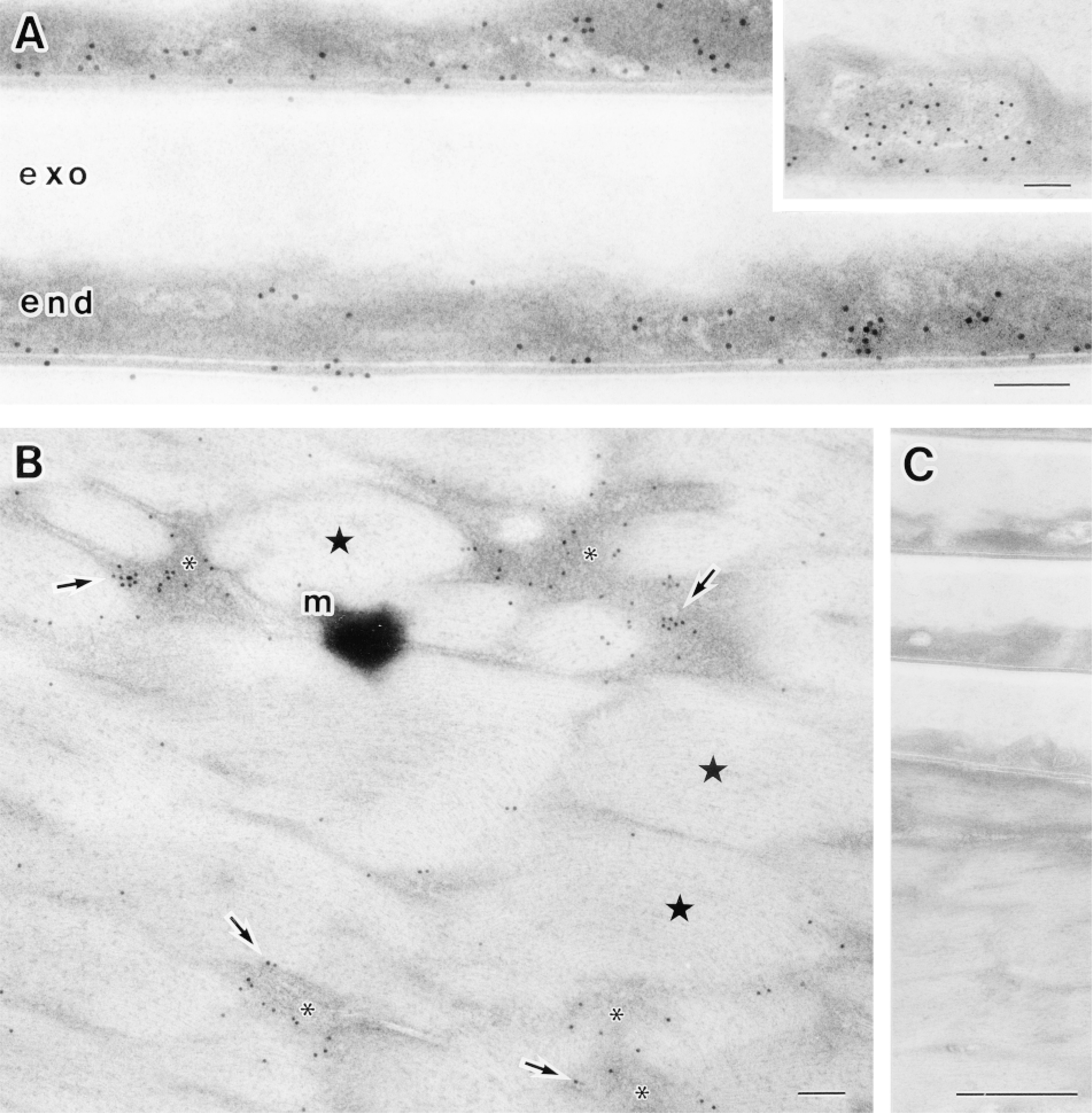
Ultrastructural localization of S100A3 in human scalp hair shafts revealed by rapid-freezing immunocytochemistry. (A) Electron micrograph of the cuticle of a rapidly-frozen hair shaft. Immunogold particles are predominantly located in the nonstructural matrix of the endocuticle (end). (Inset) Some of the immunogold particles are associated with granule-like structures in the endocuticle. The exocuticle (exo) is evident. (B) Electron micrograph of the cortex of a rapid-frozen hair shaft. Most immunogold particles demonstrating S100A3 (arrows) are present in the intermacrofibrillar matrix (∗) between macrofibril bundles (stars). A melanin granule is evident (m). Bars = 0.1 μm. (C) Control experiment in which the primary antibody was omitted; note the absence of gold labeling. Bar = 0.5 μm.
Immunoscanning Electron Microscopy
Ultrastructural localization of S100A3 in the cuticle was also confirmed by immunoscanning electron microscopy using anti-S100A3 conjugated to polystyrene beads (immunobeads). Anti-S100A3 immunobeads were associated with the inner-facing aspect (i.e., the endocuticle) of cuticle cells exposed by mechanical extension of the hair shafts (Figure 7). The immunobeads were also sometimes present on the outer surface of newly exposed cuticle cells, but this might be indicative of residual endocuticle from adjacent cuticle cells (not shown).
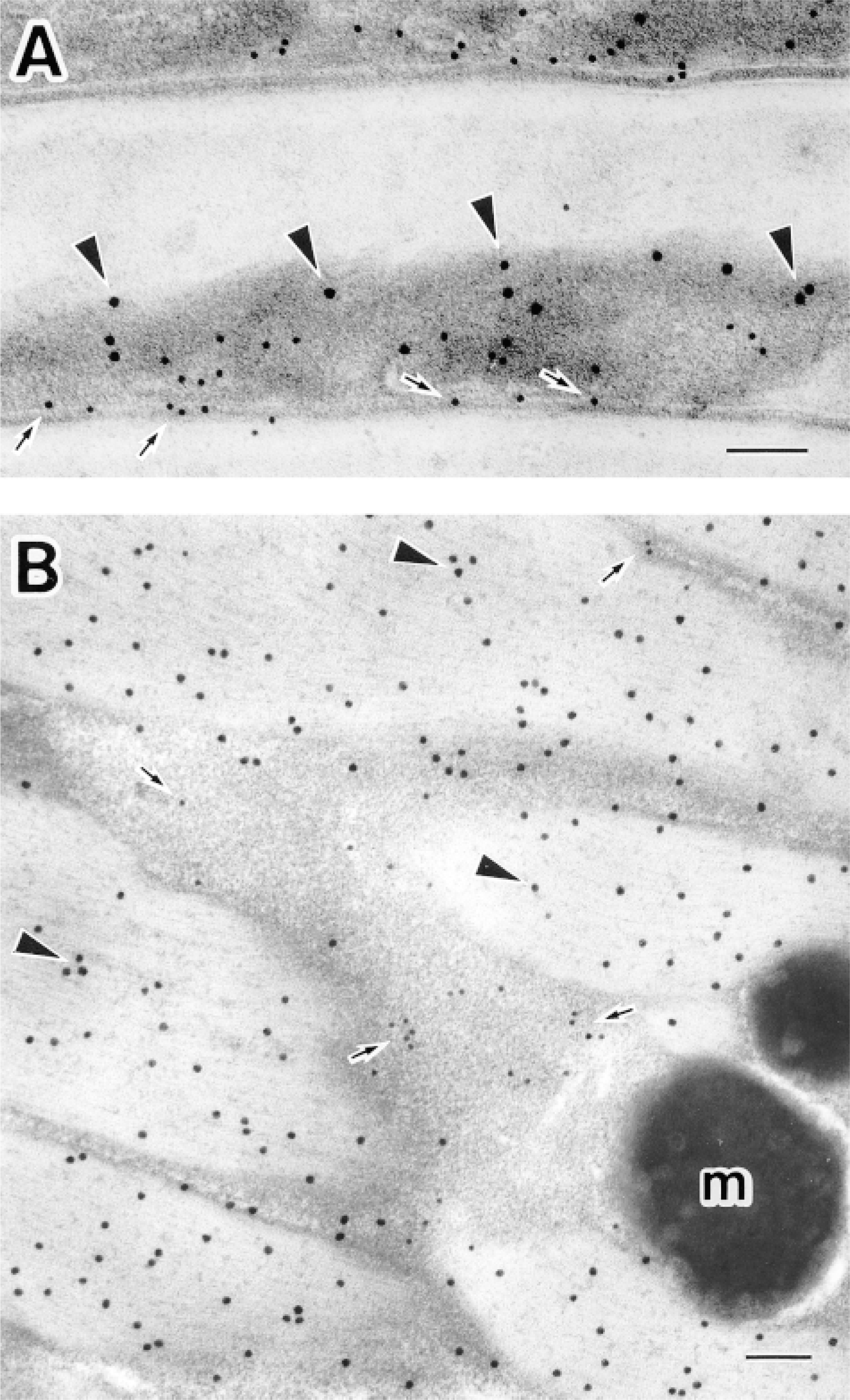
Double immunolabeling for S100A3 and hair keratins in human scalp hair shafts. (A) Electron micrograph of the cuticle of a rapid-frozen hair shaft. Both 10- and 15-nm immunogold particles indicating S100A3 (arrows) and hair keratins (arrowheads), respectively, are located within the endocuticle; few particles are present in the exocuticle. (B) Electron micrograph of the cortex of a rapid-frozen hair shaft. The 10-nm gold particles, indicating S100A3, are present mainly within the intermacrofibrillar matrix between macrofibril bundles (arrows). In contrast, most 15-nm gold particles, indicating hair keratins, are present on macrofibril bundles (arrowheads). Melanin granules are evident (m). Bars = 0.1 μm.
Morphometric analysis of the distributions of S100A3 and hair keratins in the cuticle. Both S100A3 and hair keratins are largely localized in the endocuticle but exhibit different distributions in it. S100A3 is primarily located on the inner portion of the endocuticle, whereas most hair keratins are found on the outer part. In the endocuticle, the relative distribution of immunogold particles (Dr) is defined as follows:
Discussion
There is increasing evidence that S100 proteins play important roles in calcium signaling pathways, and each member of the S100 protein family exhibits a distinct tissue-specific pattern of expression (for review see Schäfer and Heizmann 1996). In skin, the following S100 proteins are expressed in specific subsets of mammalian epithelial cells. S100A2 is present in the basal layer of the epidermis and the root sheaths of hair follicles in normal human skin (Böni et al. 1997; Shrestha et al. 1998). S100A4 is upregulated in metastatic epithelial cells in rodents (Weterman et al. 1992). S100A6 is highly expressed in melanoma cells (Weterman et al. 1992; Böni et al. 1997), although it is also present in various components of normal skin (hair follicles, keratinocytes, Langerhans' cells, melanocytes, and sweat glands) (Wood et al. 1991; Böni et al. 1997). S100A7, S100A8, and S100A9 all appear to be highly upregulated in psoriatic skin (Madsen et al. 1992; Hoffmann et al. 1994). Trichohyalin and profilaggrin, which are also members of the S100 protein family, are fused to the preforms of structural proteins synthesized in terminally differentiating epidermal tissues and are believed to serve as functional calcium binding domains (Lee et al. 1993; Markova et al. 1993). Finally, S100B has been widely used as a powerful immunopathological marker for melanoma (Cochran et al. 1993).

Immunoscanning electron micrograph of S100A3 in the cuticle. Immunobeads showing the distribution of S100A3 are associated with the inner-facing aspect of cuticle cells (i.e., the endocuticle) exposed by mechanical extension of hair shafts (arrow). Outer surfaces of cuticle cells are evident (∗). Bar = 5 μm.
S100A3 was first described as the product of one of the tightly clustered S100 genes located on the human chromosome 1q21 (Engelkamp et al. 1993). Its biochemical properties have been examined using recombinant human S100A3 (Föhr et al. 1995), yet its tissue localization remains unknown. Some tissues (e.g., skeletal muscle, heart, lung) are known to contain small amounts of S100A3 (Engelkamp et al. 1993), but the principal sites of S100A3 protein synthesis are not yet known, nor has the precise subcellular distribution of S100A3 been determined. In our previous study we detected significant amounts of S100A3 in hair cuticle both biochemically and histochemically but could not determine if it was also present in the cortex (Kizawa et al. 1996). In contrast, Böni et al. (1997) could not detect S100A3 in hair shaft cuticle using light microscopic immunocytochemistry, although they did find it in the inner root sheath cuticle of some hair follicles.
We recently discovered that rapid-freezing immunocytochemistry is an effective method for detecting the subcellular distribution and differential expression of macromolecular components within the fine structure of human hair (Takizawa et al. 1998b). In this study, we used this technique and immunoelectron microscopy to localize S100A3 in human scalp hair shafts. S100A3 was detected primarily at the interface between cells of the cuticle and, to lesser extent, at the interface between macrofibril bundles within the hair cortex. These findings were confirmed by Western blot analysis, showing that most S100A3 was present in the cuticle and that a relatively small amount was present in the cortex (see Figure 2). Analogous results were obtained from in situ hybridization studies showing that, in active hair follicles, S100A3 mRNA is predominately expressed in the cuticle and, to a lesser extent, in the cortex (Kizawa et al. 1998). Therefore, S100A3 is associated with the cells comprising the principal histological components of hair shafts: cuticle and cortex.
Double labeling experiments (see Figures 5 and 6) revealed the in situ spatial relationship between S100A3 and hair keratins (i.e., the major structural proteins of hair shafts). Both proteins were present within the endocuticle, but S100A3 was present on the inner portion of the endocuticle, whereas hair keratins were present on the outer portion. It appears that, within the endocuticle, the distribution of S100A3 corresponds to cysteine- and/or cystine-rich regions (i.e., the inner layer and its neighboring flame-shaped structure), which are revealed by silver-methenamine staining (Swift 1968; Swift and Bews 1974, 1976). Given the high cysteine content of S100A3, intra- and/or interpolypeptide disulfide bridges might be expected to develop during keratinization and maturation of hair shafts (Powell and Rogers 1994; Föhr et al. 1995; Kizawa et al. 1996).
Several questions remain to be addressed with respect to the function of S100A3 in human hair. Ultra-structural and molecular biological studies aimed at understanding the macromolecular organization of hair and the mechanisms regulating the differential expression of hair keratins and keratin-associated proteins during hair maturation are now under way. Rapid-freezing immunocytochemistry should provide the means to precisely localize subcellular components of hair cells and elucidate important new clues to better understanding of hair structure and function.
In this study, we used rapid-freezing immunocytochemistry to directly demonstrate that a S100 protein is localized in specific subcompartments in human hair shafts. These findings suggest that, in addition to their role in calcium signaling, S100 proteins may have other unique tissue-specific functions.
Footnotes
Acknowledgements
Supported in part by Grants-in-Aid for Scientific Research from the Ministry of Education, Science, Sports, and Culture of Japan (ToT).
We thank Dr William F. Goldman for improving the English of the manuscript and Dr Takashi Yashiro and Dr Shigeo Ookawara of Jichi Medical School for their encouragement of this study. We are indebted to Ms Kiyomi Inose, Ms Michiyo Soutome, and Ms Megumi Yatabe for excellent technical assistance.

