Abstract
For ultrafast fixation of biological samples to avoid artifacts, high-pressure freezing (HPF) followed by freeze substitution (FS) is preferred over chemical fixation at room temperature. After HPF, samples are maintained at low temperature during dehydration and fixation, while avoiding damaging recrystallization. This is a notoriously slow process. McDonald and Webb demonstrated, in 2011, that sample agitation during FS dramatically reduces the necessary time. Then, in 2015, we (H.G. and S.R.) introduced an agitation module into the cryochamber of an automated FS unit and demonstrated that the preparation of algae could be shortened from days to a couple of hours. We argued that variability in the processing, reproducibility, and safety issues are better addressed using automated FS units. For dissemination, we started low-cost manufacturing of agitation modules for two of the most widely used FS units, the Automatic Freeze Substitution Systems, AFS(1) and AFS2, from Leica Microsystems, using three dimensional (3D)-printing of the major components. To test them, several labs independently used the modules on a wide variety of specimens that had previously been processed by manual agitation, or without agitation. We demonstrate that automated processing with sample agitation saves time, increases flexibility with respect to sample requirements and protocols, and produces data of at least as good quality as other approaches.
Introduction
Resin embedding of cells and tissues for ultrastructural studies, element and isotope analysis, immunolabeling, or hybridization experiments require chemical fixation and dehydration. Fixation and dehydration at ambient temperatures (e.g., crosslinking with glutaraldehyde, paraformaldehyde, osmium tetroxide, and other chemicals followed by dehydration in ethanol or acetone series), however, lead to artifacts due to shrinkage, extraction, aggregation, and redistribution of biological material. Cryopreparation techniques avoid such artifacts.
Freeze substitution (FS) is a well-established method for processing rapidly cryoimmobilized cells and tissues, mainly for subsequent embedding in resins, sectioning, and studying in the transmission electron microscope (TEM; for review, see Steinbrecht and Müller 1 ; Humbel and Schwarz 2 , Humbel 3 ). FS is used to dehydrate and chemically fix samples at very low temperatures resulting in superior preservation of samples that are cryoimmobilized by high-pressure freezing (HPF) or freezing at ambient pressure. 4 During FS, samples are dehydrated by exchange of frozen cell water molecules for organic solvents at temperatures below the onset of destructive water recrystallization. The solvents usually contain additives such as osmium tetroxide, glutaraldehyde, and uranyl acetate. Subsequent warm-up of the substitution medium ensures low-temperature fixation of the dehydrated sample throughout its volume at the same time. Until recently, the dissolution of “frozen” cell water by organic solvents at temperatures between −80C and −90C was time-consuming, usually taking several days. This could be achieved best by commercial automated FS units, which have been purchased by many electron microscopy (EM) facilities worldwide.
The duration of FS depends on a number of parameters: (1) the substitution medium (e.g., acetone, methanol, ethanol, diethyl ether), (2) temperature, (3) accessibility of the samples to the substitution medium (especially, if the samples remain in the carriers that were used for HPF), and (4) size and nature of the sample itself (e.g., exoskeletons, cell walls, or egg shells may necessitate longer substitution times). Consequently, FS protocols designed either for ultrastructural analysis or immunolabeling have been notoriously slow. There are exceptions, resulting from optimization of processing parameters for certain applications. Wild et al.5,6 reported FS of high-pressure frozen cell monolayers on sapphire glass within 4–6 hr followed by a slow warm-up at 5C/hr to 0C. Hess et al. 7 processed plant samples within 8−12 hr; with a slow 24-hr warm-up. Hawes et al. 8 applied rapid FS for 1 hr, at −90C in combination with a steeper warm-up slope (20C/hr) to cell monolayers for Lowicryl embedding at −50C.
Besides optimizing individual FS protocols, efforts were made to accelerate FS on the basis of novel technical know-how. Dorward et al.9,10 introduced microwave-assisted FS combining a laboratory microwave oven with a cryo-thermal platform and accelerated the FS of chemically prefixed tissue culture cells. 11 In 2011, McDonald and Webb 12 demonstrated the tremendous potential of sample agitation for acceleration of the FS process. They performed FS of cryofixed samples within 3 hr or less in a polystyrene box filled with dry ice that was agitated on a laboratory shaker. This experimental low-budget approach reached popularity among the scientific community,13–16 despite some limitations: (1) the temperature/time schedule is determined by the heat capacity of the metallic sample holder, the mass of dry ice used, and the conditions for its evaporation; (2) defined temperature ramps during warm-up (e.g., to improve fixative crosslinking) are difficult to realize; and (3) particular care is needed for sealing of the tubes, especially if the substitution medium contains highly toxic osmium tetroxide or uranyl acetate. Therefore, it is clear that reproducibility, variability in the processing schedule, and safety issues are better addressed by agitating within automated FS units. Accordingly, RMC Boeckeler Instruments Inc. (Tucson, AZ) launched an Automatic Freeze Substitution System (AFS) that features integrated sample agitation, 17 but to our knowledge, there is no published assessment of the performance of this approach.
We, therefore, chose a straightforward way of merging the advantages of sample agitation with those of automated FS; we inserted a magnetic force-driven agitation module into the cryochamber of an AFS2 unit (Leica Microsystems; Vienna, Austria). 18 This did not interfere with the electronics and construction of the AFS itself. We demonstrated the suitability of the prototype for accelerated FS of fresh- and seawater algae from culture as well as algae internalized as endosymbionts. 19
For dissemination of our method, we followed a cost-saving concept based on three-dimensional (3D)-printed versions of the agitation module. These can be modified to fit the geometric conditions of a diversity of FS units. Here, we introduce “printed” agitation modules for two of the most common automated FS units currently in use, namely, the AFS(1) and AFS2 from Leica Microsystems. We summarize independent testing in a number of EM labs that resulted in a diversity of application protocols. These examples underline unmatched flexibility in combining temperature/time schedules with sample agitation in an automated FS unit.
Materials and Methods
FS Under Agitation Without Ethanol Bath as a Mediator of Cold Conductance
(1) Very Fast FS Under Agitation
Saccharomyces cerevisiae (for details, see Fišerová et al. 20 ), Caenorhabditis elegans, and Ostreococcus tauri were high-pressure (HP)-frozen with an EMPACT freezer (Leica Microsystems). C. elegans were harvested and transferred into 1% lecithin coated membrane carriers 100 µm in depth, prefilled with 20% bovine serum albumin (BSA) in M9 worm buffer (22 mM KH2PO4, 42 mM Na2HPO4, 86 mM NaCl, 1 mM MgSO4) and HP-frozen. O. tauri were pelleted at 4000 × g and gently resuspended with a small amount of 20% BSA in artificial seawater, transferred to a membrane carrier, and HP-frozen.
The agitation module was inserted into the chamber of the AFS(1) and precooled to −110C. HP-frozen samples were transferred to Sarstedt tubes containing the liquid nitrogen-frozen substitution medium (1% OsO4, 0.5% uranyl acetate, and 5% H2O in acetone or 1% OsO4, 2% glutaraldehyde, 0.5% uranyl acetate, 5% H2O in acetone) 16 and placed in the tube holders of the agitation module. The lid was shifted to the “open” position and replaced by the rotor unit of the agitation unit, and the transfer function (TF) for vaporizing liquid nitrogen was turned off by pressing the TF button. Agitation was set to maximum speed (24 V; ca. 70 rpm), and the AFS(1) program was started. The AFS(1) was programmed for increase in temperature from −110C to −80C at 30C/hr, then from −80C to +10C at a rate of 90C/hr. For washing, the samples were transferred to a rocker at room temperature for 15 min with 3 changes of acetone. Subsequently, the samples were infiltrated with epoxy resin, Agar 100 (Agar Scientific; Stansted, UK) and polymerized at 60C for 24 hr.
Ultrathin sections of 40–50 nm were cut with an ultramicrotome UC6 (Leica Microsystems), transferred onto Formvar-coated copper grids, and contrasted with 1% aqueous uranyl acetate for 10 min, followed by Reynolds lead citrate for 10 min. Images were acquired at 100 kV with a Hitachi H7600 TEM and an AMT Orca-ER digital camera (Deben UK Limited; Suffolk, UK).
(2) FS Under Agitation Overnight
Dried cysts of Artemia franciscana were provided by Prof. van Stappen, Laboratory of Aquaculture & Artemia Reference Center, University of Gent, Belgium. A. franciscana were reared under laboratory conditions in artificial seawater (salinity between 35 and 40 ppt) to the adult stage. For feeding, algae of the two strains, Rhodomonas sp. and Isochrysis sp. (courtesy of Dr. Abed-Navandi; Haus des Meeres; Vienna, Austria) were added to the culture.
Dissected ovisacs were transferred into carriers type A (6 mm diameter; 200 µm depth) and covered with the flat surface of carriers type B (Leica Microsystems). The carriers were coated with 1-hexadecene (Merck Sharp & Dome Corp.; Kenilworth, NJ) before use, and the inside volume of carrier A was filled up with 10% BSA. 21 The carrier sandwich was HP-frozen with the HPM100 freezer (Leica Microsystems).
FS was performed in an AFS2 (Leica Microsystems) equipped with the agitation module. Carriers containing the HP-frozen samples were placed onto 1 ml liquid-nitrogen frozen FS medium (1% OsO₄ in acetone) in 2 ml-Sarstedt tubes. Tubes were inserted into the tube holders of the agitation module within the AFS2, which had been precooled to −140C. FS took place under agitation at 56 rpm at −85C, initially for 3 hr to test how a previous protocol for algae would suit the application to oocytes and encysted embryos of A. Franciscana. 19 However, mostly, we used a 10 hr (overnight) incubation. Sample temperature was measured and recorded with a T-type thermocouple and universal serial bus (USB) data logger EL-USB-TC-LCD (Lascar Electronics; Erie, PA) inside a dummy tube filled with acetone. This was followed by warm-up to room temperature and embedding in low-viscosity epoxy resin (Agar Scientific). This approach required programming of the AFS2 unit that considered the temperature difference between the thermocouple in the dummy tube (presumed to be the same as the sample) and the temperature sensor of the AFS2: after rapid warm-up from −140C to −105C, the programmed temperature displayed by the AFS was kept at −105C to ensure freeze substitution at −85C as measured by the thermocouple in the dummy tube.
FS Under Agitation With Ethanol Bath as a Mediator for Cold Conductance
(1) FS of Anthers With Resistant Cell Walls
Immature anthers of Arabidopsis thaliana containing microspores were placed in aluminum carriers (3 mm in diameter, 150 µm in depth; Wohlwend; Sennwald, Switzerland) filled with 1-hexadecene (Merck Sharp & Dome). Carriers were covered with a lid and were HP-frozen using an HPM 010 (Bal-Tec; Balzers, Lichtenstein).
The agitation module was inserted in the cryochamber of an AFS2 filled with ethanol up to a level that mediated full contact with the cryotubes. This was precooled to substitution temperature (–90C). The frozen anthers were partially freed from 1-hexadecene and transferred into 2 ml cryotubes (# 72.694.006; Sarstedt; Nümbrecht, Germany) filled with 2.5% OsO4 (EMS; Hatfield, PA) in frozen acetone. The cryotubes were inserted into the tube holders, and the rotor unit was placed onto the lid of the cryochamber. Agitation was set at moderate speed at 20 rpm. Temperature was set to increase from −90C to 0C in 5 hr, followed by 1 hr at 0C. For comparison, cryotubes were agitated by hand every 15 min or not agitated at all. Washing with acetone at 0C and infiltration with epon resin (Carl Roth GmbH; Karlsruhe, Germany) at ambient temperature were performed without agitation. Finally, the resin-infiltrated samples were polymerized at 60C for 48 hr. Ultrathin sections were stained with 2% uranyl acetate in 50% ethanol (10 min) and lead citrate (3 min) before viewing in a JEOL JEM-1400plus TEM at 120 kV. 22 Images were taken with a 4K CMOS TemCam-F416 camera (TVIPS; Gauting, Germany).
For postembedding immunogold labeling with xyloglucan- or α-tubulin-specific antibodies, immature anthers were HP-frozen, as described above, and freeze-substituted under agitation at 20 rpm in acetone supplemented with 0.5% uranyl acetate, 2% methanol, and 2% H2O. In detail, the frozen samples were transferred at −90C into cryotubes filled with the substitution medium and, subsequently, warmed up within 2 hr to −55C. Samples were transferred to an ethanol bath cooled with an immersion cooler (–55C). After incubation for 2 hr at −55C, samples were washed 5× with acetone before they were infiltrated with Lowicryl HM20 (Polysciences, Inc.; Warrington, PA) at −55C (25%, overnight; 50% and 75%, 4 hr each; 2× pure resin, overnight). Resin infiltration was followed by ultraviolet (UV)-polymerization for 2 days at −55C. Ultrathin sections, 70 nm in thickness, were cut with an ultramicrotome UC7 (Leica Microsystems) and mounted on grids covered with Pioloform and carbon. For immunolabeling, unspecific binding sites were blocked with PBS containing 0.2% BSA (Sigma-Aldrich; Munich, Germany) and 0.2% milk powder (Granovita; Heimertingen, Germany). Sections were labeled with mouse anti-xyloglucan IgG (1:10; MAb CCRC-M1; Carbosource Services; University of Georgia, Athens, GA) or mouse anti-α-tubulin IgG (1:100; clone B-5-1-2; Sigma-Aldrich; St. Louis, MI) diluted in blocking buffer and goat anti-mouse IgG coupled to 6 nm gold (1:30 in blocking buffer; Dianova; Hamburg, Germany) for 1 hr, respectively. Gold particles were silver-enhanced using R-Gent (Aurion; Wageningen, the Netherlands) for 35–40 min. Resin sections were stained with 1% aqueous uranyl acetate for 4–5 min and lead citrate for 15–20 sec.
(2) FS of Organotypic Nerve Tissue
The cerebellum of male C57Bl/6 mice at postnatal day 7 was dissected under aseptic conditions, sliced at 400 µm using a vibratome (Leica VT1200 S; Leica Biosystems; Nussloch, Germany), placed on Millicell 0.4 µm semiporous membranes (Sigma-Aldrich GmbH; Vienna, Austria), and incubated for 21 days according to the interface method by Gähwiler et al. 23 Slices were cultured in sterile Corning 6-well plates (Sigma-Aldrich GmbH) at 37C and 5% CO2 with the following medium: 50% MEM/HEPES (Gibco by life technologies; Grand Island, NY), 25% heat inactivated horse serum (Gibco by life technologies), 25% HBSS (Gibco by life technologies), 2 mM NaHCO3 (Merck; Darmstadt, Germany), 36 mM glucose (Sigma-Aldrich GmbH), 2 mM glutamine (Merck), pH 7.2. During the culture period, the tissue was regenerated and flattened to a thickness below 200 µm for fitting in the carriers for HPF. 24
For HPF, the organotypic slices were quickly dissected without separation from the membrane insert and placed into aluminum carriers (2 mm inner diameter, indentation 200 µm; Wohlwend) with the tissue side facing down. As filler, 2% BSA in PBS (0.1 M, pH 7.4) at 37C was used. The flat side of a carrier with a 300 µm indentation, coated with 1-hexadecene, was used as a lid. 19 The sandwiched samples were HP-frozen using an HPM 010 (Bal-Tec), transferred to 2 ml cryovials (Biozym T311-2; Vienna, Austria), and stored in liquid nitrogen. The timing from dissection to freezing was about 2 min for each slice.
For FS, modified protocols for the processing of brain tissues were applied, including 2 consecutive substitution cocktails, (1) 1% tannic acid (w/v; Sigma -Aldrich GmbH) in anhydrous acetone and (2) 1% OsO4 (w/v; EMS) plus 0.2% uranyl acetate (v/v of 20% stock in methanol; Agar Scientific) in anhydrous acetone.25,26 Screw-cap Nalgene cryogenic vials of 2 ml (V4632; Sigma-Aldrich GmbH) were filled with 1 ml of substitution medium and frozen in liquid nitrogen. Frozen samples within their carriers were loaded onto the frozen substitution medium.
For comparison, two FS protocols were performed: (1) a classical slow procedure lasting more than 116 hr under static conditions, and (2) a rapid procedure using the agitation module at a rotor speed of 22 rpm, powered by a Voltcraft LSP-1403 power supply. Protocol 1 comprised 72-hr substitution in 0.1% tannic acid in acetone at −82C, followed by 3 20-min washes in acetone at −82C, 6-hr substitution in 1% OsO4 plus 0.2% uranyl acetate in acetone at −82C, raising of the temperature 3C/hr to −60C, 8-hr incubation at −60C, raising of the temperature 3C/hr to −30C, 8-hr incubation at −30C, 20-min wash 3 times in acetone at −30C, raising of the temperature 12C/hr to 4C. Protocol 2 started with 24-hr substitution in 0.1% tannic acid in acetone at −82C, followed by 3 10-min washes in acetone at −82C, 6-hr substitution in 1% OsO4, and 0.2% uranyl acetate in acetone at −82C, temperature rise 15C/hr to −60C, 3-hr incubation at −60C, temperature rise 15C/h to −30C, 3-hr incubation at −30C, 10-min wash 3 times in acetone at −30C, temperature rise 15C/hr to 4C.
Subsequently, samples were washed in acetone at 4C, separated from the carriers and embedded in epoxy resin (Durcupan ACM, Fluka; Buchs, Switzerland). For polymerization, the samples were transferred to BEEM capsules (#70020-B; Science Services; Munich, Germany), filled with freshly prepared Durcupan, and cured for 48 hr at 60C. Ultrathin sections (70–80 nm) were cut using an ultramicrotome UC7 (Leica Microsystems), collected onto Formvar-coated grids, and stained with 1% uranyl acetate in water (w/v) and 0.3% lead citrate. Sections were examined in a Tecnai 10 TEM (FEI; Hillsboro, OR) at 80 kV and equipped with a side-mounted camera Megaview III (Olympus Soft Imaging Solutions [OSIS] GmbH; Münster, Germany). Images were processed with iTEM software (OSIS GmbH).
Safety note: It is imperative that liquid nitrogen is not sealed in the cryovials during handling, because it will expand during transition into the gaseous state 700-fold and, therefore, could explode.
Results
Agitation Modules Based on 3D-Printing of Major Components
Building on experience with a handmade prototype of an agitation module for the AFS2 reported by Goldammer et al., 19 we followed a concept for cost-effective and flexible “on demand” manufacturing based on 3D-printing for generation of major components. Similar to the prototype, the “3D-printed” versions comprise (1) an insert for pivot-mounting the tube holders (Fig. 1A), (2) 8 tube holders for radial arrangement within the insert (Fig. 1B), and (3) a 24 V motor for driving of a rotor with magnets attached onto opposite positions of its blade. The motor is mounted on a Plexiglas plate and linked to a direct current (DC)-power supply (Fig. 1C). The 3D-printed insert and the tube holders consist of cryo-resistant, biodegradable plastics (polylactic acid). The insert has been adapted to be compatible with the geometry of both AFS(1) and AFS2 cryochambers. In the cryochamber of the AFS(1), the geometry is determined by a central tube for filling the unit with liquid nitrogen. The producer of the agitation modules, Cryomodultech e.U., applied the same design for agitation modules for both the AFS(1) and the AFS2. In consequence, the empty space in the middle of the insert within the AFS2-cryochamber could be used for precooling of exchange media (Supplemental Fig. 1).

Module for agitation of frozen samples during freeze substitution in an automated FS unit. (A) 3D-printed insert (1) with inner and outer recesses (2) for fitting of pivot-mounted tube holders (3) for the uptake of 8 Sarstedt cryotubes. The inner space (4) is required for fitting of the central tube in the AFS(1) (Leica Microsystems). In the AFS2 (Leica Microsystems), it offers space either for precooling of solvents or for comparative processing under static conditions. Large holes in the bottom of the insert ensure unhindered distribution of ethanol for the purpose of cold transfer between the cryochamber wall and the cryotubes. Small holes at the rim of the side wall serve as a hold for a spring catch that might be used for taking out of the whole insert. (B) Detail of a tube holder with a cryotube inserted, where (1) = axis for rotation; (2) = carbon fiber rod with a screw serving as a magnetic element; (3) = counterweight. (C) Agitation module inserted in the cryochamber of the AFS2 with its rotor unit on top. A 24 V-motor (1) is carried by a Plexiglas plate (2) mounted onto a metallic ring (blue). The rotor unit is placed on the frame of the glass lid of the cryochamber of the AFS2 (3). The space in between allows the free rotation of the rotor blade with magnets (4) attached opposite to each other at the bottom side. The motor is powered by a low-voltage supply shown on the left. Abbreviations: AFS, Automatic Freeze Substitution System; FS, freeze substitution; 3D, three dimensional.
Agitation of the sample tubes is achieved by magnets attached to the rotor blades, rotating around the central axis of the cryochamber of the AFS at a maximum speed of ca. 70 rpm. For the AFS2, the rotor unit is mounted onto a single Plexiglas plate. The rotor with the rotating magnets runs in the space between this plate and the original glass lid of the cryochamber. The magnetic field is transmitted through the glass lid of the cryochamber to the magnetic elements that are attached to the tube holders via carbon fiber rods (Fig. 1B). The magnetic elements are periodically attracted and released by the passing magnets. Because the tube holders are pivot-mounted and counterbalanced by a weight, the sample-containing tubes are agitated discontinuously (Supplemental Movie 1). Raising the rotor speed by increasing the voltage results in smoothing of fluctuations in the agitation (compare Supplemental Movies 1–3).
In contrast to the solution for the AFS2, the original glass lid for the cryochamber of the predecessor AFS(1) has to be shifted to the “open” position and replaced by a rotor unit with its rotor blade running between double glass plates serving as the new lid (Fig. 2).

Agitation module inserted in the cryochamber of the AFS(1) (Leica Microsystems) with its rotor unit on top. The original glass lid of the cryochamber (1) is shifted underneath the working table with the help of a handle (2). To agitate the samples in the insert of the agitation module, a rotor unit equipped with a 24 V motor (3) is placed onto the cryochamber, with its rotor blade (4) running between a glass and a Plexiglas plate separated by a metallic ring (red), which function as a lid for the cryochamber. Abbreviation: AFS, Automatic Freeze Substitution System.
Tube holders of the agitation modules are designed for fitting of both Sarstedt tubes (2 ml, conical bottom) and standard or ribbed skirted cryotubes (2 ml). The ribbed cryotubes allow one-handed opening and closing in the liquid nitrogen bath as the ribs around their bottom interlock with the wells in cryovial racks.
Generation of Very Short Substitution Protocols
For initial application of the prototype of the agitation module to algae, such as Chlorella sp. in symbiosis with their host Paramecium bursaria (Fig. 3A), a cautious time schedule was chosen consisting of 3 hr of FS followed by warm-up to room temperature. 19 We tested whether the deflection of the tubes from their vertical positions in the tube holders would suffice to achieve similar short FS schedules as those demonstrated by McDonald and Webb 12 with a dry-ice filled box on a laboratory shaker with horizontally arranged tubes. Within a 2 hr warm-up from liquid nitrogen-temperature to 10C at maximal rotor speed, we succeeded with samples of 3 different species tested, namely, algae O. tauri (Fig. 3B), nematode C. elegans (Fig. 4), and yeast S. cerevisiae (Supplemental Fig. 2). We applied acetone containing 5% water with OsO4 and uranyl acetate as substitution media, as used previously for quick FS of C. elegans on a laboratory shaker. 16 The water added to the substitution media aimed to increase contrast of membranous cellular components,27,28 O. tauri (Fig. 3B) showing prominent thylakoid membranes compared with algae freeze-substituted in the absence of water (Fig. 3B and Goldammer et al. 19 ). The yeast cell nucleus (Supplemental Fig. 2) shows a well-contrasted double membrane endowed with clearly recognizable nuclear pore complexes. The overall preparation quality of the intestinal absorptive epithelium of C. elegans with its characteristic microvilli brush border and glycocalyx (Fig. 4A and B) meets those previously achieved by Geisler et al., 16 who applied the method by McDonald and Webb. 12 The epithelium in Fig. 3C contained strongly contrasted rough endoplasmic reticulum and mitochondria.
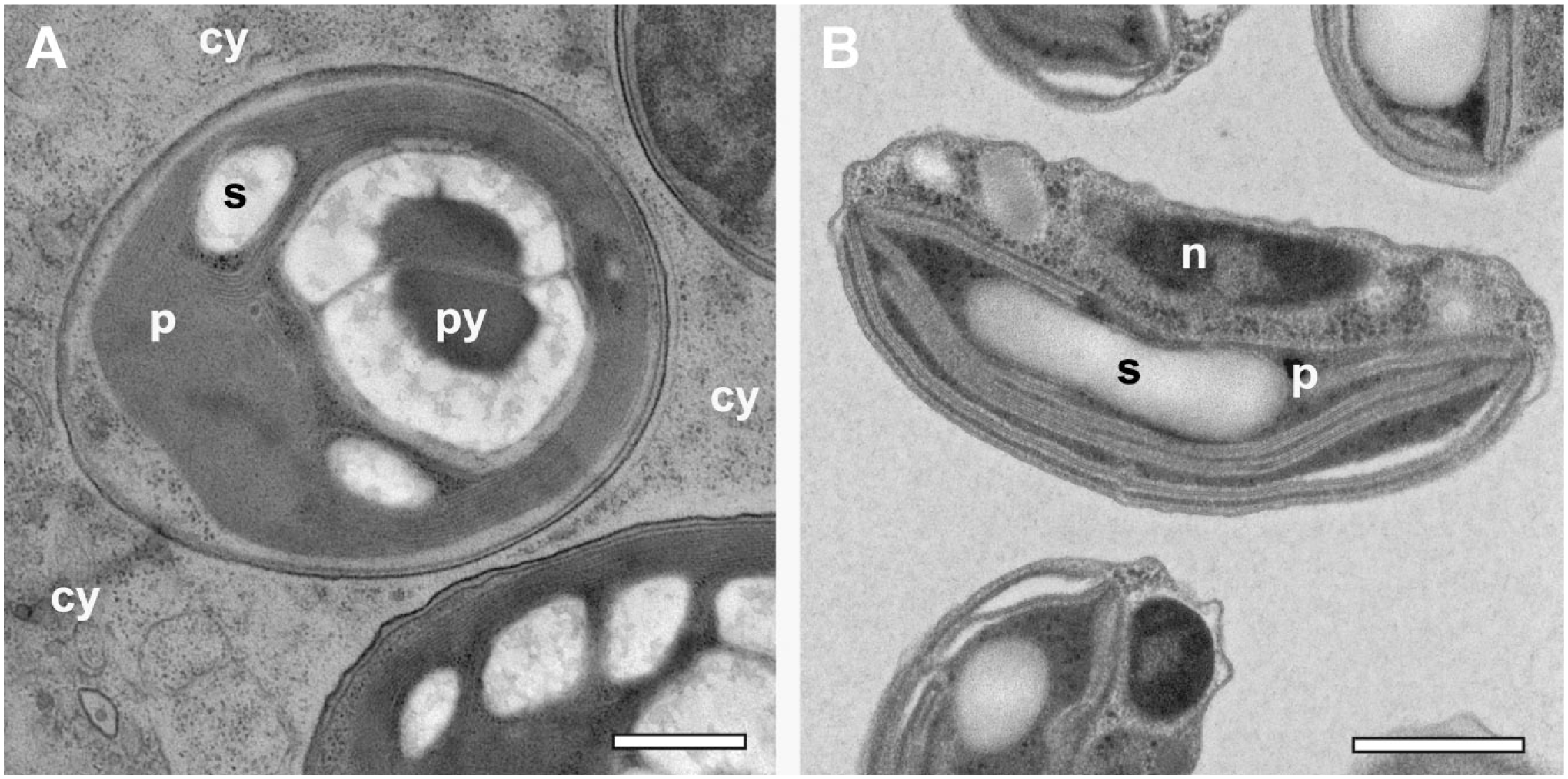
Rapid FS under agitation applied to HP-frozen algae. (A) Chlorella sp. symbiont within the cytoplasm (cy) of Paramecium bursaria. FS was performed under moderate agitation within 3 hr, followed by warm-up of the sample to room temperature. The plastid appears electron-dense, but with little detail of its internal organization. (B) Ostreococcus tauri. FS and warm-up, altogether, took place within 2 hr at maximum speed of agitation. Note that samples in A and B were exposed to different contrasting media both during FS and during staining of the resin sections. The plastid in B, but not in A, displays thylakoid membranes prominently. Contrasting in B is believed to be enhanced by the addition of 5% water to the substitution medium. Scale bars = 1 µm. Abbreviations: n, nucleus; p, plastid; py, pyreonoid; s, starch granule; FS, freeze substitution; HP, high-pressure.
HP-frozen Caenorhabditis elegans rapidly freeze-substituted within 2 hr under agitation at maximal speed. (A) Detail of enterocytes surrounding the intestinal lumen (lu), which are connected via an apical junction (arrow). Microvilli form a brush border (b) at the apical side of these cells. (B) Detail of the brush border containing microvilli (mv) embedded in glycocalyx (gl). (C) Mitochondria (m) and rough ER (er) densely decorated with ribosomes. Scale bars = 1 µm in A; 500 nm in B; 200 nm in C. Abbreviations: ER, endoplasmic reticulum; l, lipid droplets; y, yolk granule; FS, freeze substitution; HP, high-pressure.
FS Under Agitation With Ethanol Bath as a Mediator of the Cold Conductance
Ethanol is often used to fill the cryochamber of automated FS units to mediate cold conductance between the chamber wall and the cryotubes containing samples in the substitution medium. This measure minimizes differences between the programmed temperature and the actual temperature.
The effectiveness of the agitation module using ethanol in the cryochamber was demonstrated by application of a rapid FS protocol to anthers of A. thaliana. We compared the influence of agitation on a “linear” warming-up schedule over 5 hr (1) under discontinuous manual agitation every 15 min, and (2) under moderate agitation by using an agitation module in the AFS2.
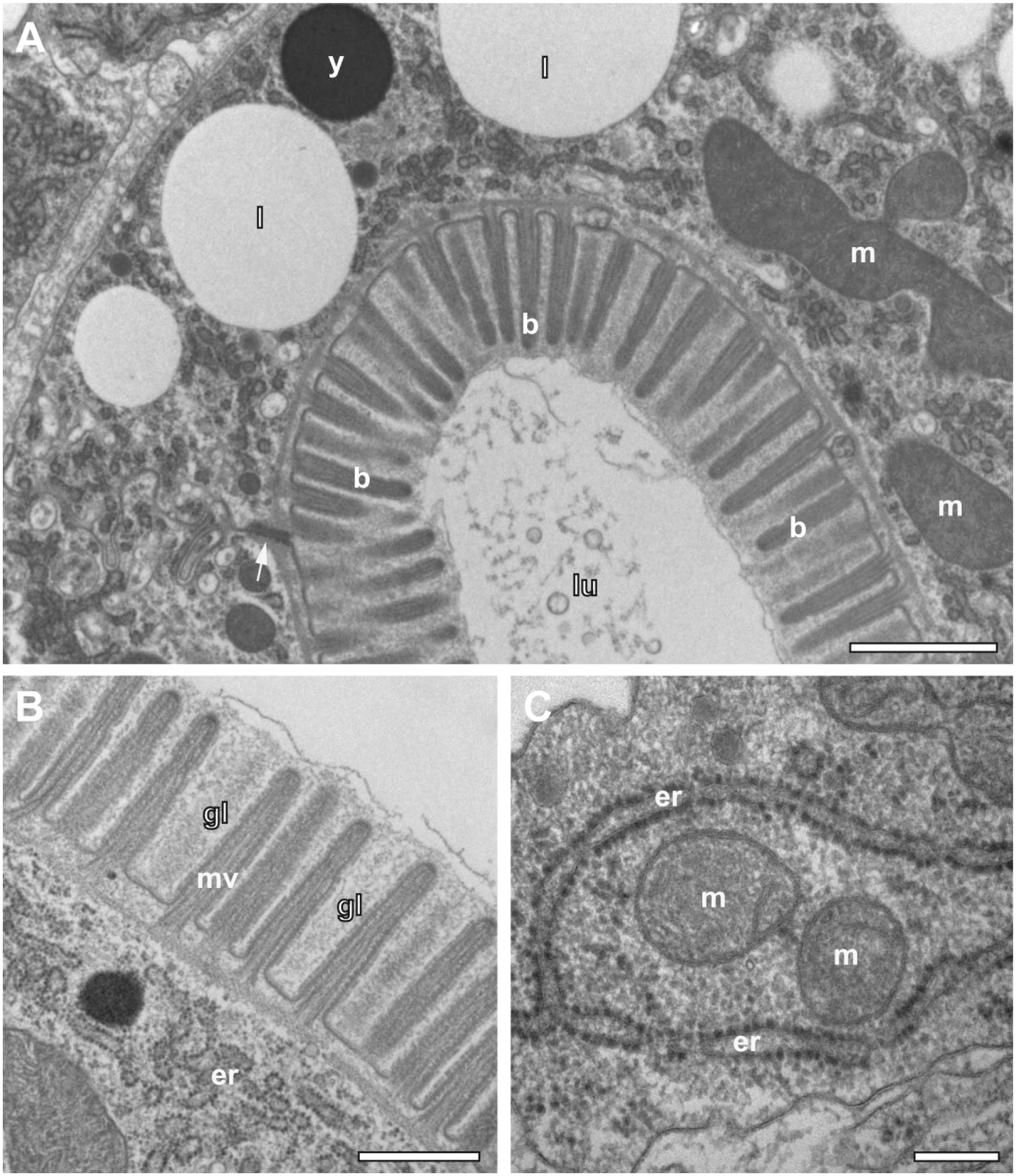
In the case of discontinuous manual agitation, most pollen grains (94%, n=81; sections from 3 different anthers) showed strong (62%; Fig. 5A and C) or weak (32%) damage by ice crystals, while only 6% were preserved in good quality. In contrast, most microspores enclosed in anthers (85%, n=48; sections from 2 different anthers) processed with the agitation module were well preserved and showed, at most, traces of microcrystalline ice as it is the case in many biological samples high-pressure frozen without cryoprotectant (Fig. 5B and D). About 15% of pollen grains displayed weak damage of mitochondria, nuclei, and generative cells by ice crystals.
The role of agitation during rapid FS. The processing of microspores enclosed in high-pressure frozen immature anthers of Arabidopsis thaliana was performed under the same temperature/time schedule within 5 hr, but using different methods of sample agitation. (A, C) Agitation by hand for every 15 min, and (B, D) agitation with the agitation module at moderate speed. Agitation by hand resulted in freezing artifacts that are already apparent at low magnification in (A). As visualized in detail (C), ice crystals led to segregation within the cyto- and nucleoplasm, and to damage of mitochondria and plastids. Application of the agitation module for FS preserved microspores with no apparent signs of ice crystal damage at low magnification (B). In consequence, organelle substructures of the Golgi complex, endoplasmic reticulum, mitochondria, and plastids are adequately preserved (D). Scale bars = 2 µm in A and B; 0.5 µm in C and D. Abbreviations: n, nucleus; gc, generative cell; m, mitochondrion; g, Golgi complex; er, endoplasmic reticulum; p, proplastid; cw, cell wall; ex, exine; FS, freeze substitution.
In a variant of the protocol, anthers of A. thaliana were also embedded in the methacrylate Lowicryl HM20 for subsequent immunogold labeling. FS and warming could be shortened to 2 hr, followed by 2 hr incubation and washing at −55C. Resin infiltration and UV-polymerization took place also at low temperature (–55C). Figure 6A displays the intense immunogold labeling of xyloglucan at the cell wall of a cell of the middle layer of an anther. Besides the expected localization at cell walls, xyloglucan was also found in association with intracellular vesicle-like structures (Fig. 6B), which are likely to be related to the xyloglucan biosynthesis located at the Golgi complex.29–31 Anthers were also immunogold-labeled for α-tubulin. The overview of the middle-layer cells shown in Fig. 6C displays labeling of cortical microtubules (MT), which is typical for non-dividing interphase plant cells. The association of the labeling with MTs oriented almost in parallel to the plane of the section became evident at higher magnification (Fig. 6D). Typical for postembedding labeling, the gold-conjugated antibodies bound to epitopes at the section surface. Consequently, α-tubulin was found to be concentrated at regions where the MTs met the surface of the ultrathin section.
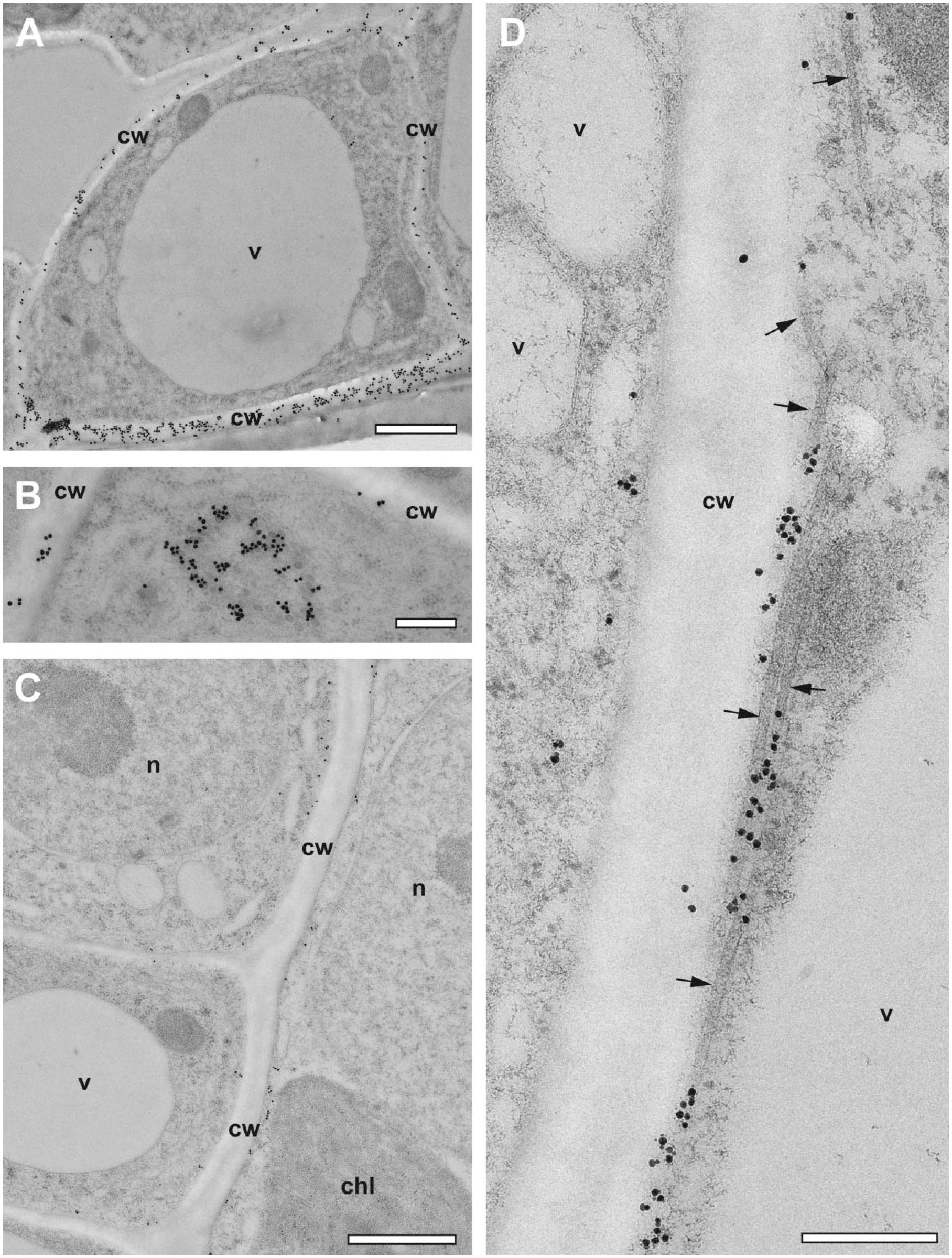
Immunogold labeling of Lowicryl HM20 sections of HP-frozen, low-temperature embedded immature anthers of Arabidopsis thaliana generated by accelerated FS under agitation. (A) Overview image showing a middle layer cell from an anther surrounded by anti-xyloglucan labeled cell wall (cw). (B) Aggregation of intracellular vesicle-like xyloglucan-positive structures. (C) Overview image showing anti-α-tubulin labeling close to the plasma membrane. (D) Details of immunolabeled cortical microtubules (arrows) aligned almost in parallel to the section plane. Typical for postembedding labeling, the gold-conjugated antibodies are concentrated at regions where the MTs meet the surface of the ultrathin section. Scale bars = 1 µm in A and C; 300 nm in B and D. Abbreviations: n, nucleus; v, vacuole; chl, chloroplast; HP, high-pressure; FS, freeze substitution; MT, microtubule.
Prolonged FS Under Agitation Applied to Encysted Embryos of A. franciscana
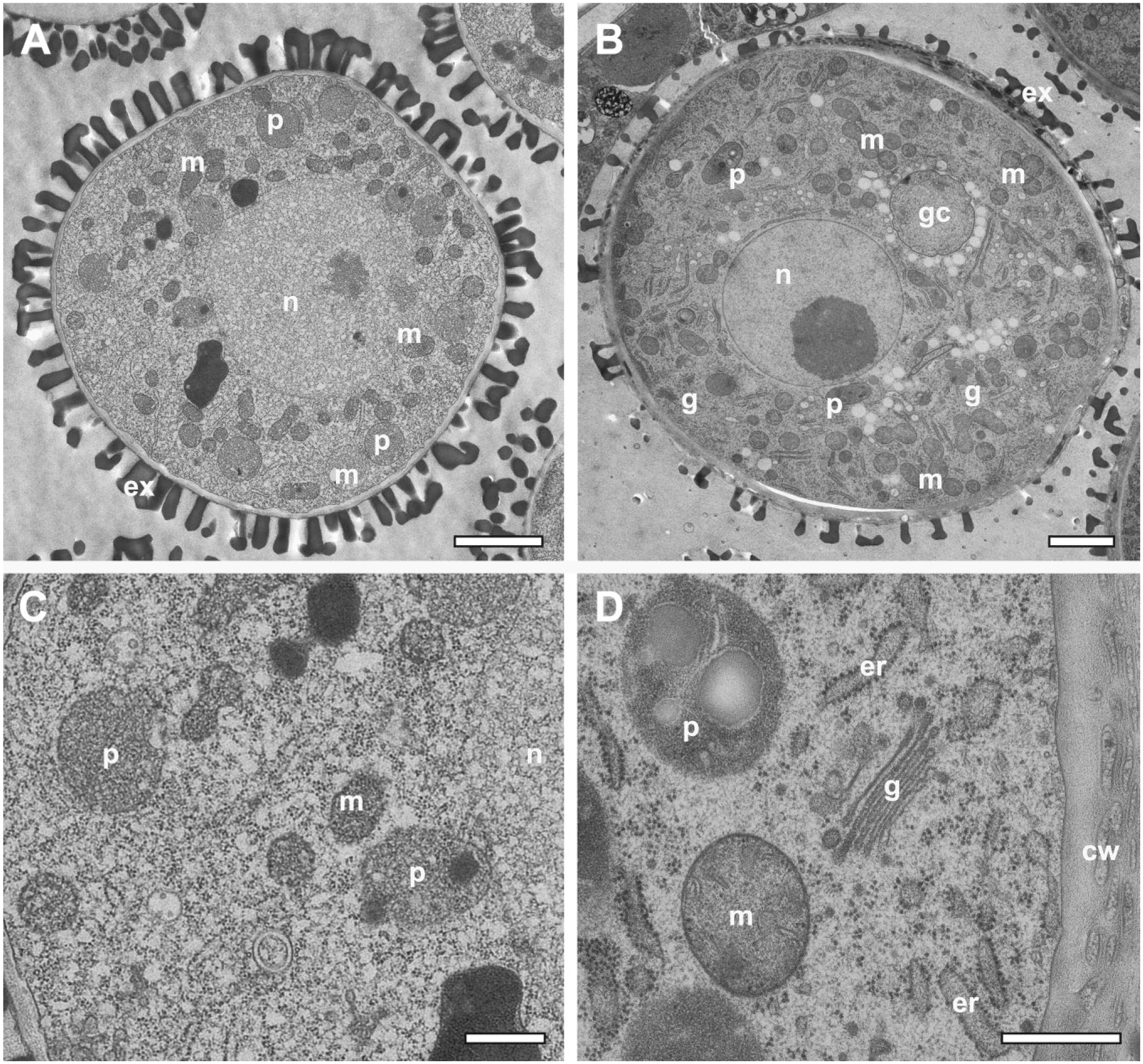
FS under agitation was applied to dissected ovisacs containing oocytes and encysted embryos of A. franciscana. The starting point of the work by Hollergschwandtner et al. was a similar protocol to that applied to algae, 19 comprising 4 hr of FS under moderate agitation followed by warm-up and embedding in low-viscosity resin. Under these conditions, the encysted eggs were completely damaged. Oocytes, however, were well preserved; their vitelline layer and a bi-layered coat with supposed jelly-like substances did not pose a hindrance for the substitution process and the infiltration with resin (inset Fig. 7A). More commonly for studies of vitellogenesis and embryonic development within the encysted eggs, however, a prolonged FS protocol (10 hr) was routinely applied. As demonstrated in Fig. 7A, the oocytes contain well-preserved electron-dense yolk platelets with para-crystalline inclusions. In addition to previously published data by Warner et al. 32 based on conventional chemical fixation at room temperature, radial arrangement of these para-crystals in some of the section profiles of the yolk platelets is evident.
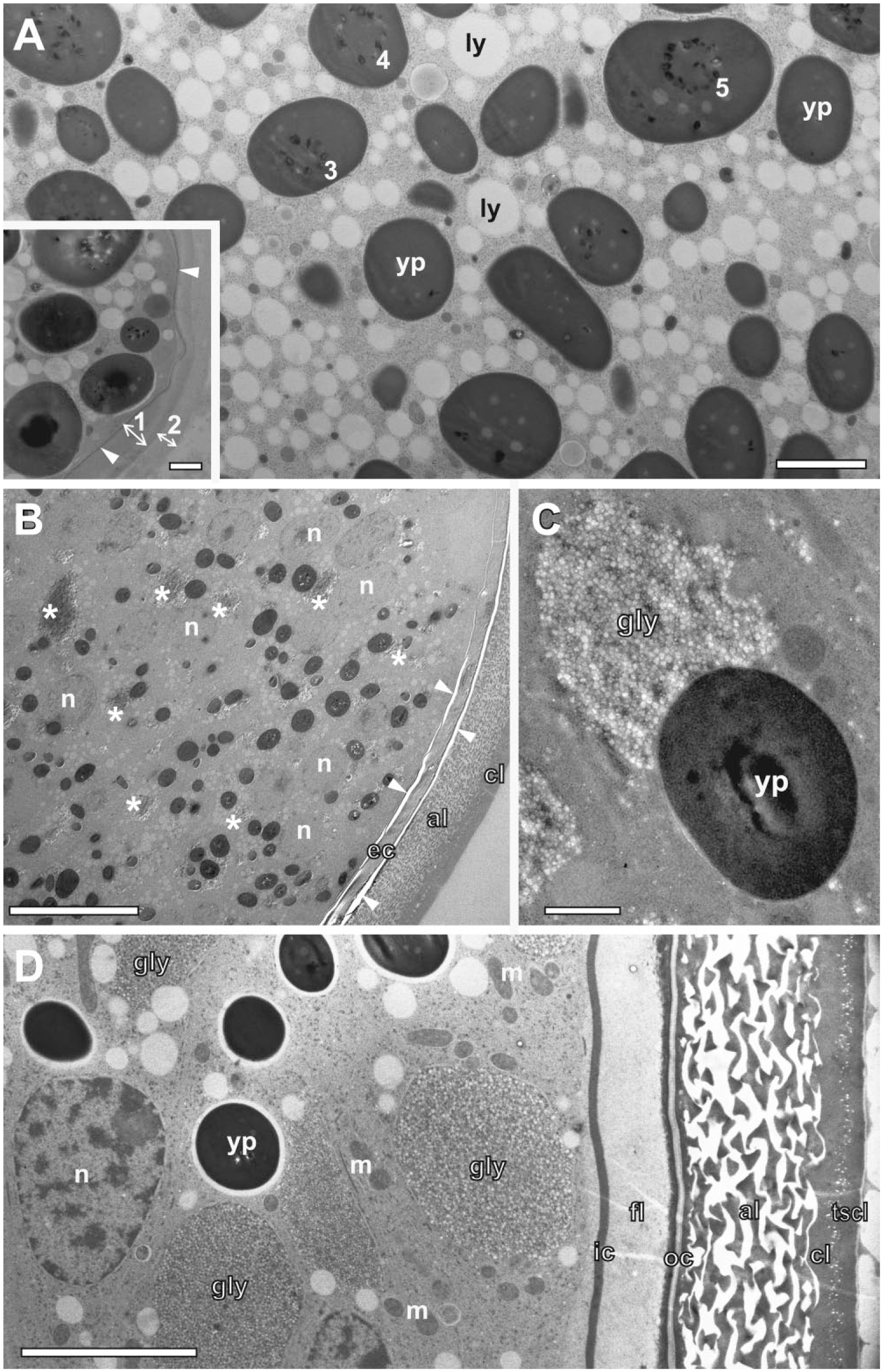
Prolonged FS under agitation for oocytes and encysted embryos in the ovisac of A. franciscana. (A) Vitellogenesis inside an oocyte, which is endowed with an electron dense vitelline layer (arrowheads) and a bi-layered jelly coat marked with double arrows 1 and 2 (inset). The inside contains numerous yolk platelets (Yp) and lipid yolk droplets (Ly). Many yolk platelets contain crystal-like inclusions, which are sometimes arranged in the form of rings (3–5). (B) Embryonic tissue in an encysted egg that did not show any cracks of the egg shell as a result of the high-pressure freezing. Major cellular components, including nuclei (n), are preserved without ice crystal damage. Glycogen clusters (asterisks) and yolk platelets (dark in contrast) display a blurred and cloudy appearance, and the embryonic cuticle (ec) appears to be damaged at its rim (possibly because of incomplete dehydration). (C) Glycogen cluster (gly) made of densely packed glycogen rosettes visualized at high resolution in association with a yolk platelet. (D) Encysted embryo that is rich in glycogen clusters (gly). Its developed embryonic cuticle is well-preserved, consisting of an inner cuticle membrane (ic), a fibrous layer (fl), and outer cuticle membrane (oc), which is confined by an alveolar layer (al), a cortical layer (cl), and a thin supra-cortical layer (tscl). Scale bars = 1 µm in inset in A; 2 µm in A; 20 µm in B; 1 µm in C; 5 µm in D. Abbreviation: FS, freeze substitution.
To preserve the embryonic tissue inside the eggs, we had to prolong the FS to ca. 10 hr. Studies of eggs without apparent cracks in their shells (that might be caused by freezing under high pressure) indicated that the prolonged time schedule preserves major cellular aspects in the absence of ice crystal damage. It made us also aware of specific artifacts: (1) the inner and outer embryonic cuticle appeared to be damaged (we speculate, because of incomplete dehydration), and (2) artifacts within numerous glycogen clusters (see Fig. 7B). Both the content of the glycogen cluster and the yolk platelets appear blurred and cloudy. Individual glycogen rosettes could only be identified at higher magnification (Fig. 7C). Their electron lucent appearance closely resembles negatively stained glycogen. 33 Similar electron lucent clusters of glycogen have been observed by FS under static conditions in a quite different biological context in epithelial cells of mouse submandibular gland and in nematodes interacting with Wolbachia endosymbionts.34,35
Singular cracks in the egg shell, presumably originating from HPF, enhanced the substitution processes and resulted in further improvement of sample preservation inside the egg. In consequence, the aggregates of glycogen became less affected by ice crystals, and the multiple layers of the egg shell, including the embryonic cuticle, were preserved (Fig. 7D).
Measurement of the Temperature Inside the Cryochamber
The way the temperature/time schedule is controlled in an automated FS unit differs from the experimental FS apparatus using a dry-ice bucket placed on a laboratory shaker. In the latter, it is convenient to measure the real substitution temperature by inserting a thermocouple into a dummy tube within the cold block loaded with the sample tubes. 12 Because the AFS(1) and AFS2 do not offer an opportunity for in situ measurement of the temperature, the developers of the agitation modules tested the conditions inside the cryochamber of the AFS2 by inserting a thermocouple into a dummy cryotube within a tube holder of the agitation module. To link the thermocouple with the USB-data logger outside the chamber, a glass lid with a feedthrough was manufactured. Figure 8 displays the real warm-up curve for the long FS substitution program (without ethanol bath) under agitation, which was used for oocytes and eggs of A. franciscana in Fig. 7, superimposed by the programmed temperature of the AFS2. In similarity to the warm-up curves published by McDonald and Webb, 12 the graph displays a characteristic discontinuity at around −95C. This discontinuity, in the form of a notable delay in the warm-up, is explained by the reversal of the crystallization of the frozen substitution medium. The release of crystallization energy at the melting point of acetone at −94.7C results in a cooling effect. Consequently, the discontinuity in the warm-up curve marks the beginning of the FS substitution process in the molten substitution medium.
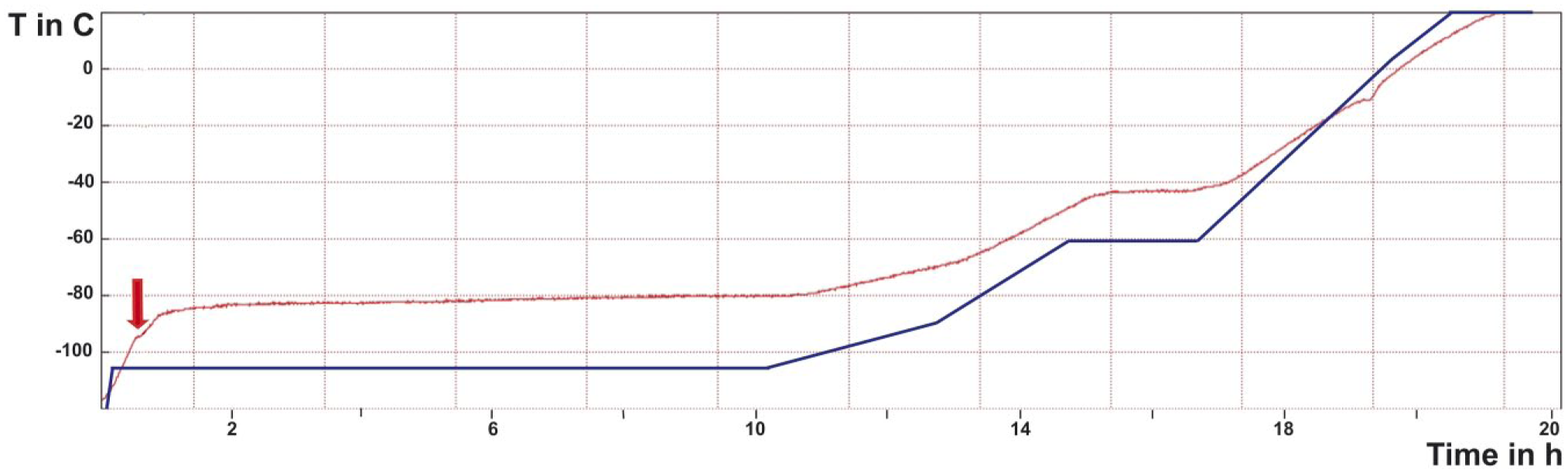
Warm-up curve for FS under agitation in an AFS2 in the absence of an ethanol bath as a mediator of cold in the cryochamber showing measured temperature changes over time (red line), compared to the programmed temperature/time schedule (blue line). This indicates a significant temperature gradient within the cryochamber. A small discontinuity at ca. −95C is reached after ca. 30 min (arrow). This indicates the transition of the frozen substitution medium to the liquid state, which happens despite a programmed temperature well below the measured temperature within the substitution medium. The warm-up curve shown here was applied to embryos of the ovisac of A. franciscana (displayed in Fig. 6). Abbreviations: AFS, Automatic Freeze Substitution System; FS, freeze substitution.
FS Under Agitation of Cryoimmobilized Organotypic Nerve Tissues
FS is favored for studying nerve tissues, as chemical fixation at ambient temperature can alter structural characteristics of synapses or even induce vesicle exocytosis. 36 We used the agitation module to shorten time-consuming FS protocols for processing of HP-frozen nerve tissues. The protocol by Siksou et al. 25 involves 4-day incubation in tannic acid-containing substitution medium, followed by incubation in osmium-containing medium, to enhance contrast. 25 We now compared the outcome of a long FS protocol under static conditions, resembling those by Siksou et al., 25 with a procedure that shortened the FS in tannic acid/acetone to 24 hr by means of sample agitation in the AFS(1). Overall, rapid processing under agitation generated samples with smoother, more electron-dense neuropils displaying less signs of tissue extraction, in particular, with respect to dendrites (compare Fig. 9A with 9C). The advantage of rapid processing under agitation became more apparent by comparison of ultrastructural details in excitatory spine synapses at higher magnification. The fine structure of the long-term freeze-substituted organotypic samples in Fig. 9B shows a grainy and extracted appearance, similar to data obtained for presynaptic terminals of HP-frozen rat nerve tissue. 25 Shorter FS under agitation (Fig. 9D) resulted in smooth, continuous membranes, including those forming synaptic vesicles, and in clearer, better discernible fine structural details such as filamentous material in the presynaptic active zone tethering synaptic vesicles, the presynaptic grid of dense projections, trans-synaptic protein complexes, and postsynaptic densities.
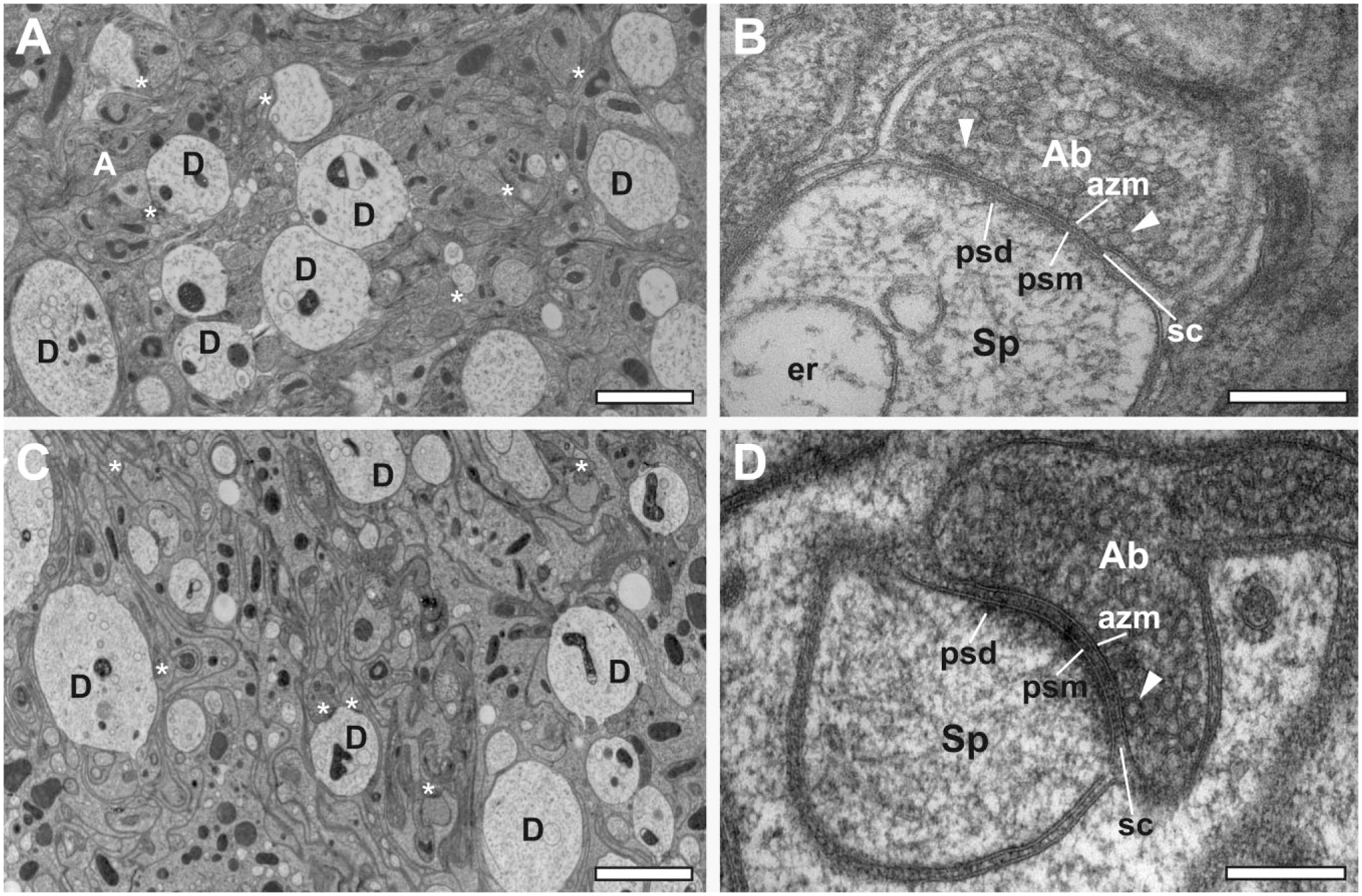
Organotypic slice cultures of mouse cerebellum, HP-frozen and freeze-substituted by using two consecutive substitution cocktails. (A, B) Slow FS under static conditions: (A) Overview image of a 70-nm section through the molecular layer displaying Purkinje cell spine and shaft synapses marked with asterisks. (B) Detail of an excitatory spine synapse in a 40-nm section of the same sample material as in Fig. A. Synaptic vesicles are identifiable in the axon bouton. In some instances, vesicles are docked at the active zone with vesicle membrane touching the presynaptic plasma membrane (arrowheads). Electron-dense material of the active zone compartment is retained. The trans-synaptic protein complexes spanning the synaptic cleft are indicated and postsynaptic densities are visible. (C, D) Accelerated FS under agitation: (C) Overview image of a 70-nm section of a sample generated by use of the agitation module yielding a well-preserved cerebellar molecular layer. The tissue ultrastructure displays less extraction of biological material than what is apparent for dendritic profiles, and cell membranes are smooth and intact. (D) Higher magnification of a 40-nm section through the same material containing an excitatory spine synapse. The synaptic vesicles and membrane specializations are clearly distinguishable. The membranes themselves are particularly smooth and intact when compared with Fig. B. The presynaptic active zone membrane and postsynaptic membrane run parallel to each other, and the synaptic cleft is regular. The whole tissue appears less grainy and washed-out. Scale bars = 2 µm in A and C; 200 nm in B and D. Abbreviations: a, axon; ab, axon bouton; azm, active zone membrane; sp, spine; d, dendrite; er, endoplasmic reticulum; m, mitochondrion; psd, postsynaptic density; psm, postsynaptic membrane; sc, synaptic cleft; HP, high-pressure; FS, freeze substitution.
Discussion
The applications presented here demonstrate that agitation modules are an appropriate means to speed up the FS process in automated FS units. Although the FS protocols differ in their temperature/time schedules and other parameters, they all have in common the shortening of protocols that were applied previously to similar samples. The saving of time was achieved with a mode of agitation that differs from the physically more intense agitation in horizontally arranged substitution tubes introduced by McDonald and Webb. 12 Apparently, the vertical arrangement of the sample tubes in the tube holders for discontinuous agitation by rotating magnets sufficed to achieve substantial acceleration of the FS process. Notably, such vertical arrangement is safer with respect to leakage of substitution medium than horizontally arranged tubes in an experimental setup on a laboratory shaker. The latter might leak if one does not follow the detailed safety instructions by McDonald. 37
Agitation Modules Enable Very Short FS
Recently, Goldammer et al. 19 reported a significant acceleration of the FS of cryoimmobilized algae but they did not compare it with the very rapid protocols published by McDonald and Webb. 12 Here, we performed very rapid FS in a 2-hr run in the AFS(1). The results met our previous experience with the rapid FS of nematodes C. elegans performed on a laboratory shaker. 16 Application of the same rapid program to yeast and algae, O. tauri, underlined that processing times as short as the experimental approach performed on a laboratory shaker can be achieved for a variety of species by using agitation modules in the AFS. The excellent results for O. tauri are equivalent to previous HPF/FS by Henderson et al. 38 using a similar FS protocol, but lasting more than 50 hr in total, instead of 2 hr. Thylakoid membranes were particularly prominent. Nuclei, ribosomes, mitochondria, and plasma membranes are well preserved and visualized, depending on the plane of section. Other organelles/structures, such as the Golgi apparatus, were sometimes recognized, but due to the small size and dense nature of O. tauri, identification was not as clear as with thinner cryo-tomographic slices. 38
We consider the continuous mode of the agitation essential to accelerate the FS tremendously, as it is expected to disperse water accumulated in the transition zone between the frozen-hydrated and the fully dehydrated state. At the same time, it promotes the movement of water through natural hindrances (lipid membranes, cell walls, cuticles, egg shells, etc.) and its final release from the bulk sample into the surrounding substitution medium. Manual shaking at intervals, demonstrated for the processing of cell wall-confined anthers of Arabidopsis, will not speed up FS as effectively as continuous agitation, because it gives water the opportunity to reaccumulate in the transition zone soon after each shaking step. Moreover, a time schedule optimized for FS under continuous agitation, but applied under conditions of manual shaking will result in recrystallization, as it is too short to complete the timely release of bulk water from the samples into the substitution medium before the temperature rises above the recrystallization point. (The anthers reached a temperature of −54C already after 2 hr FS and 7× manual shaking.) An attempt to apply the same rapid time schedule for FS under static conditions, inevitably, led to the destruction of the samples.
The FS Schedules Under Agitation Have to Be Adapted to the Nature of the Samples
Certain biological samples are confined by natural barriers for the substitution and fixation processes, and/or they contain particular water-binding components that pose a particular challenge for FS. For these, sample agitation has been proved to be an advantage as it shortens otherwise excessively long FS protocols.
Microspores and pollen grains enclosed in anthers are protected by particularly resistant cell walls. 39 Tetrad-stage microspores were previously freeze-substituted under static conditions for 5 days followed by rapid stepwise warm-up; older stages were freeze-substituted for 3 or 7 days followed by a slow warm-up.39–42 A previous application of FS to anthers of A. thaliana by Stierhof and El Kasmi 43 lasted 2 to 3 days. In contrast, here, we achieved FS of microspore-containing anthers of A. thaliana under agitation within 5 hr.
Even more challenging natural barriers are built up during the embryonic development of A. franciscana in the form of multilayered egg shells. 44 Recent studies of the egg shells did not include cryofixation and FS.45–48 This could be because of technical difficulties in establishing cryopreparation methods for this field of research. Application of prolonged temperature/time schedules lasting 10 hr indicates that FS under agitation could become an appropriate means for systematic studies of both the egg shell development and internal processes during embryogenesis. For such an approach, one has to take into account not just the barrier of the egg shell itself, but also time needed to substitute frozen water that is associated with large clusters of glycogen, which are located inside the A. franciscana embryos.48–50 Glycogen clusters of similar size have been reported previously for developing mouse submandibular gland and nematodes hosting Wolbachia endosymbionts.34,35 Both authors chose HPF and FS under static conditions, because of the detrimental effect of conventional chemical fixation and dehydration at room temperature on the preservation of the glycogen clusters. The details of clustered glycogen rosettes are clearly visible after FS for 60 hr by Matsuura et al., 34 but barely recognizable after FS for 10 hr by Fischer et al. 35 The preservation of densely packed glycogen clusters within the encysted embryonic tissue of A. franciscana by FS under agitation opens new, effective opportunities for studying the glycogen and glycogenolysis even in encysted species.
More generally, we argue that any application requiring prolonged FS under agitation (e.g., 10 hr or more) is not possible in simple experimental setups. Automated FS units, in contrast, provide a long-lasting, controlled cold reservoir that is needed for such special applications.
An Ethanol Bath May Be Used as a Mediator of Temperature Conductance But Is Not Essential
FS protocols in automated FS units may differ in the way the cold transfer to the sample is achieved. For some of our applications, the substitution medium-filled cryotubes were agitated in the cold, gaseous atmosphere, for others, they were immersed in cold ethanol filling the cryochamber of the FS unit. The former leads to a strong discrepancy between the programmed temperature and the real one in the substitution medium. This discrepancy is caused by imperfect thermal insulation of the chamber; in consequence, the samples are exposed to warmer temperatures than those measured at the outside bottom of the cryochamber. (Measuring the temperature with a thermocouple inside a cryotube dummy within the AFS2 programmed at −85C resulted in FS at −80.5C when the chamber was filled with 200 ml ethanol; in the absence of ethanol, the temperature in the cryotube was just −73.5C.) Without ethanol, the samples might be exposed to temperatures well above the temperature of water recrystallization.51,52 At temperatures above −80C, this could lead to conversion of small cubic ice crystals into large, destructive hexagonal ice. 4
Our applications in the absence of an ethanol bath as a mediator of the cold indicate that agitation enables FS at a speed high enough to prevent excessive growth of ice crystals. Similar to the experimental approach by McDonald and Webb, 12 we started almost every FS with frozen samples placed onto the frozen medium. The volume of the frozen substitution medium in the cryotube (ca. 1 ml or more) provided a cold sink, which prevented initiation of crystallization at an early time point. In consequence, and in similarity with the approach by McDonald and Webb, 12 measurement of the actual substitution temperature is not required for successful FS with the agitation module. Nevertheless, the opportunity for recording the temperature/time slope might be helpful for deeper understanding of the FS itself.
Agitation Conditions Are Not Critical
The intensity of the sample agitation is not directly related to rotor speed. Running of the rotor at low or medium speed results in particularly discontinuous tube vibrations (compare Supplemental Movies 1–3). Initially, Goldammer et al. 19 aimed to use these discontinuities under undamped conditions, and, therefore, avoided ethanol as a cold mediator. Here, we achieved very rapid FS (within 2 hr) in some protocols in the absence of ethanol at maximum rotor speed, indicating that discontinuity in the tube vibrations is not a precondition for accelerated FS. In applications to microspores enclosed in anthers, we demonstrated that even moderate agitation of the cryotubes in an ethanol bath suffices for acceleration of the FS process. Taking all our applications together, we conclude that the speed and the mode of the agitation module are not decisive for successful acceleration of the substitution process. However, agitation might be optimized if it is aimed to perform FS as quickly as possible. Less intense agitation might be chosen for applications to very delicate and fragile samples.
The Agitation Module Offers Flexibility in Organizing Lab Work
FS is part of a long processing chain starting with freezing at ambient or high-pressure and ending with the embedding of the samples in resin. Processing of the frozen samples within a working day becomes feasible for many applications if the FS under agitation is combined with accelerated resin-embedding techniques. 53 However, to our experience, not all applications fit in the time schedule of a working day. The use of agitation modules in automated FS units offers the flexibility of programming. The HPF session can easily be combined with rapid FS of samples overnight, and their embedding in resin on the following day. Samples requiring prolonged FS under agitation, such as those for encysted embryonic tissues or those of organotypic mouse brain slices necessitating treatment with tannic acid in preparation of the low-temperature fixation might run even longer (e.g., over the weekend).
Extension of the Application of the Agitation Modules for Analytic and Labeling Purpose
The prospect of less extraction of biological material achieved by a rapid FS is of advantage not just for the preservation of ultrastructural features, but also for application of analytical methods such as energy dispersive X-ray analysis (EDX), energy loss spectroscopy (EELS), or nano secondary ion mass spectrometry (NanoSIMS). Recently, Hollergschwandtner et al. 54 applied EDX to semi-thin sections of optically active mesostructured cellular inclusions in the epidermal layer of the ovisacs of A. franciscana that were preserved by HPF and FS under agitation in an AFS.
However, for many samples collected in the field and infectious samples, cryofixation is not an option. In such cases, the aldehyde-fixed samples might benefit from subsequent FS.55,56 The usefulness of such a hybrid approach for NanoSIMS was demonstrated by Volland et al., 57 who plunge-froze aldehyde-fixed ciliates Zoothamnium niveum harboring bacterial endosymbionts and freeze-substituted them by using an agitation module. The resin-embedded samples were used for correlating TEM studies with the localization of carbon isotopes.
With respect to immunolabeling, FS under agitation shortens the procedure of sample preparation for postembedding labeling significantly, as demonstrated here, by application to low-temperature embedding of cells from an anther in Lowicryl HM20 resin. As resin infiltration and UV-polymerization at low temperature in the automated FS units require large amounts of liquid nitrogen, an ethanol bath cooled by an immersion cooler was used as an alternative. Where appropriate, the freeze-substituted samples for immunolabeling or in situ-hybridization might be transferred, instead, to LR White resin for heat polymerization.
Given the widespread distribution of AFS in EM labs worldwide, we conclude that the acceleration of FS with agitation modules offers a cost-effective way to generate cryopreparations in much shorter times, at high quality. The application of the agitation modules might increase the use of cryopreparation methods in the overall sample preparation for light and electron microscopy, also correlated with analytical techniques.
Supplemental Material
DS_10.1369_0022155418786698 – Supplemental material for Agitation Modules: Flexible Means to Accelerate Automated Freeze Substitution
Supplemental material, DS_10.1369_0022155418786698 for Agitation Modules: Flexible Means to Accelerate Automated Freeze Substitution by Siegfried Reipert, Helmuth Goldammer, Christine Richardson, Martin W. Goldberg, Timothy J. Hawkins, Elena Hollergschwandtner, Walter A. Kaufmann, Sebastian Antreich and York-Dieter Stierhof in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
Thanks to the Core Facility Cell Imaging and Ultrastructure Research (University of Vienna), Helen Grindley (Durham University), Rebecca Stahl (University of Tübingen), Dr. Harumi Harada (Institute of Science and Technology–Austria), and Dr. Vanessa Zheden (Institute of Science and Technology–Austria) for excellent technical assistance. We are also grateful to Prof. Gilbert van Stappen from the Laboratory of Aquaculture & Artemia Reference Center, University of Gent, Belgium, for providing Artemia cysts and to Dr. Daniel Abed-Navandi from the Haus des Meeres aquarium, Vienna, Austria, for providing algae. The freeze substitution (FS) agitation module is protected under Austrian patent number AT515423 and PCT application WO2015154118.
Competing Interests
The author(s) declared the following potential competing interests with respect to the research, authorship, and/or publication of this article: S.R. and H.G. and the University of Vienna are patent holders. H.G. manufactures agitation modules on demand. Beyond that, the authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: S.R. designed the study, drafted the manuscript, initially tested prototypes of the agitation modules, designed protocols for algae and A. franciscana, and generated electron microscopy (EM) data. H.G. developed and manufactured prototypes of the agitation modules used in the EM labs of the coauthors. E.H. and M.E. helped design protocols for algae and A. franciscana, generated EM data, and contributed to interpretation. S.A. participated in prototype testing and experimental design. C.R., M.W.G., and T.J.H. provided material, helped design protocols for S. cerevisiae, C. elegans, and O. tauri, helped interpret the EM data, and revise the manuscript. C.R. carried out the protocols and imaging. W.A.K. designed protocols for organotypic nerve tissues, generated, and interpreted EM data. Y.-D.S. designed protocols for anthers of A. thaliana, carried out comparative studies of freeze substitution under agitation, performed immunogold labeling, generated, and interpreted EM data. Y.-D.S., W.A.K., C.R., and M.W.G. participated in revision of the draft. All authors have read and approved the manuscript as submitted.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: S.R. was supported by the Austria Wirtschaftsservice (P1404894). M.W.G. was supported by Biotechnology and Biological Sciences Research Council, UK (grant numbers BB/E015735/1 and BB/G011818/1). Y.-D.S. was supported by the Deutsche Forschungsgemeinschaft through SFB1101.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
