Abstract
Endocrine cells require several protein convertases to process the precursors of hormonal peptides that they secrete. In addition to the convertases, which have a crucial role in the maturation of prohormones, many other proteases are present in endocrine cells, the roles of which are less well established. Two of these proteases, dipeptidyl peptidase IV (EC 3.4.14.5) and membrane dipeptidase (EC 3.4.13.19), have been immunocytochemically localized in the endocrine pancreas of the pig. Membrane dipeptidase was present exclusively in cells of the islet of Langerhans that were positive for the pancreatic polypeptide, whereas dipeptidyl peptidase IV was restricted to cells positive for glucagon. Both enzymes were observed in the content of secretory granules and therefore would be released into the interstitial space as the granules undergo exocytosis. At this location they could act on secretions of other islet cells. The relative concentration of dipeptidyl peptidase IV was lower in dense glucagon granules, where the immunoreactivity to glucagon was higher, and vice versa for light granules. This suggests that, in A-cells, dipeptidyl peptidase IV could be sent for degradation in the endosomal/lysosomal compartment during the process of granule maturation or could be removed from granules for continuous release into the interstitial space. The intense proteolytic activity that takes place in the endocrine pancreas could produce many potential dipeptide substrates for membrane dipeptidase.
Keywords
E
In this study we confirm that DPP-IV is strictly confined to pancreatic A-cells and that MDP is also present in only one type of islet of Langerhans cell in the pig, the pancreatic polypeptide (PP) cells. Like DPP-IV, MDP is also present in the content of secretion granules. Finally, both enzymes were observed in granules undergoing exocytosis for their release within the islet interstitial space, where they could act on secretory products of neighboring cells.
The location of MDP in the endocrine pancreas is better justified than in the exocrine pancreas, because intense proteolytic processing takes place in the islets, thus producing numbers of potential substrates.
Materials and Methods
Antibodies
Antibodies were generated against affinity-purified pig kidney MDP and against pig kidney DPP-IV as previously described (Littlewood et al. 1989; Howell et al. 1993). Antibodies specific to porcine glucagon (NCL-GLUCp) were from Novo Castra Labs (Newcastle, UK). Antibodies to somatostatin (S309-JA3-81), a kind gift of Dr. R. Benoit (McGill University, Montreal, Canada), were previously characterized as specific for somatostatin 28 (Ravazzola et al. 1983). Antibodies to serotonin (B56-1) and PP (B32-1) were from Euro-Diagnostica (Malmö, Sweden). Anti-insulin antibodies (A564) were from Dako (Carpinteria, CA). All the primary antibodies were produced in rabbits.
Tissue Fixation and Embedding
Immediately after sacrifice, pieces of tissue (1 mm3) were taken from different regions of the pancreas of a 47-day-old pig (20 kg) and were fixed at room temperature (RT) for 120 min in 2% formaldehyde, 0.25% glutaraldehyde buffered with 100 mM PIPES, pH 7.4. After washing in the same buffer, samples were progressively dehydrated in ethanol at −35C and embedded at the same temperature in Unicryl for 24 hr. Polymerization was brought about by diffuse UV irradiation at 360 nm for 24 hr at −35C, 24 hr at −20C, 24 hr at −10C, 24 hr at 4C, and finally 24 hr at RT. Experimental procedures on animals in this study were performed in compliance with Canadian Council on Animal Care guidelines.
Immunocytochemistry
Thin sections were immunocytochemically stained with protein A-gold using 10-nm gold particles at a dilution of OD520 nm = 0.1 in PBS for 25 min at RT as previously reported (LeBel et al. 1998) and were counterstained with uranyl acetate and lead citrate for observation by electron microscopy. Three control incubations were performed: without primary antibodies, with preadsorbed antibodies, and with preimmune serum. The antigens used for preadsorption were the corresponding porcine hormones, or kidney microvilli in the case of MDP and DPP-IV antibodies. Under these control conditions, only a few randomly distributed gold grains were observed. Quantitative evaluation of labeling concentration over secretory granules was carried out by counting gold particles over 15 granules. Differences between groups were tested with Student's t-test and only significant differences (p>0.005) are mentioned. For double staining, sections labeled with one primary antibody (anti-MDP or anti-DPP-IV) and protein A-gold particles of 10 nm were submitted to controlled silver enhancement (AURION R-Gent Silver Enhancement Kit; Cedarlane Labs, Hornby, ON, Canada) for 3 min. Sections were rinsed and submitted to immunocytochemical staining with the second antibody (anti-PP or anti-glucagon) using 10-nm colloidal gold particles.
Results
Immunocytochemistry of MDP in the exocrine pancreas gave a clear localization of the protein in the membranes of the acinar cell secretory pathway, and particularly in the granule membrane and content (LeBel et al. 1998). In the endocrine portion of the pancreas, MDP was restricted to only one cell type, a type that is not a major constituent of the islet of Langerhans. As shown in Figure 1, the identity of the cell could not be determined from the morphology of its granules, one of the reasons being that the morphology of the pig endocrine pancreas differs markedly from that of rodents. For example, the typical morphology observed in rodent B-cell β-granules, with a dense core surrounded by a clear halo, was not observed in the pig endocrine tissue (Figure 2a). The morphology of the pig β-granule is very similar to that of the human (Like and Orci 1972). To determine the identity of the MDP-positive cell, immunocytochemical localization of five endocrine secretory products was carried out in the pig tissue. Figures 2b and 2c show that only A-cells have granules with a prevailing round morphology. Granules of the other cell types have a less homogeneous morphology, like those of D-cells (Figure 2d), PP cells (Figure 2e), and enterochromaffin cells (EC cells) (Figure 2f). As previously shown by Poulsen et al. (1993), we were able to confirm that the only endocrine pancreatic cell expressing DPP-IV was the A-cell (Figure 2c). DPP-IV is also totally absent from the exocrine pancreas (not shown).
Double labeling immunocytochemistry of MDP separately with each one of the five endocrine pancreatic secretory products led to the exclusive co-localization of MDP and PP (Figure 3). In PP cells, MDP could be observed in structures that may represent poorly preserved Golgi cisternæ (Figure 3b), in the granule contents, and in granules very close to the membranes, and could therefore be undergoing exocytosis (Figure 3c). In all these locations, MDP was mostly present in the lumen of these organelles (Figures 3a and 3c) and was consequently seen secreted along with PP (Figure 3c). The presence in the lumen of these organelles of a presumably soluble form of a GPI-anchored membrane protein is somewhat unexpected but has previously been observed in the exocrine pancreas with GP-2 (Paquette et al. 1986) and MDP (Hooper et al. 1997a; LeBel et al. 1998). Therefore, it was interesting to examine the localization of a differently anchored membrane protein.
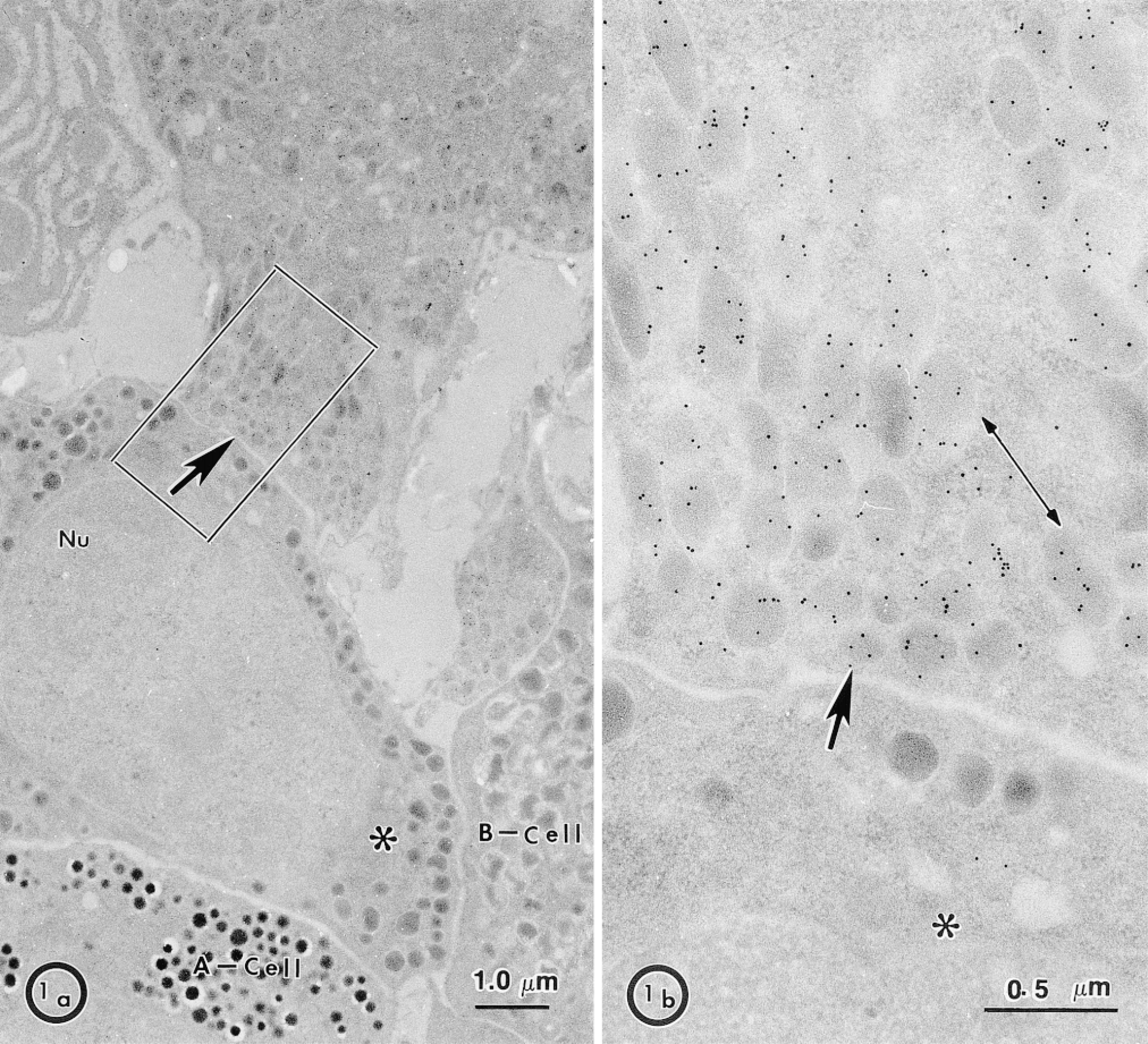
Localization of MDP in the pig endocrine pancreas. Under low magnification (a) MDP immunoreactivity is confined to only one cell type (arrow) distinct in morphology from adjacent A-cells, B-cells, and from another unidentified cell (∗) (b) An enlarged area of the positive cell in a shows MDP immunoreactivity associated with the secretory granule matrix. Labeling is uniform from one granule to another. Note the low background of labeling in the adjacent cell (∗). Nu, nucleus.
Localization of DPP-IV, which is a Type II membrane protein anchored by an uncleaved signal pep-tide, was performed. Figure 4 shows that, like MDP in PP cells, DPP-IV is present in the lumen of A-cell granules. The concentration of gold particles per μm2 for DPP-IV was separately determined for granules that were electron-lucent and those that were electron-dense. The concentration was 195.24 ± 15.60/μ2 in lucent granules, and 145.64 ± 15.25/μm2 in dense granules, showing a significant decrease of DPP-IV immunoreactivity in dense granules. This decrease in reactivity could be due to masking of the antigen in denser granules but, on the contrary, glucagon immunoreactivity in the same granules increased very significantly. The concentration of gold particles for glucagon was 42.71 ± 7.47/μm2 in electron-lucent granules, and 485.72 ± 29.28/μm2 in electron-dense granules. Such a positive concentration of glucagon immunolabeling over dense granules suggests that maturation of the granules is taking place, as previously observed in the exocrine pancreatic secretory pathway (Bendayan 1984). Therefore, in contrast to glucagon, DPP-IV did not undergo concentration in the A-cell dense granules.
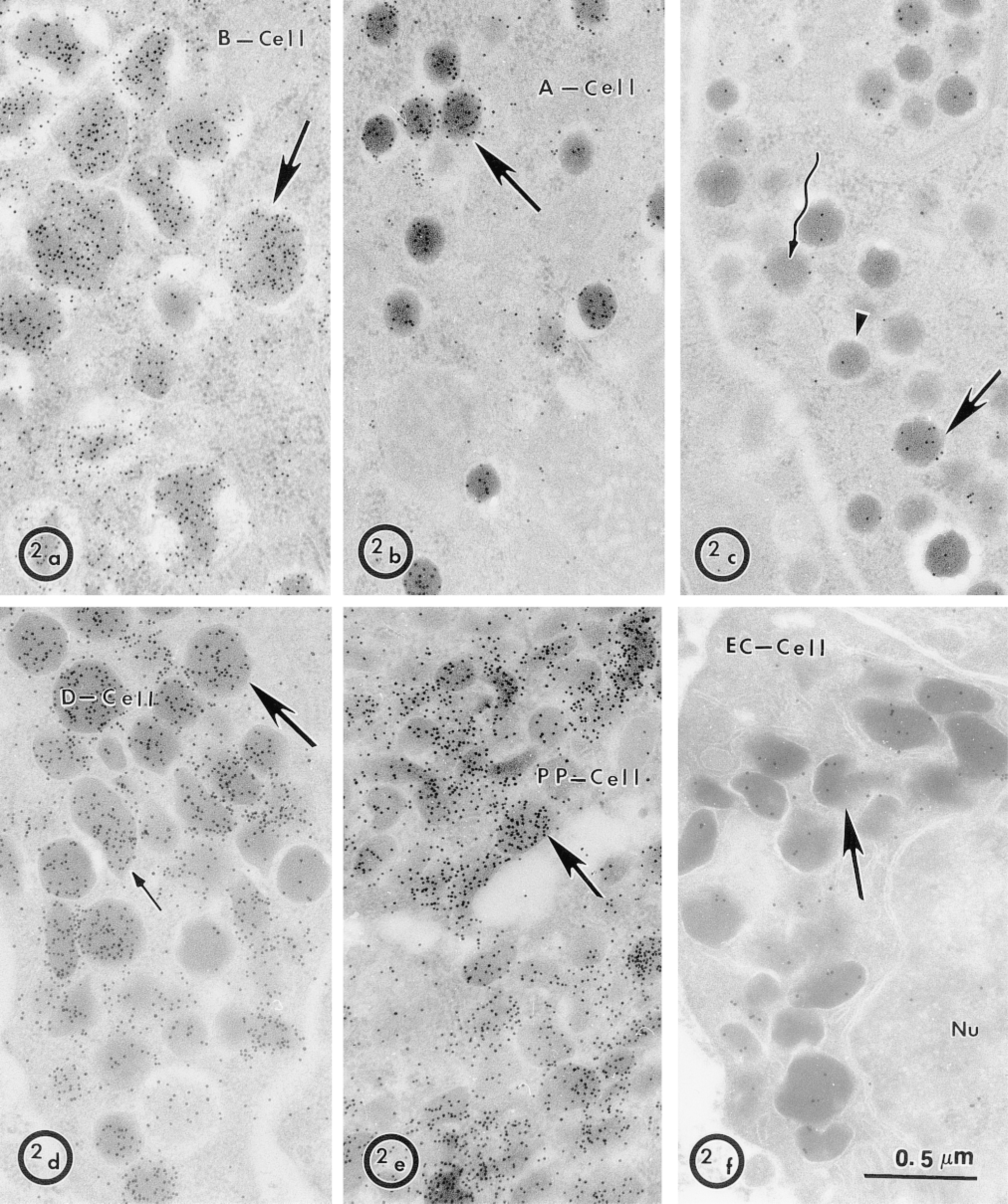
Characterization of five pig pancreatic endocrine cell types. To clearly identify the MDP-positive cell, antibodies to five secretory products of pancreatic islet cells were used to distinguish the morphology associated with the secreted hormone. Anti-insulin strongly labeled B-cell granules (a) anti-glucagon A-cell granules (b) anti-somatostatin-28 D-cell granules (d) anti-pancreatic polypeptide PP-cell granules (e) and anti-serotonin EC cell granules (f). (c) The specific immunolocalization of DPP-IV in A-cells (Poulsen et al. 1993) was confirmed, showing that the labeling is low in some granules (arrowhead, winding arrow) and high in others (arrow). In D-cells, round dense granules (large arrow) and irregular light granules (small arrow) were similarly labeled for somatostatin-28.
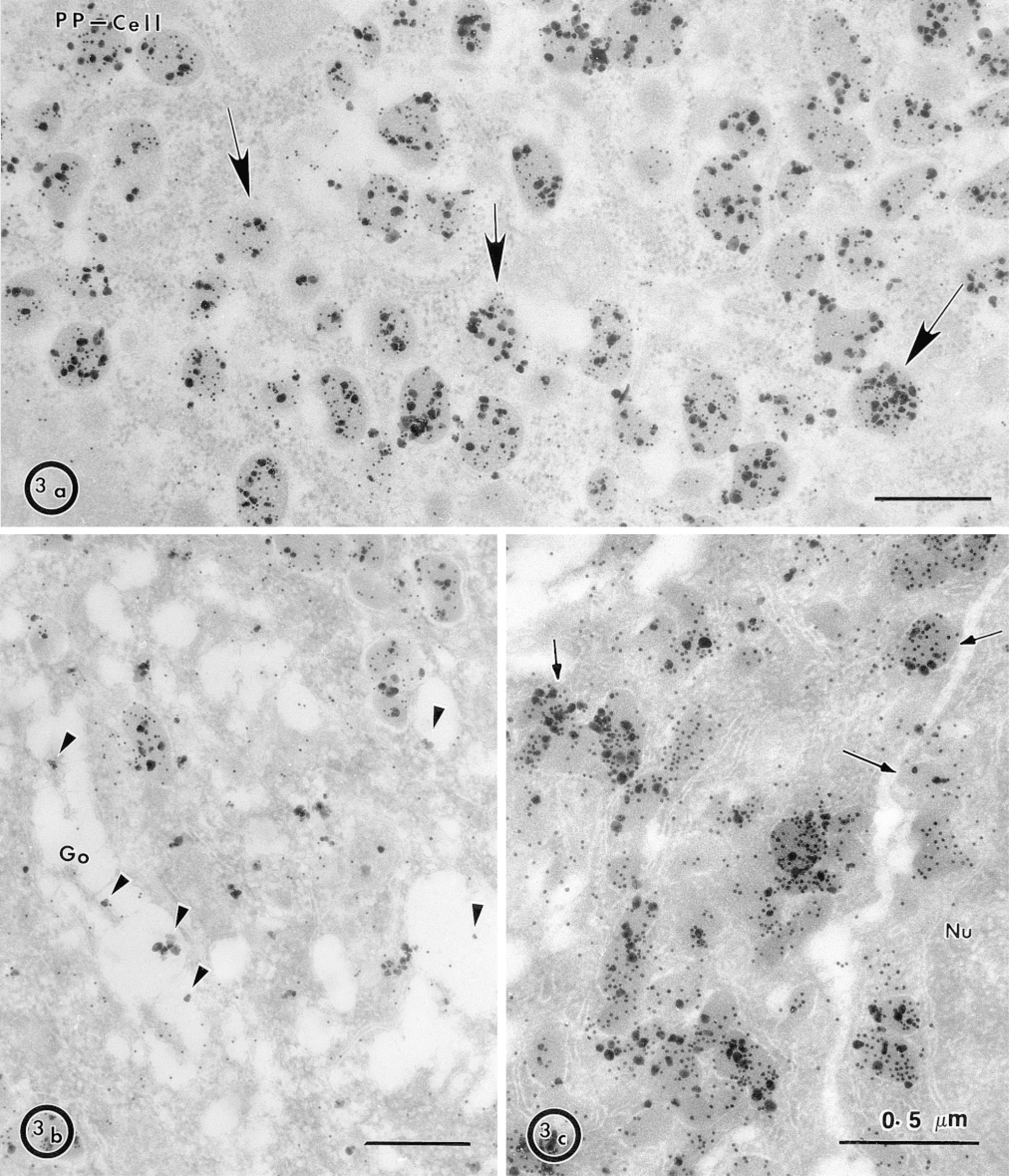
Co-localization of MDP and PP in pig pancreatic islet cells. (a) Double immunolabeling of PP cells with MDP antibodies (large particles) and PP antibodies (small particles) was performed. Most granules were positive for MDP (arrows). (b) Arrowheads show MDP in structures that could be Golgi cisternae, most of the signal being in the lumen of the cisternæ and little being associated with the membrane. (c) Granules in the close vicinity of the plasma membrane that may be in the process of exocytosis (arrows) are positive for PP and MDP. Nu, nucleus.
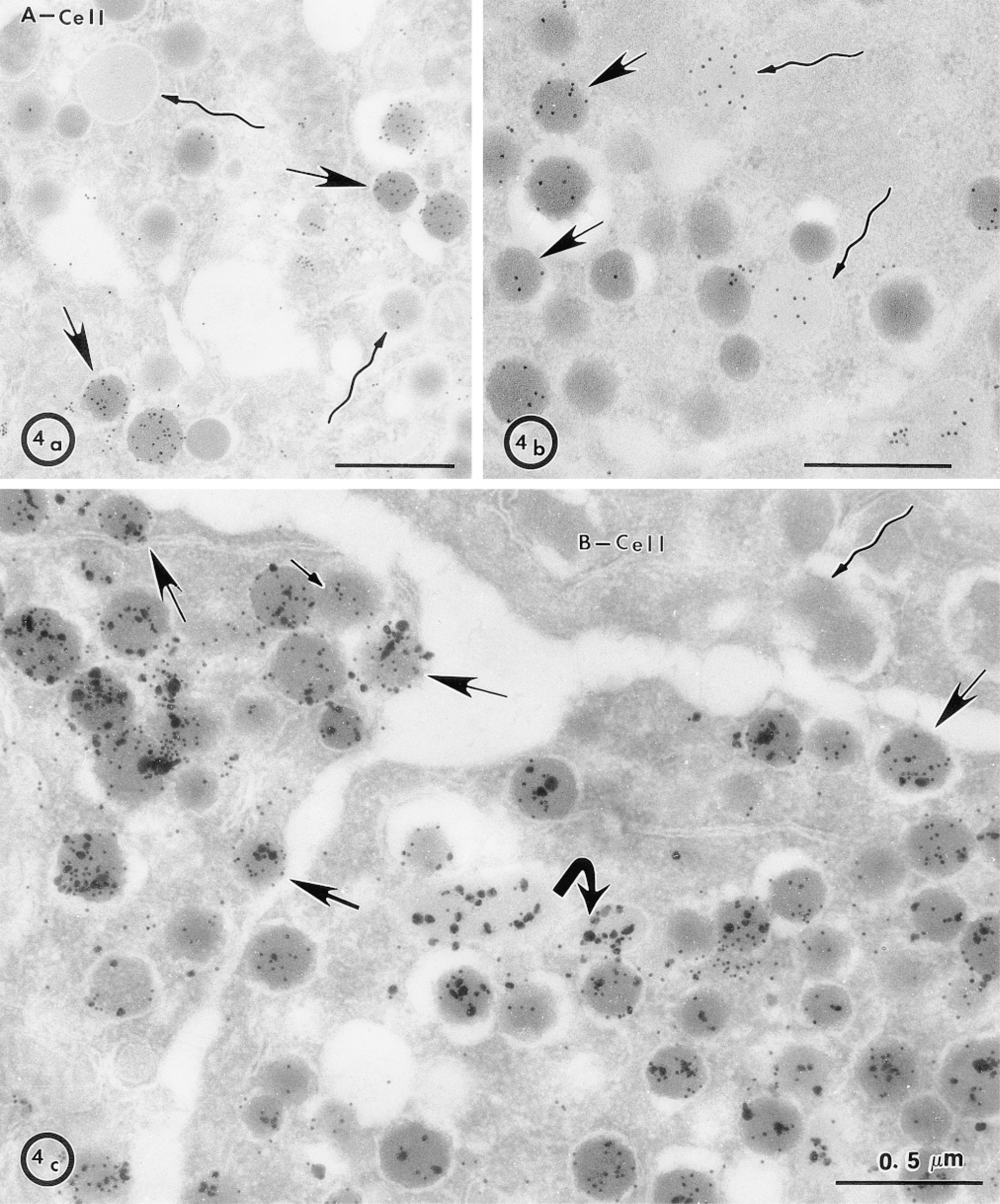
Association of DPP-IV with A-cell electron-lucent granules. (a) Glucagon immunodetection in A-cells was much more intense in electron-dense granules (arrows) compared to electron-lucent granules (winding arrows). Conversely, immunocytochemical detection of DPP-IV in A-cells (b) was much stronger and uniform in lucent granules (winding arrows) than in dense granules (arrows), where it was very heterogeneous. (c) Co-localization of DPP-IV (large particles) and glucagon (small particles) in A-cells confirmed that light granules have a much higher signal for DPP-IV than glucagon (bent arrow). Many granules positive for glucagon and DPP-IV are very close to the membrane and could be in the process of exocytosis (arrows). A B-cell with a granule located close to the plasma membrane (winding arrow) shows the specificity of detection for DPP-IV and glucagon, as well as the low level of background.
In divergence with a previous report in the mouse (Rombout et al. 1987), no PP immunoreactivity could be detected in the pig A-cell granules (not shown).
Discussion
Maturation and processing of hormone precursors in endocrine cells are performed by a number of exoand endoproteolytic proteases (Halban and Irminger 1994). In this respect, the endocrine pancreas is a particularly rich gland, secreting five different secretory products that are synthesized by five specialized cells. In addition to being endowed with the well-described protein convertase family of endoproteases, carboxypeptidases and the ubiquitous furin (Hook et al. 1994), the A-cell specifically contains DPP-IV in its secretory granules (Poulsen et al. 1993). Here we show by immunocytochemistry that DPP-IV is mostly soluble in granules of A-cells and consequently is secreted in the interstitial space of the islets of Langerhans. DPP-IV releases dipeptides from the N-terminus of proteins. Its cleavage specificity requires a Pro residue (or exceptionally Ala) to be at the C-terminus of the released dipeptide (see Trugnan et al. 1997 for review). According to its specificity, primary substrates for the peptidase in the α-granule would be glucagon-like peptide-2 and the truncated glucagon-like peptide-1, two C-terminal byproducts of proglucagon processing. Although both peptides are good substrates for DPP-IV in vivo (Kieffer et al. 1995; Drucker et al. 1997), the acidic pH of the granule would make their hydrolysis by DPP-IV less likely in this location. The secretory nature of DPP-IV that we report, and the optimal pH of its activity, suggest that the protease could act on the secretory products released into the interstitial space by any of the five types of endocrine cells. In addition to the previously mentioned glucagon maturation byproducts, two other secretory products could be substrates for DPP-IV. These are the propeptides of somatostatin and PP that are specifically secreted by D-cells and PP-cells, respectively. In support for such a role of the secreted DPP-IV on PP are reports that the major form of neuropeptide Y immunoreactivity in the circulation is a 34-residue peptide constituted of residues 3–36 (Medeiros and Turner 1996). Neuropeptide Y is a peptide very similar in sequence and function to PP, which possesses the consensus Ala-Pro at the N-terminus for recognition by DPP-IV. For the somatostatin propeptide, there is only one report for such trimming of its N-terminus (Baldissera 1994). Finally, the release of DPP-IV in the interstitial space also suggests a role for the protease in the inactivation of cytokines and chemokines, many of which have the N-terminal consensus sequence (X-Pro) (Vanhoof et al. 1995). Evidence for an involvement of DPP-IV in anti-inflammatory and antiviral responses has accumulated (Proost et al. 1998), supporting such a role for DPP-IV in the endocrine pancreas.
The release of DPP-IV from the membrane is not a new feature for this protease. It has long been known that acidic conditions favor autolysis of the protein (Macnair and Kenny 1979) and that a soluble form of the protein is present in the circulation (Trugnan et al. 1997). The simple mode of membrane anchorage of DPP-IV by an uncleaved signal peptide reduces the task of releasing the peptidase to the hydrolysis of a single peptide bond. A number of proteins anchored by a single membrane-spanning polypeptide are also known to exist in soluble forms as a result of proteolytic cleavage by a family of proteases termed secretases (Hooper et al. 1997b). It is very likely that the conditions of pH found in vivo in mature [pH 5.5 according to Dittié et al. (1997)] and most particularly in immature granules, where it is lower (Orci et al. 1987), are close to those used in vitro (pH 4.5–5.0) to generate a soluble form of DPP-IV by autolysis (Macnair and Kenny 1979). The higher amount of DPP-IV in electron-lucent granules compared to electron-dense ones is a very interesting finding regarding the biogenesis of secretory granules. If the lower level of DPP-IV immunodetection in dense granules was not due to antigen masking, this suggests that the protein is sorted out of immature granules during maturation, a phenomenon that has been observed with proteins that are soluble under the acidic conditions of granules and that have weak or no affinity for the aggregated cargo. This pathway is called the constitutive-like secretory pathway. It was elegantly demonstrated with the insulin C-peptide (Arvan et al. 1991). The pathway operates when granules are in the process of maturation. According to this scenario, DPP-IV could then be released almost continuously from A-cells. An alternative to the former pathway to explain the decrease of DPP-IV in dense granules could be a degradation of DPP-IV in the endosomal/lysosomal compartment.
Like DPP-IV, MDP is also secreted in the interstitial space. To find a substrate for MDP is easier because the only identifiable one in the endocrine pancreas would be the dipeptide made of the two basic residues that specify the cleavage site of convertases. However, the release of these two basic residues from the C-terminal end of these proteolytic fragments has thus far been attributed to sequential hydrolysis by carboxypeptidases (Loh et al. 1993). Because exocrine pancreatic cells are capable of a full range of prohormone processing reactions without any need for it (Dickinson et al. 1993), it would not be surprising that in some endocrine cells MDP could be an example of such a remaining activity without real utility in processing endogenous secretory products. As for the release of MDP into the endocrine granule lumen, it is quite conceivable that a phospholipase could be involved in cleaving its GPI anchor and releasing the protein from the membrane. A phospholipase A activity has indeed been identified in the exocrine pancreatic acinar cell granule membrane that promotes such a release of MDP (Hooper et al. 1997a; LeBel et al. 1998), but its purification and characterization have not yet been achieved.
Footnotes
Acknowledgements
Supported by the Canadian Cystic Fibrosis Foundation, NSERC (Canada), FCAR (Québec) to DL, by the Medical Research Council of Great Britain to NMH, and by a NATO Collaborative Research Grant.
