Abstract
The GPI-anchored membrane dipeptidase is the major peptidase activity of the secretory granule membrane in the exocrine pancreas. The enzyme is also found in the granule content and in pancreatic secretions. Immunocytochemical localization confirmed its location in the granule membrane and in the acinar cell apical plasma membrane. In the endoplasmic reticulum and Golgi, membrane dipeptidase was strictly membrane-bound. There was no membrane dipeptidase in duct cells. The release of membrane dipeptidase from the membrane starts in the immature granule. To identify the mechanism responsible for its release, secretions were collected from cannulated conscious pig under basal conditions and atropine perfusion. The latter treatment caused complete inhibition of protein secretion but had a negligible effect on membrane dipeptidase activity in the secretions. In secretions, membrane dipeptidase partitioned into the detergent-rich phase on phase separation in Triton X-114, whereas treatment with bacterial phosphatidylinositol-specific phospholipase C caused the peptidase to partition into the aqueous phase, indicating that the secreted enzyme could come from shedding of membrane fragments at the apical surface or via the action of a previously characterized phospholipase A activity.
T
Here we show, using immunocytochemistry at the electron microscopic level, that MDP is localized all along the secretory pathway of the exocrine pancreatic cell, from the endoplasmic reticulum (ER) to the lumen of the acinus, and that its release from the zymogen granule membrane starts in the immature granule. The endogenous mechanism by which MDP is released into pancreatic secretions has also been assessed by characterization of the secreted protein.
Materials and Methods
Materials
Cilastatin was a gift from Merck, Sharp & Dohme (Rahway, NJ). Bacillus thuringiensis phosphatidylinositol-specific phospholipase C (PI-PLC) was a gift from Dr. M.G. Low (Columbia University; New York, NY). Gly-
Atropine Perfusion of Pigs
Large White hogs (~50 kg live weight) were fitted with a permanent cannula into the pancreatic duct under halothane anesthesia as previously described (Viau et al. 1994). After an overnight fast with free access to water, cannulated pigs were perfused at 10 ml.h-1 with saline containing 0.5% porcine serum albumin (saline-PSA) for a period of 1 hr. Stimulation was then initiated by
MDP enzymic activity was determined as previously reported (Hooper et al. 1987) with Gly-
Incubation with PI-PLC and temperature-induced phase separation in Triton X-114 were performed as reported in Hooper et al. (1997) on 50 μl of secretions.
Microscopy and Immunocytochemistry
Immediately after sacrifice, pieces of tissue (1 mm3) were taken from different regions of the pancreas of a 47-day-old pig (20 kg) and fixed at room temperature (RT) for 120 min in 2% glutaraldehyde buffered with 100 mM cacodylate, pH 7.4. After washing in the same buffer, samples were progressively dehydrated in ethanol at −35C (Carlemalm et al. 1985) and embedded at the same temperature in Unicryl for 24 hr. Polymerization was brought about by diffuse
Results
A high specific activity has been shown for MDP in the secretory granule membrane of the exocrine pancreas in the pig and human (Hooper et al. 1997). It was also evidenced in secretions and granule content. Immunocytochemical localization of MDP was therefore performed on pig exocrine pancreatic tissue to ascertain the presence of MDP in acinar cells and absence in the duct system, and also to determine where the GPI-anchored MDP started to be released from the membrane in the secretory pathway. Figure 1A confirms biochemical observations that MDP is associated with the granule membrane and that it is also present in the matrix and in the secretory material released into the apical acinar lumen. In the granule content, the concentration of label is not uniform from one granule to another (Figure 1A) or from one cell to another (not shown). Quantitation of the label in mature granules gives a total of 8.45±0.62 particles per granule, 2.31±0.21 particles at the membrane and 6.14±0.56 in the matrix. Controls using blocked primary antibodies give a signal of 0.16±0.01 particles per granule. This quantitation shows that 72.7% of the label is in the granule matrix and 27.3% on the membrane. Figure 1 shows that MDP is strictly membrane-associated from its site of synthesis in the rough ER (Figure 1B) and up to the Golgi (Figure 1C). At the level of immature granules, MDP is explicity released into the lumen (Figure 1C). No signal for the CRD epitope could be detected in the whole pancreas, and no MDP could be observed in duct cells (result not shown).
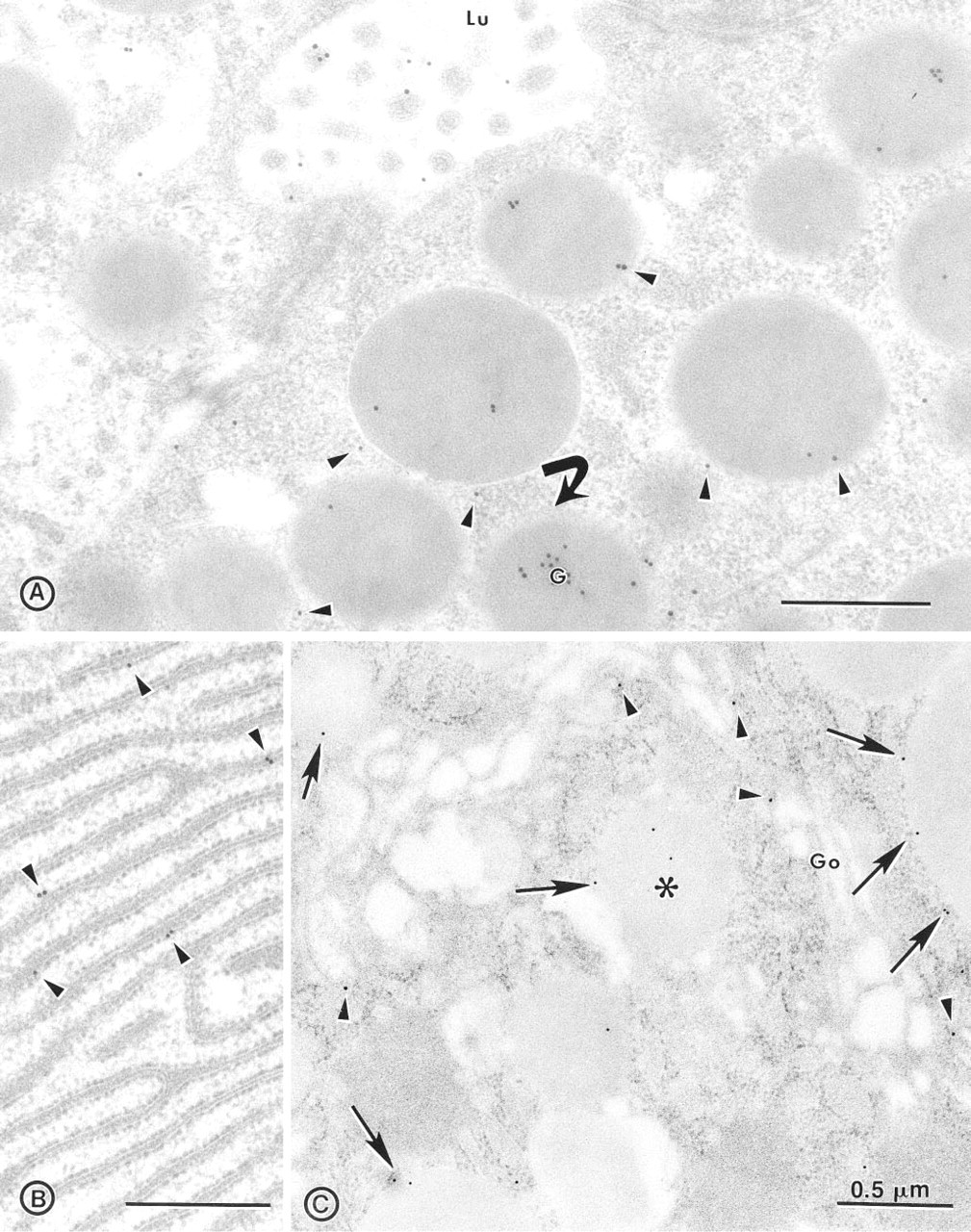
Immunolocalization of MDP along the secretory pathway of the pig exocrine pancreas. (
The release of MDP in pancreatic secretions was further examined using the cannulated pancreas of conscious pigs to document its mode of release. A protocol similar to the one previously used to study GP-2 secretion was used (Viau et al. 1994). In short, the cannulated pancreatic secretion of conscious pigs was harvested under constant
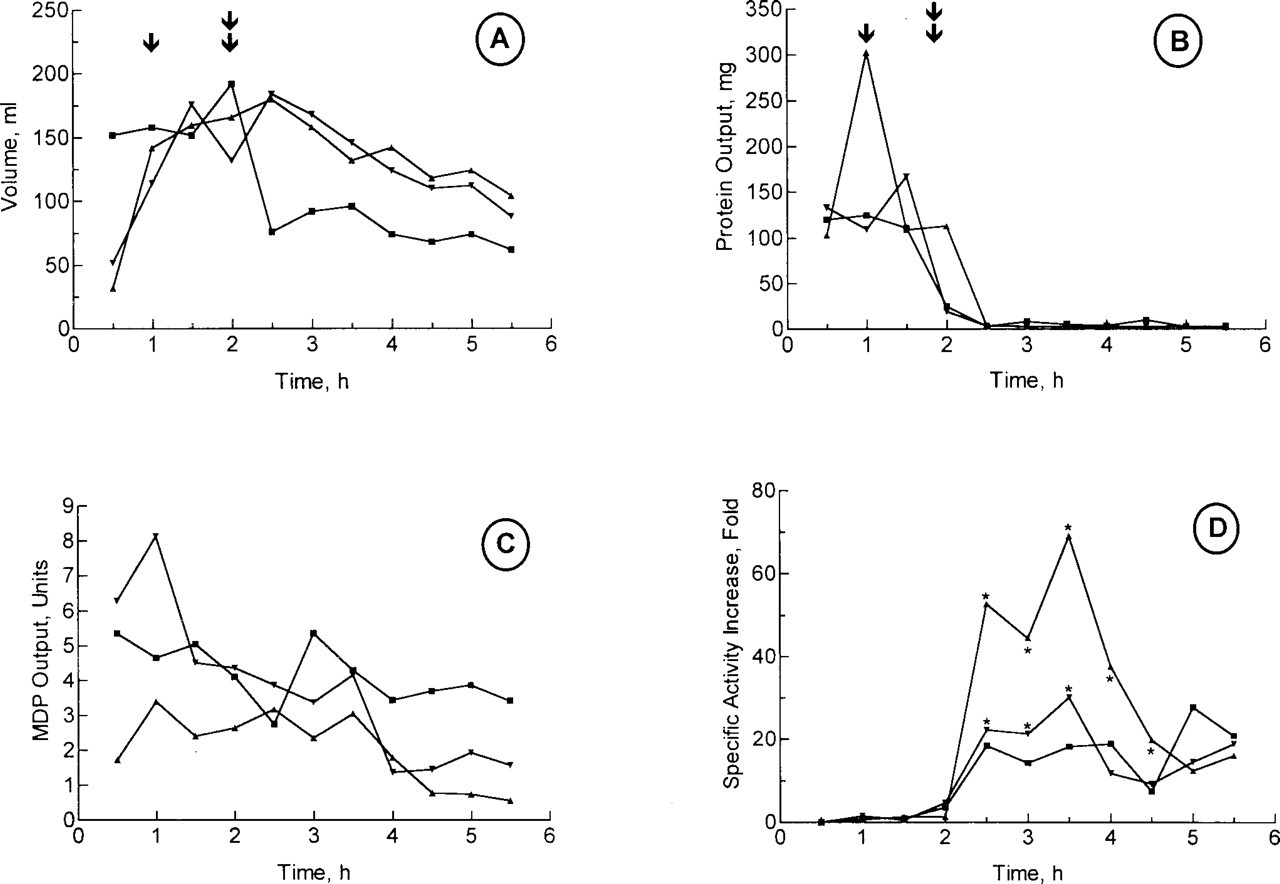
Effect of atropine on MDP secretion and secretory response of cannulated pig pancreas under secretin perfusion. Cannulated conscious pigs with permanent pancreatic cannula were perfused IV with 36 pmol.kg-1.h-1 of secretin starting at time 1 hr (↓) and ending at time 4 hr. Saline-PSA was perfused for 1 hr before secretin perfusion. Pancreatic secretion was sampled in 30-min aliquots. Atropine (50 mg.kg-1.h-1) was perfused starting at time 2 hr (double ↓) and lasted 2 hr. Three separate experiments on two different pigs are shown. Secretion was monitored for volume (
Total amount and specific activity of MDP in porcine samples a
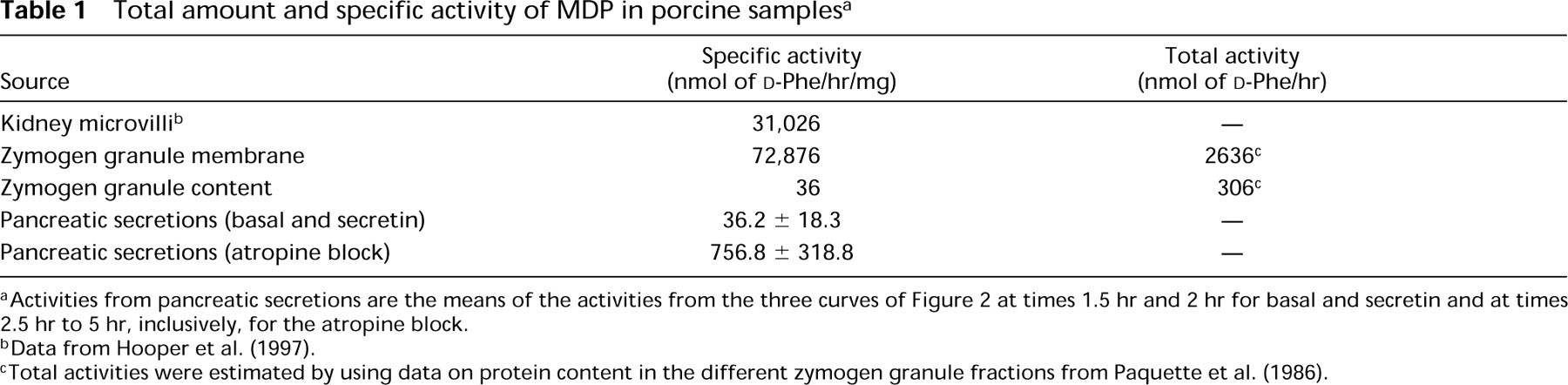
Activities from pancreatic secretions are the means of the activities from the three curves of Figure 2 at times 1.5 hr and 2 hr for basal and secretin and at times 2.5 hr to 5 hr, inclusively, for the atropine block.
Data from Hooper et al. (1997).
Total activities were estimated by using data on protein content in the different zymogen granule fractions from Paquette et al. (1986).
The form into which MDP is secreted was examined using temperature-induced phase separation in Triton X-114. Table 2 shows that in secretions under the atropine block, MDP partitioned predominantly (>75%) into the detergent-rich phase, suggesting the presence of a hydrophobic moiety of the GPI anchor still attached to the enzyme. Treatment with bacterial PIPLC caused MDP to partition predominantly (>67%) into the aqueous phase, confirming that at least one fatty acid chain is present on the GPI anchor. In contrast, treatment with pancreatic phospholipase A2 (PLA2) did not alter partitioning of MDP (results not shown) as previously observed (Hooper et al. 1997). Therefore, the secreted MDP retains a GPI anchor with an intact diacylglycerol or monoacylglycerol insensitive to hydrolysis by PLA2.
Discussion
MDP is the major constituent membrane peptidase of the zymogen granule in the exocrine pancreas (Hooper et al. 1997). The pig granule membrane is one of the membranes in which the enzyme reaches its highest specific activity of 72,876 nmol
In the rough ER, MDP is neatly confined to the membrane (Figure 1). This is consistent with the mode of addition of the GPI anchor to proteins that takes place in the ER. This is the first time that such an association of a GPI-anchored protein with the rough ER membrane has been so explicitly confirmed by immunocytochemistry at the electron microscopic level. From the ER up to the Golgi along the secretory pathway, MDP is exclusively membrane-bound. However, as mentioned above, a significant fraction of MDP is not membrane-bound in granules and the enzyme is secreted by the pancreas. Therefore, two questions were addressed. (a) Where in the secretory pathway does the release of MDP from the membrane start? (b) By which enzymatic process is it accomplished? Immunocytochemistry answered the first question, showing that MDP is first released in immature granules (Figure 1C) and downstream in the secretory pathway in the mature granule and the acinar lumen.
Effect of PI-PLC on the phase separation in Triton X-114 of MDP in pancreatic secretions (% MDP in aqueous phase a )
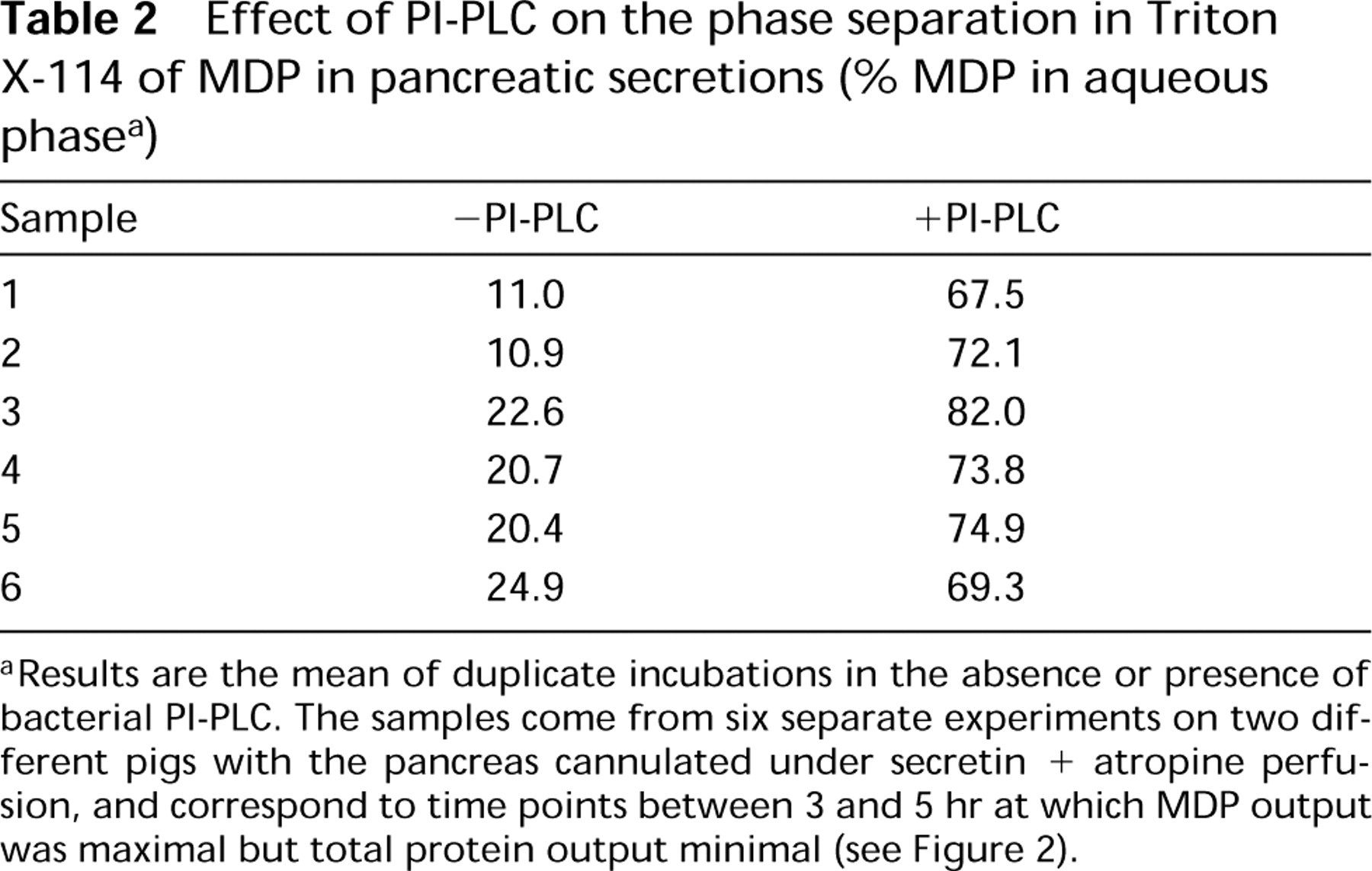
Results are the mean of duplicate incubations in the absence or presence of bacterial PI-PLC. The samples come from six separate experiments on two different pigs with the pancreas cannulated under secretin + atropine perfusion, and correspond to time points between 3 and 5 hr at which MDP output was maximal but total protein output minimal (see Figure 2).
For the mode of release of MDP from the membrane in vivo, two mechanisms are known: a PI-PLC (Movahedi and Hooper 1997) and a PI-PLD (Metz et al. 1991). For the involvement of a PI-PLC, we have never been able to demonstrate such an activity in the granule membrane (Hooper et al. 1997), and we now confirm that the CRD epitope [inositol-1,2-(cyclic)monophosphate] exposed by the action of a PI-PLC on GPIanchored proteins is not detectable in the exocrine pancreas even if two GPI-anchored proteins are present on the zymogen granule membrane (GP-2 and MDP). As far as pancreatic secretions are concerned, they have always been negative for the CRD epitope (Viau et al. 1994; Hooper et al. 1997). For the involvement of a PI-PLD activity, we and others have never been able to observe such activities in the granule membrane or pancreatic secretions (M.G. Low and D. LeBel, unpublished observations; Metz et al. 1991) or to detect the product of these activities in secretions (Hooper et al. 1997). This conclusion is also supported by the observation that MDP could not be isolated from either the granule content or secretions by affinity chromatography on cilastatin-Sepharose (Hooper et al. 1997). A different phospholipase, such as a PLA, might therefore be involved in the release of MDP from the membrane. Although we cannot directly distinguish whether the GPI anchor on the secreted form of MDP is intact or has been cleaved by PLA, with the recent identification of a PLA activity in the zymogen granule membrane (Hooper et al. 1997) and the present observation that the secreted MDP possesses at least one fatty acid chain (Table 2), it is possible that this endogenous activity promotes the release of MDP from the acinar cell. The other mechanism that is compatible with the properties of secreted MDP would be the shedding of intact membrane fragments in secretions.
Another question can be raised about the secreted MDP: from which intracellular compartment does it originate? The first obvious source would be the granule content because the specific activity of secreted MDP under basal conditions and secretin stimulation is very similar to that found in the granule content (Table 1). However, under atropine block, the specific activity of MDP increases tremendously, reaching very high values, values only found in membranes (Table 1), but with almost no change in the amount of activity secreted (Figure 2C). Considering also that only 10% of the granule active MDP is in the content (Table 1), the origin of the active MDP in secretions would therefore essentially exclude the granule content as a source. Therefore, one way to explain why the secretion of active MDP is unaffected by the complete inhibition of regulated secretion is that the apical plasma membrane might be the main source of secreted MDP activity. The low amount actually secreted could be compatible with the membrane as the source of MDP. This conclusion confirms two previous studies on the secretion of GP-2 that identified the apical plasma membrane as the main reservoir and site of release of this GPI-anchored protein (Freedman et al. 1994; Viau et al. 1994). Taken together, previous observations on GP-2 and the present observations of MDP in pig pancreatic secretions still having a hydrophobic portion of its GPI anchor suggest that the release of MDP activity would take place at the apical surface of the acinar cell by the action of a PLA or by the shedding of intact membrane fragments.
Footnotes
Acknowledgements
Supported by the Canadian Cystic Fibrosis Foundation, NSERC (Canada) to DL and by the Medical Research Council of Great Britain to NMH, and by a NATO Collaborative Research Grant.
