Abstract
The vgf gene has been shown to be involved in several metabolic pathways. Because the pancreas is crucial to metabolism and food intake, we studied the VGF peptides in bovine, rat, and pig Langherans islets using antisera raised against specific sites along the primary sequence of the rat/mouse and human VGF protein precursor. Whereas almost all of the pancreatic endocrine cells expressed vgf mRNA, when using the VGF antisera a different staining pattern became apparent. VGF556–565 and VGF282–291 immunoreactivity were exclusively found in δ somatostatin-producing cells, whereas the human C-terminus antiserum selectively immunolabeled α glucagon and pancreatic polypeptide cells. The same cells were decorated with the VGF443–588 antiserum, which also weakly labeled β insulin-secreting cells. Finally, the VGF298–306 peptide and the rat C terminus were found in virtually all pancreatic endocrine cells. Using bovine, swine, and rat pancreatic extracts, data from chromatography and ELISA assay showed the presence of a high molecular mass form compatible with the proVGF and lower molecular mass fractions corresponding to short VGF peptides. In conclusion, selective VGF distribution may suggest a multifaceted cell type-specific processing of proVGF, resulting indifferent peptides probably involvedin neuroendocrine regulatory metabolic mechanisms.
The vgf gene was discovered in PC12 rat pheochromocytoma cells because of its specific “delayed early” response to nerve growth factor (NGF) occurring at a later stage, compared with immediate-early genes (Levi et al. 1985; Salton et al. 2000). The relevant gene product is the proVGF polypeptide, or VGF protein, composed of 617 or 615 amino acids (in rat/mouse and in man, respectively, with >85% identity) (Canu et al. 1997; Salton et al. 2000) and migrating as an ~90-kDa molecular mass band in Western blot. Ten stretches of paired basic amino acid residues are conserved across the rat, mouse, and human coding sequences and represent potential cleavage sites (Salton et al. 2000). Processing of proVGF to intermediate and small molecular mass products has been shown in vivo (Possenti et al. 1989; Trani et al. 1995, 2002), and an endogenous peptide corresponding to the C-terminal 30-amino acid segment of human proVGF has been identified from bovine posterior pituitary (peptide V) (Liu et al. 1994). Moreover, processing at the Arg553-Pro554-Arg555 cleavage site allowed different VGF fragments to be isolated in rat brain. Peptides derived from the proVGF C-terminal region were studied in some detail and shown to be preferentially released in response to secretory stimuli from PC12 cells (Possenti et al. 1989), cerebellar granule cells (Trani et al. 1995), and insulinoma INS-1 cells (Possenti et al. 1999). In animal tissues, VGF immunoreactivity was restricted to central and peripheral neurons (Van den Pol et al. 1989; Ferri et al. 1992), as well as to endocrine cells of pituitary, adrenal medulla, gut, and pancreas (Ferri et al. 1992).
The vgf gene modulation was demonstrated in the mouse hypothalamic arcuate nucleus in response to fasting and feeding (Hahm et al. 2002). In vgf knockout mice, the most striking defect observed is an altered energy homeostasis. The VGF-deficient mice displayed a significant decrease in body weight and body fat, appeared hyperactive and hypermetabolic, and showed derangement of the hypothalamic pathways that regulate peripheral metabolic tissues and energy balance (Hahm et al. 1999, 2002).
More recently, VGF function was also investigated by examining the effect that vgf gene ablation had on glucose homeostasis in VGF mutant mice and in doublemutant mice generated by crossing VGF-deficient mice with well-characterized genetically obese mouse models. These studies showed that targeted deletion of VGF reduced the hyperinsulinemia and hyperglycemia resulting from high carbohydrate/high fat diets, gold thio-glucose lesions, and genetic defects in the hypothalamic melanocortin pathways (Watson et al. 2005).
Interestingly, chronic intracerebroventricular (ICV) injection of a VGF peptide named TLQP-21 increased resting energy expenditure and temperatureinmice. Furthermore, in mice fed a high-fat diet, the same peptide prevented the increase in body and white adipose tissue (WAT) weight as well as hormonal changes associated with a high-fat regimen (Bartolomucci et al. 2006).
Thus, a potential role has been proposed for VGF in the neuroendocrine regulation of several metabolic mechanisms. In the present study, we investigated the localization of VGF protein and its peptides within the endocrine pancreas, which is crucial to metabolism and food intake regulation. Because immunolocalization studies regarding the endocrine pancreas have so far been largely limited only to peptides containing the C-terminal portion of proVGF (Possenti et al. 1989), we developed and applied antisera to a range of further peptides, in connection with proVGF processing sites.
Materials and Methods
Tissue Samples and Processing
Samples of bovine and swine pancreas were taken from a local abattoir. Tissues were collected from 12 animals (males and females) for each species. Each animal was killed between the 10th and 12th month after birth. Sprague Dawley rats (n = 7) weighing ~ 250–300 g were killed by decapitation. Experimental protocols were approved by the Ethical Committee at the University of Cagliari and performed in strict accordance with the care and use of animals approved by the American Physiological Society and EEC Council Directive of 24 November 1986 (86/609).
For each case, areas corresponding to the body and tail regions have been taken. Tissues were immersion fixed in 40 g/liter (w/v) paraformaldehyde (in 0.1 mol/liter PO4, pH 7.2, 3–4 hr at 0–4C) and, after fixation, were washed in PBS (0.01 mol/liter PO4, pH 7.2–7.4, 0.15 mol/liter NaCl) containing 70 g/liter sucrose and 0.2 g/liter NaN3. Specimens were oriented in small aluminum foil molds in an embedding media composed of PVA 56–98 59 g/liter, Tween-20 10 g/liter, and Peg-400 40 g/liter in PBS-NaN3 1 g/liter (Cocco et al. 2003) and subsequently frozen in melting freon cooled with liquid nitrogen.
Five-μm sections were carried out by HM-560 cryomicrotome (Microm; Walldorf, Germany) and collected on slides coated with poly-
For chromatography and ELISA experiments, pancreas from rat, bovine, and pig were dropped into a tube containing distilled water (~10 ml/g tissue), kept in a boiling water bath (10 min), homogenized (3–5 min), briefly centrifuged (10–15 min, 3000 × g), and supernatants stored at −20C.
Antisera
A number of antisera against the precursor protein or specific sequences in connection with proVGF processing sites have been produced (Figure 1).
VGF antisera have been raised against the human and rat proVGF C terminus (respectively, VGF607–615 and VGF609–617) as previously described (Ferri et al. 1995; Brancia et al. 2005). In the C-terminal region of proVGF, a single amino acid substitution is found at position −3 with an arginine (Arg613) in man, as opposed to a histidine (His615) in rat and mouse (Salton et al. 2000). These nonapeptides were synthesized (Affiniti-Biomol; Exeter, UK) and conjugated to bovine thyroglobulin via an additional N-terminal d-tyrosine, for immunizations. The VGF-β-galactosidase fusion protein antiserum encompassing longer domains within rat proVGF (rat VGF443–588) was also used (Possenti et al. 1989; Ferri et al. 1992). The rat VGF556–565 peptide corresponds to the N-terminal portion of VGF fragment/s found in rat brain that resulted from processing at the VGF553–556 “Arg-Pro-Arg” cleavage site (Trani et al. 2002). This peptide was synthesized (Affiniti-Biomol) and conjugated to keyhole limpet hemocyanin (KLH) via an additional C-terminal cysteine, to expose the N-terminal region during immunizations. The amidate peptide corresponding to the human VGF298–306 sequence, and containing an amide group at its C terminus, was synthesized (Affiniti-Biomol) and conjugated to bovine thyroglobulin via an additional N-terminal d-tyrosine, for immunizations. The human VGF282–291 synthetic peptide containing the amino acid cys-teine at the C-terminus portion has been conjugated to KLH. All antisera used are listed in Table 1.
Immunohistochemistry
Sections of pancreas were treated with Triton X-100, 1 g/liter in H2O for 1 hr (Merck; Milan, Italy), to facilitate access of the antibodies. VGF antisera (Table 1) were diluted (1:200–1:10,000) in PBS (0.01 mol/liter PO4, pH 7.2–7.4, 0.15 mol/liter NaCl) containing 3% normal donkey serum, 3% normal serum of the species being immunostained (porcine, rat, or bovine), and 0.2 g/liter NaN3. Sections were incubated overnight at room temperature in a humid chamber. Blocking with unconjugated avidin-biotin was also used to reduce background staining (Vector Laboratories; Burlingame, CA). After the primary incubation, sections were treated with the affinity-purified biotinylated anti-rabbit or anti-guinea pig IgG diluted in the same medium as the primary, followed by Cy3-conjugated avidin (both from Jackson ImmunoResearch Laboratories; West Grove, PA). Endocrine cell populations were characterized on the basis of their hormonal content by means of double and triple immunofluorescence using primary antibodies against (i) insulin (Sigma), (ii) somatostatin (Chemicon International; Temecula, CA), (iii) glucagon (Sigma), and (iiii) pancreatic polypeptide (Serotec; Milan, Italy). Bounding was revealed with secondary antibodies (from donkey; Jackson ImmunoResearch Laboratories) conjugated with the following: (i) cyanin 3.18 (Cy3, emitting in yellow/red), (ii) 7-amino-4-methylcoumarin-3-acetic acid (AMCA, blue), and (iii) either cyanin 2.18 (Cy2, green) or biotin followed by Alexa 488-labeled avidin (green, Molecular Probes; Leiden, The Netherlands). Routine controls included substitution of each antiserum/antibody or conjugate, in turn with PBS. Absorption controls were carried out by incubations of each antibody with its homologous peptide and fusion proteins (up to 30 nmol/ml) as well as the unrelated peptide/s.
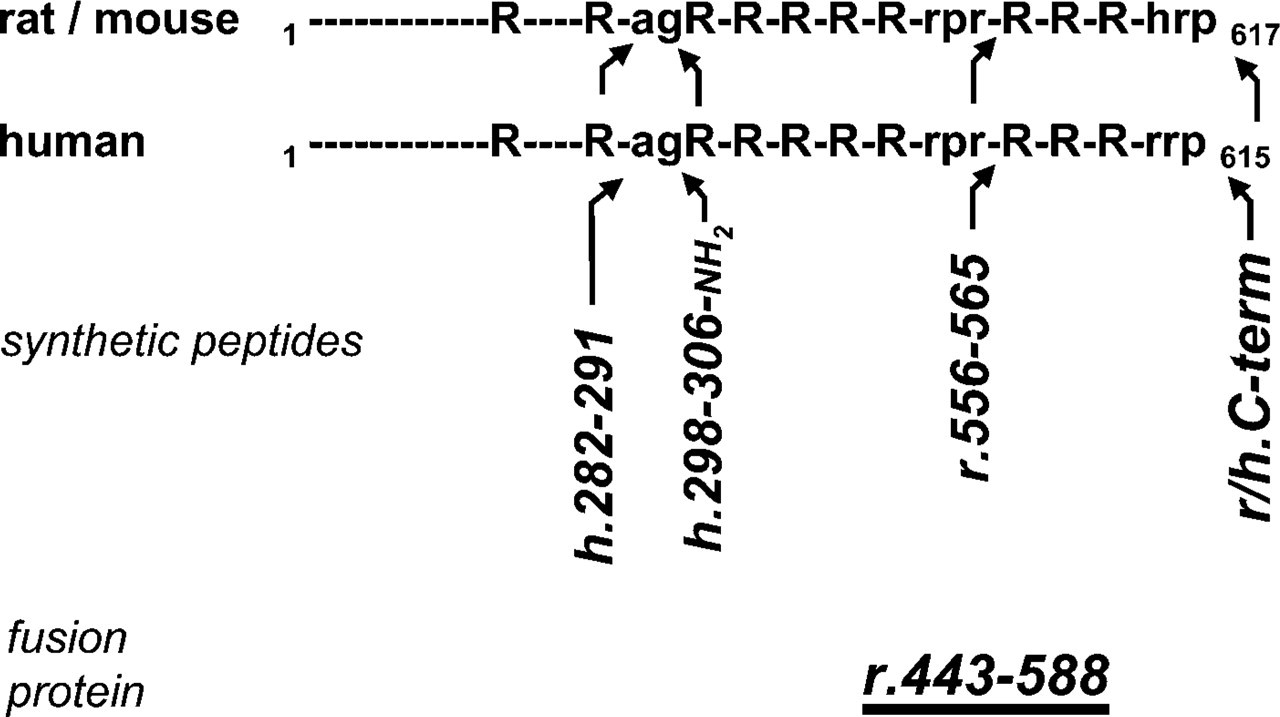
VGF peptides and fusion proteins used to raise antisera. Putative and confirmed processing sites within rat/mouse and human proVGF are indicated as “R” (two or more Arg/Lys basic residues), “rpr” (ratArg553-Pro554-Arg555 processing site; Trani et al. 2002), or “agR” (putative processing-amidation site: Ala306-Gly307-Arg308-Arg309); the last three C-terminal amino acids are indicated in single-letter notation. h.282–291, human VGF282–291. h.298–306, human VGF298–306; r.556–565, rat VGF556–565; r/h. C-term, rat and human pro-VGF C terminus. Arrowheads are bent according to expected antiserum reactivity.
Homologous peptides resulted in prevention of immunostaining, whereas unrelated peptides and carrier protein had no significant effect, confirming the specificity of our antibodies. Slides were coverslipped with PBS-glycerol. BX60 and BX50 microscopes (Olympus Italia; Milan, Italy) equipped with standard 100-W mercury lamps were used for observations and T-Max 400 film (Kodak; Rochester, NY) and PM-30 systems (Olympus Italia) for photography.
In Situ Hybridization
An ×600-bp fragment of rat VGF cDNA (BamHI-XhoI 235–854 bp from the ATG of the coding sequence) was cloned in both orientations, downstream of the T7 polymerase promoter. Labeled antisense and sense probes were obtained by in vitro transcription of the linearized plasmids with T7 polymerase using Megascript labeling kits (Ambion; Austin, TX) and digoxigenin-11-UTP (Roche; Monza, Italy). Probe quality was assessed by 20 g/liter of agarose electrophoresis. The hybridization buffer contained 50% deionized formamide, 1% blocking solution, 5 mmol/liter EDTA, 5X SSC, 0.1% Chaps, 0.1 mg/ml heparin, 1 mg/ml yeast total RNA, and 10 mg/ml denatured sheared herring sperm DNA (reagents from either Roche or Sigma, as relevant) (see Moorman et al. 2001).
Sections were treated with Triton X-100 (1 g/liter in PBS, 0.01 mol/l PO4, pH 7.2–7.4, 0.15 mol/liter NaCl), then digested with proteinase K (1 μg/ml, in PBS containing 1 mmol/liter EDTA, for 20 min) (ICN; Milan, Italy) followed by treatment with glycine (2 g/liter in PBS, 5 min) and postfixation with paraformaldehyde (40 g/liter, in PBS, 5 min). After a preincubation step with hybridization buffer (at 70C, 1 hr), preparations were hybridized overnight with the relevant probe dissolved in hybridization buffer (at 70C). RNase digestion (100 μg/ml; ICN) and stringency washes (70C, 2X SSC, 2 × 10 min; 1X SSC, 0.5X SSC, 0.25X SSC, 10 min each; 1X SSC = 0.15 mol/liter NaCl, 15 mmol/liter sodium citrate, pH 7.2) were used. Hybridization was carried out also with lower stringency (at 55C, 2X SSC, 2 × 10 min; 1X SSC). The bound probe was revealed using alkaline phosphatase-labeled anti-digoxigenin Fab fragment antibodies (Roche) diluted in AP1 buffer (1 mol/liter Tris-HCl, pH 7.6, containing 5 mol/liter NaCl, 0.5 mol/liter MgCl2, and 30 g/liter BSA). Overnight development was carried out using a 5-bromo-4-chloro-3-indolyl phosphate/nitroblue tetrazolium kit with inclusion of levamisole, according to the kit's instructions (Vector Laboratories). Controls included incubation with sense probe and probe omission, as well as predigestion of sections with RNase A (100 μg/ml, 37C, 1 hr).
VGF antisera used in the study

Antigens were conjugated to
b.thyrogl-dY (bovine thyroglobulin via an additional d-tyrosine) or
KLH (keyhole limpet hemocyanin) via an additional C-terminal cysteine, and
β-galact (β-galactosidase).
h. VGF, human VGF; r. VGF, rat VGF; r/h., rat/human.
Chromatography
Extracted proteins from bovine, swine, and rat pancreas (~2 ml) were transported through a Sephadex G-50 (Sigma) column equilibrated with 50 mM ammonium bicarbonate and connected with a pressure pump (5 impulses/min, 4C). Void volume (V0) and standard curve were calculated by an apposite kit (Sigma) containing the following molecular markers: 66 kDa (bovine albumin), 29 kDa (carbonic anhydrase), 12.4 kDa (cytochrome c), and 6.5 kDa (aprotinin).
Eluted pancreatic proteins were collected in 3-ml fractions corresponding to specific molecular masses and analyzed using ELISA assay.
ELISA
To demonstrate which and how specific molecular mass fragments are revealed by our antibodies, we carried out an ELISA competitive binding assay.
Plates (NUNC-Immuno Plate F96 MaxiSorp; Milan, Italy) were coated (100 μl/well, 4 hr at 37C) with the following specific antigens diluted in carbonate/bicarbonate buffer (pH 9.6): (i) human VGF607–615 C terminus, (ii) rat VGF609–617 C terminus, (iii) synthetic peptide resulting from processing at the VGF553–555 “Arg-Pro-Arg” cleavage site, and (iiii) amidate peptide correspondent to the human VGF298–306 sequence containing an amide group at its C terminus.
Regarding the other VGF antisera used in immunohistochemistry (Table 1), preliminary experiments using the ELISA assay failed to obtain detectable signals. Plates were washed in PBS (0.01 mol/liter PO4, pH 7.2–7.4, 0.15 mol/liter NaCl) containing 0.5 g/liter Tween 20, overcoated with 9% normal donkey serum (4 hr), washed as above, and stored at −20C.
For each single assay, wells were incubated (100 μl/well, 4 hr at room temperature) in 9% normal donkey serum with the primary antibody (VGF607–615, VGF609–617, VGF556–565, or VGF298–306) and the pancreatic fractions competing with the substrate-attached synthetic peptide for the VGF antibody binding.
The human C-terminus antiserum was used exclusively with bovine and swine pancreatic extracts, whereas the rat VGF609–617 C terminus was used with rat pancreatic extracts. Standard duplicates and blanks were run for each plate to ensure accuracy. After the primary incubation, wells were treated (100 μl/well, 1 hr at room temperature) with the biotin-conjugated secondary antibodies (anti-rabbit and antiguinea pig from donkey; Jackson ImmunoResearch Laboratories, West Grove, PA) diluted in 9% normal donkey serum.
For detection, streptavidin-peroxidase was diluted in PBS (0.01 mol/liter PO4, pH 7.2–7.4, 0.15 mol/l NaCl) containing 1 g/liter BSA and incubated (100 μl/well for 30 min) at room temperature. After washes (10 min), tetramethylbenzidine (TMB; Sigma) and 1 M HCl were added to the wells. The assay was then quantified by measuring (450 nm absorbance) the amount of the secondary antibody bound to the substrate-attached synthetic peptide using a Plate CHAMELEON multilabel detection platform (HIDEX; Turku, Finland). Absorbance was converted to units of product formation using a standard curve obtained by carrying out the ELISA procedure with known quantities of each VGF peptide (500–0.005 nmol/ml).
In the assays, 50% inhibitory concentrations were 0.1 pmol/ml for the rat C terminus peptide, 1 pmol/ml for the amidated peptide, and 2 pmol/ml for the VGF556–565 and the human C-terminus peptides.
Results
Immunofluorescence and In Situ Hybridization
Pancreatic endocrine cells were effectively labeled in varied numbers and intensities of staining in bovine, swine, and rat tissues, whereas exocrine pancreas was not decorated. The selective positive reaction within the islets was suddenly apparent using the VGF antisera, which labeled single or multiple cells in the islet periphery (Figures 2A and 2E), as well as inside (Figure 3A) or throughout it (Figures 2I and 2O). A few antisera also immunostained nerve fibers running in the acinar tissue and, even more so, encircling the islets, as well as occasional ganglia between pancreatic lobules (Figure 3C). By in situ hybridization, we also detected vgf mRNA in almost all pancreatic endocrine cells of the rat and, with less intensity of staining, in bovine islets (Figure 3E). On the contrary, vgf mRNA was not visible within rat and bovine exocrine pancreas and in all of the swine pancreatic cells.
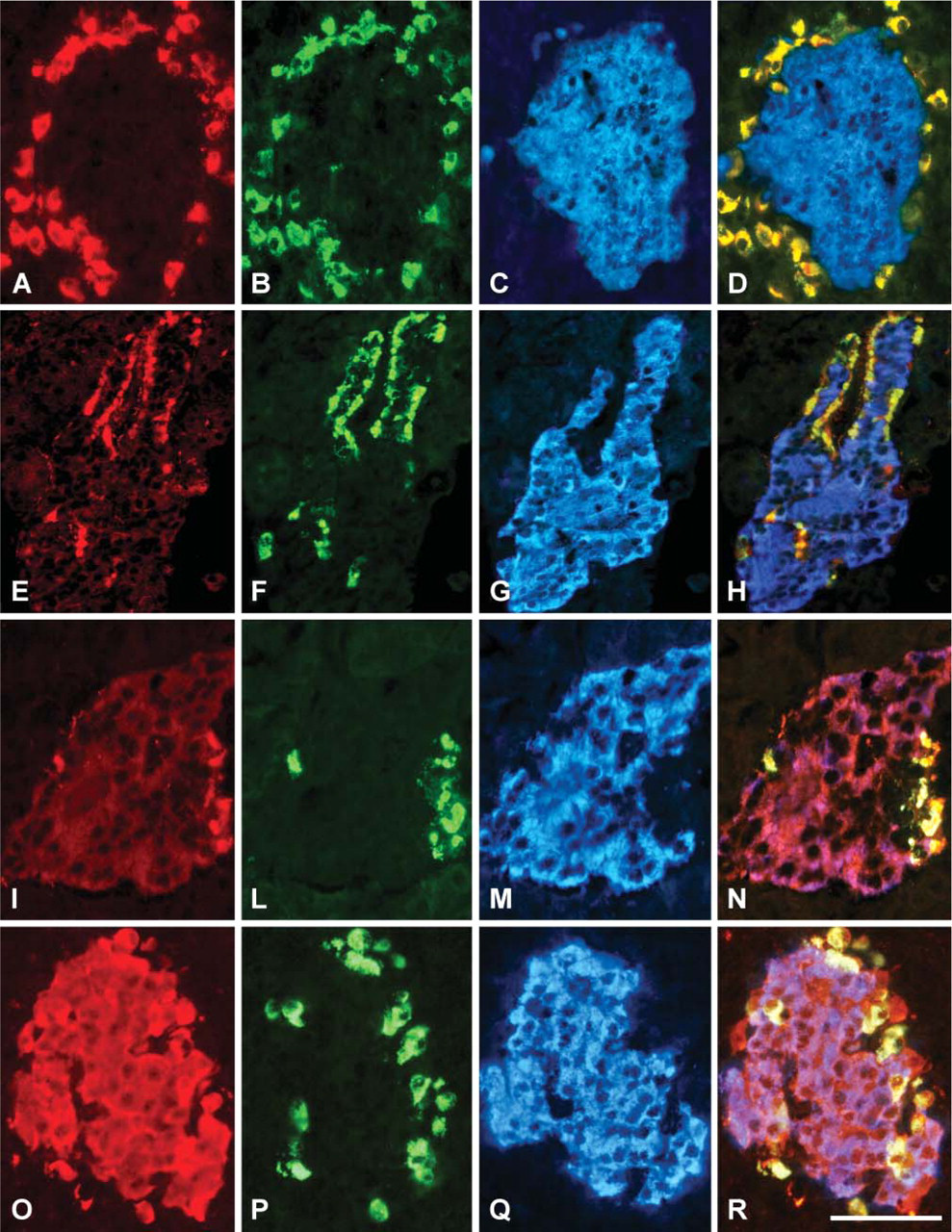
Differential localization shown using VGF antisera in triple staining. (
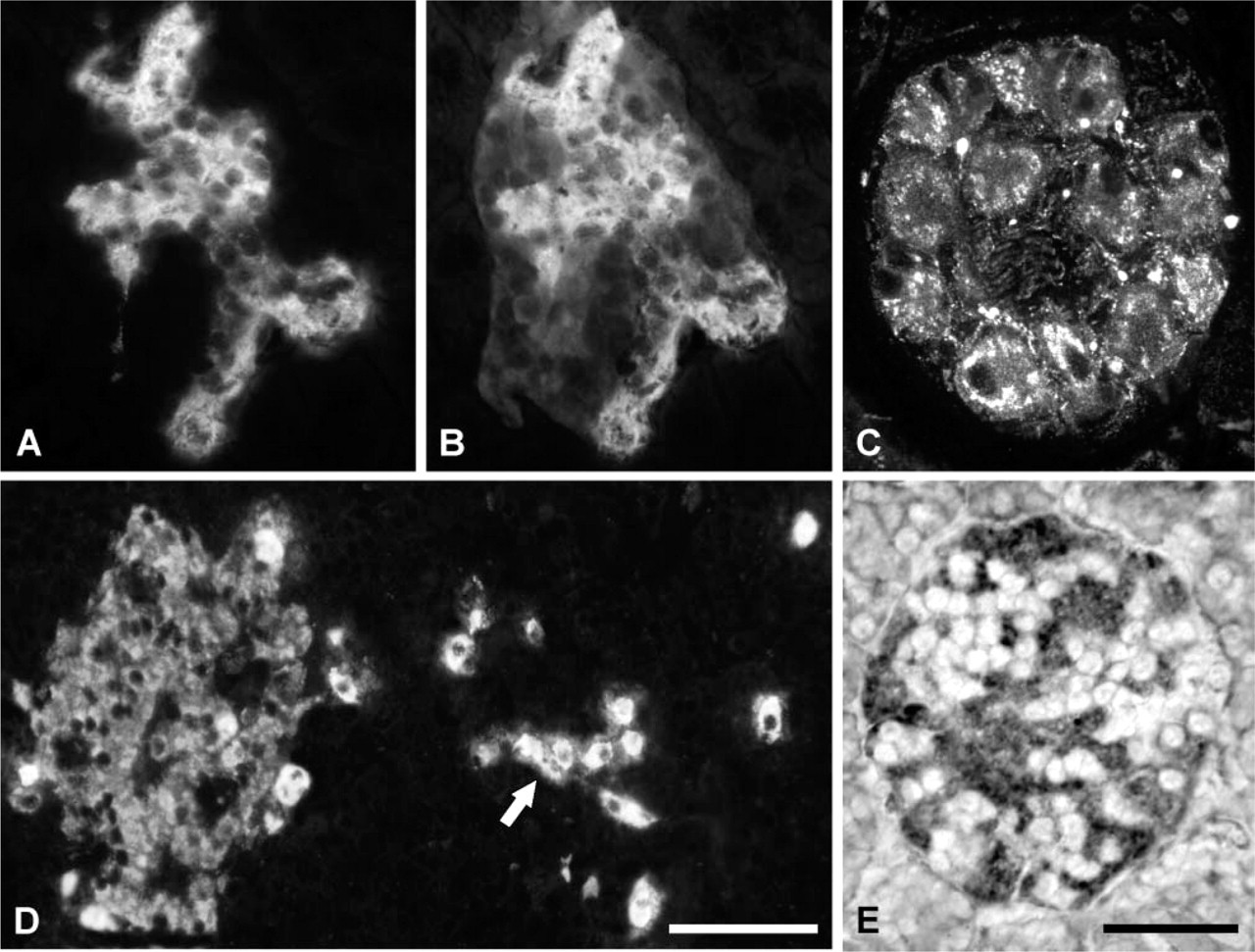
VGF immunofluorescence and in situ hybridization. (
In rat tissue, antisera specific for the rat and human C terminus (respectively, VGF609–617 and VGF607–615) were tested; however, immunoreactivity was found only using the antiserum specific for the rat C-terminus portion. Instead, the human VGF C-terminus immunostaining was found only in swine and bovine tissues.
When using the different VGF antisera, diverse staining patterns became apparent, with no significant differences between the pancreatic areas as well as the tissue species tested.
Selective immunolabeling was found using the VFG556–565 (Figure 2A) and VFG282–291 (not shown) antisera, which brightly decorated peripheral cells of the islets. A similar pattern was shown with human VGF607–615 antiserum (Figure 2E) which, in the ventral region, also labeled endocrine cells inside the islets (Figure 3A). The same antiserum also stained nervous components including fibers and ganglia (Figure 3C). Conversely, virtually all of the rat pancreatic islet cells were weakly immunolabeled with the rat proVGF C-terminus antiserum (not shown). Furthermore, intense labeling of all swine and bovine islets was found with the antiserum raised against the amidate peptide (VGF298–306, Figure 2O), whereas only feeble staining was found in rat using the same antiserum. Finally, the antiserum raised against a fusion protein (VGF443–588) brightly labeled peripheral cells and, even more so, the rest of the islet in a weak manner (Figure 2I). A few axons surrounding the islets were also immunolabeled with the same antiserum (not shown).
Interestingly, in the ventral region, using the VGF443–588 and the human/rat C-terminus antisera, we found a few differences among the species tested. In fact, whereas in rat and bovine these antisera labeled several cells inside the islets (Figures 3A and 3B) using swine tissues, staining was also found in single endocrine cells within the exocrine pancreas (Figure 3D).
VGF antisera vs pancreatic hormones
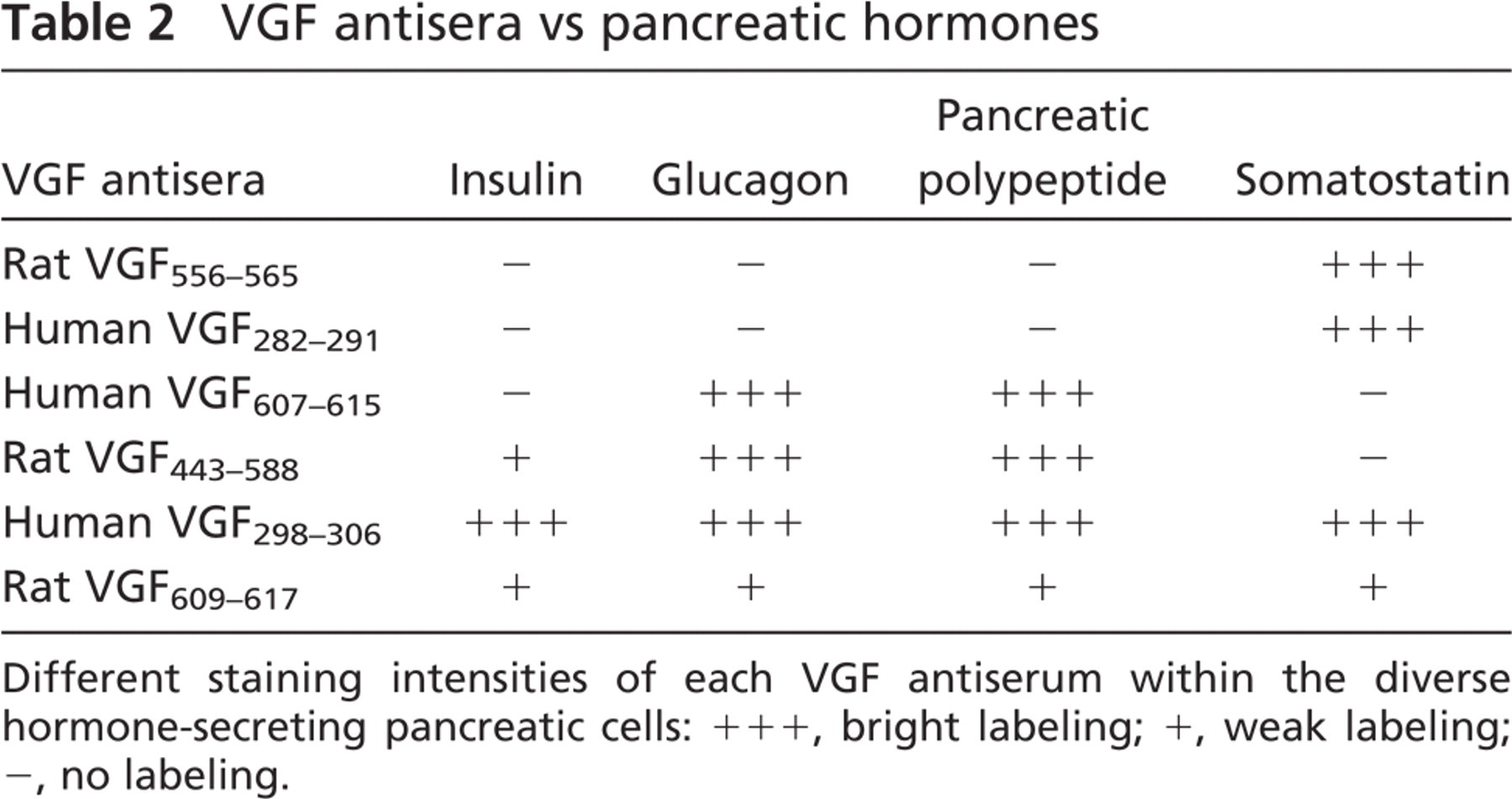
Different staining intensities of each VGF antiserum within the diverse hormone-secreting pancreatic cells: +++, bright labeling; +, weak labeling; −, no labeling.
Double- and triple-immunolabeling experiments confirmed that the above patterns were linked to specific endocrine cell populations (Table 2).
Thus, antisera to the rat VGF556–565 peptide (Figures 2A-2D) and VGF282–291 (not shown) exclusively decorated only somatostatin-secreting cells. The human VGF C-terminus (VGF607–615) antiserum immunolabeled glucagon cells (Figures 2E-2H) and, in the ventral area, pancreatic polypeptide cells (Figures 3A and 3B). It is rather interesting to notice that, in swine tissues, we found the pancreatic polypeptide cells scattered within the exocrine pancreas (Figure 3D), whereas in the other species tested the same cells were found inside the islets, as widely noted. Moreover, the latter cells were stained with the VGF443–588 antiserum (not shown), which was also found labeling insulin and glucagon cells (Figures 2I-2N). Finally, the VGF298–306 (Figures 2O-2R) and the rat proVGF C-terminus peptides (not shown) were localized within all the endocrine cells.
ELISA
Data from chromatography and ELISA assay showed the presence of a high molecular mass form of ~70–90 kDa, compatible with proVGF, with the addition of lower molecular mass fractions (in all species tested).
Regarding the VGF607–615 C-terminus assay, using swine (not shown) and bovine pancreatic extracts (Figure 4A), the consistent high level of the presumptive proVGF within the high molecular mass form was suddenly apparent. Instead, a lower concentration of C-terminus-containing peptides was found within low molecular mass fractions of ~3–4 kDa and 2–3 kDa that were previously identified with, respectively, the peptide V (Liu et al. 1994) and the VGF2 (Trani et al. 2002) (Table 3).
Using rat pancreatic extracts (Figure 5A), comparing with bovine and swine levels, a lower amount of the presumptive proVGF was found within the high molecular mass form. In addition, smaller molecular forms were revealed, including the 3–4 kDa, 2–3 kDa, 15–20 kDa, and 7–8 kDa fractions, which may be compatible with, respectively, the peptide V and VGF2 (Liu et al. 1994; Trani et al. 2002), as well as the VGF20 and 10 bands previously identified in extracts of insulinoma cell line using the same C-terminus antibody (Possenti et al. 1999) (Table 3).
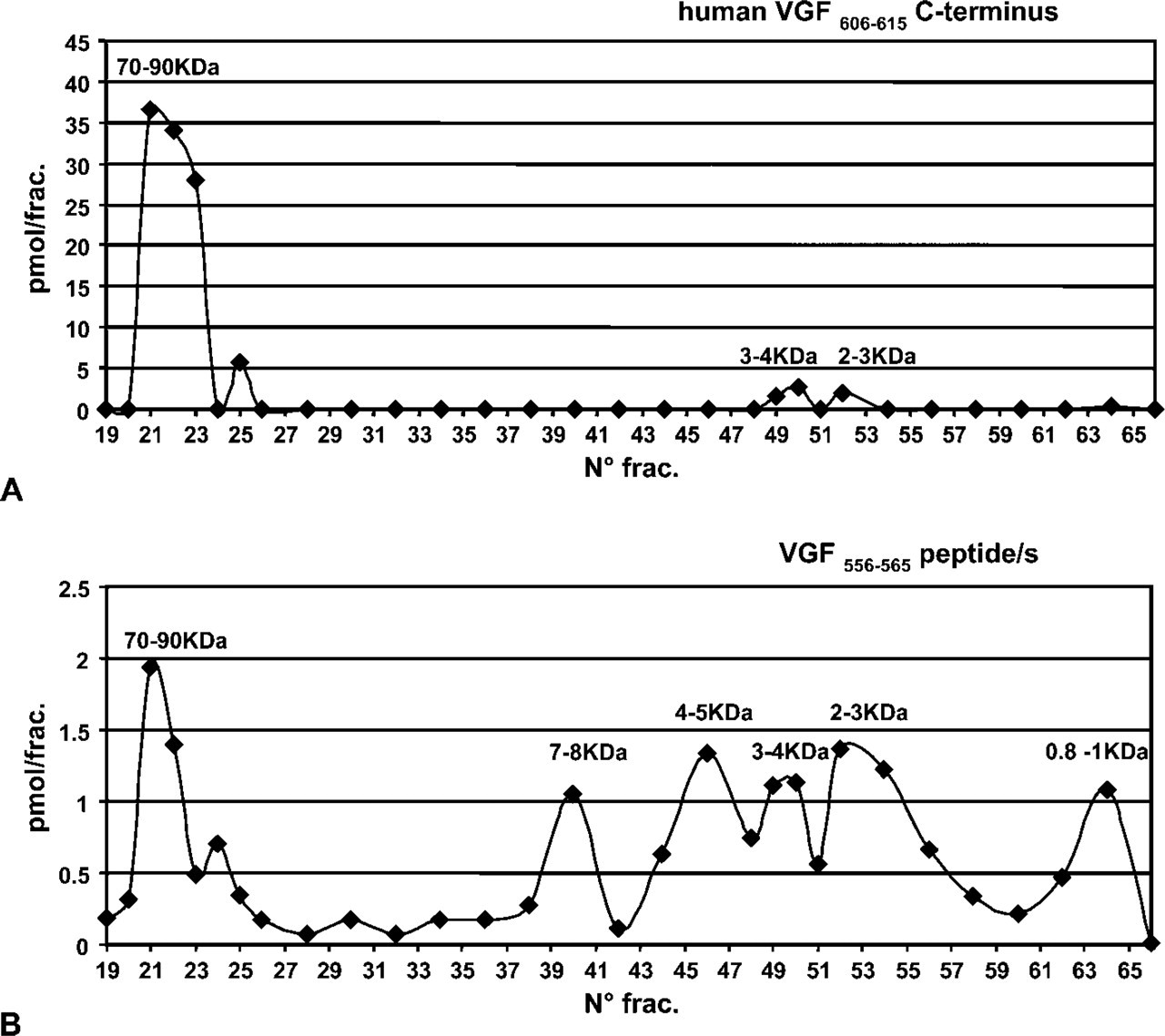
Chromatography and ELISA assay of the human C terminus and the VGF556–565 antibody. (
Calculated mass of expected peptides and the molecular mass fractions (MMF) recognized by the VGF antisera
The rat/human C-terminus antisera recognized the high molecular mass fraction corresponding to the proVGF as well as other small forms compatible with previously identified C-terminus-containing peptides. VGF556–565 recognized fractions of different molecular masses compatible with peptides containing the TLQP-sequence adjacent to the VGF553–555 cleavage site as well the proVGF-containing form. The antibody against the VGF298–306 amidate peptide recognized the proVGF and a fragment compatible with a peptide corresponding to a portion from the proVGF N terminus up to the VGF307–309 cleavage site. Bold type indicates amino acids sequence used for immunization and cleavage sites. r.C-term, rat C terminus; h.C-term, human C terminus.
Using bovine, rat, and swine pancreatic extracts, the antibody VGF556–565 (Figure 4B), other than a small portion of the presumptive proVGF, mainly recognized a number of low molecular mass fractions, compatible with the calculated mass of expected pep-tides resulting from processing at the VGF553–555 “ArgPro-Arg” cleavage site (Table 3), including the rat VGF556–576 peptide (Trani et al. 2002) also noted as TLQP-21 (Bartolomucci et al. 2006).
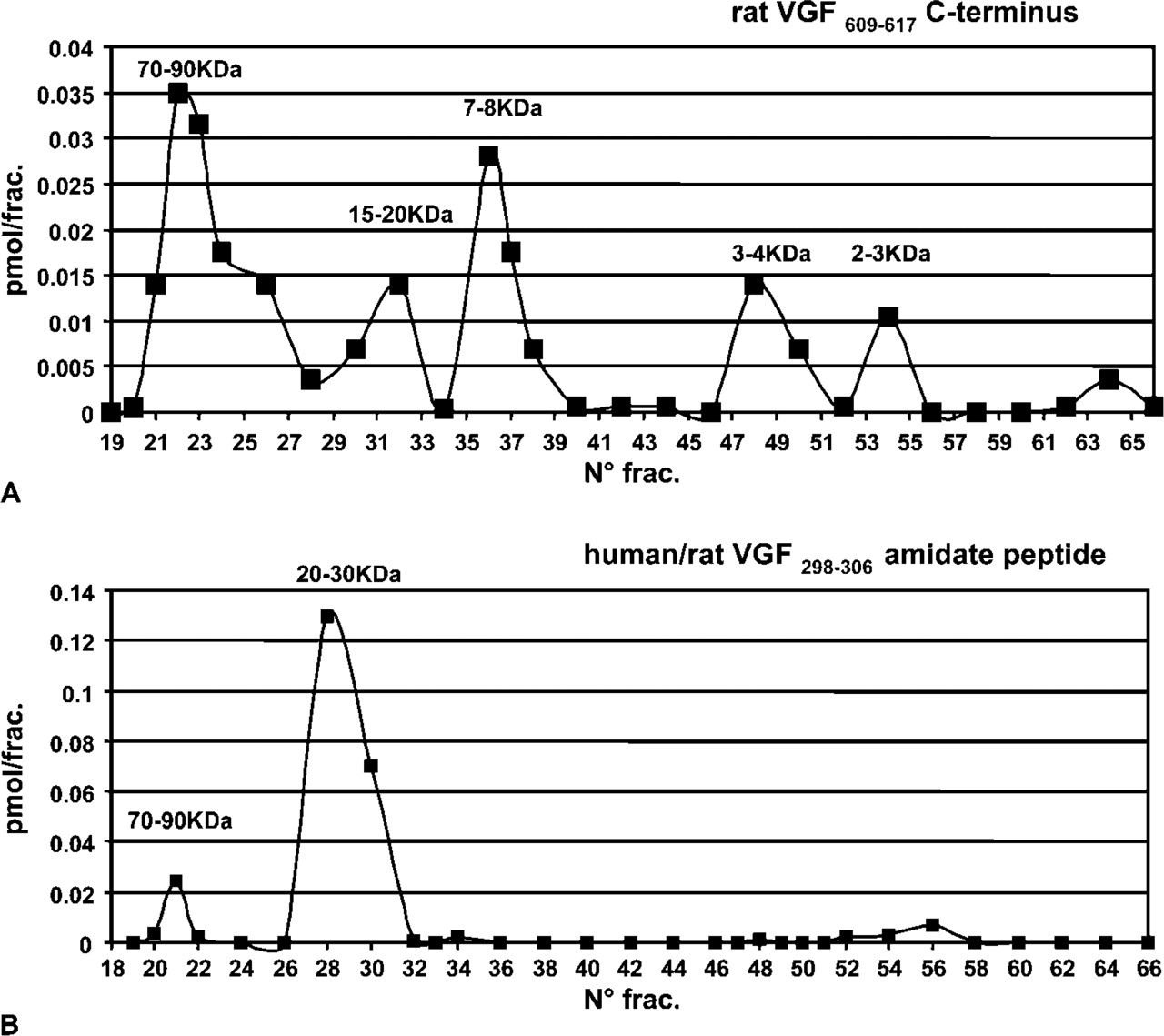
Chromatography and ELISA assay of the rat C terminus and the VGF298–306. (
Finally, the antibody against the VGF298–306 amidate peptide (Figure 5B) selectively recognized a form (20–30 kDa) compatible with the calculated mass, corresponding to a portion from the proVGF N terminus up to the VGF307–309 cleavage site (Table 3). As expected, only a low concentration of the presumptive proVGF was labeled by the same antiserum (Figure 5B). The latter antiserum was not used with rat pancreatic extracts in view of the feeble signal found in immunofluorescence.
Discussion
In the present study we demonstrated the presence of different VGF-related products in the mammalian species as normal bovine, rat, and swine endocrine pancreas. Our study was done using antibodies raised against the presumptive precursor protein as well as specific sequences in connection with proVGF processing sites.
By hybridization in situ, vgf mRNA was brightly revealed in rat, weakly shown in bovine, and not visible in swine pancreatic endocrine cells. Nevertheless, vgf gene has not yet been sequenced in pig and bovine. It seems that there would be a low and near to no affinity of the probe raised against the rat sequence, with bovine and swine tissues, respectively. Additionally, human proVGF C-terminus (VGF607–615) immunoreactivity was not visible in rat, but resulted in bright staining in bovine and swine pancreas. These findings may indicate that, at least in its C-terminal region, the proVGF in bovine and swine may be closely related to the human sequence.
Using bovine and swine pancreatic extracts, our data from chromatography and ELISA assay showed a consistently high level of the presumptive proVGF within the high molecular mass form of 70–90 kDa. Conversely, using rat, the amount of precursor was lower, according to immunofluorescence data showing that all endocrine pancreatic cells were weakly labeled by the VGF609–617 antiserum.
Furthermore, the presumptive human proVGF was selectively found only in glucagon and polypeptide pancreatic cell types, contrary to the expected wide distribution of the precursor. Thus, within the remaining unlabeled cells, the presumptive VGF precursor could be immediately processed and/or released. Indeed, in a female rat, in the anterior pituitary after estrus, a striking loss of VGF immunoreactivity was found in gonadotropes associated with a high level of VGF mRNA in the whole gland (Ferri et al. 1995), suggesting that VGF production may be associated with enhanced release or degradation upon different stimuli (Possenti et al. 1989, 1999).
Multiple immunostaining with antibodies against pancreatic hormones showed that the VGF peptides were well represented within the different hormone cell types with a selective localization. Specificity of the immunostaining obtained using the various VGF antisera was confirmed by absorption controls.
Concerning the VGF443–588 antiserum, previous Western experiments showed an intense labeling in a band of ~90 kDa compatible with the proVGF and, even more so, a weak band of ~20 kDa (Brancia et al. 2005). These findings are in accordance with our immunofluorescence data which, in bovine and swine, showed that the VGF443–588 antiserum brightly labeled the same cells as the human.
C-terminus antiserum and, even more so, additional weak cells, do not contain the precursor (Table 2). Such weak cells were identified by double staining as insulin-secreting cells, which were also labeled by other antisera such as the VGF298–306 and the rat C terminus.
Interestingly, some studies suggest that maturation of VGF is the consequence of proteolytic cleavage by the tissue-specific prohormone convertases PC1/3 and PC2 (Trani et al. 2002). These convertases are the same as those detected in insulinomas and pancreatic islets and are responsible for processing the pro-insulin (Neerman-Arbez et al. 1993). Furthermore, activation of either PKA or PKC, which promotes the secretion of insulin, is also responsible for VGF-derived peptide secretion, suggesting that the mature forms of VGF and insulin may be stored in the same type of vesicles (Wollheim et al. 1996).
The involvement of VGF in the control of circulating glucose and insulin levels is confirmed by recent interesting studies. The latter indicate that VGF deficiency lowered circulating glucose and insulin levels in several murine models of obesity and that VGF mutant mice resisted developing obesity and hyperglycemia in response to several stimuli (Watson et al. 2005).
Using immunofluorescence, the VFG556–565 antiserum was found selectively labeled within somatostatin cells, and a similar localization was also found within the hypothalamus and stomach (Brancia C, et al., unpublished data).
Using pancreatic extracts, we showed that the same antibody recognized a number of low molecular mass fractions including a form corresponding to the rat VGF556–576 peptide, differing from VGF556–565 by 11 amino acids and also noted as TLQP-21 (Bartolomucci et al. 2006).
This peptide seems to specifically elicit dose-dependent contractions on rat stomach fundus (La Corte G, et al., unpublished data).
Interestingly, chronic ICV injection of the TLQP-21 increases energy expenditure and temperature in mice and seems to prevent the increase in body and WAT weight and hormonal changes associated with a high-fat regimen (Bartolomucci et al. 2006).
The presence of some VGF peptides (VGF606–615 and VGF443–588) was found in both endocrine cells and nerve structures. In the pancreas, a number of neuronal markers have been detected in β-cells and insulinoma-derived cell lines (Atouf et al. 1997).
Thus, VGF proteins may be involved in regulating cleavage, stability, and/or secretion of other hormones or neuropeptides. In the anterior pituitary, the selective modulation of VGF peptides seems to be connected with seasonal variations (Brancia et al. 2005).
In conclusion, the different endocrine localization of the VGF peptides in parallel to the wide expression of vgf mRNA that we have shown may suggest an intriguing cell type-specific processing of proVGF, probably involved in the regulation of multifaceted neuroendocrine metabolic mechanisms.
Footnotes
Acknowledgements
This study was supported in part by research grants from the Italian Ministry of Education, Universities and Research, Fondo per gli Investimenti della Ricerca di Base (MIUR FIRB): RBNE013XSJ_002 and RBNE01JKLF_002) and by the Ministry of Health, Italy.
We thank G. Boi, F. Incollu, the COALBE Company, M.C. Mostallino at the National Research Council, Institute of Neuroscience, Cagliari, and M. Collu for tissue samples.
