Abstract
We investigated human thymic tissue by immunohistochemistry and in situ hybridization for the presence of synaptophysin-producing cells. Our results indicate that anti-synaptophysin antibody detected immunoreactive material in nerve fibers around vessels located in major thymic septa, in a relevant number of cortical epithelial cells, and in scattered epithelial cells in the medulla. The epithelial nature of synaptophysin-positive cells was documented by the co-expression of cytokeratins as revealed by double immunofluorescence. In situ hybridization studies revealed the presence of synaptophysin mRNA in cells mainly located in the cortex, the specific fluorescent signals being localized in the cell cytoplasm. Western blot analysis using an affinity-purified polyclonal antibody revealed an immunoreactive band of about 38 kD in the extracts from unfractionated thymic tissue and from epithelial cell-enriched fractions. No staining was observed in isolated thymocytes. The expression of synaptophysin in epithelial cells of the thymic cortex suggests that this protein may be involved in secretory activities related to T-cell maturation.
THYMIC EPITHELIUM is the major component of the thymic microenvironment and has important regulatory influences on T-cell maturation. These influences are mainly mediated through the production of cytokines, neuropeptides, and peptide hormones by the epithelial cells (Dardenne and Savino 1994). Thymic hormones such as thymopoietin, thymulin, thymosin-α1, and thymic humoral factor produced by thymic epithelium are modulators of thymocyte differentiation (Goldstein et al. 1972,1977; Bach et al. 1976; Hadden 1993). In addition, oxytocin, vasopressin, β-endorphin, and some pituitary hormones have been found in animal and human thymus (Geenen et al. 1986; Piantelli et al. 1990; Batanero et al. 1992), although their exact significance is still unknown. The exocytotic activity of epithelial cells is therefore a crucial step in the modulatory function on microenvironment.
The integral membrane protein synaptophysin is one of the major polypeptide components of the small electron-translucent vesicles of neurons and neuroendocrine cells (Leube 1994). The observation that synaptophysin belongs to a gene family whose expression is not restricted to neuronal and neuroendocrine cells suggests a fundamental contribution of these gene products to basic controls of the exocytotic process (Leube 1994; Leube et al. 1994). Because synaptophysin may play a role in the secretory function of thymic cells, we investigated the expression of this protein in the human thymus.
Materials and Methods
Specimens
Fragments of thymic tissue were obtained from 12 patients (seven boys and five girls, 6 months to 7 years old) undergoing open cardiac surgery for congenital malformations. In some experiments, thymocytes and thymic epithelial cell fractions were obtained as previously reported (Wolf and Cohen 1992). Briefly, viable thymocytes were isolated by Ficoll-Hypaque (Pharmacia; Uppsala, Sweden) density centrifugation. Stromal cell cultures were washed daily with fresh RPMI 1640 medium supplemented with 10% FCS to remove nonadherent cells. During the first days of culture, 0.2 mM deoxyguanosine was added to the medium to remove thymocytes, macrophages, and dendritic cells. After about 2 weeks, stromal cells were detached by trypsinizing and after washing with PBS were utilized for Western blot assay (see below).
Immunohistochemical Analysis
Specimens were fixed in Bouin's fixative or phosphate-buffered formalin, pH 7.2. Tissue fixation times were 4 hr and 18 hr for both Bouin's and formalin fixative. Preliminary experiments showed that, in thymic tissue, an antigen unmasking procedure was necessary for optimal synaptophysin immunodetection. For antigen retrieval, sections were microwave-treated in 0.01 M citric acid buffer at pH 6.0 for 10 min and allowed to cool for 20 min. Sections were mounted on slides freshly coated with 3-aminopropyltriethoxy-silane (Sigma; Deisenhofen, Germany), treated with 0.3% H2O2 in methanol for 10 min to block endogenous peroxidase activity, and incubated for 1 hr with an affinity-isolated rabbit anti-human synaptophysin polyclonal antibody (Dako; Glostrup, Denmark) at dilutions ranging from 1:200 to 1:1000 or with a mouse monoclonal anti-synaptophysin antibody SY-38 (Dako) at 1:100 dilution. Consecutive sections were also incubated with a mouse monoclonal antibody to human chromogranin A (DAK-A3 clone; Dako) at 1:100 dilution. Indirect immunostaining was achieved using the ABC (Vector Laboratories; Burlingame, CA) technique (Hsu et al. 1981). Endogenous biotin was saturated by a biotin blocking kit (Vector Laboratories). The peroxidase was developed with a DAB substrate kit (Vector Laboratories). Negative controls were performed using normal rabbit or normal mouse serum and omitting the primary antibodies. As positive controls, normal human pancreas obtained at pancreatectomy was used. Sections were lightly counterstained with hematoxylin. For immunohistochemical double staining, after incubation with polyclonal anti-synaptophysin antibody, sections were incubated with a monoclonal anti-cytokeratin antibody (clones AE1/AE3; Dako). These antibodies were detected by goat anti-mouse tetraethylrhodamine isothiocyanate (Dako) and goat anti-rabbit fluorescein isothiocyanate (Dako) for anti-keratin and anti-synaptophysin antibodies, respectively. Negative controls were performed by omitting either the anti-synaptophysin or the anti-cytokeratin antibody.
In Situ Hybridization
Specimens were immediately fixed in 4% paraformaldehyde in PBS for 4 hr at 4C, embedded in paraffin, and stored at −20C as previously reported (Viaene and Baert 1994). In situ hybridization was carried out as previously reported (Garson et al 1987; Maggiano et al. 1994). Briefly, tissue sections were cut and mounted on glass slides freshly coated with 3-aminopropyltriethoxy-silane. All the buffers and glassware were RNase-free. Dewaxed sections were rinsed in PBS, immersed in 0.2 M HCl for 10 min, and treated with 3.5 μg/ml proteinase K (Sigma) in 100 mM Tris-HCl, 5 mM EDTA, pH 8.0, for 15 min at 37C. After enzyme digestion, the slides were further fixed with 4% paraformaldehyde. After washing in double-strength standard saline citrate (2 × SSC), slides were incubated with RNase-free DNase (100 mg/ml in 2 × SSC) for 60 min at 37C in a humidified box, rinsed twice in 2 × SSC, dehydrated through an ethanol gradient series, and air-dried. As the probe for synaptophysin, the EcoRI insert of the rat synaptophysin cDNA clone pSR5 (a generous gift from Rudolf E. Leube, Joannes Gutenberg-University, Mainz, Germany) was labeled with fluoresceinconjugated 11-dUTP-digoxigenin. Hybridization procedures were carried out as previously detailed (Maggiano et al. 1994). Tissue samples from human pancreas were used as positive controls. Digoxigenin-labeled synaptophysin cDNA signals were inhibited by addition of homologous unlabeled synaptophysin cDNA. After tissue fixation, some sections were pretreated for 60 min with 100 mg/ml RNase A (Sigma) in 2 × SSC. A significant loss of signal after RNase treatment indicated that the probe was hybridizing to RNA. The specimens were observed and digitalized by a fluorescence Zeiss Axioskop (Zeiss; Jena, Germany) equipped with an intensified CCD camera system, series 200 Macintosh configuration (Photometrics; Tucson, AZ).
Western Blot Analysis
Lysates were prepared by homogenizing thymic samples in lysis buffer (0.05 M Tris-HCl, pH 7.4, 1% Triton X-100, 0.15 M NaCl, 5 mM MgCl2, 1 mM phenylmethylsulfonyl fluoride, 1 μg/ml leupeptin, 1 μg/ml aprotinin). Protein concentrations were determined by the Bio-Rad protein assay (Bio-Rad; Munich, Germany). Equal amounts of proteins (20 μg) were mixed with equal volumes of sample buffer (0.05 M Tris-HCl, pH 6.8, 10% 2-mercaptoethanol, 2% SDS, 30% v/v glycerol, 0.025% bromophenol blue, and a few crystals of methyl green), boiled for 5 min, and separated by SDS-PAGE on a 12.5% polyacrylamide gel. Proteins were transferred in transfer buffer (25 mM Tris-HCl, pH 8.3, 192 mM glycine, 20% v/v methanol) onto a 0.45-μm nitrocellulose filter (Schleicher & Schuell; Dassel, Germany) by low-field electroblotting for 24 hr. The filters were incubated with a blocking solution of 4% BSA, 0.1% IGSS quality gelatin (Janssen; Olen, Belgium), 2 mM NaN3, and 150 mM NaCl in 10 mM PBS, pH 7.4. After incubation at 37C for 60 min followed by washing with 0.1% BSA and 2 mM NaN3 in 10 mM PBS, the filters were transferred into bags with anti-synaptophysin polyclonal antibody (Dako) at 1:500 dilution in incubation buffer (0.8% BSA, 0.1% IGSS quality gelatin, 2 mM NaN3, and 1% rabbit serum) and incubated overnight at 4C. After washing, the reaction was developed using the peroxidase-ABC kit. Peroxidase activity was visualized using the DAB substrate kit.
Results
As shown in Figures 1A–1C, the polyclonal anti-synaptophysin antibody detected immunoreactive materials in nerve fibers around vessels located in major thymic septae, in a relevant number of cortical epithelial cells, and in scattered epithelial cells in the medulla.
The majority of immunolabeled cortical epithelial cells were located at the corticomedullary junction and showed cytoplasmic projections. Furthermore, labeled dendritic cells were also detected in the outer cortex. Occasionally, isolated cells with rounded profiles and devoid of projections, containing synaptophysin-immunoreactive materials, were observed in the cortex.
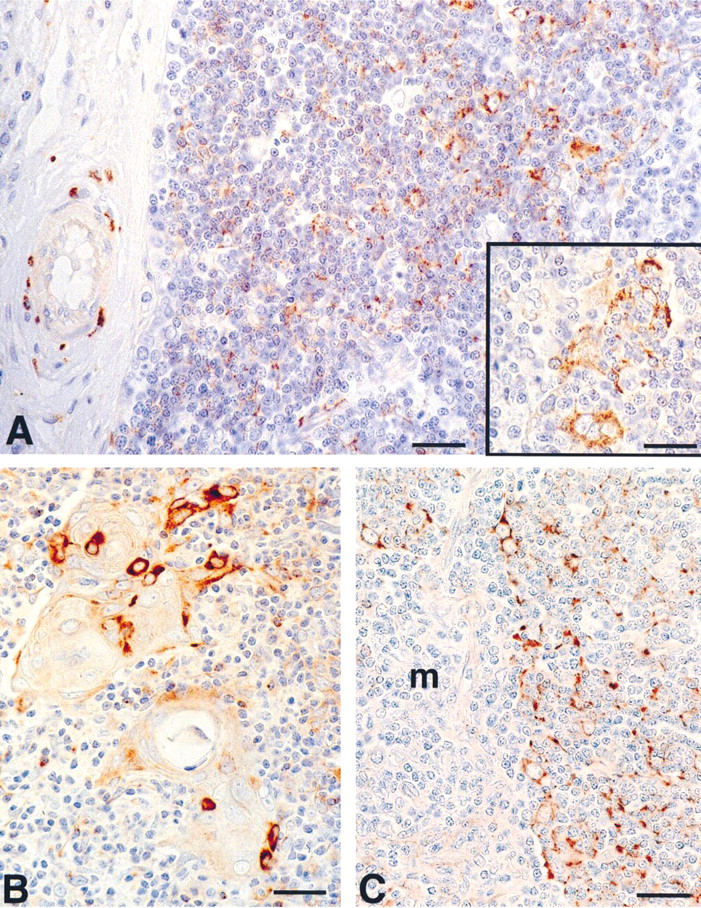
Localization of cells immunoreactive with polyclonal anti-synaptophysin antibody in human thymus. (A) Synaptophysin-positive cells appear in the cortex. Note the synaptophysin-positive perivascular nerve fibers within an interlobular septum. (B) Scattered synaptophysin-positive cells are located in the medulla, mainly adjacent to Hassal's bodies. (C) Synaptophysin-positive cells in the juxtamedullary cortex are arranged to form a cytoreticulum. m, medulla. Bars = 35 μm; inset = 20 μm.
The immunolabeled cells in the medulla appeared preferentially isolated and were located at the periphery of Hassal's corpuscles or scattered in the medulla.
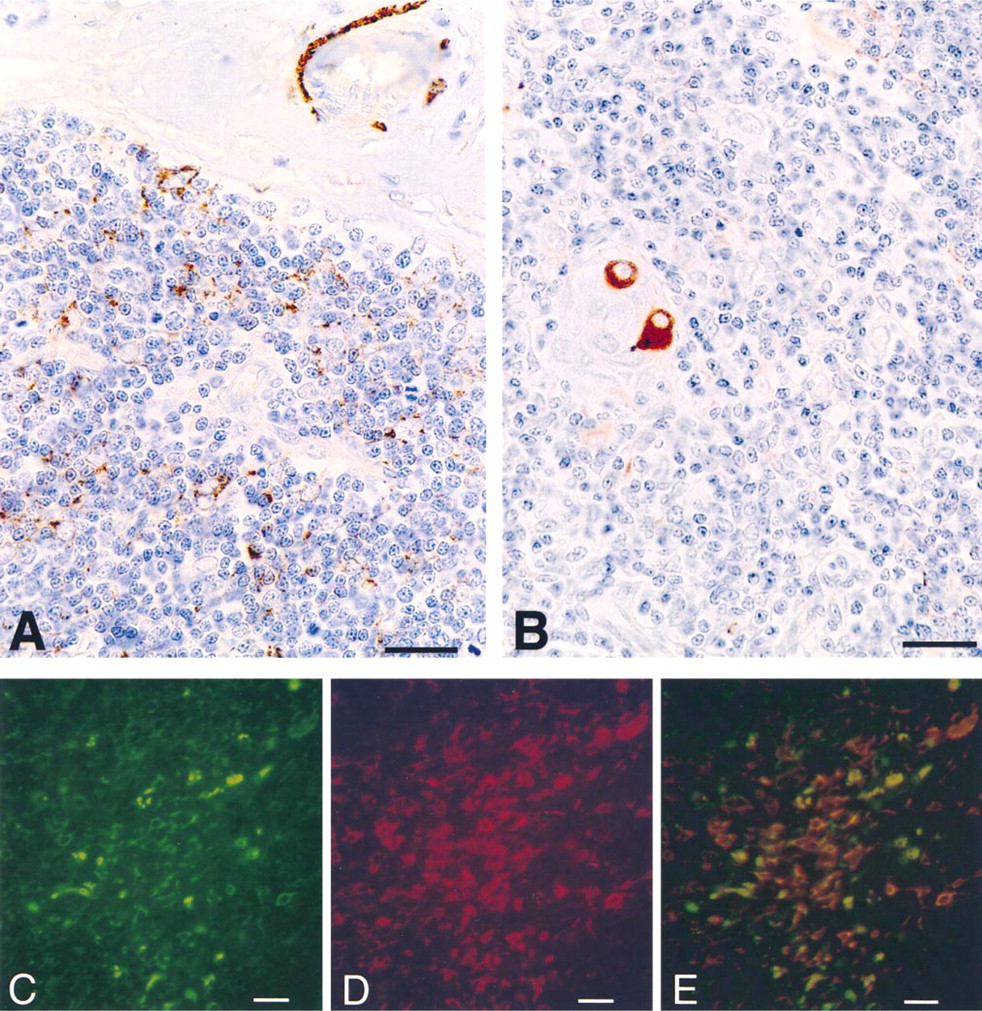
Localization of cells immunoreactive with monoclonal anti-synaptophysin antibody (SY-38) in human thymus. (A) Synaptophysin-positive cells in the cortex and synaptophysin-positive perivascular nerve fibers in an interlobular septum. (B) Immunostained cells at the periphery of a Hassal's body. Bars = 35 μm. (C-E) Double label immunofluorescence showing detection of rabbit polyclonal anti-synaptophysin antibody (C, green fluorescence) and mouse monoclonal anti-cytokeratin antibody (D, red fluorescence) in human thymus. The individual recordings were used to create the image in E, showing the simultaneous detection of synaptophysin and cytokeratins. Bars = 50 μm.
A similar pattern of immunoreactivity was observed after staining with the monoclonal antibody SY-38 (Figures 2A and 2B). However, compared to the polyclonal anti-synaptophysin antibody, the monoclonal antibody stained a minor proportion of cells. To verify whether both polyclonal and monoclonal antibodies share the same specificity of immunoreaction, we used human pancreas as a tissue in which exocrine cells can be compared directly with endocrine cells. As shown in Figure 3, consistent with the reported pattern of synaptophysin expression in normal human pancreas (Wiedenmann et al. 1986), both antibodies immunostained the insular neuroendocrine tissue but not the exocrine portion of the gland. Apart from specificity, we observed that the immunoreactivity with the monoclonal antibody SY-38 faded out after a fixation time longer than 4 hr, according to the findings of Hoog et al. (1988). On the other hand, the polyclonal antibody was also effective in staining thymic tissue after overnight fixation, suggesting that it recognized fixative-resistant synaptophysin epitopes. Independently of the anti-synaptophysin antibody employed, maximal intensity of immunostaining was obtained when the tissue was fixed with Bouin's fixative.
Double immunofluorescence microscopy of synaptophysin (Figure 2C) and cytokeratins (Figure 2D) revealed a considerable degree of co-localization (Figure 2E), suggesting the epithelial nature of synaptophysinexpressing cells.
After staining with the anti-chromogranin antibody, only occasional immunostained cells were observed in the medulla (not shown).
In situ hybridization studies revealed the presence of synaptophysin mRNA in cells located both in cortex and medulla. The specific fluorescent signals were localized in the cell cytoplasm (Figure 4A). The number of synaptophysin mRNA-expressing cells appeared lower than the number of cells containing synaptophysin-like immunoreactive materials. Treatment of sections with RNase resulted in complete loss of fluorescent signal (not shown). As a positive control, we used human pancreas. Consistent with the reported pattern of synaptophysin expression in normal human pancreas (Wiedenmann et al. 1986), only neuroendocrine pancreatic islet cells appeared labeled with the synaptophysin cDNA probe (Figure 4B).
To further characterize the anti-synaptophysin antibody-immunoreactive polypeptide, proteins extracted from total thymic tissue, from isolated thymocytes, and from a thymic fraction enriched in epithelial cells were examined by immunoblotting. As shown in Figure 5, affinity-isolated polyclonal antibody revealed an immunoreactive band of about 38 kD in the extracts from unfractionated thymic tissue and from the epithelial cell-enriched fractions. No staining was observed in the isolated thymocyte fraction. The intensity of staining was strongest in the epithelial cell-enriched fraction.
Discussion
Synaptophysin is one of the major polypeptide components of the membrane vesicles of neurons and neuroendocrine cells (Leube et al. 1994). In this study we documented the presence of synaptophysin in thymic epithelial cells. Apart from nerve fibers, synaptophysin-immunoreactive cells were located mainly in the cortex and, in lesser amounts, in the thymic medulla. The epithelial nature of the synaptophysin-containing cells was confirmed by the co-expression of cytokeratin. Moreover, the presence of synaptophysin in the epithelial component of human thymus was further documented by Western blot analysis on thymic isolated cell fractions, showing that a 38-kD immunoreactive band was present in the epithelial cell but not in thymocyte component.
Synaptophysin is a member of a growing multigene family. Recently, it has been demonstrated that the pantophysin gene is closely related to that of synaptophysin but that its mRNA and protein are ubiquitously expressed in tissues and cultured cells (Leube 1994; Haass et al. 1996). Moreover, expression of the individual family members is not mutually exclusive, as shown by the observation that pantophysin and synaptophysin can co-localize in the same vesicle (Haass et al. 1996), suggesting overlapping functions of the different family members. This possibility is also supported by the lack of dramatic defects in pantophysinexpressing synaptophysin-deficient mice (Eshkind and Leube 1995). The above observations suggest that pantophysin and synaptophysin are co-expressed in human thymus. Our results did not concern the possibility that pantophysin is present in human thymus. However, they indicate that epithelial thymic cells express synaptophysin. This possibility is supported by the following findings. First, the immunostaining with SY38 monoclonal antibody is specific for synaptophysin because this antibody recognizes the carboxy terminus of synaptophysin (Knaus and Betz 1990), which lacks the pantophysin molecule (Haass et al. 1996). Furthermore, the specificity for synaptophysin of both the monoclonal (SY-38) and the polyclonal antibody was confirmed by the observation that the pattern of staining of human pancreas was consistent with the reported localization of synaptophysin, i.e., neuroendocrine islet cells were labeled (Wiedenmann et al. 1986), whereas the pantophysin-expressing exocrine tissue (Haass et al. 1996) was unlabeled by these antibodies. Moreover, Western blot analysis revealed the presence of an immunoreactive band at approximately the expected molecular size of synaptophysin (38 kD), whereas no immunolabeling was detectable in the molecular size region of pantophysin (#28.5 kD). Finally, in situ hybridization analysis with a cDNA probe specific for synaptophysin mRNA clearly showed that thymic cells contained this mRNA species.
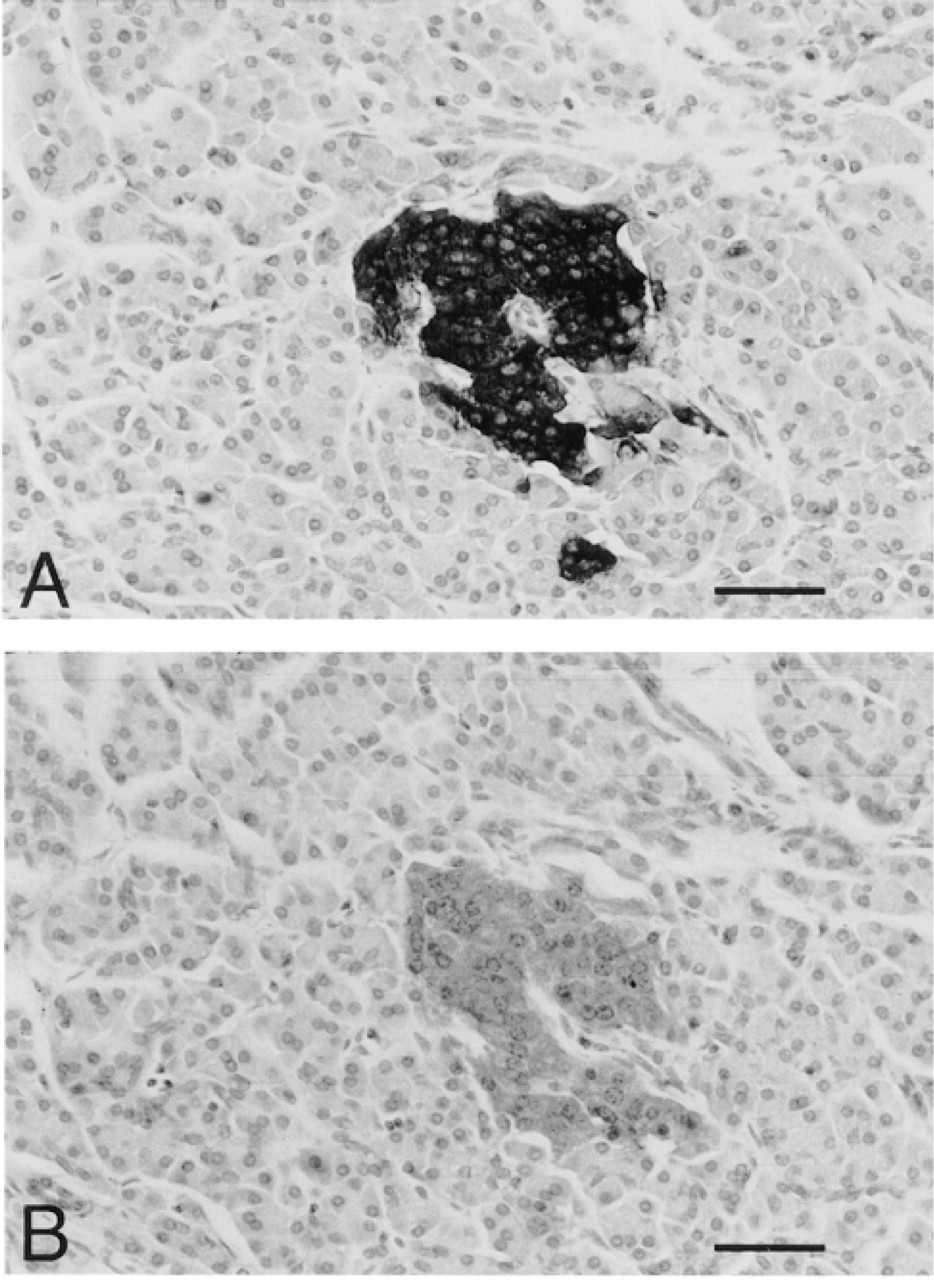
Consecutive sections of human pancreatic tissue showing the immunostaining with polyclonal (A) and monoclonal SY-38 (B) anti-synaptophysin antibodies. The immunolabeling is confined to the neuroendocrine insular cells; the exocrine acinar tissue is negative. Bars = 60 μm.
Synaptophysin is a well-accepted neuroendocrine cell marker (Gould et al. 1986). Neuroendocrine cells are presently recognized as a minor but constant component of the normal thymus (Suster and Rosai 1990). Histochemical and immunohistochemical studies have documented the presence of neuroendocrine cells in the medulla of human and animal thymus and ultra-structural investigations have found dense-core granule-containing cells in this location (Ciaccio 1942; Frazier 1973; Hakanson et al. 1974). More recently, it has been reported that epithelial cells containing both neurohypophysial (Geenen et al. 1986; Piantelli et al. 1990) and some adenohypophysial hormones (Batanero et al. 1992; Maggiano et al. 1994) are located mainly in the thymic cortex. Interestingly, the thymic nurse cells also express several neuropeptides that may reflect a neuroendocrine origin (Geenen et al. 1988).
Here, we observed that synaptophysin and chromogranin A are expressed by cells located in the cortex and the medulla of the thymus, respectively. The localization of synaptophysin and chromogranin A in different thymic cell types is consistent with the observation that neuroendocrine cell markers are independently expressed by neuroendocrine cells belonging to different normal and neoplastic tissues (Jensen et al. 1990; Loy et al. 1995; Senden et al. 1997), probably reflecting nonoverlapping functions of these substances in different neuroendocrine cell types (Gould et al. 1986).
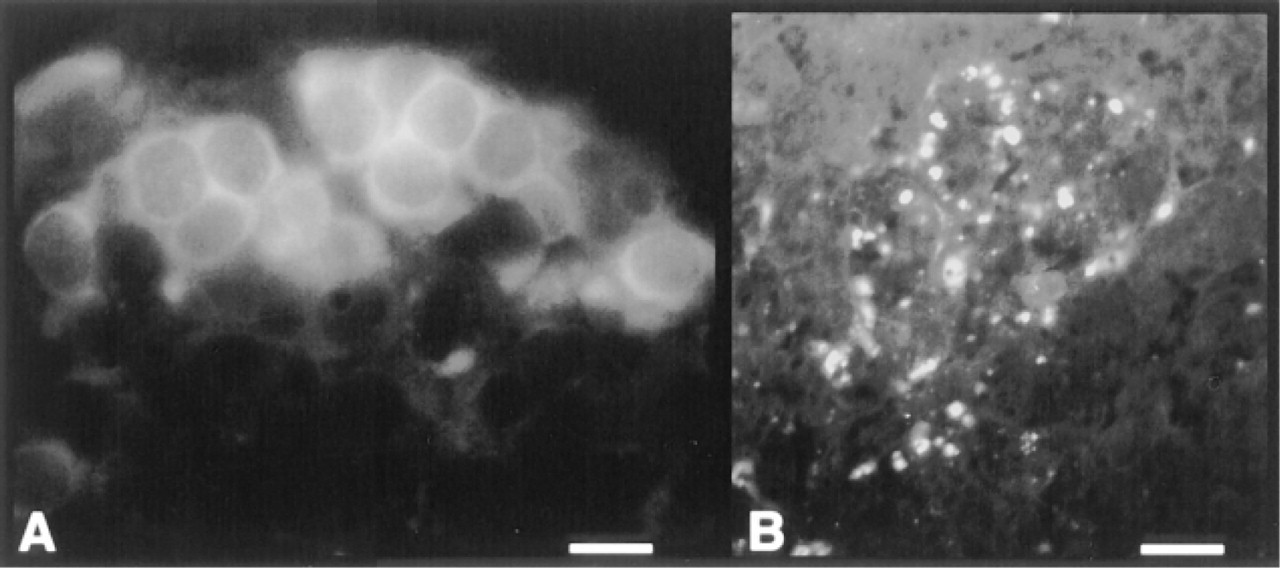
Localization of synaptophysin mRNA by in situ hybridization. (A) A cluster of cells with cytoplasmic fluorescence in human thymic cortex. Bar = 15 μm. (B) Labeled cells in a human pancreatic insula (positive control). Bar = 60 μm.

Western blot probed with an anti-synaptophysin polyclonal antibody, showing a signal at about 38 kD in Lane 1 (unfractionated thymic tissue) and Lane 3 (enriched epithelial cell fraction). No staining is present in Lane 2 (isolated thymocytes).
The synaptophysin-immunoreactive cells located in the cortex are probably engaged in secretory activities that are relevant for early events of T-cell maturation. In this context, it is interesting that synaptophysincontaining cells display a cortical distribution pattern very similar to that of oxytocin- and vasopressin-containing cells, which are engaged in particular neuroendocrine-immune interactions through a cryptocrine cell-cell signaling pathway (Martens et al. 1996).
Footnotes
Acknowledgements
Supported in part by a grant from M.U.R.S.T.
We thank Prof Rudolf Leube for the generous gift of the rat synaptophysin cDNA clone pSR5 and for helpful suggestions.
