Abstract
Adenocarcinomas are malignant tumors with glandular growth and/or supposed intracellular mucin as identified by periodic acid-Schiff (PAS) positivity. Gastric signet ring cell carcinomas are classified as diffuse type. A proportion of diffuse-type adenocarcinomas have previously been suggested to be of neuroendocrine origin. In the present study we examined gastric signet ring cell carcinomas for neuroendocrine differentiation. Of 11 gastric signet ring cell carcinomas, 8 contained areas with PAS-positive signet ring cells that also were immunoreactive for one or several neuroendocrine markers: synaptophysin, chromogranin A, and histidine decarboxylase, the latter an enterochromaffin-like (ECL) cell marker. Whereas PAS positivity was located in the central cytoplasm, neuroendocrine immunoreactivity was often located as a rim surrounding an otherwise non-immunoreactive cytoplasm, presumed to represent the area with PAS-positive material. These findings indicate that signet ring cell carcinomas could be of neuroendocrine origin. We propose that signet ring cell carcinomas develop by gradual dedifferentiation from ECL cells via signet ring cells with neuroendocrine immunoreactivity toward signet ring cells where the cytoplasm mainly consists of PAS-positive material. This finding could have implications for the classification and understanding of gastric carcinogenesis.
Keywords
A
Materials and Methods
Eleven gastric carcinomas located at the oxyntic mucosa containing signet ring cells were selected from our patient files. Two of these were obtained from different parts of the stomach from the same patient (case 4). Four-μm-thick serial sections were cut from formalin-fixed, paraffin-embedded tissue. Signet ring cells were diagnosed in sections stained with hematoxylin and eosin (HE). After deparaffinization, sections for immunohistochemical labeling were immersed in 3% hydrogen peroxide to block endogenous peroxidase activity. Antigen retrieval before immunolabeling was achieved by boiling the sections in 10 mM Tris/EDTA-buffer, pH 9.0 [synaptophysin and histidine decarboxylase (HDC)] or 10 mM citrate buffer, pH 6.0 [chromogranin A (CgA), somatostatin, and 5-hydroxytryptamine (5-HT)] for 15 min. The sections were incubated with antibody against synaptophysin (1:200, Code A010; Dakocytomation, Glostrup, Denmark) and HDC (Code B260-1, 1:15,000; Eurodiagnostica, Malmo, Sweden) for 1 hr at room temperature, and CgA (1:2000, Code MO869; Dakocytomation), somatostatin (Code A0566, 1:200; Dako Corporation, Carpinteria, CA), and 5-HT (Code AB16007, 1:175; Abcam Ltd, Cambridge, UK) for 18 hr at 4C. Tyramide signal amplification was used to increase sensitivity of CgA labeling as described previously (Qvigstad et al. 2002).
Neuroendocrine expression in gastric carcinoma cells with signet ring phenotype

PAS, periodic-acid Schiff; CgA, chromogranin A; HDC, histidine decarboxylase.
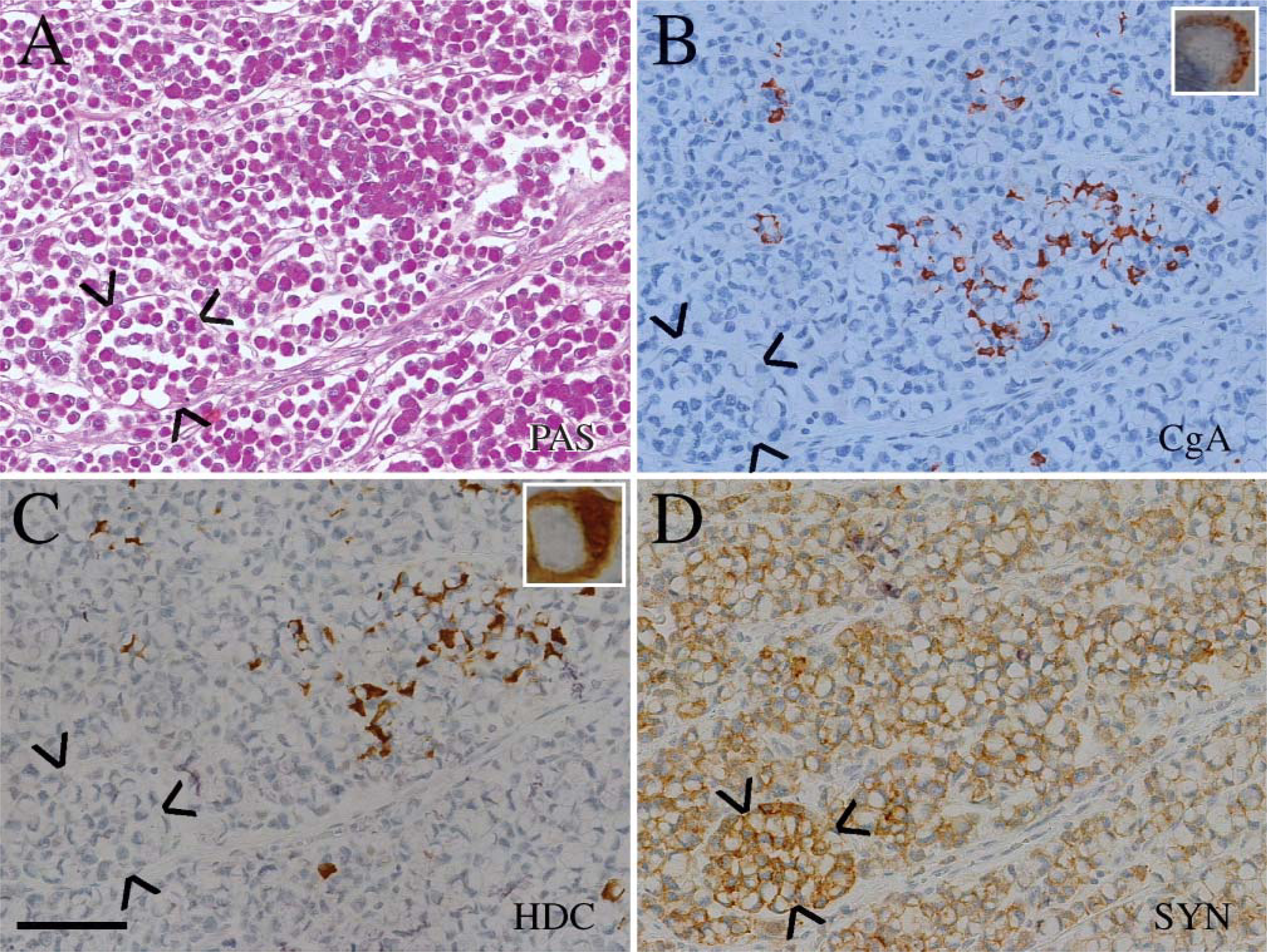
Serial sections of periodic-acid Schiff (PAS)-positive signet ring cells
Antigen-antibody complexes were visualized using the Envision-HRP kit (K5007, Dakocytomation) and AEC (SK4200; Vector Laboratories, Burlingame, CA) or DAB+ (K5007, Dakocytomation). Finally, the sections were counterstained with hematoxylin.
PAS staining was performed with a commercial kit (395B; Sigma, St Louis, MO). To confirm localization to the oxyntic mucosa, samples were examined with antibodies to H+K+ ATPase (Code MA3-923; Affinity Bioreagents, Golden, CO) and HE staining to verify the presence of parietal cells in the mucosa adjacent to the tumor. The study was approved by the regional Ethical Committee.
Results
Localization of the Tumors in the Gastric Mucosa
Of the 11 tumors, 9 were surrounded by a mucosa containing H+K+ATPase-positive cells, parietal cells, demonstrating that they originated from the oxyntic mucosa. Two of the tumors, both located at the cardiac region, were negative for H+K+ATPase (Table 1).
Neuroendocrine Markers
By definitions, all tumors contained neoplastic cells with an eccentric nucleus and central accumulation of PAS-positive material (Table 1). In 8/11 tumors, a subset of signet ring cells was positive for a neuroendocrine marker. In 4/11 cases, the signet ring cells were immunoreactive both for synaptophysin and CgA (Table 1). The three tumors with HDC immunoreactive tumor cells belong to these four tumors (Table 1; Figure 1 and Figure 2). Three of the tumors contained signet ring cells that were positive for synaptophysin but negative for CgA (Figure 3 and Figure 4), whereas only one tumor had signet ring cells positive for CgA but negative for synaptophysin (Table 1). In three of the tumors the signet ring cells were negative both for synaptophysin and CgA (Table 1).
In some of the tumors, a subset of the PAS-positive cells was also positive for the general neuroendocrine marker synaptophysin and CgA as well as for the ECL cell marker HDC (Figure 1 and Figure 2), whereas in other tumors only a minor proportion of PAS-positive cells were immunoreactive for these markers (Figure 5). Synaptophysin and CgA immunoreactivity were often localized to the perinuclear area or in a rim of cytoplasm between the plasma membrane and PAS-positive vacuole (Figure 1 and Figure 5), but a granular distribution of synaptophysin was also observed (Figure 3 and Figure 4).
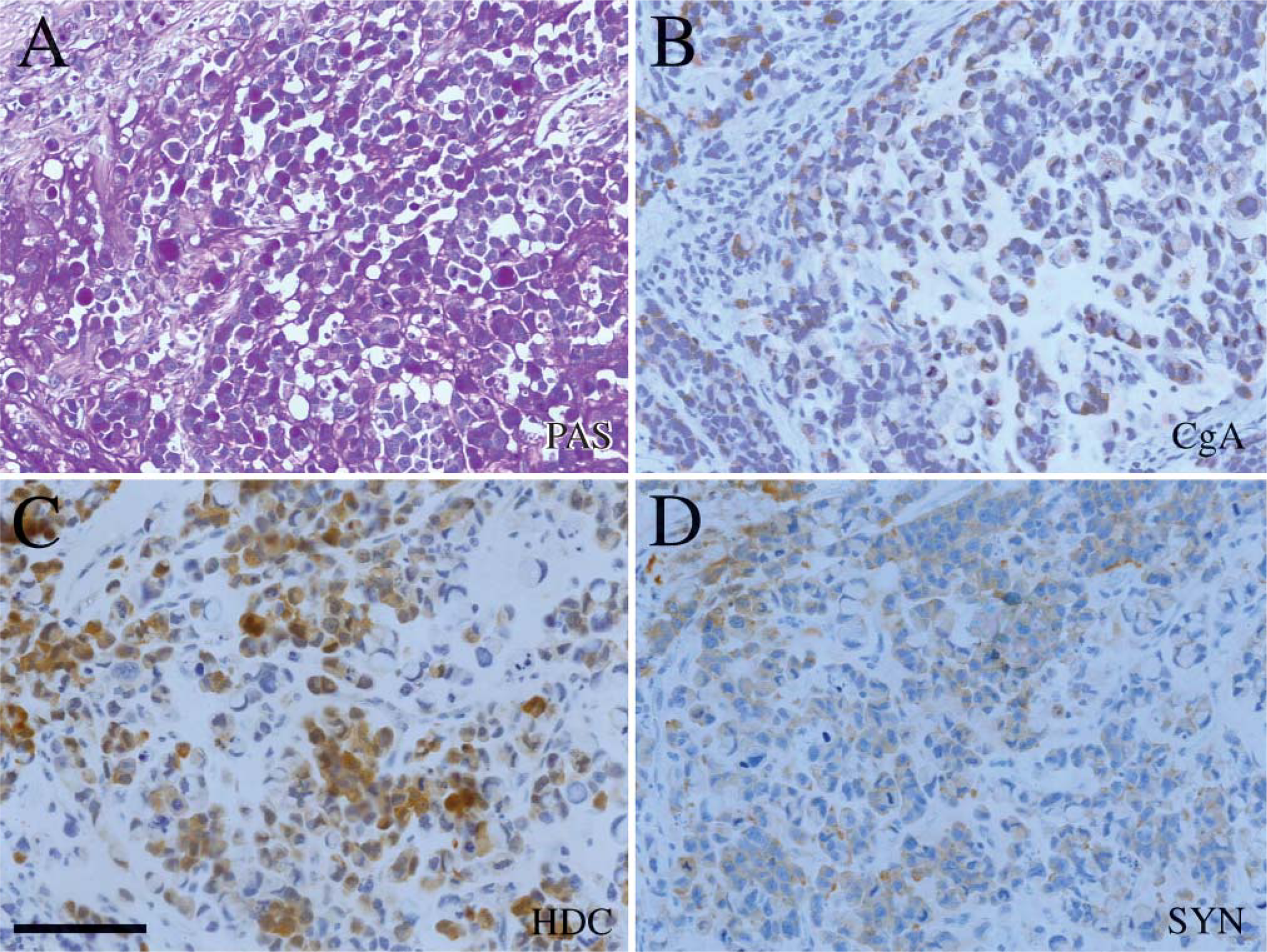
Serial sections of PAS-positive signet ring cells
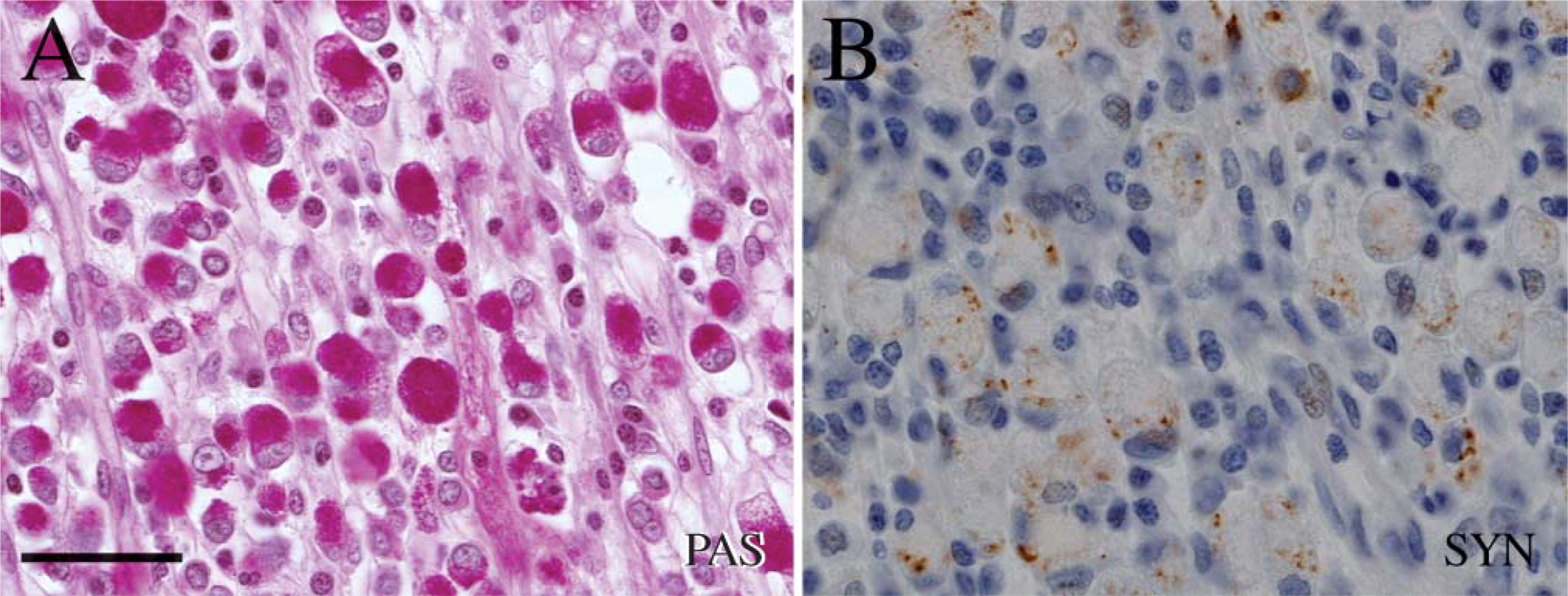
Serial sections of PAS-positive signet ring cells
The amount of PAS-positive material seemed to vary in the signet ring cells between tumors, but also within the tumor (Figure 4), and could appear as PAS-positive granula.
None of the tumors was positive for somatostatin or 5-HT.
Discussion
This study demonstrates that in 8/11 diffuse gastric cancers with signet ring cells, a proportion of PAS-positive signet ring cells express one or more neuroendocrine markers, a finding previously reported to be rare. Previously, two cases of gastric cancer with neuroendocrine differentiation in neutral and acid mucin-negative signet ring cells have been reported (Morii et al. 1999; Sugihara et al. 2004), and a few case reports have described mucin-positive gastric signet ring cells with neuroendocrine markers (Tahara et al. 1975; Prade et al. 1982).
The PAS-positive material has been classified histo-chemically as well as immunohistochemically and related to type of mucins found in normal gastrointestinal cells and found to vary among different tumors (Akamatsu and Katsuyama 1990). Type of glycoproteins in the tumor cells may change with time (Yamachika et al. 1997; Bamba et al. 2001), and hybrid expression may also occur (Yamachika et al. 1997). PAS-positive material in signet ring cancer cells is not a specific marker for mucin and therefore not for exocrine cells either. On the other hand, CgA and synaptophysin are markers specific for cells of neurological and neuroendocrine origin (Wiedenmann et al. 1986; Bordi et al. 1988). Their presence therefore suggests that signet ring cancer cells originate from neuroendocrine cells. The occurrence of HDC in some of these tumor cells further indicates that these cancer cells are derived from ECL cells, the most prevalent neuroendocrine cells in the oxyntic mucosa (Simonsson et al. 1988). Among epithelial cells in the oxyntic mucosa, only ECL cells express HDC, which is specific for that cell type (Rubin and Schwartz 1979). PAS and AB positivity is based on the presence of glycosylated proteins and thus not only mucins. It is known that many neuroendocrine cells produce glycohormones that also may stain positive with these methods (Karsten 1960). When stimulated by gastrin, the ECL cell produces human chorionic gonadotropin (hCG) which is a glycohormone (Bordi et al. 1998). hCG may show positivity with AB and possibly PAS (Cefis et al. 1983). We have no indications that the patients at the time of operation were hypergastrinemic, but it is conceivable that malignant transformation could induce similar changes in the ECL cell as seen after continuous gastrin overstimulation. The occurrence of varying PAS positivity within the tumor could be tumor cells at different stages of dedifferentiation.
We have accordingly shown that the greater proportion of signet ring cancer cells, a cell type expressing nonspecific glycoproteins, express specific general neuroendocrine markers, indicating a neuroendocrine origin, and that at least a proportion of these tumors are derived from ECL cells based on HDC positivity.
Signet ring cell carcinomas of the stomach arise in a gastric mucosa not affected by intestinal metaplasia (Sugano et al. 1982). The prevalent theory to date has been that these carcinomas, like other gastric carcinomas, originate from stem cells in the glandular neck region (Hattori and Fujita 1976). There are reports describing that signet ring cell carcinomas are prevalent in the fundic region of the stomach (Kim et al. 1994). Most of these carcinomas, therefore, originate from oxyntic mucosa where the ECL cell is the most prevalent neuroendocrine cell, and its proliferation is regulated by gastrin (Simonsson et al. 1988; Waldum et al. 1991b). Hypergastrinemia regularly induces ECL cell hyperplasia by increased self-replication (Ryberg et al. 1990) and when prolonged, ECL cell carcinoids (Havu 1986; Sjoblom et al. 1988; Cadiot et al. 1995). Chronic hypergastrinemia is also associated with gastric carcinomas (Sipponen et al. 1985), and when we examined human gastric carcinomas occurring in hypergastrinemic individuals we found that the larger proportion of these tumors actually were derived from ECL cells (Waldum et al. 1998a; Qvigstad et al. 2002). The finding that no signet ring cells contain immunoreactivity against somatostatin and 5-HT further strengthens the ECL cell origin of these neuroendocrine-derived malignant cells.
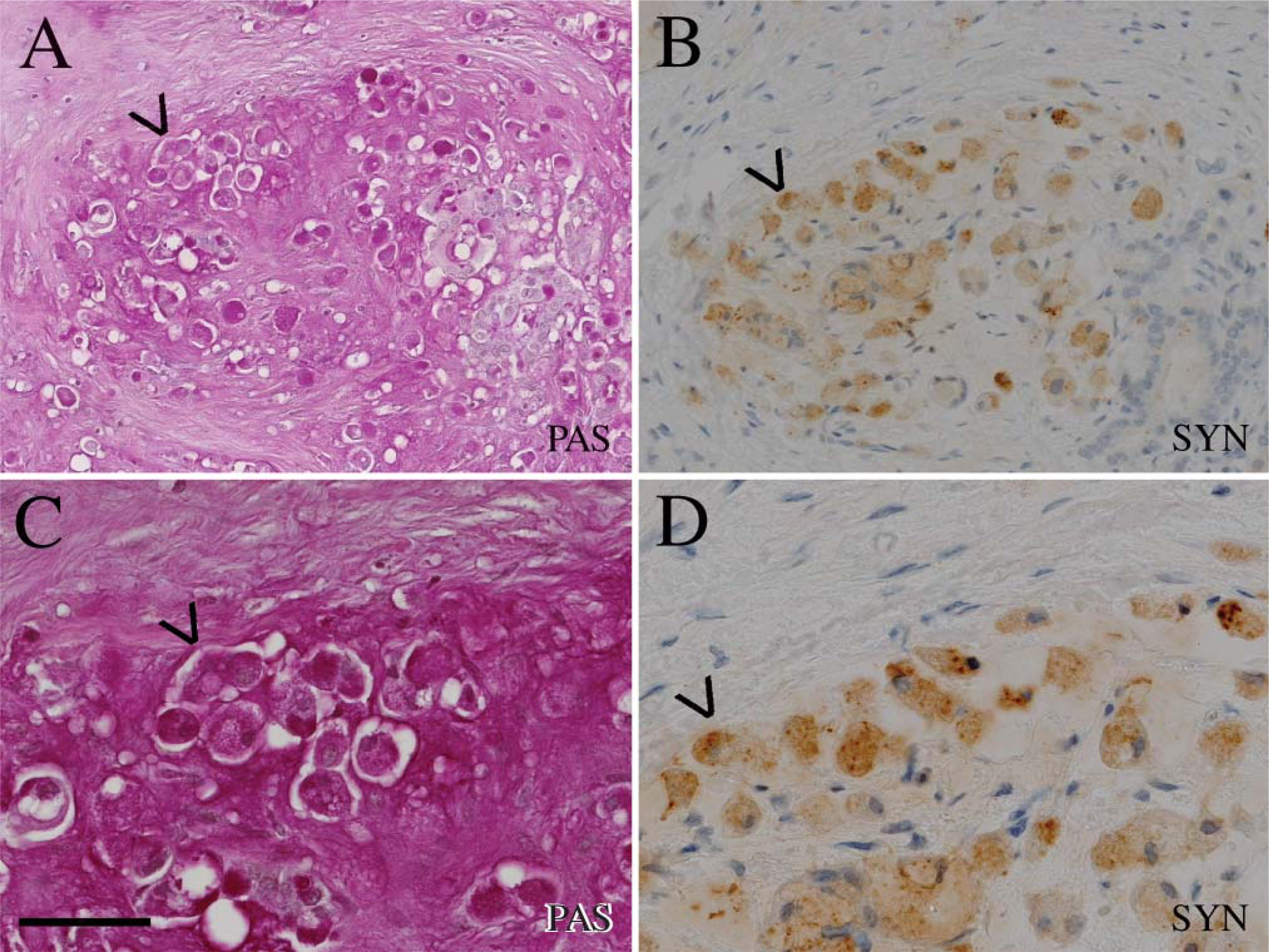
Serial sections of PAS-positive signet ring cells
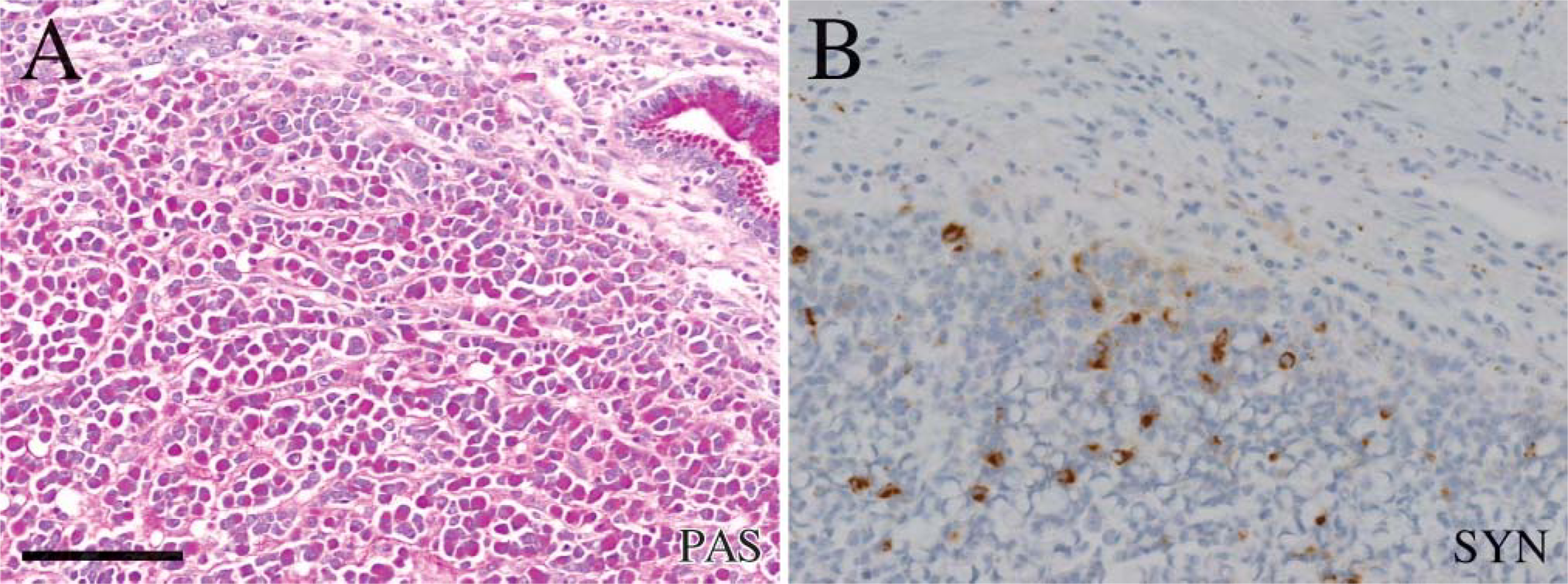
Serial sections of PAS-positive signet ring cells in (A) in a carcinoma (case 8) simultaneously positive for synaptophysin (B). Bar=100μm
To conclude, our study shows that an important proportion of gastric cancers with signet ring cells occurring in the oxyntic mucosa are of neuroendocrine origin. At least a portion of these seem to be derived from the ECL cells.
