Abstract
Pre-embedding immunocytochemistry for the active form of glutamate decarboxylase (GAD67) and postembedding staining for γ-aminobutyric acid (GABA) were compared as markers for central GABAergic terminals in the phrenic motor nucleus, in which phrenic motor neurons had been retrogradely labeled with cholera toxin B-horseradish peroxidase. Nerve terminals with or without GAD67 immunoreactivity were identified in one ultrathin section. GABA was localized with immunogold in an adjacent section after etching and bleaching. GABA labeling density was assessed over 519 GAD67-positive and GAD67-negative nerve terminals in the phrenic motor nucleus. Frequency histograms showed that statistically higher densities of gold particles occurred over most GAD67-positive terminals. However, some GAD67-negative terminals also showed high densities of gold particles, and some GAD67-positive terminals showed low densities. Preabsorption of the anti-GABA antibody with a GABA-protein conjugate, but not with other amino acid-protein conjugates, significantly reduced gold labeling over both GAD67-positive and GAD67-negative terminals. These results show that the presence of GAD67 immunoreactivity correlates strongly with high densities of immunogold labeling for GABA in nerve terminals in the phrenic motor nucleus. Preabsorption controls indicate that authentic GABA was localized in the postembedding labeling procedure. Only a small proportion of intensely GABAimmunoreactive terminals lack GAD67, suggesting that both GAD67 and GABA are reliable markers of GABAergic nerve terminals.
GABAergic nerve terminals throughout the central nervous system have been identified using either preor postembedding immunogold methods to localize γ-aminobutyric acid (GABA), or pre-embedding immunocytochemistry to visualize glutamate decarboxylase (GAD), the enzyme required for synthesis of GABA from
Using similarly processed tissue in this study, we determined whether GAD67-containing nerve terminals labeled by pre-embedding immunocytochemistry showed high densities of gold particles after postembedding immunogold staining for GABA in adjacent etched and bleached ultrathin sections.
Materials and Methods
Retrograde Tracing
The right hemidiaphragms of three male Wistar-Kyoto rats (250-350 g) were injected with cholera toxin B-subunit conjugated to horseradish peroxidase (CT-HRP; List Biological Laboratories, Campbell, CA) (Murphy et al. 1996).
Tissue Processing
After 2-3 days, the rats were anesthetized with sodium pentobarbital (80 mg/kg ip) and perfused with phosphate-buffered 2.5% glutaraldehyde. Cervical spinal cords were divided in two, postfixed for 60-90 min, and cut parasagitally at 50μm on a Vibratome. The sections were washed in 50% ethanol for 45 min (Llewellyn-Smith and Minson 1992). Retrogradely transported CT-HRP was visualized with a tetramethylbenzidine-tungstate (TMBT) reaction, which was subsequently stabilized with cobalt-diaminobenzidine (Llewellyn-Smith et al. 1993).
Sections were incubated in the following reagents: 10% nonimmune horse serum (NHS) for 30 min, rabbit anti-GAD67 antiserum (Chemicon; Temecula, CA) 1:20,000 in 10% NHS in Tris-PBS for 48-72 hr, biotinylated donkey anti-rabbit IgG (Jackson ImmunoResearch, West Grove PA; 1:500 in 1% NHS-TPBS) for 16-24 hr, and finally ExtrAvidin-horseradish peroxidase (Sigma, St Louis, MO; 1:1500 in TPBS) for 4-16 hr. A nickel-intensified diaminobenzidine reaction revealed GAD-immunoreactive nerve fibers (Llewellyn-Smith et al. 1992b). The primary antibody to GAD is well characterized and recognizes only GAD67 (Kaufman et al. 1991).
Stained sections were osmicated, exposed to uranyl acetate, dehydrated, and embedded flat in Durcupan (Sigma). At least two sets of paired serial sections were cut from one tissue block from each of the three animals. Serial ultrathin sections were collected on formvar-coated nickel single-slot grids. Grids were kept in pairs and in serial order. One grid of each pair was stained with Reynold's lead citrate to identify GAD67-immunoreactive nerve terminals. GABA was localized by postembedding immunocytochemistry in the other.
Postembedding Immunocytochemistry
Sections were etched in 2% aqueous periodic acid for 7 min and then bleached for 7 min in 2% aqueous sodium metaperiodate [adapted from Somogyi and Soltész (1986)]. Between etching and bleaching, the sections were rinsed in three changes of distilled water, followed by immersion for 5 min. Sections were then processed for immunogold localization of GABA (Llewellyn-Smith et al. 1992a). After washing in Trisbuffered saline with Triton X-100 (TBST), pH 7.6, the grids were immersed overnight in rabbit anti-GABA antiserum (Arnel Products, Brooklyn, NY; 1:2500 or 1:5000 in TBST, pH 7.6). The primary antibody recognizes GABA but not a wide range of other amino acids (Hepler et al. 1986). The grids were washed in TBST, pH 7.6 then pH 8.2, before incubation for 1 hr in secondary antibody conjugated to 15-nm gold particles (Amersham, Arlington Heights, IL; 1:25 GAR Auroprobe EM in TBST, pH 8.2). After washing, the grids were stained with uranyl acetate and lead citrate.
Absorption Controls
Controls for the specificity of GABA immunogold labeling included omitting the primary or secondary antibody and replacing the primary antibody with normal rabbit serum. Overnight preabsorption of the primary antibody with a variety of amino acid-hemocyanin-glutaraldehyde conjugates was also carried out to test crossreactivity (Seguela et al. 1984). Hemocyanin conjugates of glutamate, glycine, and glutamine were prepared, resuspended in 500μl distilled water, and diluted to a final conjugate concentration of 1:100 with 1:2500 anti-GABA antibody in TBST, pH 7.6. A GABA-hemocyanin conjugate was similarly diluted with primary antibody to give final conjugate concentrations of 1:100 to 1:2500. The conjugate-antibody mixtures were stored overnight at 4C, as was 1:2500 anti-GABA without conjugate. Postembedding staining was done on two sections from runs of three serial sections, with the third section left unstained. Primary antibody preabsorbed with one of the four amino acid-hemocyanin conjugates (GABA, glutamate, glycine, or glutamine) was used on the first section and unabsorbed anti-GABA antibody on the second. After staining, the density of gold particles over 20 nerve terminals in the phrenic motor nucleus was measured for each treatment. The nerve terminals assessed for GABA immunoreactivity were then located in the third unstained section and assessed for GAD67 immunoreactivity. In separate control experiments, the primary antibody was replaced with amino acid-glutaraldehyde-hemocyanin conjugate (1:2500).
Data Analysis
One pair of serial ultrathin sections per block was randomly selected for identification of GAD67-immunoreactive varicosities in the phrenic motor nucleus. In each section postembedding-stained for GABA, nerve terminals in the vicinity of retrogradely labeled phrenic motor neurons were photographed randomly, numbered, located in the adjacent section on the unlabeled grid, and scored for GAD67 immunoreactivity. Terminals that could not be identified in the untreated sections were not analyzed.
GABA immunoreactivity was assessed on micrographs printed to × 37,000-40,000. The areas of axon terminals and of mitochondria within them were measured with a digitizing tablet. Gold particles over axoplasmic matrix plus synaptic vesicles and over mitochondria were counted manually. GABA labeling density (gold particles/μm2) was then calculated for axoplasmic matrix plus synaptic vesicles (excluding mitochondria), for mitochondria, and for the whole terminal (axoplasmic matrix, synaptic vesicles, and mitochondria). Labeling over blank resin was determined from gold particle densities over the lumina of at least four blood vessels per sample and ranged from 0.5 to 1.6 gold particles/μm2. Because this labeling was present at such a low level, we did not correct for it in any of our measurements.
Terminals identified as GAD67-positive or GAD67-negative were sorted into groups on the basis of their gold particle labeling density for GABA. Frequency distributions for the densities of immunogold labeling over synaptic vesicles and axoplasmic matrix were constructed for all analyzed terminals from each section because this tissue compartment appeared most likely to reflect the distribution of neurotransmitter GABA.
Statistics
All statistical analyses were carried out according to Sokal and Rohlf (1995) and Rohlf and Sokal (1995).
Nerve terminals from GABA-labeled sections were sorted into GAD67-positive and GAD67-negative categories. In the four sets of data collected, tests for homogeneity of variance between the GAD67-positive and GAD67-negative groups showed that the variances of these two groups were heterogeneous in almost all cases. Where this occurred, these data were log-transformed before unpaired t-tests were used to compare the density of labeling of the two groups of nerve terminals. The mean densities are given after back-transformation from their log values.
In the preabsorption control experiments, comparisons of the density of gold labeling were made over groups of GAD67-positive and GAD67-negative nerve terminals. The same terminals were identified in adjacent sections. One section of each pair was stained for GABA and the other with anti-GABA antibody that had been preabsorbed overnight with one of the four amino acid-hemocyanin conjugates. In these cases, the density of labeling over GAD67-positive or GAD67-negative nerve terminals over the area of synaptic vesicles and axoplasmic matrix was compared with a paired t-test. In several cases, the variances of the densities of gold labeling were heterogeneous, so data were log-transformed before the statistical test was performed.
Results
Four pairs of adjacent ultrathin sections through the phrenic motor nucleus were examined from the three rats that had received injections of CT-HRP into the diaphragm. One section of each pair was untreated so that nerve terminals could be classified as GAD67-positive or GAD67-negative. The other section was stained for GABA after being etched to remove resin and bleached to remove DAB reaction product. An initial sample of 697 GAD67-immunoreactive terminals in the phrenic motor nucleus was photographed; 25% of these (178) made synapses and direct contacts with retrogradely labeled phrenic motor neurons. Five hundred and nineteen of the 697 terminals could be identified in both GABA- and GAD67-labeled sections and were analyzed further for their density of gold labeling and their GAD67 content.
When GAD67-positive and GAD67-negative terminals were assessed for GABA gold labeling in the adjacent section, GAD67-positive nerve terminals generally appeared to have higher levels of GABA labeling than GAD67-negative nerve terminals (Figures 1, 2A, and 2B). Frequency distributions for gold labeling density over synaptic vesicles and axoplasmic matrix were constructed first for all nerve terminals examined in each section (Figure 3A) and then for nerve terminals that had been sorted into GAD67-positive and GAD67-negative classes (Figure 3B). These histograms confirmed that GAD67-positive nerve terminals generally showed higher densities of gold labeling for GABA than GAD67-negative terminals. The presence of two distinct peaks in the overall frequency distribution (Figure 3A) suggested a population of nerve terminals with a low GABA labeling density and a population with a high GABA labeling. However, when frequency distributions were constructed after sorting terminals into GAD67-positive and GAD67-negative classes, the two populations were not as well separated as the overall frequency distribution suggested, i.e., there was some degree of overlap between the labeling densities of GAD67-negative and GAD67-positive nerve terminals (Figure 3B). There were nerve terminals that lacked GAD67-immunoreactivity in the plane of section but showed a high density of GABA labeling. There were also GAD67-positive nerve terminals that displayed low levels of GABA labeling. Nevertheless, in all four pairs of sections from the three animals, unpaired to-tests showed that the density of gold labeling was significantly higher over GAD67-positive nerve terminals than over GAD67-negative nerve terminals (Table 1). This was true regardless of whether the density of gold labeling for GABA was measured over synaptic vesicles and axoplasmic matrix, over mitochondria alone, or over the entire nerve terminal.
Control experiments confirmed that the heavy labeling over GAD67-immunoreactive terminals was likely to be authentic GABA immunoreactivity. Overnight preabsorption of the primary antibody with increasing concentrations of GABA-hemocyanin resulted in a graded decrease in labeling density over both GAD67-positive and GAD67-negative nerve terminals (Figures 2C and 2D), with the highest concentration (1:100) virtually abolishing gold labeling. This reduction in gold particle density was statistically significant (paired t-test p < 0.001; Table 2). In contrast, incubation of the anti-GABA antiserum with glutamate-, glycine-, or glutamine-hemocyanin conjugates did not significantly alter gold labeling density (Table 2), although the density of labeling over both GAD-positive and GAD-negative nerve terminals showed a decrease with all conjugates tested. When the GABA-glutaraldehyde-hemocyanin conjugate diluted 1:2500 was substituted for primary antibody or when primary antibody was omitted, no gold labeling was observed. Substitution of normal rabbit serum for the primary antibody resulted in very sparse labeling, with densities similar to that seen in sections incubated with GABA-hemocyanin-preabsorbed primary antibody.
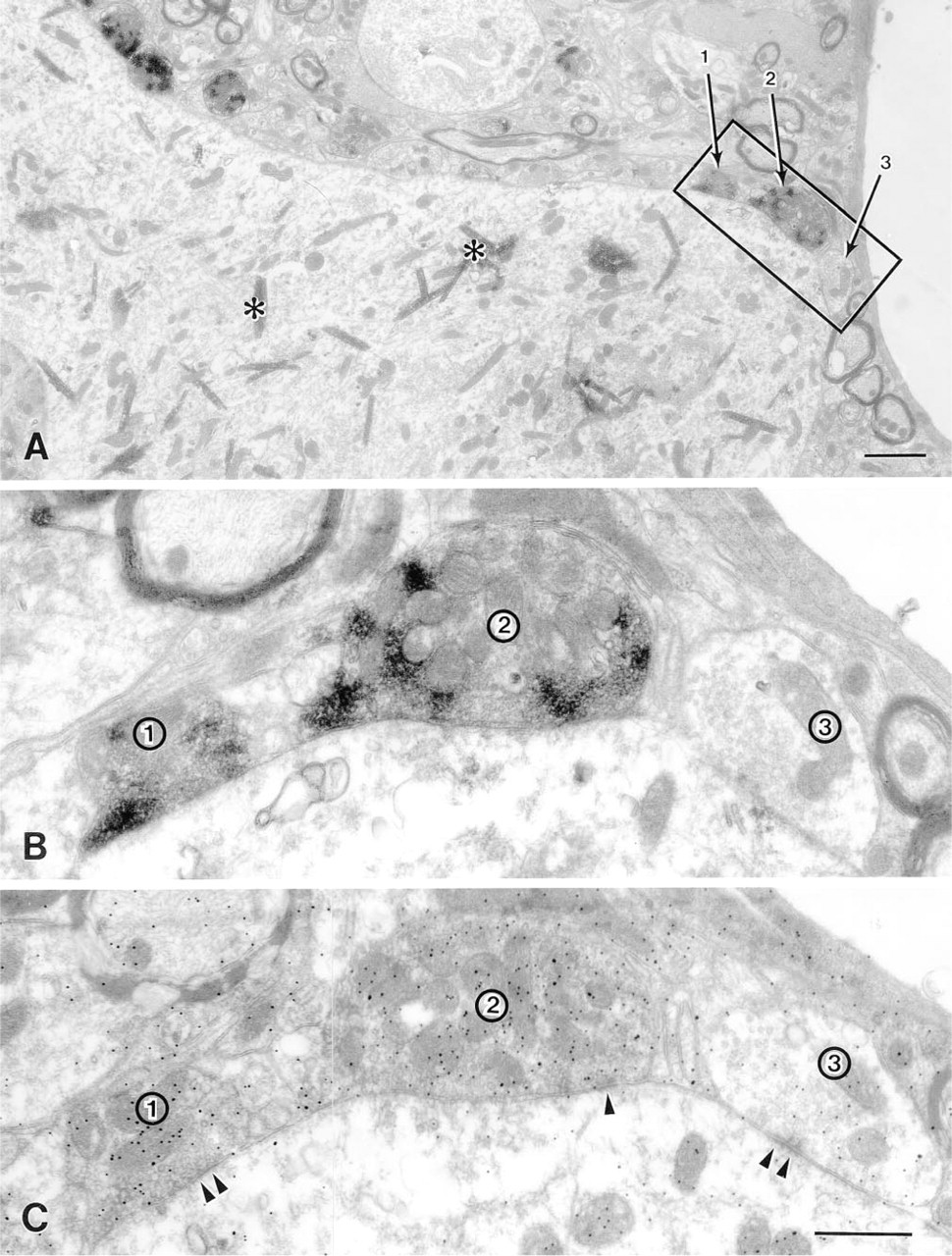
After postembedding immunogold localization of GABA, GAD-positive nerve terminals that synapse on phrenic motor neurons show a higher density of gold particles than GAD-negative nerve terminals. (
Discussion
Our correlation of the densities of GABA labeling with the occurrence of GAD67 immunoreactivity in adjacent sections of nerve terminals in the phrenic motor nucleus and of terminals synapsing on retrogradely labeled phrenic motor neurons has demonstrated that GAD67-positive nerve terminals have a statistically higher density of GABA gold labeling than GAD67-negative terminals. Although some GAD67-positive terminals showed low levels of GABA labeling and some GAD67-negative terminals showed high levels, the overall correspondence between the presence of immunoreactivity for GABA and GAD67suggests that both are useful markers of GABAergic terminals.
In this study, GAD67-positive nerve terminals showed statistically higher GABA labeling over synaptic vesicles and axoplasmic matrix, over mitochondria, and over the whole nerve terminal, than was seen over GAD67-negative nerve terminals. This difference between GAD67-positive and GAD67-negative nerve terminals was apparent in frequency distribution histograms based on gold particle density. Nevertheless, all of the frequency distributions for GABA labeling densities over the GAD67-negative and GAD67-positive groups of terminals showed some degree of overlap. Some GAD67-positive terminals showed low levels of GABA labeling and some GAD67-negative terminals showed levels of GABA labeling that were as high as those seen in GAD67-positive terminals. GAD67 terminals were marked by peroxidase reaction product, the presence of which unequivocally identifies them as possessing the active form of the enzyme necessary for GABA synthesis. However, the absence of GAD67 immunoreactivity from a nerve terminal, particularly from a single ultrathin section through a nerve terminal, does not prove that the terminal cannot synthesize GABA. Because the primary antibody used here recognizes GAD67 and not GAD65 (Kaufman et al. 1991), we may have underestimated the true proportion of GABAergic input. Terminals that lacked GAD67 mmunoreactivity but showed high levels of GABA labeling might contain GAD65 and therefore be capable of producing GABA under the appropriate intracellular conditions (Kugler 1993).
Only one previous study has directly examined GABA immunogold labeling over GAD-positive and GAD-negative nerve terminals (Maxwell et al. 1989). However, it did not show quantitatively whether all GAD-positive nerve terminals were heavily labeled with GABA and all GAD-negative terminals were lightly labeled. In synaptosomes, GABA concentrations and GAD activity are generally well correlated (Löscher et al. 1985; Santiapillai et al. 1989), but these data cannot be compared with our present results because they do not provide information about individual nerve terminals. In the opossum piriform cortex, the presence of GABA immunoreactivity correlated with [3H]-GABA uptake in nerve terminals (Haberley et al. 1987), but the relationship between the presence of GAD immunoreactivity and [3H]-GABA uptake was not as consistent, possibly because the antiserum used to localize GAD did not recognize both forms of the enzyme (Feldblum et al. 1993). We believe that, in this study, the impact of any possible underestimation of the number of GAD67-containing nerve terminals is small because we saw relatively few GAD67-negative nerve terminals with high densities of gold labeling. For example, beyond the midpoint of the frequency distribution in Figure 3B, 10 of 11 (92%) terminals most heavily labeled for GABA were GAD67-positive and only one was GAD67-negative. Across the other sections examined, GAD67-positive varicosities comprised 62%, 80%, and 83% of all the varicosities that exceeded the midpoint of the range of gold particle density. It is possible that the small number of GAD67-negative terminals that display high levels of GABA labeling are a consequence of inadequate penetration of antibodies in the pre-embedding immunocytochemical procedure. More problematic are the nerve terminals at the lower ends of the frequency distribution, those that were GAD67-positive but showed low levels of GABA gold labeling equivalent to the levels for some GAD67-negative nerve terminals. If these terminals contain the active form of GAD, why should they have levels of GABA immunoreactivity similar to nerve terminals that lack GAD67? Possibly the presence of some GABA in GAD67-negative nerve terminals is due to production of GABA by other enzymatic reactions that do not require GAD67, such as the conversion of putrescine to GABA through aldehyde dehydrogenase (Kugler 1993), or by uptake of GABA via GABA transporters (Iversen and Kelly 1975).
The data from our control experiments indicate that we have localized authentic GABA immunoreactivity in both GAD67-positive and GAD67-negative nerve terminals. First, there was no gold labeling over any tissue structures in the absence of primary antibody, and gold labeling was minimal in experiments in which the primary antibody was replaced with normal rabbit serum. We also showed concentrationdependent reductions in the density of gold labeling when the primary antibody to GABA was preabsorbed with increasing concentrations of a GABA-protein conjugate. This anti-GABA antibody has been used in our previous studies on amino acid content of synaptic inputs to rat sympathetic preganglionic neurons, and to bulbospinal neurons of the rostral ventrolateral medulla, in which we showed that terminals that contained high densities of immunogold labeling for GABA contained low levels of GLU labeling, and vice versa (Llewellyn-Smith et al. 1992a,1995). Furthermore, the labeling density produced by this antiserum was not significantly affected by preabsorption with glutamate-, glycine-, or glutamine-protein conjugates.
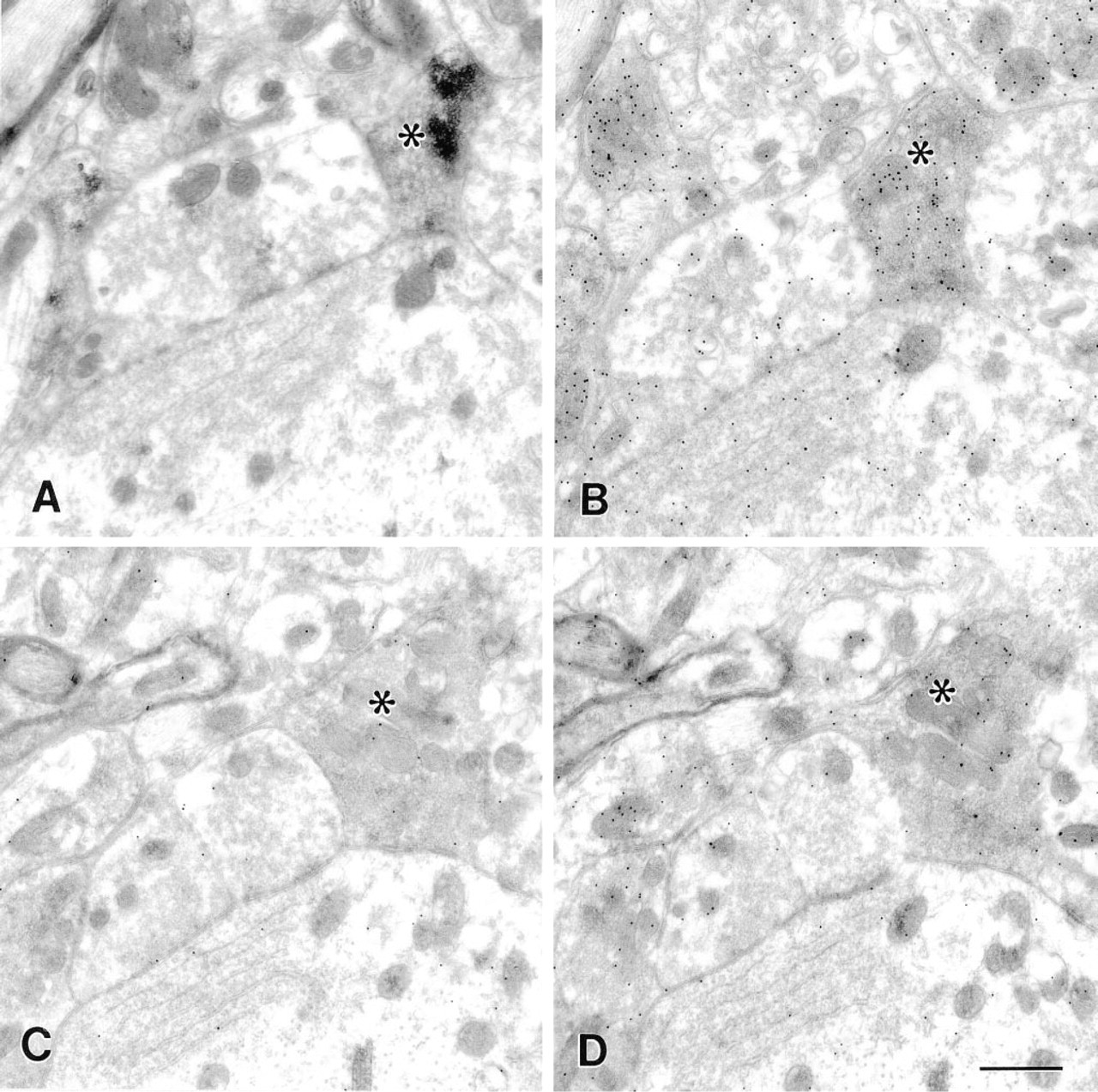
Control experiment showing effect of overnight preabsorption of the anti-GABA antibody with a GABA-glutaraldehyde-hemocyanin conjugate. (
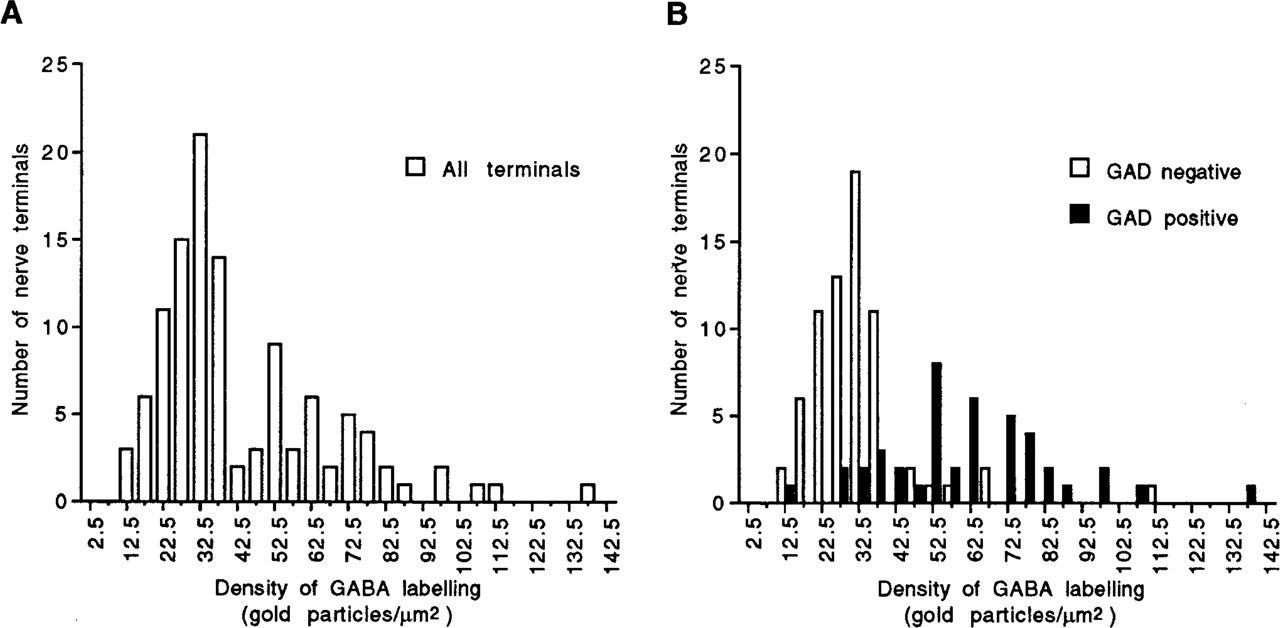
Frequency distributions of gold particle densities over synaptic vesicles and axoplasmic matrix for all nerve terminals (
Mean densities of GABA immunogold labeling (gold particles/μm2) over GAD-positive and GAD-negative terminals in the phrenic motor nucleus a
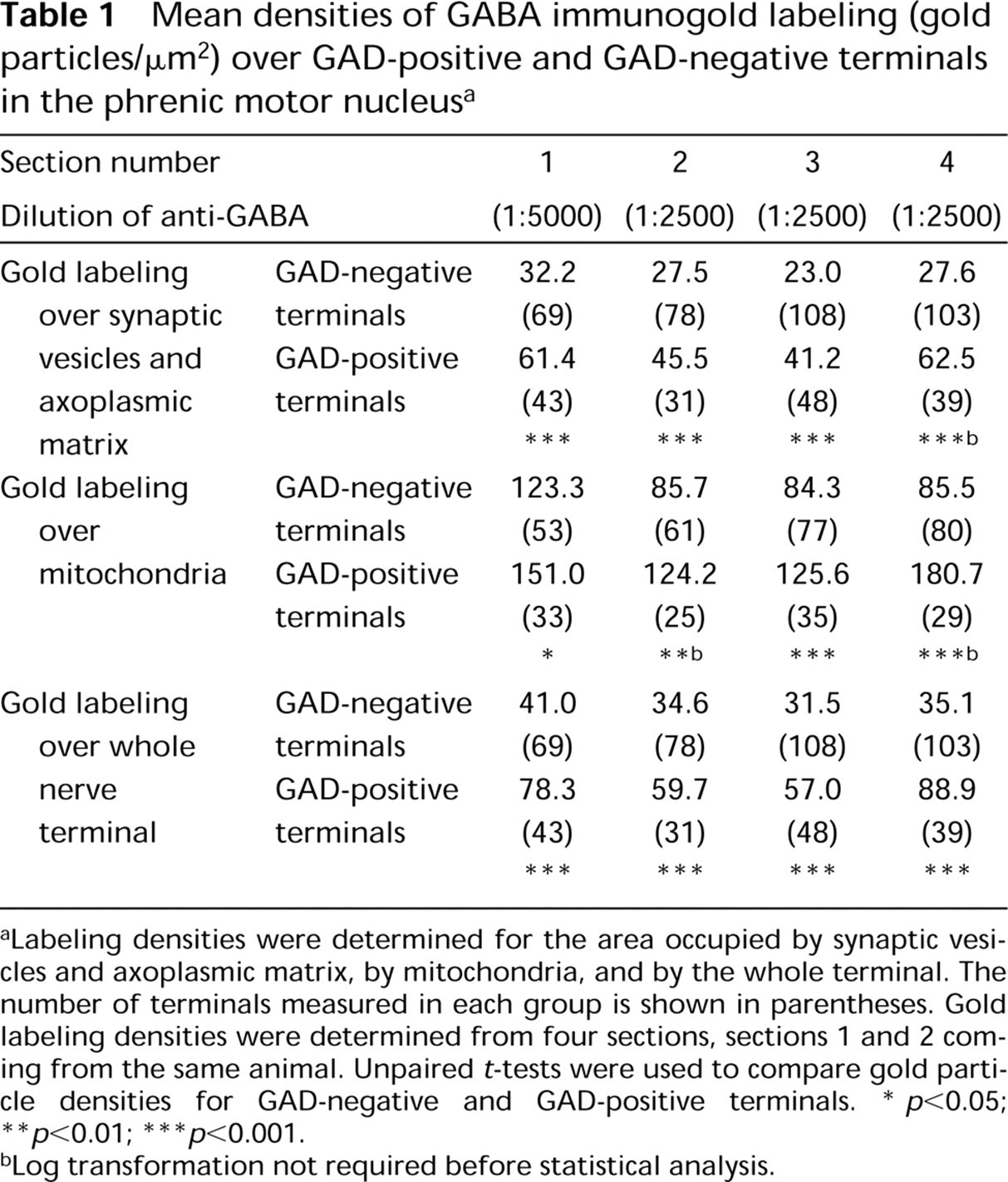
Labeling densities were determined for the area occupied by synaptic vesicles and axoplasmic matrix, by mitochondria, and by the whole terminal. The number of terminals measured in each group is shown in parentheses. Gold labeling densities were determined from four sections, sections 1 and 2 coming from the same animal. Unpaired t-tests were used to compare gold particle densities for GAD-negative and GAD-positive terminals.
p <0.05
p <0.01
p <0.001.
Log transformation not required before statistical analysis.
Mean density of GABA immunogold labeling (gold particles/μm2) with or without overnight preabsorption of the primary antibody a
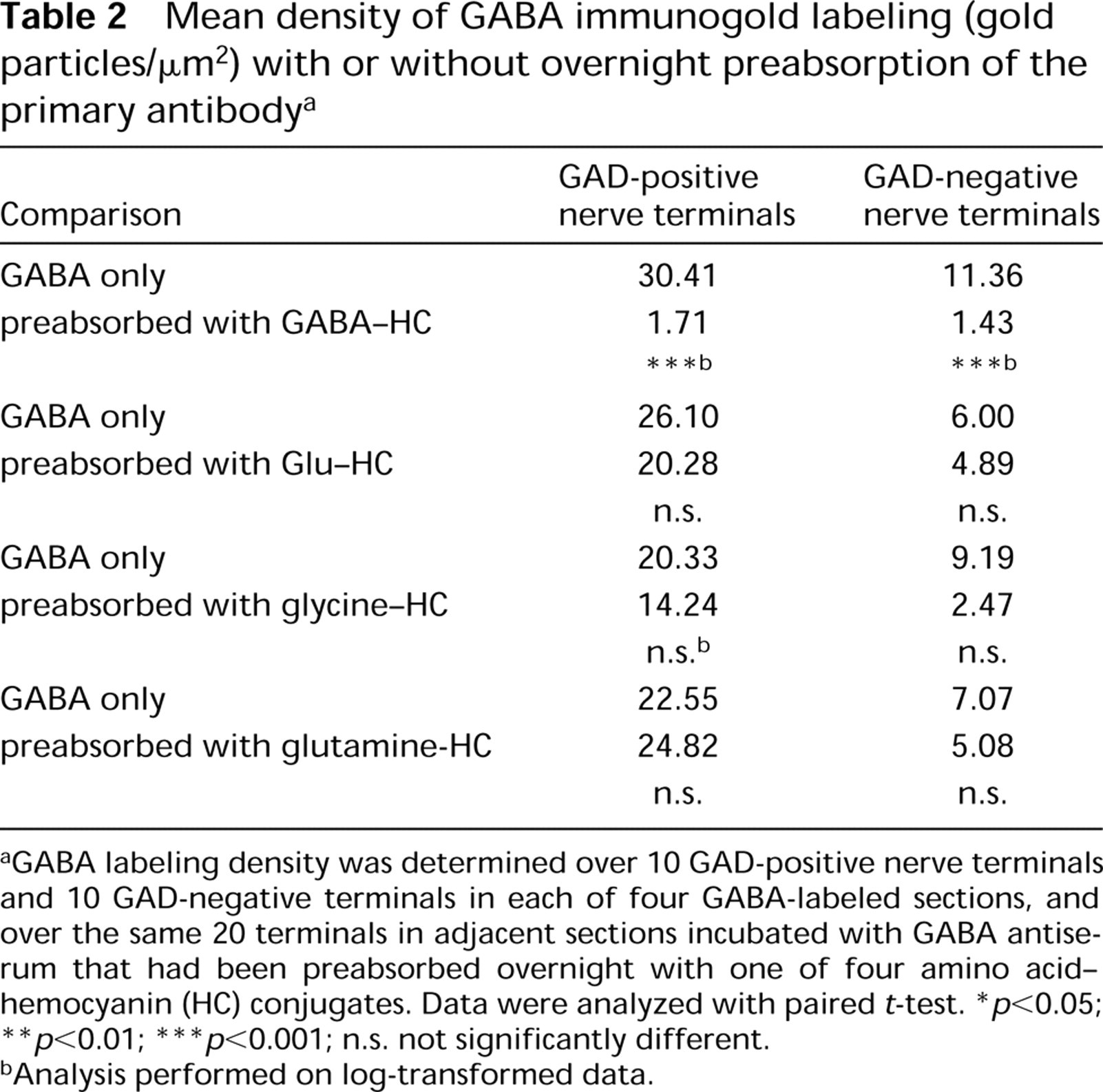
GABA labeling density was determined over 10 GAD-positive nerve terminals and 10 GAD-negative terminals in each of four GABA-labeled sections, and over the same 20 terminals in adjacent sections incubated with GABA antiserum that had been preabsorbed overnight with one of four amino acid-hemocyanin (HC) conjugates. Data were analyzed with paired t-test.
p <0.05
p <0.01
p <0.001; n.s. not significantly different.
Analysis performed on log-transformed data.
In this study, the presence of peroxidase reaction product in GAD67-labeled nerve terminals did not appear to cause any significant quenching of immunogold labeling for GABA, as has been reported in other studies (e.g., Ji et al. 1991) because there was statistically higher GABA immunogold labeling over peroxidase-containing (i.e., GAD67-positive) nerve terminals. Moreover, our quantitative results support the general observation of Maxwell et al. (1989) that GAD-positive nerve terminals were more heavily labeled for GABA than GAD-negative terminals.
In summary, our postembedding immunogold studies have shown that, in the phrenic motor nucleus, GAD67-immunoreactive nerve terminals, including many terminals that synapse with phrenic motor neurons, are more heavily stained for GABA than terminals that lack GAD67 immunoreactivity. Our results indicate that both immunoreactivity for GABA and immunoreactivity for GAD67 are good markers for central GABAergic terminals.
Footnotes
Acknowledgments
Supported by grants from the National Health and Medical Research Council of Australia, the National Heart Foundation of Australia, and the National Sudden Infant Death Syndrome Research Council of Australia. SMM was supported by a Post-doctoral Research Fellowship from the National SIDS Research Council.
We would like to thank Carolyn Martin for expert technical assistance.
