Abstract
We analyzed the internalization of the receptor subtype 2 (sst2) for the neuropeptide somatostatin in glioma cells at the ultrastructural level using an antibody against an extracellular amino acid sequence. Intact cells derived from solid human gliomas or those of the human glioma cell line U343 were receptor-labeled (a) by classical gold immunocytochemistry using a 15-nm gold-labeled second antibody, (b) directly with the sst2 antibody adsorbed to 5-nm colloidal gold, and (c) with the physiological ligand somatostatin conjugated to 5-nm colloidal gold. The receptor was predominantly internalized via uncoated vesicles budding from the cell membrane but only rarely via coated pits, which has been mostly reported for G-protein-coupled, seven transmembrane-domain receptors. In the presence of ligand and sst2 antibody vesicles, tubule-like structures, and multivesicular bodies were labeled in superficial and in perinuclear portions of the cells within the first 30 min. Lysosomal labeling was observed after 30 min and especially after an hour of internalization time. This internalization route is also used to study the directly labeled sst2 antibody or the labeled ligand. However, the late endosomal compartment appears to be reached more rapidly in these latter experiments.
E
Despite the complexity of these internalization processes most investigations were performed with subcellular fractionation techniques or by confocal laser microscopy and used mostly transfected cells that were not the physiological target for the ligand (Garland et al. 1994; Ashworth et al. 1995; Nouel et al. 1997; Roosterman et al. 1997). Studies using high-resolution electron microscopic techniques are lacking up to now. Therefore, the aim of the present study was to analyze the agonist-induced internalization of a seven-transmembrane domain G-protein-coupled receptor at the ultrastructural level in a nontransfected cell. We investigated the receptor subtype 2 (sst2) for the widely distributed inhibitory neuropeptide somatostatin in human glioma cells. These cells are known to express sst2 in large amounts compared to the natural target cell, the nonmalignant astrocyte (Feindt et al. 1995). Therefore, glioma cells appear to be a good model for investigating internalization processes by electron microscopic analysis.
The internalization of the sst2 receptor is described at the ultrastructural level by use of an antibody against an amino acid sequence in the extracellular part of the receptor molecule and with an electrondense conjugate of the ligand. Visualization of the dynamics of the receptor internalization on intact cells was accomplished by (a) classical gold immunocytochemistry using the sst2 antiserum as first antibody and a gold-labeled anti-rabbit immunoglobulin as second antibody, (b) labeling the sst2 antibody directly with colloidal gold, and (c) adsorption of the physiological ligand somatostatin to colloidal gold and affinity-labeling of the receptor.
Materials and Methods
Antisera
The sst2 receptor antibody was raised in rabbits against a synthetic peptide corresponding to residues 33-43 (QTEPYYDLTSN) plus a C-terminal cysteine that was conjugated via SH groups to keyhole limpet hemocyanin (Eurogentec; Seraing, Belgium). This sequence does not occur in any of the other sst subtypes, but is common to the splice variants A and B of the human sst2. The antibody was affinity-purified (affinity gel-peptide coupled to EAH Sepharose; Pharmacia, Uppsala, Sweden) and analyzed by SDS-PAGE and ELISA. A further anti-peptide antibody against the C-terminal sequence of sst2A (ETQRTLLNGDLQTSI) was obtained from Gramsch Laboratories (Schwabhausen, Germany); compare Feindt et al. (1997).
Membrane Fractionation and Western Blotting
A total of 1-2 × 106 cells were lysed in 10 ml 5 mM Hepes buffer, pH 7.4, with 1 mM phenylmethanesulfonyl fluoride, 0.1 mM
Reverse Transcription-Polymerase Chain Reaction (RT-PCR) and cAMP Assay
RT-PCR for detection of sst2A and sst2B was performed (Feindt et al. 1995) with the sense primer 5′-TGGTGGTCCTCACCTATG-3′ in combination with the anti-sense primers I 5′-CTGTATAGGCATTACCTACG-3′or II 5′-CCTCCAAACCAGTATCTGA-3′ (Taylor et al. 1994). PCR products were identified by agarose gel electrophoresis and controlled by cycle sequencing using the DyeDeoxy Terminator Cycle Sequencing Ready Reaction Kit (Applied Biosystems; Foster City, CA). The expected length of the PCR products with the anti-sense primer I is 623
Labeling of Antibody and Ligand with Colloidal Gold
A total of 100 μg affinity-purified sst2 antibody dialyzed vs 2 mM borate buffer, pH 9.0, was stirred for 1 hr with 20 ml colloidal gold sol (EM GC 15 or EM GC 5; British Biocell, Cardiff, UK) previously adjusted with 0.2 M K2CO3 to pH 9.0. Then 1 ml of 10% bovine serum albumin (BSA) in bidistilled water adjusted to pH 9.0 was added. After stirring for another 10 min the conjugate was concentrated to 1 ml by ultrafiltration (YM 10 membrane; Amicon, Temecula, CA), purified by gel filtration (Superdex S-300 1.6 × 60-cm column equilibrated and eluted with 0.14 M NaCl, 0.2% BSA, 10 mM Hepes, pH 8.0), and concentrated again by ultrafiltration. Labeling of somatostatin has been previously described (Mentlein et al. 1990). In brief, 10 ml of 5-nm gold sol adjusted to pH 9.0 was incubated with 100 μl (100 nmol) of 1 mM somatostatin stock solution in water for 24 hr at 4C. After stabilization with 1 ml 10% BSA, excess peptide and other substances were removed by gel chromatography and repeated ultrafiltration as above. Particle concentrations of colloidal gold sols were calculated from their absorbance at 520 nm (Mentlein et al. 1990).
Cell Culture
Astrocytes were obtained from dissociated cerebral cortices of 2–day-old Fisher F344 rats, purified, and subcultivated (McCarthy and De Vellis 1980). Cells from surgical human gliomas (classified as glioblastoma WHO Grade IV) and the human glioma cell line U343 (Deutsches Krebsforschungszentrum; Heidelberg, Germany) were purified and cultivated in DMEM containing 10% fetal calf serum (Feindt et al. 1995). All cultures used were more than 95% immunopositive for the astroglia/glioma marker glial fibrillar acidic protein (GFAP) and less than 1% reactive with the microglia/ macrophage marker CD68 (Lucius et al. 1995).
Receptor Labeling and Internalization
Subconfluent cells (105 U343 or glioma cells) grown on poly
Controls
Cells were incubated only with gold-labeled second antibody (anti-rabbit IgG-Au), or without anti-sst2, with anti-sst2 replaced by preimmune serum, or with antigen-inactivated anti-sst2 (1 μM peptide, 30 min, 37C) and fixed after 10 min at 37C. Internalization was performed without preincubation with somatostatin and after washing in 20 mM citrate, 140 mM NaCl, pH 5.5. Under hyperosmolar conditions (0.4 M sucrose in GBSS) or in potassium-free buffer (140 mM NaCl, 1 mM CaCl2, 1 mM MgCl2, 1 mg/ml glucose, 20 mM Hepes, pH 7.4), internalization times of 5 and 10 min were used.
Results
Glioma Cells Express the sst2A Somatostatin Receptor
Expression of the sst2 receptor in human glioma cells could be verified by Western blotting and RT-PCR experiments. The affinity-purified sst2 antiserum directed to the N-terminal extracellular portion of the receptor strongly reacted with a 75-kD band in U343 human glioma cells (Figure 1). The immunoreactivity was abolished completely in the presence of the immunization peptide or by omitting the primary antibody. A corresponding 75-kD band was also stained by a different sst2A-directed antiserum raised against the C-terminal 15 intracellular residues of this splice variant of the receptor. The mRNA for sst2A could be detected in U343 glioma cells by RT-PCR with two different primer pairs, whereas mRNA for the splice product sst2B was undetectable (not shown).
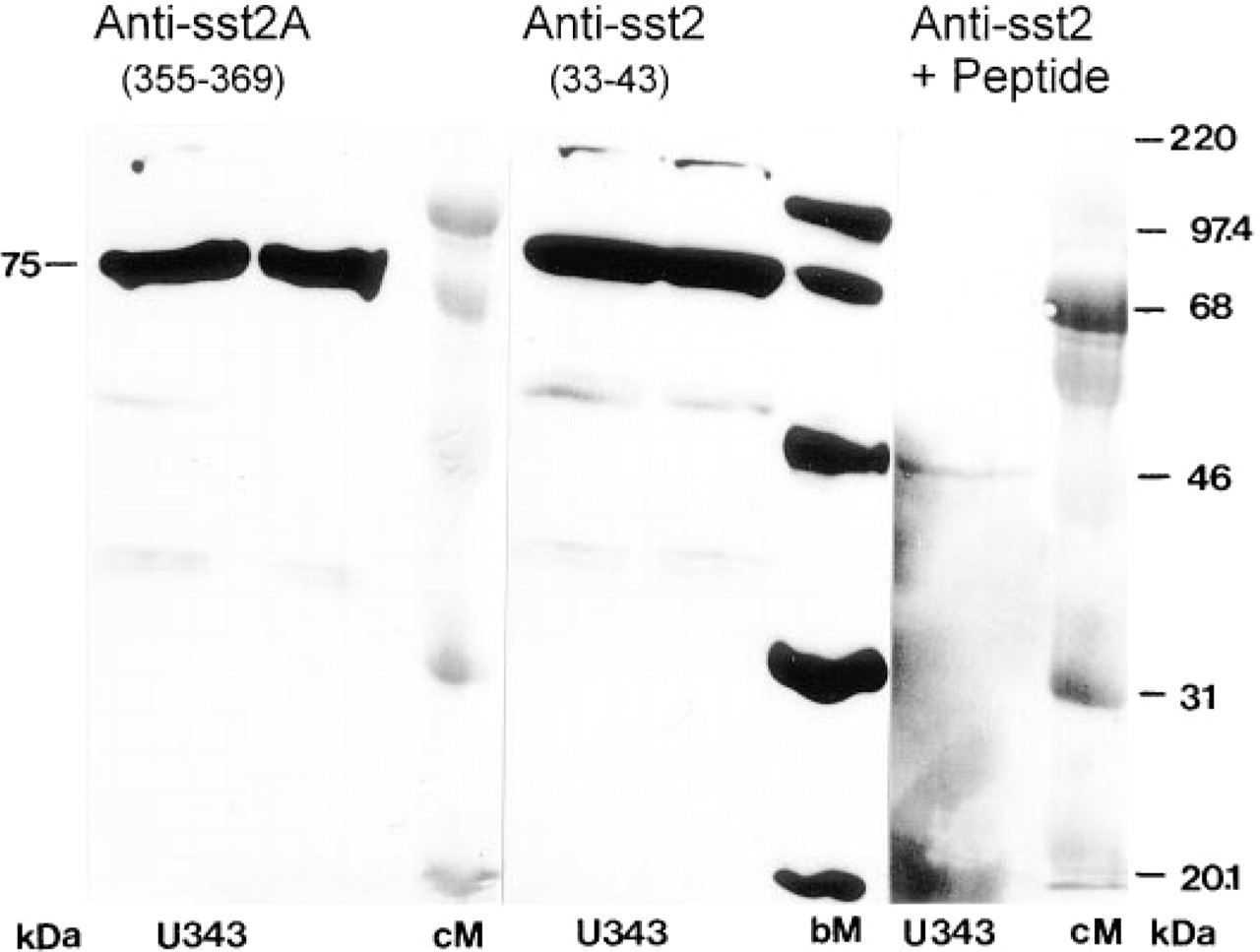
Western blot analysis of the somatostatin receptor sst2 in U343 glioma cell membranes using different antibodies. Different blots with two electrophoresis lanes were stained with the sst2-antibody directed to the extracellular part of the receptor (anti sst2 33-43) or with an sst2A antibody generated to the intracellular C-terminal amino acids of the splice variant A (anti sst2A 355-369) and detected by chemiluminescence. (Control) Immunostaining in the presence of the peptide antigen. Rainbow-colored (cM) and biotin-labeled (bM) proteins were used as molecular mass markers.
Binding of the antibody to sst2 had no influence on its signal transduction. The somatostatin analogue octreotide (sst2-specific in glioma cells at 1 nM due to the absence of sst3 and sst5) reduced intracellular cAMP levels in U343 cells elevated by the adenylyl cyclase activator forskolin (50 μM at 37C for 10 min; compare Feindt et al. 1995) irrespective of whether anti-sst2 was present or absent (not shown).
The sst2 Receptor Can be Visualized on the Surface of Glioma Cells
After cooling to 8C subconfluent cultures either of the glioma cell line U343 or from solid human gliomas, the somatostatin receptor could be always stained with the sst2 antibody at the light or electron microscopic level on the cell surface, but with different intensity on single cells (Figure 2). Unstained and heavily stained cells were in direct juxtaposition. When the antibody was inactivated with the immunization peptide, no labeling was observed. At the electron microscopic level, the labeling on U343 cells was rather uniformly distributed mainly on the cell somata, whereas the delicate processes of human glioma cells were labeled up to their finest tips (Figure 3).
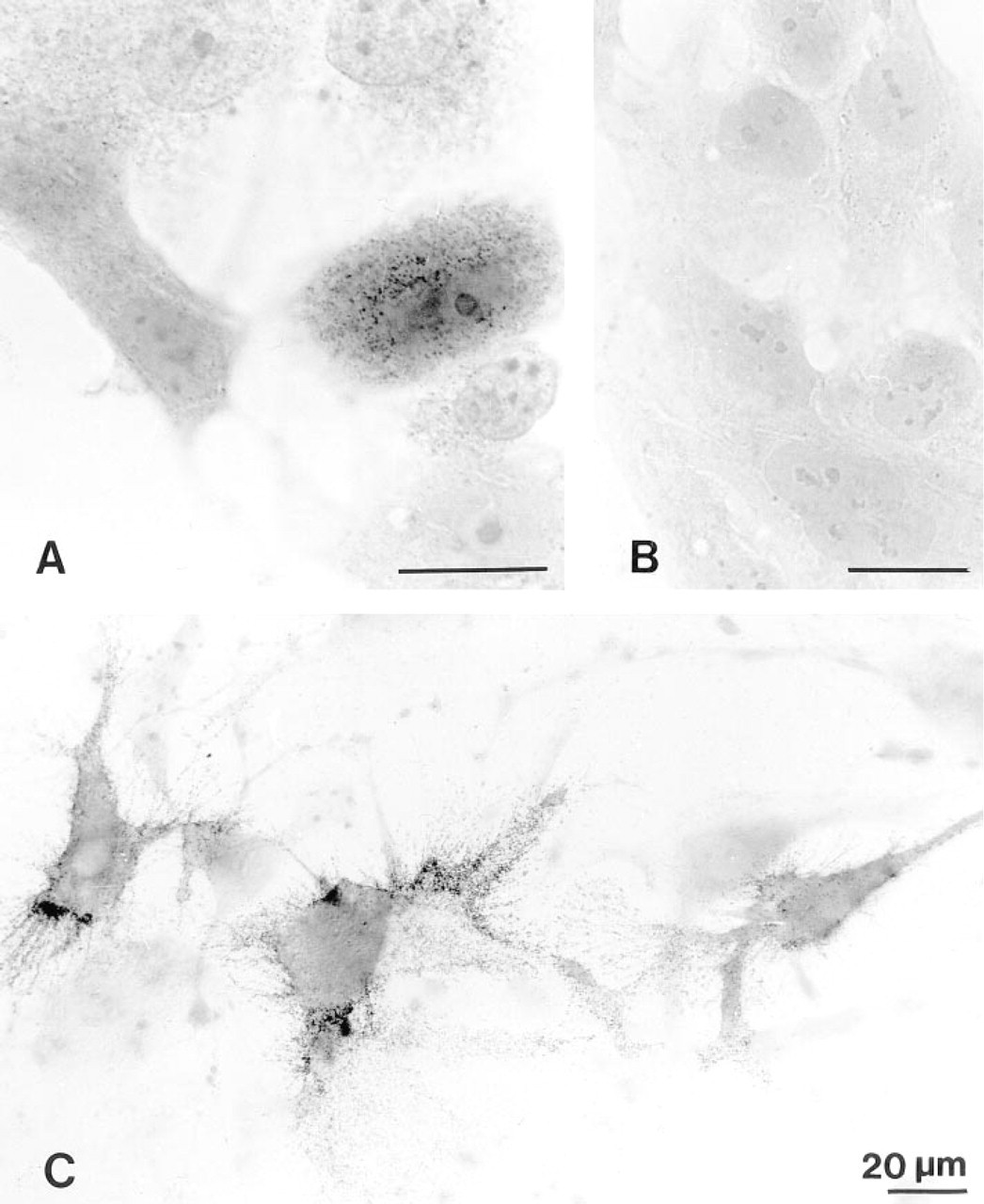
Light microscopic immunocytochemistry of the sst2 receptor on intact glioma cells. (
The Antibody-labeled Somatostatin Receptor is Internalized via Endosomal Vesicles and Not via Coated Pits
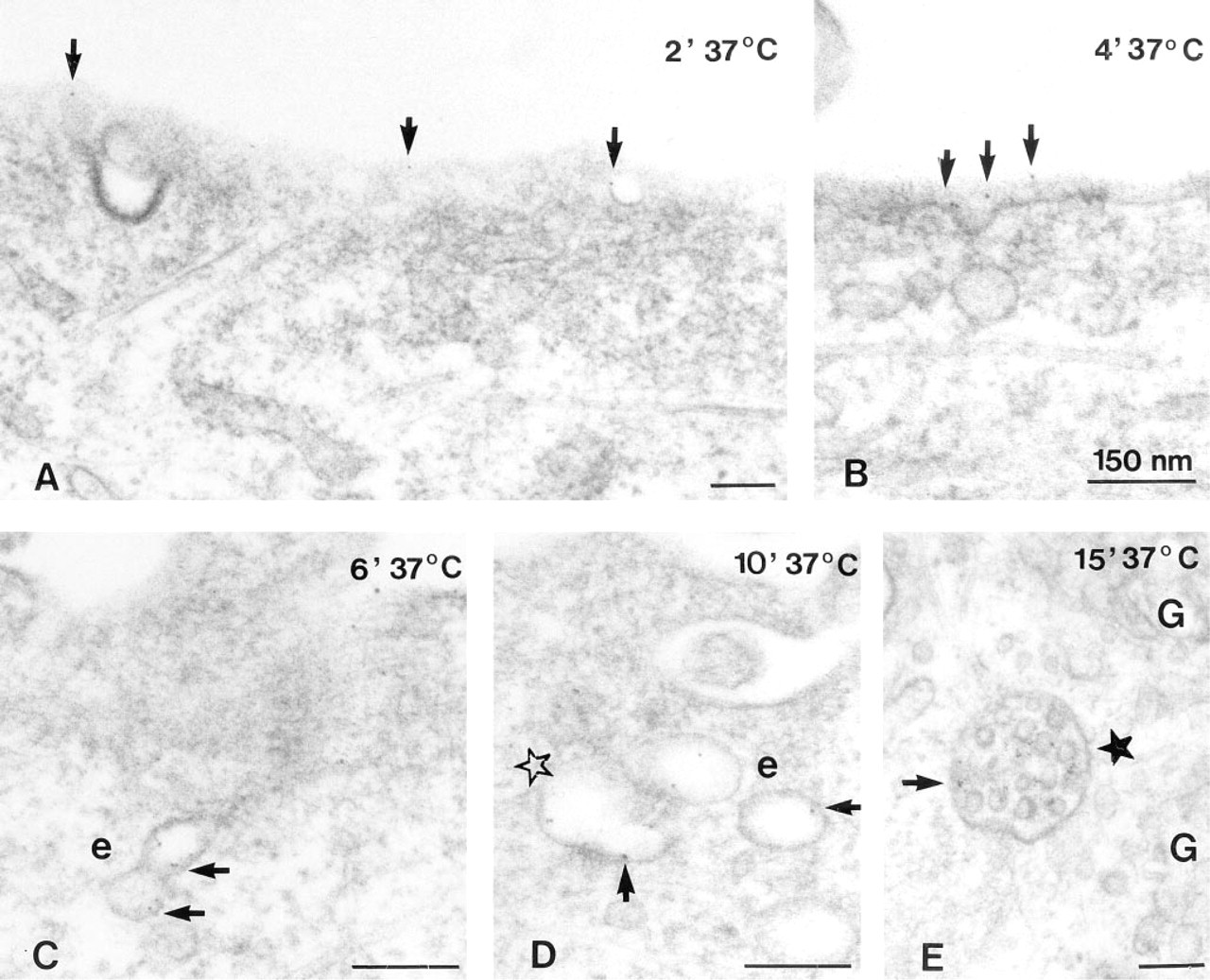
Although U343 cells and cells of human glioblastoma differ in their surface morphology (Figure 3), there were no fundamental differences in their internalization pattern. Cells fixed without warming carried singly dispersed gold particles exclusively on the surface of short, coarse processes in U343 cells or on the filiform network of glioblastoma cells (Figure 3). After warming at 37C for 5 min, assembly of gold particles was observed on the cell somata between the processes, and a few of them were found in inconspicuous vesicles opening at or positioned directly beneath the cell surface (Figure 4A). On tangential sections through the superficial parts of the cell, a high regional amount of gold assembly on limited areas of the cell surface was even more evident (not shown).
The labeling intensity of these vesicles increased with increasing internalization times; the vesicles sometimes became elongated to little tubules (Figure 4B). They still were most often found in the superficial parts of the cells. Coated pits were only rarely labeled, at most in about 1% of labeled vesicles. After 15 min at 37C a few gold particles were included in multivesicular bodies (Figure 4C). Their number and the number of engulfed gold particles enlarged with increasing internalization times. In addition, after 30 min at 37C, labeled multivesicular bodies were located in the vicinity of Golgi areas and lysosome-like bodies containing gold particles were first observed (Figure 4D). Thereafter, their number and labeling intensity increased, but even after 60 min of internalization labeled lyso- some-like bodies were clearly less common than labeled multivesicular bodies. Binding and internalization continued even for longer times at 37C, however, with decreasing labelling intensity at the cell membrane after 45 min and still less intensity after 60 min at 37C. Then, heavily labeled lysosomal bodies were conspicuous although staining was dense in superficial cytoplasmic vesicles, tubules, and multivesicular bodies.
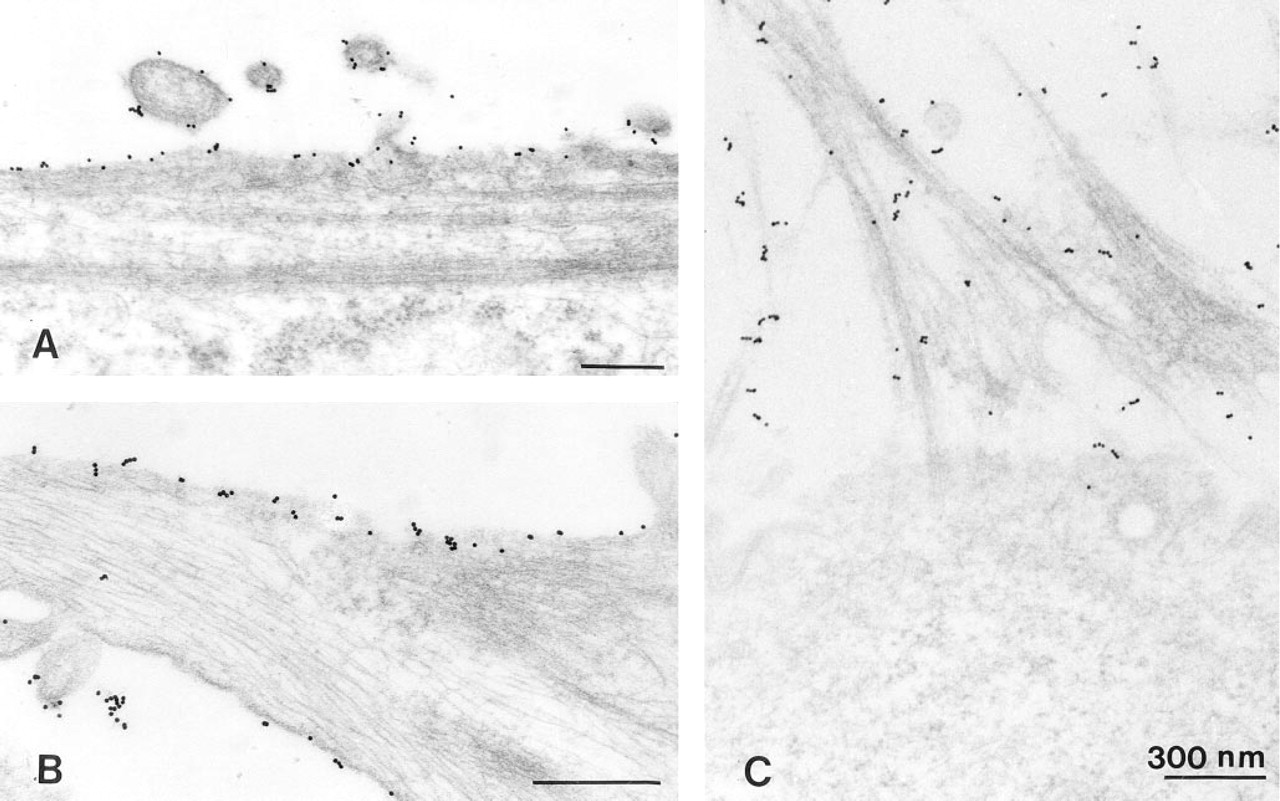
Electron microscopic immunocytochemistry of the sst2 receptor on glioma cells at 4C. (
With 5-nm gold-labeled sst2 antibodies, fundamentally the same internalization pattern was observed as with unlabeled primary and 15-nm gold-labeled secondary antibodies. Gold particles were mostly engulfed in uncoated vesicles, moving slowly with longer incubation times into the perinuclear region. Multivesicular bodies took the same route with a slight shift in time. After 5 min the first labeled multivesicular bodies were observed, which is clearly earlier than in experiments using 15-nm gold-IgG as indicator for the bound sst2 antibody. However, no differences were evident regarding the appearance of gold-containing lysosome-like bodies.
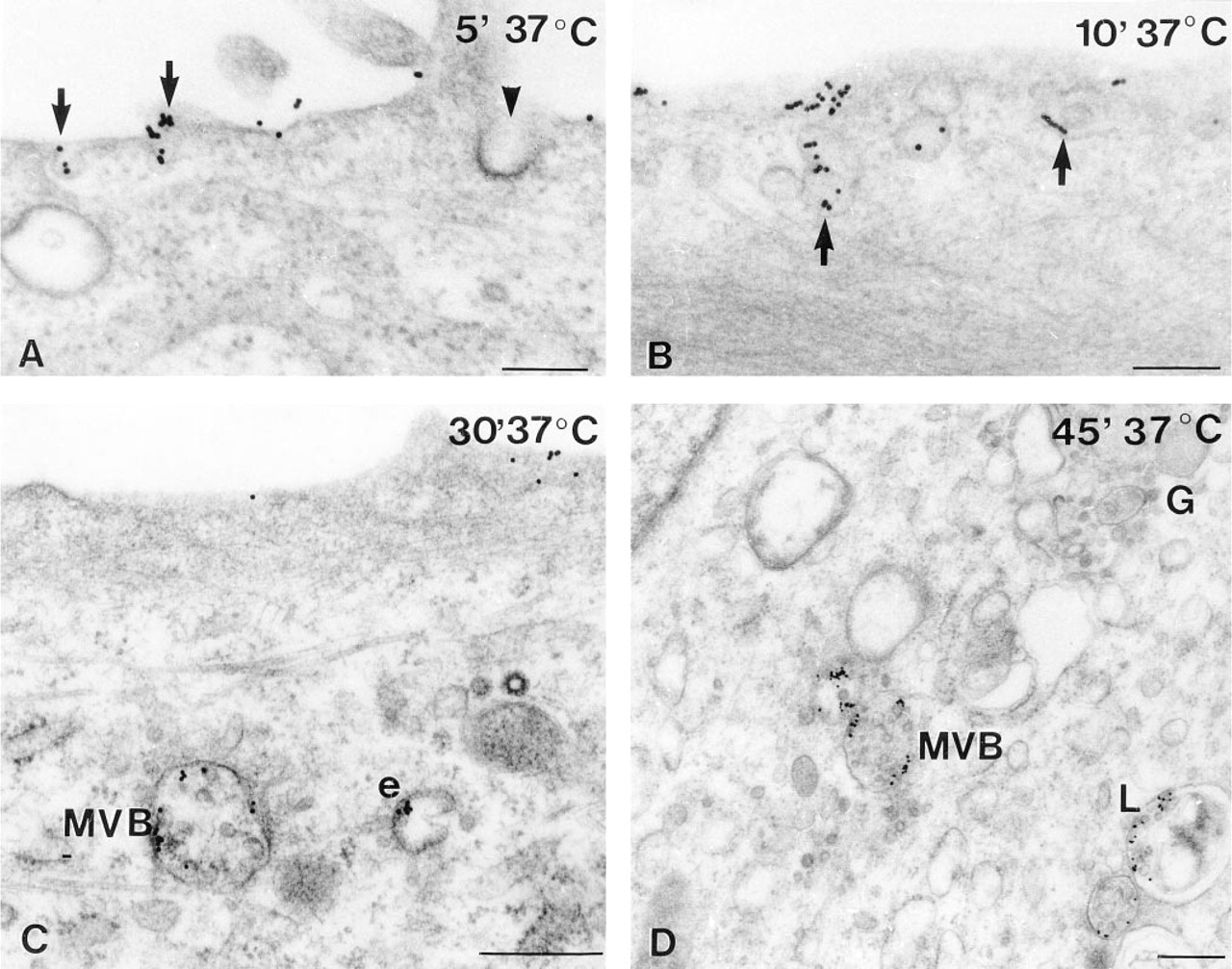
Electron microscopic immunocytochemistry of the sst2 receptor on glioma cells after different internalization times in the presence of the ligand. (
Labeled Somatostatin Is Internalized via Endosomal Vesicles and Not via Coated Pits
The gold-labeled ligand was internalized in the same way as the antibody-labeled receptor. In previous studies we have shown that this electron-dense conjugate of somatostatin to 5-9-nm colloidal gold is still biologically active in displacing radiolabeled peptide from pituitary or cortical binding sites and in reducing basal or growth hormone-releasing hormone-induced somatotropin release from adenohypophyseal cells (Mentlein et al. 1989,1990). Moreover, the conjugate was physically stable for at least 1 hr at 37C at pH values between 4 and 8.6 (Mentlein et al. 1989). After 5 min of internalization, the ligand was observed at the cell surface and in omega-shaped uncoated vesicles (Figure 6), but coated pits were only rarely labeled. As early as 5 min after incubation at 37C, multivesicular bodies contained the small gold particles along with early endosomal vesicles and tubular structures. The lysosomal compartment was not reached earlier than in experiments using 15-nm gold-labeled anti-rabbit IgG as tag for receptor antibodies.
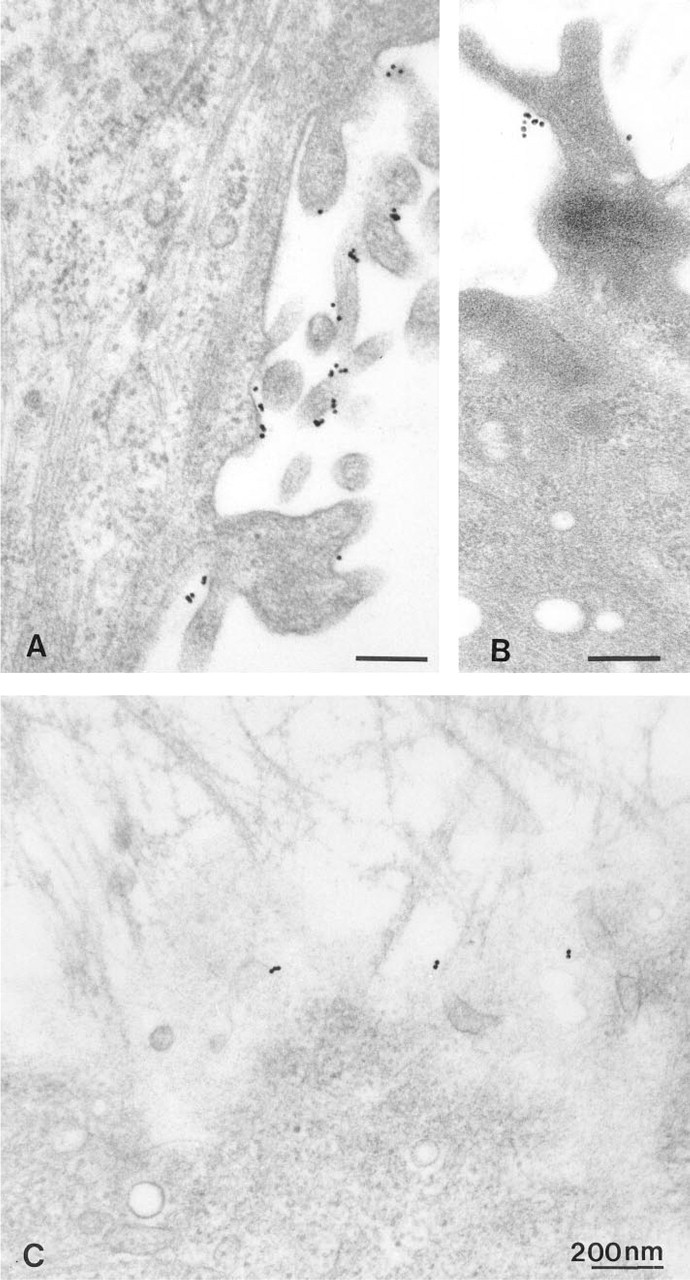
Incubation controls, internalization time 10 min. (
Internalization of 5-nm gold-labeled somatostatin (arrows) in U343 glioma cells at different internalization times. (
Finally, we attempted to double label the receptor with the 5-nm gold-labeled ligand and the 15-nm gold-labeled antibody (either directly or via the second antibody). However, in these combination studies only the 5-nm gold-tagged ligand could be demonstrated. The preparations were completely devoid of 15-nm gold particles, irrespective of whether ligand and receptor antibody were applied together or in sequence.
Discussion
With a triple electron microscopic approach (classical immunocytochemistry with gold-labeled second antibodies, directly gold-labeled antibody, and affinitylabeling with gold-tagged ligand), the internalization pathway of ligand-activated sst2 was studied in glioblastomas cultivated after neurosurgery and in a human glioblastoma cell line (U343). The results achieved in these three approaches are congruent. The predominant internalization route is via uncoated vesicles budding from the cell membrane. Under permanent internalization conditions for half an hour, labeled vesicles and tubule-like structures as well as labeled multivesicular bodies (MVB) are observed in superficial and perinuclear parts of the cells. Lysosomal labeling begins after 30 min and is fully expressed after an hour of internalization time. Only then is labeling intensity at the cell surface decreased. Slight differences between the results achieved with labeled receptor antibodies and labeled ligand appear to concern internalization dynamics. The ligand appears more rapidly in the late endosomal compartment (MVB) than the labeled antibodies. It does not, however, enter the lysosomal compartment more rapidly.
Neither the gold-labeled ligand nor the sst2 antibody is suitable to discriminate between the splice variants sst2A and sst2B which differ in the intracellular C-terminal residues. However, in the U343 cell line and in gliomas, only the subtype 2A is detectable, as has been reported for other tumors, e.g., carcinomas and melanomas (Taylor et al. 1994). In the presence of antibody the receptor was still able to transduce intracellular signals, i.e., inhibition of the adenyl cyclase.
Internalization processes terminate after very short periods, with sorting in early endosomes being completed after 2-3 min (Mellman 1996). Coated pits can lose their coats so rapidly that the presently used internalization times are too long to observe them. We did not intend to investigate the time course of the different internalization steps but rather to observe the organelles involved. Therefore, the internalization conditions were kept unchanged over a rather long time so that all internalization steps could be expected to be observable at least during the first 5-10 min.
Internalization of receptor-selective ligands (Koenig et al. 1997; Nouel et al. 1997) or somatostatin receptor subtypes (Hipkin et al. 1997; Roosterman et al. 1997) has been described in transfected cells on the basis of confocal fluorescence microscopy. A main internalization route via coated pits has been proposed from the pattern of fluorescent signals in incubations with phenylarsine oxide, hypertonic sucrose, or in potassium-deficient conditions. These additions have been reported to block internalization processes via coated pits (Larkin et al. 1983; Heuser and Anderson 1989; Hansen et al. 1993). These studies, however, were focused exclusively on coated pits, disregarding all other membrane-associated vesicles. Hence, it can not be taken for granted that these conditions similarly impede other internalization routes. Clear ultrastructural demonstration has been lacking up to now. Using this superior technique, these previous findings on somatostatin receptors cannot be corroborated, at least for human glioma cells.
The alternative internalization route via caveolae or other endosomal vesicles has been suggested only for a few of the other seven transmembrane domain receptors (Raposo et al. 1989; Chun et al. 1994; Roettger et al. 1995; Wu et al. 1997). Caveolin, the integral membrane protein of caveolae, co-purifies with G-proteins and receptors with tyrosine kinase activity like that of insulin and neurotrophin receptors (Wu et al. 1997). Moreover, a specific interaction between caveolin and inactive Gα-subunits has been described (Li et al. 1995), the activation of the latter being impeded by caveolin. A cytosolic amino terminal domain of caveolins binds to trimeric Gα-subunits (Gαα and particularly Giα2), thus preventing activation of the Gα-subunit (Couet et al. 1997). According to Gu and Schonbrunn (1997), sst2A couples to Giα1, Giα2, and Giα3. In astrocytes, caveolae have been demonstrated by immunofluorescence microscopy of caveolin-1 and by conventional electron microscopy (Cameron et al. 1997).
In the present electron microscopic study we did not observe the receptor directly but only indirectly via one or two antibodies or via the labeled ligand. However, given a pH of 6.3-6.8 in early endosomal vesicles (Mellman 1996), antibody and ligand binding to gold can be accepted to be stable (Mentlein et al. 1990). Therefore, our experimental protocol appears to be a suitable model to determine possible internalization pathways of a G-protein-coupled receptor. In initial experiments on the internalization of somatostatin- gold by cultivated somatotrophs, we were also unable to find labeled coated pits, although the gold-labeled ligand was functionally active in inhibiting growth hormone release (Mentlein et al. 1989).
The expression of somatostatin receptors on gliomas cannot be associated only with malignancy. In addition to neurons (Krisch 1994), astrocytes also bear somatostatin receptors on certain subpopulations in situ and in vitro (Mentlein et al. 1990; Krisch et al. 1991). In cultured astrocytes, somatostatin has been shown to induce several signal transduction pathways. Activation of sst inhibits adenylate cyclase activity (Feindt et al. 1995), stimulates phospholipase A2 activity (Krisch and Mentlein 1994), and activates a protein phosphatase that inhibits the autophosphorylation of the receptor for epidermal growth factor (unpublished observations). As a result of the reduction in cAMP levels, somatostatin inhibits interleukin 6 release from astrocytes (Grimaldi et al. 1997). Antisecretory effects on gliomas have not yet been described.
In conclusion, in glioma cells the sst2A receptor is an example of a further G-protein-coupled seven transmembrane-domain receptor that after ligand binding is not internalized via coated pits but via uncoated endosomal vesicles.
Footnotes
Acknowledgments
Supported by grants from the Deutsche Forschungsgemeinschaft (Kr 569/7-1) and the Hensel Foundation.
We are greatly indebted to Ms Martina Burmester, Ms Dagmar Freier, and Ms Helga Prien for skillful technical assistance.
This work is dedicated to Prof Dr Helmut Leonhardt on the occasion of his eightieth birthday.
