Abstract
Somatostatin (somatotropin-release inhibitory factor, SRIF) exerts multiple inhibitory actions throughout the central nervous system and the periphery by binding to specific membrane-bound SRIF receptors (sstrs) of which five subtypes (sstr1–5) have now been identified. Individual sstr subtypes have been suggested to mediate selective biological actions of SRIF. Although the adrenal gland is a known target of SRIF action, the sstr subtypes involved in its actions are unclear. This study examined the expression of sstr1–5 in rat adrenal gland by RT-PCR analysis and in situ hybridization (ISH) histochemistry. Using RT-PCR expression combined with Southern blotting, sstr1, −2, −4, and −5 mRNAs were shown in the adrenal gland. ISH histochemistry revealed strong expression of sstr2 mRNA alone localized to the zona glomerulosa of the adrenal cortex and moderate labeling in scattered cells of the adrenal medulla, indicating a possible role for sstr2 in mediating SRIF physiology in this tissue by altering adrenal aldosterone and catecholamine secretion. These data also point to potential roles for sstr subtypes sstr1, −4, and −5 in the adrenal gland.
S
The adrenal gland is a known target of SRIF action, where it inhibits angiotensin II-stimulated aldosterone secretion in collagenase-dispersed rat adrenal glomerulosa cells (Aguilera et al. 1981) and acetylcholine-stimulated catecholamine secretion (Patel 1999). Immunoreactive SRIF has also been demonstrated in the adrenal capsule (Aguilera et al. 1981), and subsequent localization of SRIF receptors to the zona glomerulosa (Aguilera et al. 1982; Srikant and Patel 1985) suggests that locally produced SRIF may exert autocrine and/or paracrine regulatory effects on target cell responses to endocrine stimulation. SRIF-binding sites have also been visualized in the adrenal medulla, but little or no SRIF-like immunoreactivity has been described in these cells (Maurer and Reubi 1986a,b).
Contradictory information regarding individual sstr mRNA distribution in the rat adrenal gland has been reported (Kong et al. 1994; Raulf et al. 1994; Patel et al. 1995), and the sstr subtypes mediating SRIF actions in this tissue are unclear. Determination of the sites of expression of the mRNAs of the sstr subtypes may enable a correlation to be made between receptor gene expression with known physiology and pharmacology and thus help define the potential role of each subtype in mediating the physiological effects of somatostatin in the adrenal. To this end, the receptor distribution and the exact cellular localization of the five sstr subtypes in the male adrenal gland was investigated by RT-PCR and by in situ hybridization (ISH) histochemistry using subtype-specific cRNA probes.
Materials and Methods
RNA Extraction and RT-PCR
Total RNA from rat pituitary and rat adrenal gland was extracted by the RNAzol B method and quantified by UV absorption. Before RT, residual genomic DNA was removed by digestion of with RQ DNase (Promega; Madison, WI). RQ DNase (5 U) and RNasin (10 U; Promega) were added to the RNA samples (∼2 μg) and digestion was carried out at 37C for 15 min. Reactions were terminated by phenol extraction and the RNA was precipitated with ethanol and resuspended in 15 μl of distilled water. The DNase-treated rat pituitary and adrenal RNAs were reverse transcribed in 20 μl reaction volumes using random hexamer oligonucleotide primers (100 pmol) and Superscript II reverse transcriptase (100 U; BRL, Gaithersburg, MD). Corresponding control samples containing no reverse transcriptase were included for each tissue. After an initial incubation for 10 min at room temperature (RT), the reactions were allowed to proceed at 42C for 45 min and were then heated to 95C for 5 min. cDNA was stored at −20C and 1-μl aliquots were used for PCR.
PCR was carried out using primers for rat sstr1–5 (see Park et al. 2000) and β-actin (sstr1 sense, 5′-CTA CTT TGC CGC CTG GTG CTC-3’ and antisense 5′-TGG CAA TGA TGA GCA CGT AAC-3′, GenBank accession number (ACC) X62314; sstr2 sense, 5′-TTG ACG GTC ATG AGC ATC G-3’ and antisense 5′-ACA GAC ACG GAC GAG ACA TTG-3’ (ACC M93273); sstr3 sense 5′-GGC CGC TGT TAC CTA TCC TTC-3’ and antisense 5′-GGC ACT CCT GAG AAC ACA ACC-3’ (ACC X63574); sstr4, sense 5′-CGG AGA CGC TCA GAG AAG AAG-3’ and antisense 5′-TGG TCT TGG TGA AAG GGA CTC-3’ (ACC M96544); sstr5 sense 5′-CAT GAG TGT TGA CCG CTA CC-3’ and antisense 5′-GGC ACA GCT ATT GGC ATA AG-3’ (ACC L04535); β-actin sense 5′-CGT GAA AAG ATG ACC CAG AT-3’ and antisense 5′-ATT GCC GAT AGT GAT GAC CT-3’ (ACC V01217)). The expected sizes of PCR products were 354 base pairs (bp) for sstr1, 449 bp for sstr2, 555 bp for sstr3, 409 bp for sstr4, 508 bp for sstr5, and 411 for β-actin. Amplifications were carried out in a reaction volume of 50 μl with 10 pmol of each primer, 0.2 mM deoxyNTPs, and Advantage 2 polymerase (Clontech; Palo Alto, CA). The thermal cycling profile for sstr1–5 was 95C for 3 min, 30 cycles of 94C for 45 sec, 68C for 1 min, and 72C for 45 sec, followed by a final extension of 72C for 10 min. Additional amplifications for sstr4 were performed as above using 40 PCR cycles. The thermal cycling profile for β-actin was 95C for 3 min, 25 cycles of 94C for 45 sec, 60C for 1 min, and 72C for 30 sec, followed by a final extension of 72C for 10 min. Products were analyzed on 1.4% agarose gels containing 1 μg/ml ethidium bromide. The authenticity of the products was verified by both restriction digestion and sequence analysis after subcloning into the pGEM-4Z vector (Promega).
Southern Blotting
Standard protocols (Bonner et al. 1987) were followed for Southern blotting onto nitrocellulose membranes. Several 48-bp oligonucleotides (sstr1, 5′-GTC TTC TCA CGC ACC GCA GCC AAC AGC GAT GGC ACG GTG GCC TGC AAC-3′; sstr2, 5′-ATA TAC GCT GGC CTC CGG AGC AAC CAG TGG GGT AGG AGC AGC TGC ACC-3′; sstr3, 5′-GAC ACG TCC CTG GGG AAT GCA TCT GCT GGC ACT AGC CTG GCA GGA CTG-3′; sstr4, 5′-CGC TGC TGT CTC CTG GAA ACA ACT GGA GGT GCT GAG GAA GAG CCC CTG-3′; sstr5, 5′-CGG ATG TCC AGG AGG GCT GGG GCA CCT GCA ACC TGA GCT GGC CAG AGC-3′), specific for each of sstr-1–5, were synthesized and labeled using [γ-32P]-ATP and T4 polynucleotide kinase. Membranes were washed at 60C in 1 X SSC/0.1% SDS and exposed to Kodak XAR film.
Preparation of Probes for ISH Histochemistry
The rat sstr constructs used to generate 35S-labeled riboprobes were made as follows. sstr1 (631 bp) and sstr4 (436 bp) probes were generated by PCR using ∼1 μg of plasmid DNA from a rat cortex cDNA library, and while the sstr-3 probe (656 bp) was generated using ∼1 μg of rat genomic DNA (Promega). The PCR products were gel-purified and subcloned into pGEM-4Z. The resulting transcripts were complementary to bases 798–1429 of rat sstr1, bases 1621–2277 of rat sstr3, and bases 1028–1464 of rat sstr4. sstr-2 (789 bp) and sstr5 (800 bp) probes were generated by PCR using ∼1 μg of plasmid DNA from a rat pituitary cDNA library plasmid DNA. The PCR products were gel-purified and subcloned into pGEM-3Z (Promega). The resulting transcripts were complementary to bases 1214–2003 of rat sstr2 and bases 844–1644 of rat sstr5.
For all five sstr subtypes, sense and antisense riboprobes were generated using T7 and SP6 polymerases with [35S]-UTP and the MAXIscript in vitro transcription kit (Ambion; Austin, TX). The integrity of the probes was checked by restriction enzyme digests and DNA sequencing. The specificity of the probes had been described previously (O'Carroll and Krempels 1995).
ISH Histochemistry (ISHH)
Sections (12 μm) of tissues from adult (180–200 g) Wistar rats were thaw-mounted onto polylysine-coated slides and stored at −80C until hybridization. Contralateral adrenals of the same rats used for RT-PCR were used for ISHH. Hybridization was carried out as described previously (Bradley et al. 1992) using 35S riboprobes to detect all five sstr mRNAs. Briefly, the 35S-labeled probe (∼1 × 106 cpm) was applied to the sections for 18 hr at 55C. After hybridization the sections were washed four times for 5 min with 4 X SSC at RT, 30 min at 37C in RNase solution (20 μg/ml RNase A in 0.5 M NaCl, 10 mM Tris-HCl, pH 8.0, 0.25 mM EDTA), twice for 5 min in 0.1 X SSC/1 mM DTT at RT, and twice for 30 min at 65C in 0.1 X SSC/1 mM DTT. All slides were quickly rinsed in distilled water, dehydrated in a graded alcohol series, and dried. Slides were exposed to Kodak XAR film for 5 weeks at RT, dipped in Ilford K5 nuclear emulsion to localize the cellular distribution of silver grains, and stored desiccated at 4C for 4 months before development using Kodak D19 at RT. Tissue sections were counterstained with toluidine blue and slides were viewed by light/darkfield microscopy. No specific hybridization was detected with the sense probes.
Analysis
Five adjacent sections from adrenals from four animals were used for ISH with the five sstr riboprobes. Cells positive for sstr mRNA were identified by clusters of silver grains over cells.
Results
RT-PCR Expression of sstr Subtypes in Adult Adrenal Gland
RT-PCR, using subtype-specific primers, was used to characterize the expression of mRNA encoding sstr1–5 in the rat adrenal gland. The expression of sstr1–5 in the rat pituitary was included as a positive control. Expression of all five sstr subtype mRNAs was seen in the pituitary (Figure 1A) as previously reported (O'Carroll and Krempels 1995). The signal for sstr4 mRNA expression in the pituitary using 30 PCR amplification cycles was weak but was clearly visualized after PCR for 40 cycles. Strong sstr2 mRNA expression was identified in the adrenal gland while a weaker signal for sstr5 mRNA was observed (Figure 1B). mRNAs for sstr1, −3, and −4 were not detectable in this tissue after either 30 or 40 PCR amplification cycles. Corresponding control samples subjected to PCR without prior reverse transcription yielded no detectable levels of sstr1–5 mRNAs. β-Actin mRNA was also amplified from the same cDNA samples and was present in all samples tested (Figures 1A and 1B).
Southern Blotting
Southern blotting hybridizaton of the RT-PCR products with internal sstr subtype-specific probes was performed to increase the detection limit of the PCR products compared to ethidium bromide staining and to introduce an additional subtype specificity check. Expression of all five sstr subtype mRNAs was seen in the pituitary (Figure 1C, Lane 1). Strong sstr2 mRNA expression was identified in the adrenal gland and weaker signals for sstr1, −4, and −5 mRNAs were observed after Southern blotting of RT-PCR products (Figure 1C, Lane 2). mRNAs for sstr3 were not detectable in the adrenal gland.
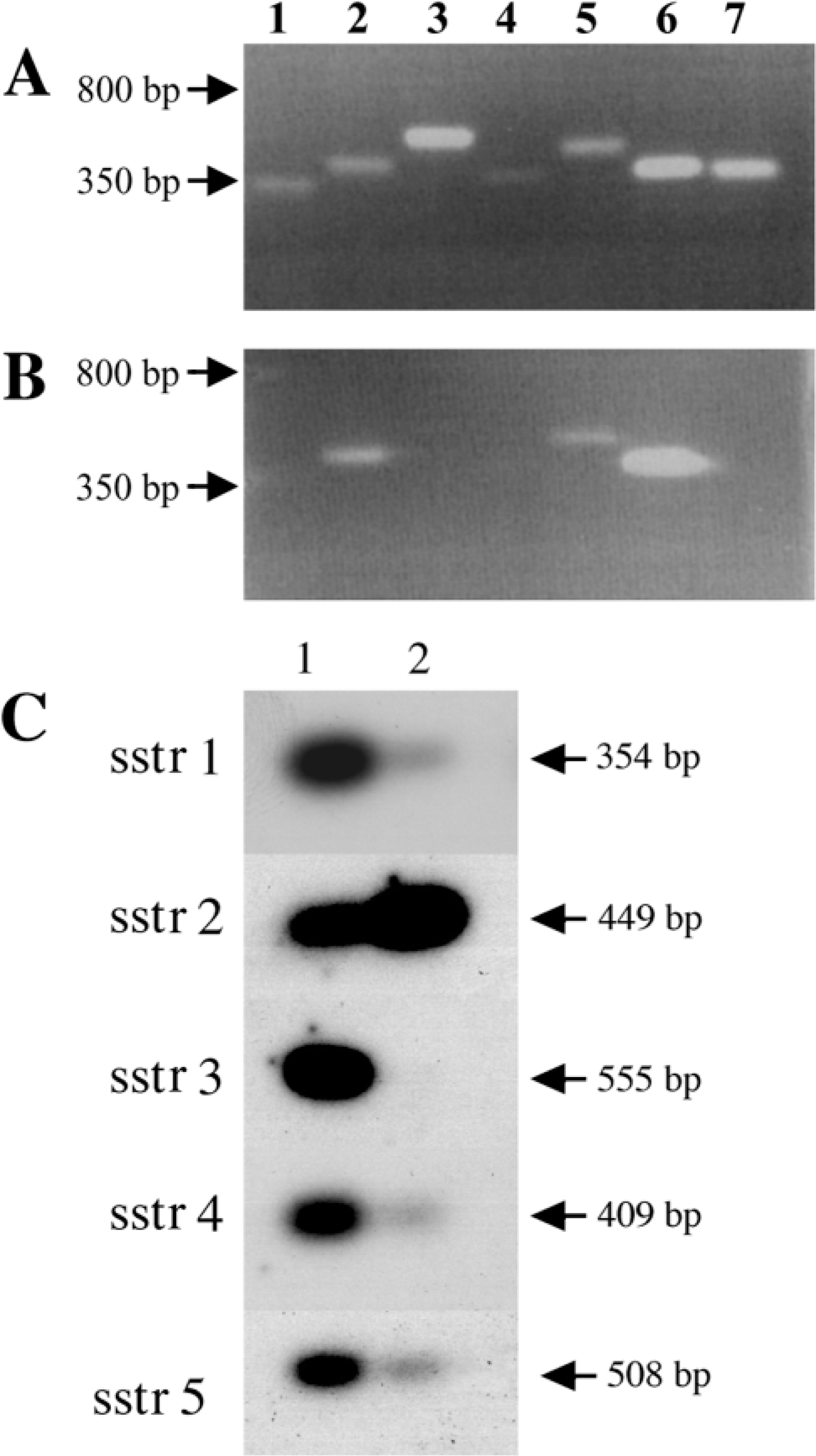
RT-PCR of sstr-1–5 expression in adult rat. PCR products of sstr subtype-specific reactions in adult rat (
Localization of sstr Subtypes in Adult Adrenal Gland
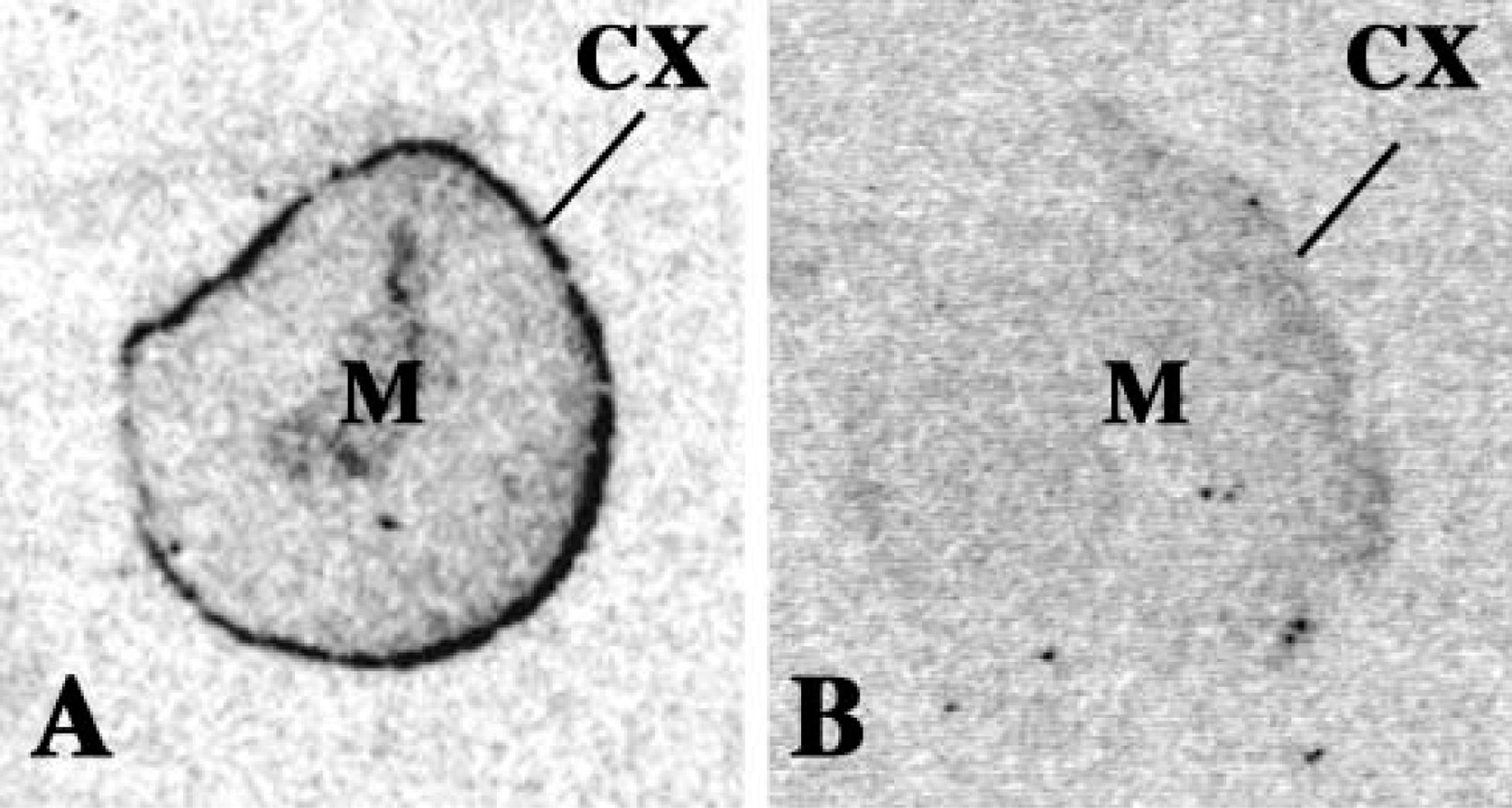
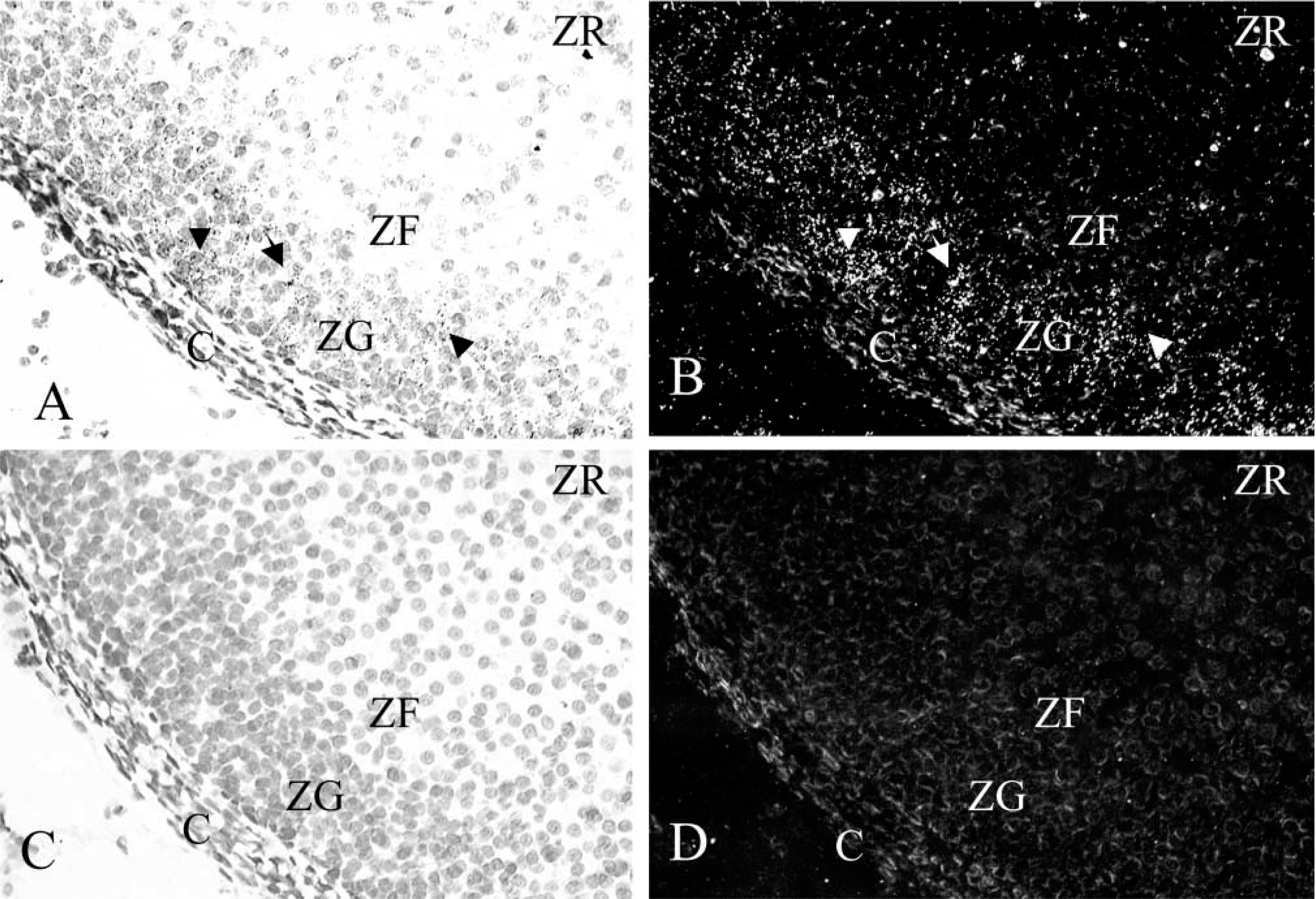
The localization and distribution of sstr1–5 mRNAs in the rat adrenal gland was also studied by ISHH. 35S-Labeled antisense and sense riboprobes were used for this study. Distribution of sstr1–5 mRNA expression in classical SRIF target tissues, i.e., brain, pituitary, kidney, and stomach, was examined in parallel to the adrenal as positive controls and expression patterns were as described in the literature (data not shown). Strong hybridization of the sstr2 antisense probe was seen in the cortical region of the adrenal gland (Figure 2), whereas the mRNAs for sstr1, −3, −4, and −5 were not detectable over nonspecific background (not shown). sstr2 labeling was also observed in isolated cells scattered throughout the adrenal medulla (Figure 2). High-power micrographs show sstr2 mRNA distribution localized to the zona glomerulosa of the cortex, where intense labeling was observed (Figures 3A and 3B), but no labeling was seen in the zona fasciculata, zona reticularis, or adrenal capsula.
Discussion
SRIF is a potent physiological regulator of endocrine secretion and exerts its biological effects via five receptor subtypes (sstr1–5). The presence of high-affinity binding sites for SRIF has been demonstrated in homogenates of rat adrenal zona glomerulosa (Aguilera et al. 1982; Srikant and Patel 1985), consistent with the demonstration of high in vivo uptake of labeled SRIF in the adrenal capsule (Aguilera et al. 1982). Binding sites for SRIF have also been described in autoradiographic studies on frozen rat adrenal medulla sections, but [125I-Tyr11]-SRIF binding has not been detected in rat adrenal medulla membrane preparations (Maurer and Reubi 1986a,b). Moreover, SRIF-like immunoreactivity has been demonstrated in the zona glomerulosa of the rat adrenal cortex (Aguilera et al. 1981) and in the norepinephrine- and epinephrine-containing cells of the adrenal medulla (Morel et al. 1990). The localization of SRIF receptor binding and SRIF-like immunoreactivity in the zona glomerulosa and in the adrenal medulla is consistent with the known inhibitory actions of SRIF on angiotensin II-stimulated aldosterone production and inhibition of acetylcholine-stimulated catecholamine secretion in the rat adrenal gland, respectively.
Localization of sstr2 mRNA expression by ISH histochemistry in rat adrenal gland. The figures were scanned directly from the X-ray film. (
Typically, individual target cells for SRIF express multiple sstr subtypes, suggesting that the effects of SRIF may represent the composite activation of more than one sstr. However, there is evidence that individual sstr subtypes may mediate selective biological actions of SRIF. Recent studies suggest that most if not all of the antihormonal actions of SRIF are mediated via either sstr2 and/or sstr5 (Lamberts et al. 1996). SRIF receptors are expressed in a large number of tumors (Kennedy and Dluhy 1997; Hofland et al. 1999; Kaltsas et al. 2001; van der Harst et al. 2001). Conformationally stabilized SRIF analogues, such as octreotide (which binds with high affinity to sstr2 and −5), are used clinically to control humoral hypersecretion and tumor growth in patients with acromegaly, carcinoids, and pheochromocytomas (Berlowitz 1995). The majority of these tumors express mRNA for sstr2, and sstr5 mRNA is also expressed in the majority of GH-secreting (Greenman and Melmed 1994a,b) and gastroenteropancreatic tumors (Buscail et al. 1995). A recent study by Reubi et al. (2001), using receptor autoradiography with subtype-selective ligands, has identified predominantly sstr2 expression in pheochromocytomas and normal human adrenal medulla tissue, while a study by Hofland et al. (1999), using IHC detection of sstr1 and sstr2, reports expression of both these receptor subtypes in adrenal medulla tumors.
Conflicting information regarding individual sstr mRNA distribution in the rat adrenal gland has been reported in studies employing methodologies that have different detection sensitivities, e.g., RT-PCR, RNase protection assays, and Northern blotting analyses, and therefore the receptor subtypes involved in the actions of SRIF in this tissue are unclear. Specific expression of sstr-2 mRNA has been described in rat adrenal in one study (Kong et al. 1994), sstr-1, −2, and −5 mRNAs in another (Raulf et al. 1994), and a further study has described the expression of sstr1–3 mRNAs (Patel et al. 1995). sstr2 and −3 immunoreactivity has been visualized in the adrenal medulla (Mitsuma et al. 1996,1997) and, recently, sstr2 immunoreactivity has been described in the adrenal cortex (Kimura et al. 2001). Consistent with the expression of sstr2 and −5 in the adrenal gland is a study by Kong et al. (1994), which identified SRIF receptors in the rat adrenal cortex with a high affinity for MK 678 (Seglitide), an SRIF agonist that displays a high affinity only for sstr2 and −5 subtypes. The present study undertook to provide a comprehensive description of the distribution of the five sstr subtypes in the male rat adrenal gland to determine the receptor subtype(s) that may be responsible for mediating the effects of SRIF in this tissue and thus contribute to the establishment of treatments using SRIF analogues for neuroendocrine tumors.
High-power micrographs of sstr-2 mRNA expression in the adrenal cortex. Detailed labeling of the adrenal cortex shown at magnification X 400. (
In the present study, RT-PCR in combination with Southern blotting demonstrated that mRNA for sstr1, −2, −4, and −5 is expressed in the rat adrenal gland, suggesting a possible role for these subtypes in mediating SRIF physiology in this tissue. By ISH histochemistry, which determines the exact cellular localization of receptor mRNAs, expression of sstr2 mRNA alone could be demonstrated in the adrenal gland. No expression of sstr1, −3, −4, or −5 mRNAs could be detected above nonspecific background. High levels of sstr2 mRNA were found in the adrenal cortex, with expression localized to the cells of the zona glomerulosa, the exclusive site within the adrenal gland for the synthesis and secretion of aldosterone. sstr2 labeling was also observed in isolated cells scattered throughout the adrenal medulla. These observations are consistent with the known effects of SRIF on the adrenal gland and suggest a specific role for the sstr2 subtype in aldosterone production and in the regulation of catecholamine secretion. Based on the RT-PCR results from this study, however, it is likely that sstr1, −4, and −5 mRNAs, also present in the adrenal gland, are in a position to contribute to SRIF effects in this tissue. The lack of detection of these sstr subtypes by ISH histochemistry can be accounted for by the presence of their mRNAs at levels below the detection limit of this technique in the adrenal gland but which are detected with more sensitive methods such as RT-PCR.
In conclusion, these results demonstrate expression of sstr1, −2, −4, and −5 mRNAs in the rat adrenal gland. sstr2 mRNA expression in the zona glomerulosa and adrenal medulla of the rat adrenal gland indicates a possible role for this sstr subtype in altering adrenal aldosterone and catecholamine secretion. The roles of sstr1, −4, and −5 in regulating adrenal function have yet to be defined and may be clarified by IHC studies using receptor subtype-specific antibodies.
Footnotes
Acknowledgements
The author is grateful for the financial support of the Wellcome Trust.
