Abstract
Neuroendocrine neoplasias (NENs) are known to express somatostatin receptors (SSTRs) 1–5, which are G-protein-coupled cell membrane receptors. Somatostatin receptor imaging and therapy utilizes the SSTR expression. Synthetic somatostatin analogs with radioligands are used to detect primary tumors, metastases, and recurrent disease. Receptor analogs are also used for treating NENs. Furthermore, commercially available SSTR antibodies can be used for the immunohistochemical (IHC) detection of SSTRs. We investigated different SSTR antibody clones applying diverse IHC protocol settings to identify reliable clones and feasible protocols for NENs. A tissue microarray including NENs from 12 different primary sites were stained. Only UMB clones were able to localize SSTR on the cell membranes of NENs. SSTR2 (UMB1) emerged as the most common subtype followed by SSTR5 (UMB4) and SSTR1 (UMB7). SSTR3 (UMB5) expression was mainly cytoplasmic. Yet, SSTR4 expression was weak and located primarily in the cytoplasm. Thus, appropriate IHC protocols, including proper positive and negative controls, represent requirements for high-quality NEN diagnostics and for planning personalized therapy.
Introduction
Somatostatin receptors (SSTRs) are located on the plasma membrane and activated by an extracellular pan-inhibitor ligand, the 14- or 28-amino acid peptide hormone somatostatin. Binding of the ligand changes the receptor morphology and leads to the dissociation of the G-protein molecule. Cytoplasmic subunits (Gα and βγ) activate multiple intracellular signaling pathways with a broad range of physiological actions, including hormone production, cell proliferation, and angiogenesis. 1
In humans, five different SSTRs (SSTR1–5) are widely expressed, although that expression varies in the central nervous system and peripheral tissues. For example, pancreatic islet cells2–6 and dispersed neuroendocrine (NE) cells of the gastrointestinal tract express different SSTR subtypes.4–7 In addition, lymphoid cells and blood vessels have shown positivity for SSTRs.7,8
Neuroendocrine neoplasias (NENs), comprising neuroendocrine tumors (NETs) and neuroendocrine carcinomas (NECs), represent a rare and varying group of tumors showing characteristic neuroendocrine-specific histomorphology and immunoprofile. 9 NENs arise from diffusely distributed or clustered NE cells. Furthermore, several NENs express SSTRs.10–12 The expression profile and coexpression of various SSTR subtypes differ according to the NEN location, histological differentiation, and tumor grade.11,12
Somatostatin radioligands are used for tumor imaging, playing a major role in localizing primary tumors and in the identification of metastases. Moreover, somatostatin analog–based therapies of NENs utilize the expression of SSTRs. 13 The effect of SSTR targeting depends on the presence of appropriate SSTR subtypes. Positron emission tomography (PET) imaging with DOTA-coupled peptides is a sensitive and specific method to select patients for peptide receptor radionuclide therapy when an NEN diagnosis is confirmed. 13 An in vitro method, autoradiography, requires frozen tissues and a specialized laboratory setting for radioactive material and is, thus, not feasible for the SSTR subtype characterization in routine diagnostics.
Alternatively, immunohistochemistry (IHC) represents an ideal in vitro method for determining the SSTR expression in formalin-fixed, paraffin-embedded tissues since the tissue staining of NENs shows a good correlation with imaging and in vitro autoradiography.14–16 However, the IHC methodology has thus far proved problematic. Commercially available monoclonal antibodies against SSTRs exist, but the quality of such stainings remains variable and partly unreliable. Therefore, IHC protocols need to be optimized and verified. Here, we aimed to identify and verify suitable IHC protocols for studying the different SSTR subtypes by using different primary antibody clones and staining instruments coupled with high-quality polymer-based detection kits on a multi-NEN tissue microarray (TMA) platform.
Materials and Methods
Tissue Material
Tumor material from 2002 to 2010, except one rectum NET G2 from 1997, were retrieved from the Department of Pathology (HUSLAB, Helsinki University Hospital, Helsinki, Finland). 17 Tumors were re-evaluated by an experienced endocrine pathologist (JA) on hematoxylin-eosin slides and their NE differentiation was confirmed through immunohistochemical chromogranin A and synapthophysin stainings. A multi-NEN TMA consisting of 12 different primary NENs (lung, thymus, gastric mucosa, pancreas, appendix, small intestine, rectum, thyroid, parathyroid, adrenal gland, paraganglia, and skin; n = 91) was constructed as previously described. 17 Tumor material as well as control tissues (normal colon, small intestine, pancreas, pituitary, adrenal gland, and thyroid including C cells) were dissected from surgical material. All the tissues were fixed in 10% formalin for 24–48 h followed by routine tissue processing, microtomy cutting and hematoxylin-eosin staining. The study protocol followed the principles of the Helsinki Declaration of 1975 and was approved by the Ethics Committee of Helsinki University Hospital (3990/04/046/07).
Immunohistochemistry
Table 1 summarizes the details of the antibodies and protocols tested. Briefly, normal tissue and multi-NEN TMA sections (3 µm) were cut onto TOMO® hydrophilic adhesion slides (Matsunami Glass, Osaka, Japan) and dewaxed. The expression of SSTR subtypes 1–5 was studied using nine different commercially available monoclonal antibodies applying different pre-treatment protocols and antibody dilutions. Polymer-based detection kits (Envision, Agilent, Santa Clara, CA, and UltraVIEW DAB and OptiVIEW DAB, Ventana, Tucson, AZ) eliminating cross-reactions to endogenous biotin were used. Staining was performed using two different staining instruments (LabVision Autostainer 480; Thermo Scientific, Fremont CA, and BechMark XT; Ventana, Tucson, AZ).
Primary Antibodies and Staining Settings in the Verification of Immunohistochemical Somatostatin Receptor 1–5 Staining Protocols.
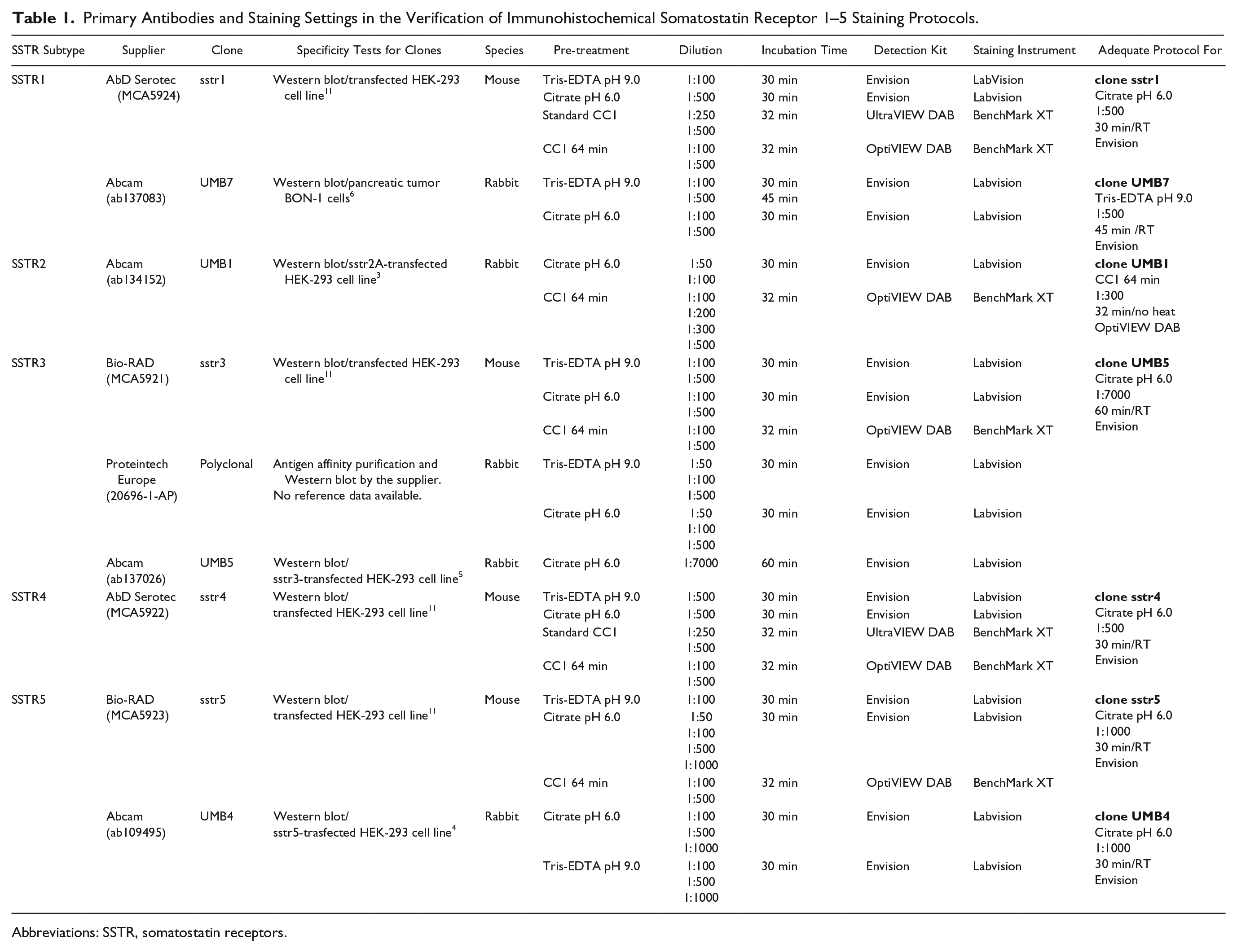
Abbreviations: SSTR, somatostatin receptors.
Standard protocol for Envision included blocking of endogenous peroxidase for 5 min before primary antibody, incubation of secondary antibody for 30 min and DAB for 10 min in room temperature. Washes were done with 1XPBS with 0.5% Tween 20 (Acros; Geel, Antwerp, Belgium) after incubation steps. With UltraVIEW and OptiVIEW DAB, standard protocols included inhibition of endogenous peroxidase for 4 min, incubation of secondary antibody for 8 min in UltraVIEW DAB protocol or 8–12 min in OptiVIEW DAB protocol, incubation of DAB chromogen with substrate and copper for 8 and 4 min, respectively. Washes were done with Reaction Buffer (Ventana, Tucson, AZ) according to instruments programming.
The quality assessment of the protocol settings and the clones used were based on normal tissues known to express SSTRs (the pancreas and the small intestine), as well as on an internal negative control. The parameters of the protocol were adequate when the maximum staining was in the correct tissue localization with none or a minimal amount of non-specific background staining. 18
IHC Scoring
SSTRs were scored independently by two researchers (SR and HL). Both membranous and cytoplasmic staining were observed and scored according to the criteria adapted from Elston et al. 19 and Körner et al. 15 Membranous staining was scored as 0 = no staining, 1 = weak membrane positivity of <10%, 2 = weak to moderate membrane positivity of ≥10%, 3 = moderate to strong complete circumferential membrane positivity, and 4 = strong membrane complete circumferential positivity >95% (Fig. 1). Cytoplasmic staining was scored as 0 = negative, 1 = weak, 2 = moderate, and 3 = strong. Figure 2 visualizes the different SSTR stainings. The highest consensus score for a membranous pattern (scores 2–4) and cytoplasmic staining, intensity levels 2 and 3, were considered appropriate.
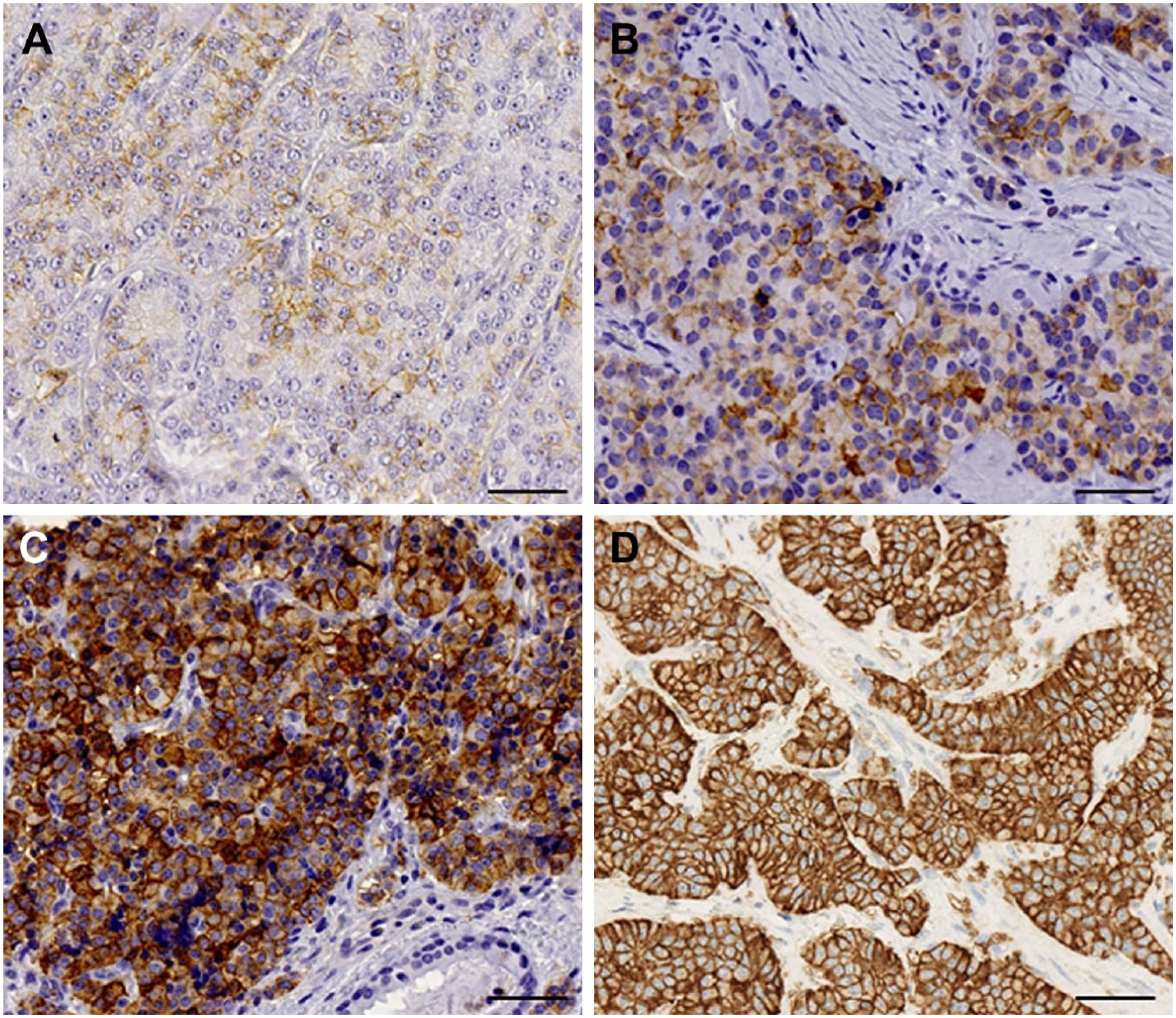
Scoring criteria for membranous staining. Score 1, weak partial membranous positivity in less than 10% of tumor cells (A). Score 2, weak to moderate partial membranous positivity in more than 10% of the tumor cells (B). Score 3, moderate to strong circumferential membranous positivity (C). Score 4, strong circumferential membranous positivity in more than 95% of the tumor cells (D). Scale bar: 50 µm; magnification: 40×.
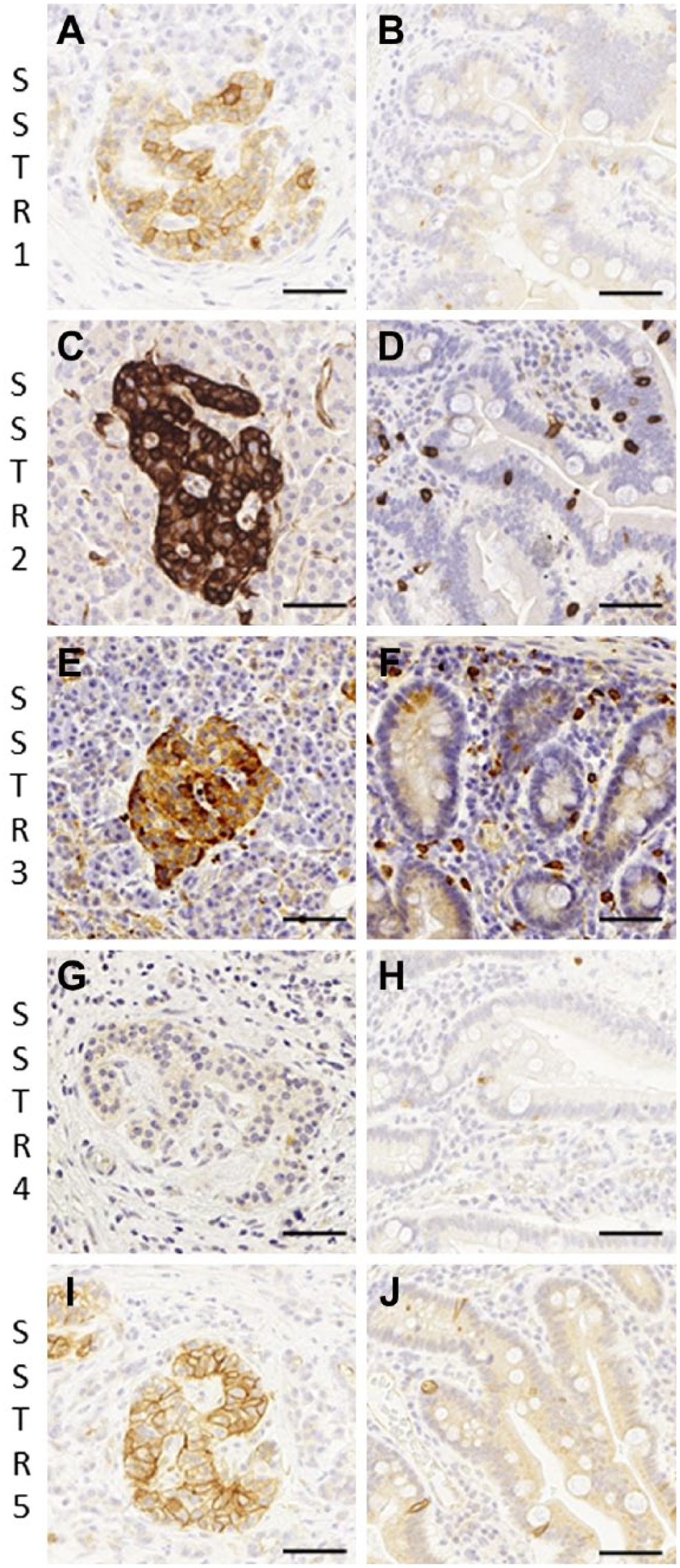
Somatostatin receptor 1–5 expression in pancreatic (A, C, E, G, I) and small intestine (B, D, F, H, J) neuroendocrine cells. Scale bar: 50 µm; magnification: 40×.
Results
SSTR Expression in Normal Tissues
Overall, we found that different tissues expressed SSTR subtypes differently, where SSTR2 represented the most common receptor type, using preferable clones and protocols. The staining pattern was membranous and/or cytoplasmic depending on the receptor subtype. NE cells in the large intestine expressed SSTR2 and SSTR5, while NE cells in the small intestine expressed SSTR1, SSTR2, SSTR4, and SSTR5. SSTR3 expression was largely absent. The pituitary gland expressed SSTR2, SSTR3, and SSTR5, while SSTR1 and SSTR4 expression was absent. The adrenal medulla stained positively for SSTR2 and SSTR3, while some adrenocortical cells were also positive for SSTR subtypes 2, 3, and 5. SSTR1 showed a nuclear positivity in the adrenal medulla and cortex. Furthermore, the thyroid C cells were weakly positive for SSTR2, SSTR3, and SSTR5. The pancreatic islet cells were positive with varying intensities for SSTR1, SSTR2, SSTR3, and SSTR5, while SSTR4 was found mainly in the exocrine pancreas. Depending on the receptor subtype and tissue, we found that the nerves, ganglion cells, the luminal site of ducts or mucosal surface, as well as mast cells or endothelial cells were also stained. Figure 2 shows the SSTR subtype distribution in pancreatic and small intestine NE cells.
Expression of SSTRs in NENs
The expression profile and pattern of different SSTRs depended of the antibody clone used: both membranous and cytoplasmic staining were seen as summarized in Table 2. The preferable clone showing membranous staining for SSTR1 was clone UMB7, for SSTR2 clone UMB1, for SSTR3 clone UMB5, and for SSTR5 clone UMB4. SSTR4 clone sstr4 yielded cytoplasmic staining.
Somatostatin Receptor 1–5 Expression in Neuroendocrine Tumors.
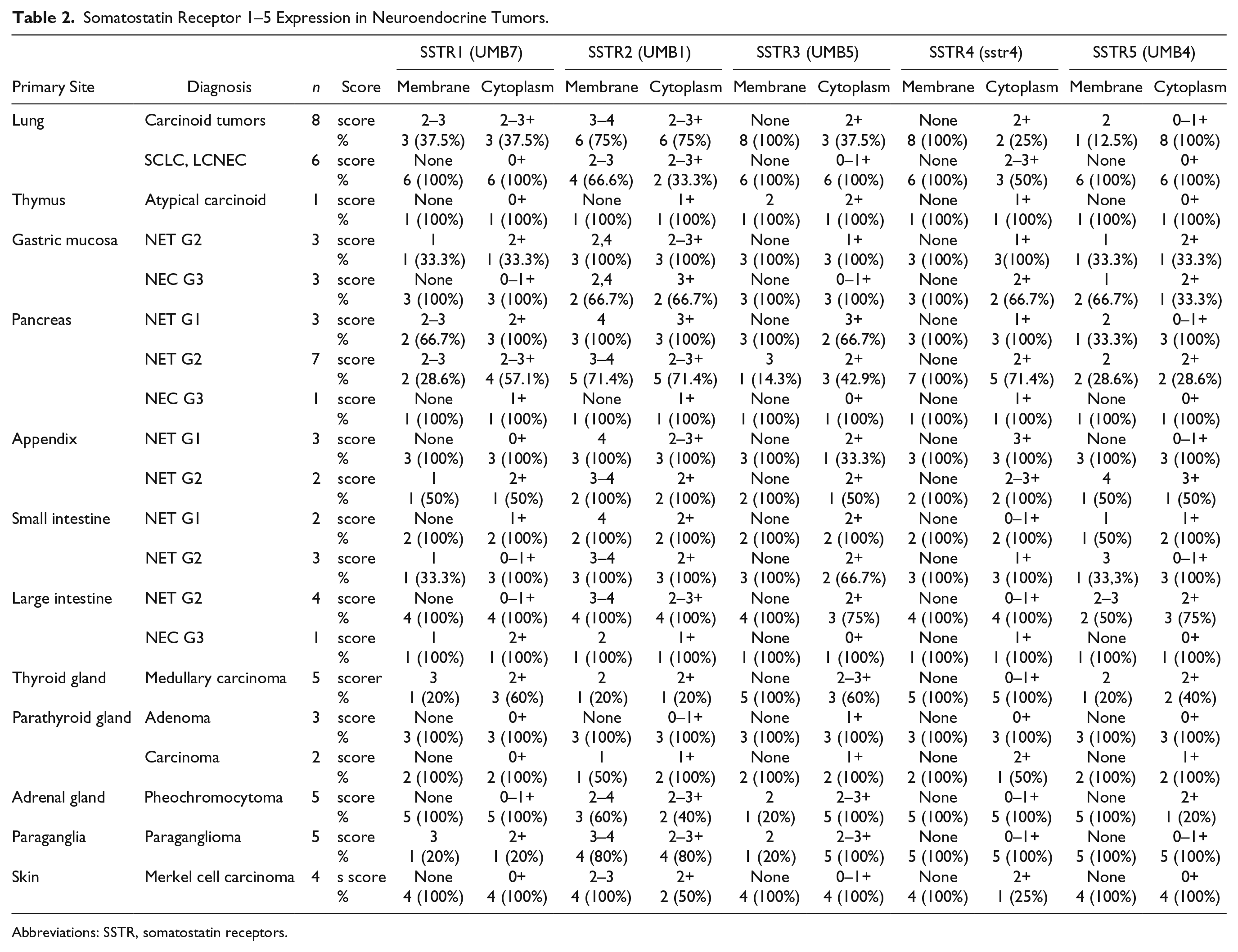
Abbreviations: SSTR, somatostatin receptors.
Briefly, SSTR2 (UMB1) was found in the majority of NENs. SSTR5 (UMB4) and SSTR1 (UMB7) were the next most common subtypes expressed, although at a weaker level than SSTR2. In addition, SSTR3 was negative in most NENs. However, the thymus, pheochromocytoma, and paraganglioma showed mainly cytoplasmic staining (Table 2). Furthermore, SSTR expressions decreased in high-grade tumors with a poor differentiation (small-cell lung cancer, large-cell NEC, gastric mucosa, pancreas, and colorectum; Table 2).
Discussion
The present study compared several commercial primary antibody clones for SSTR subtypes, revealing the IHC expression profiles in normal tissues and in a large NEN material from different primary sites. IHC protocols for each SSTR clone were verified based on the use of positive control tissues known a priori to express SSTRs. We found that different SSTR clones exhibit different staining patterns, thereby affecting the overall staining results. Overall, SSTR2 represented the most universal receptor subtype in NENs. In addition, tumors from different primary origins exhibit different SSTR profiles.
SSTRs are located on cell membranes containing seven transmembrane-spanning alpha helical regions, the extracellular C-terminal, and the intracellular N-terminal. In our normal tissue and NEN material, we found both membranous and cytoplasmic staining, supporting previous observations.11,19,20 The staining result depended on the choice of clone; UMB clones showed mostly membranous staining, as shown in earlier studies.10,19–21 Interestingly, clone sstr5 stained cell membranes in normal tissues, but in tumor cells the staining pattern was only cytoplasmic despite using the same staining protocol. The cytoplasmic staining pattern was also common in certain UMB clones, particularly in UMB5 (SSTR3), which showed mostly cytoplasmic staining in our normal tissue and tumor material. Other antibodies for SSTR3 yielded too strong background staining without a proper IHC expression profile. The clone sstr4 used in this study was not suitable for a demonstration of SSTR4 given our staining location requirements.
The SSTR expression profiles vary according to the NEN origin, differentiation, and tumor grade, an observation supported in our studies.10–12,22 Cytoplasmic staining has been accepted as sufficiently specific in previous reports on SSTR expressions, although we considered only membranous positivity as the correct and reliable staining pattern for SSTRs, 23 even though we also scored the cytoplasmic staining. Furthermore, SSTR2 (UMB1) was the most frequently detected membranous receptor subtype followed by SSTR5 (UMB4) and SSTR1 (UMB7). SSTR3 (UMB5) showed membrane positivity only in NENs of the thymus and the pancreas as well as in pheochromocytomas and paragangliomas. No NENs showed a membrane positivity for SSTR4. After binding the receptors to a natural ligand or an agonist, SSTRs are internalized to the cytoplasm. 24 Thus, the cytoplasmic staining seen in our tumor material might result from the biological SSTR internalization or from features of the available antibodies. It is also possible that a somatostatin analog therapy given to some patients before surgery affected the tissue expression of SSTRs.
SSTR antagonist therapy and tumor load imaging rely on the membranous expression of the corresponding receptor. Patients suitable for radionuclide and/or SSTR-analog treatment are selected using scintigraphy or PET/computed tomography scanning. 13 Treatment outcomes depend on the SSTR expression profile and the intensity of the tumor. Unfortunately, some NENs, such as insulinomas, remain difficult to detect through imaging. The expression of SSTRs may also decrease as the disease progresses. 13 Several studies found a correlation between IHC and autoradiography,15,25 scintigraphy, 16 and PET/CT. 14 These methods rely on the tumor receptor density on the cell membranes. IHC demonstrating the membranous SSTR-staining intensity and distribution in resected NEN specimens appears to represent a convenient method predicting treatment success.
A high-quality setting for an IHC protocol is advisable. Because SSTRs are membranous receptor proteins, only membranous positivity can be considered useful for predictive purposes. 23 We found that all tested clones, except for sstr4 for SSTR4 and the polyclonal SSTR3s, could localize to the correct cell types, although not all clones could stain the cell membranes. Using proper IHC protocol methods and clones, together with the appropriate quality control and interpretation,18,23 we argue that analysis of SSTR status using IHC is a useful method to identify the NEN receptor profiles.
One strength of our study lies in the well-characterized NEN TMA panel used. 17 However, the number of each tumor type remained quite limited and the results with standardized IHC settings need to be confirmed in a larger series of primary organ specific tumors. Furthermore, our group has extensive experience in IHC and the optimization of staining protocols supporting our results and conclusions on the staining specificity.
In conclusion, NENs exhibit different and variable SSTR profiles depending on their differentiation as well as site of origin. The verification of protocols and the choice of clones together with proper control tissue are mandatory for the standardization of SSTR IHC analyses. Further research aimed at determining the role of SSTR IHC positivity in predicting treatment responses to different SSTR analogs is needed.
Footnotes
Acknowledgements
We gratefully acknowledge Eija Heiliö and Päivi Peltokangas for their technical assistance.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SR performed the immunohistochemistry. SR, HL, and TV interpreted the staining results. JA and CH designed the study. SR and TV wrote the manuscript. All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Finnish Cancer Foundation (no grant number available) and the Helsinki University Hospital Research Fund (grant number TYH2017204).
