Abstract
Gross cystic disease fluid protein-15 (GCDFP-15)/prolactin-inducible protein (PIP) is present at moderate levels in human submandibular and sublingual glands and is barely detectable in human parotid gland. The rodent homologue, PIP, has previously been identified in adult submandibular and lacrimal glands. Here we present the molecular characterization of rat PIP and show that this protein is a product of neonatal and adult rat submandibular, sublingual, and parotid glands. cDNA clones encoding rat PIP were isolated and sequenced. The deduced amino acid sequence of rat PIP shows 56% overall identity and 80% similarity with mouse PIP. By SDS-PAGE, secreted rat PIP has an apparent Mr of 17,000, with a minor proportion present as Mr 20–22,000 N-glycosylated forms. PIP was localized in rat salivary glands by immunogold silver staining. PIP was identified in acinar cells of developing and mature submandibular and parotid glands and at very low levels in sublingual gland serous demilunes. Typically, rat submandibular gland secretory proteins are produced by either acinar cell progenitors (Type III cells) or mature acinar cells. The expression pattern observed for PIP is similar to that previously reported for salivary peroxidase, an important component of nonimmune mucosal defense.
Keywords
G
By cDNA (Haagensen and Mazoujian 1986; Murphy et al. 1987) and protein (Schaller et al. 1991) sequence analyses, human PIP was shown to be 146 amino acids in length, with probable signal peptide cleavage after residue 28. The resulting secreted protein has a calculated molecular mass of 13,506, and contains one N-glycosylation consensus sequence. Secreted PIP contains from 10 to 22% carbohydrate (Haagensen 1981; Rathman et al. 1989; Schaller et al. 1991), present exclusively as N-linked oligosaccharides (Rathman et al. 1989; Autiero et al. 1991; Schaller et al. 1991; Schenkels et al. 1994). Carbohydrate microheterogeneity results in the presence of multiple PIP forms in human secretions (Autiero et al. 1991; Schenkels et al. 1991, 1994) and T-47D breast carcinoma cells. The protein forms dimers in saliva (Rathman et al. 1989) and tetramers in breast cyst fluid (Haagensen et al. 1979) and seminal plasma (Autiero et al. 1995).
Several functions for PIP have been suggested. PIP binds to many proteins, including fibrinogen (Haagensen 1981), actin (Akiyama and Kimura 1990), keratin, myosin, and tropomyosin (Schenkels et al. 1994). Salivary PIP also binds to hydroxyapatite, the predominant component of tooth enamel (Rathman et al. 1989), suggesting that it may participate in formation of the enamel pellicle. PIP may modulate bacterial colonization because it binds to oral bacteria of the genera Streptococcus and Gemella, and to Staphylococcus hominis isolated from human skin and ear canal (Schenkels et al. 1997). PIP from seminal plasma binds the T-cell transmembrane glycoprotein CD4 with high affinity. A possible function of the observed CD4 binding is modulation of maternal immunity at insemination. However, PIP interferes with HIV envelope protein gp 120 interaction with CD4 and therefore may also inhibit HIV infection (Autiero et al. 1991,1995,1997). A different role for PIP was suggested by the finding that this protein is mitogenic for several breast cancer cell lines (Cassoni et al. 1995).
PIP gene expresion is hormonally regulated in human breast cancer cells. PIP expression is increased by prolactin and steroid hormones, with dihydrotestosterone the most effective (Murphy et al. 1987). Estrogen, however, decreases basal PIP expression levels and inhibits the stimulatory effects of androgen (Simard et al. 1989). PIP expression is also regulated by interleukins; expression is increased by IL-1, IL-4 and IL-13 and is decreased by IL-6 (Blais et al. 1994,1995,1996).
cDNA clones for the mouse homologue of PIP have been identified (Windass et al. 1984; Myal et al. 1994). In rat and mouse, PIP transcripts were demonstrated in adult submandibular and lacrimal glands, but not in skin containing sweat glands (paws, ears, tail) or in lactating mammary gland or mammary gland tumors. Hormonal regulation of PIP expression in rodent submandibular and lacrimal glands is also different from that in human breast cancer cells. Neither androgen nor prolactin affects PIP expression in rodent submandibular gland. In lacrimal gland, androgen inhibits PIP expression, whereas prolactin, again, has no effect (Myal et al. 1994).
Our laboratories have studied gene expression in developing rodent salivary glands. Neonatal rat sub-mandibular glands contain acinar cell progenitors that produce the developmentally regulated secretory proteins parotid secretory protein (PSP), SMGB (glycoforms SMGB1 and SMGB2), and common salivary protein-1 (CSP-1). None of these proteins is present in the seromucous acinar cells of the adult gland, but each is a product of the parotid gland during postnatal development and of the sublingual serous demilunes (Ball et al. 1988; Moreira et al. 1990; Girard et al. 1993; Mirels et al. 1998; Sivakumar et al. submitted for publication; MS Wolff and ARH, unpublished observations). We have isolated PIP cDNA clones in a library screen designed to identify secretory products of both the neonatal and the adult submandibular gland. Here we present the cDNA and deduced amino acid sequences of rat PIP, demonstrate its presence in submandibular and parotid gland secretions, and immunohistochemically localize PIP in developing and adult rat submandibular, sublingual, and parotid glands. Unlike the neonatal proteins mentioned above, PIP is present in both immature (proacinar; Type III) and adult seromucous acinar cells of the submandibular gland. The PIP expression pattern is similar to that reported for salivary peroxidase (Yamashina and Barka 1974; Bodner et al. 1983; Redman and Field 1993; Moriguchi et al. 1995; Redman et al. 1998; Kruse et al. 1998).
Materials and Methods
Materials
All molecular biological reagents were purchased from New England Biolabs (Beverly, MA) or from Boehringer-Mannheim (Indianapolis, IN), unless otherwise specified. Radioactive nucleotides [α-32P]-dCTP, 6000 Ci/mmol, and [α-35S]-dATP (1000–1500 Ci/mmol, Sequenase grade) were obtained from New England Nuclear (Boston, MA).
Animals
Care and manipulation of animals were performed according to the guidelines of the Animal Care and Use Committees, University of California, Berkeley, and University of Connecticut Health Center. Adult male and untimed (Figures 2 and 3) or timed (Figure 4) pregnant female Sprague- Dawley rats were purchased from Harlan (Indianapolis, IN). Pregnant female rats were observed daily at 1700 hr; the first day pups were seen was designated as Day 0. For harvesting of organs, rats were anesthetized with Metofane and sacrificed by cardiac puncture. Secretion products were collected in vitro as described in Ball et al. (1988) but into Dulbecco's modified Eagle's medium (DMEM). Secretion products were elicited from salivary glands of neonatal rats by 1 mM dibutyryl AMP and from salivary glands of an adult male rat by 20 μM isoproterenol.
RNA Preparation and Northern Blot Analysis
Organs to be used for RNA preparation were frozen immediately in liquid nitrogen. RNA was prepared according to the method of Chomczynski and Sacchi (1987). Poly(A)+ RNA was selected by affinity chromatography on oligo d(T) cellulose (Aviv and Leder 1972). Northern blot analysis was performed as described previously (Mirels and Ball 1992). RNA was separated by electrophoresis through 1.5% agarose formaldehyde gels and transferred to nitrocellulose. PIP clone gIIG4 cDNA probes were labeled with 32P by random priming (Feinberg and Vogelstein 1983) and hybridized to the filters under high-stringency conditions. Final washing of the filters was performed with 0.2 X SSC, 0.1% SDS at 65C.
Screening of cDNA Libraries
The adult male Wistar rat submandibular gland cDNA library was synthesized and propagated in plasmid pBR322 and screened by filter colony hybridization as described previously (Mirels et al. 1987). The probe for library screening was firststrand cDNA prepared from RNA of submandibular glands of 1-day-old rats. The first-strand cDNA synthesis reaction, containing 1 μg of poly(A)+ RNA from 1-day-old rat submandibular glands and 50 U of M-MuLV reverse transcriptase (New England Biolabs) was completed as described in Sambrook et al. (1989). After neutralization, the probe was isolated by chromatography on Bio-Gel P10. Filter colony hybridization took place for 17 hr at 42C in Northern blot hybridization buffer (Mirels and Ball 1992), using 106 cpm/ml first-strand cDNA. Filters were washed at highest stringency in 0.4 X SSC, 0.1% SDS at 65C. Clone gIIG4 was subcloned into pGEM 1 using standard methodology.
A 5–day-old Sprague-Dawley rat submandibular gland cDNA library was prepared in the vector λ ZapII and screened under high stringency conditions using 32P labeled adult submandibular gland cDNA clone gIIG4 as previously described (Girard et al. 1993). Five-day submandibular gland cDNA clone zIIG4, isolated in this screen, was excised by phagemid rescue according to protocols provided by Stratagene (La Jolla, CA).
DNA Sequence Analysis and 5′Race
Clones gIIG4 and zIIG4 were sequenced completely on both strands using a Sequenase version 2.0 kit according to instructions provided by the manufacturer (Amersham; Arlington Heights, IL). Plasmids were sequenced from either end using T3 and T7 (zIIG4) or SP6 and T7 (gIIG4) primers, and bidirectionally from the Hind III site (
5′ ends of IIG4 transcripts from 5-day submandibular gland, adult submandibular gland, and adult parotid gland poly (A)+ RNA were cloned using a Marathon cDNA Amplification Kit (Clontech; Palo Alto, CA) as specified by the manufacturer. The IIG4-specific primer was TCCGGGATCCAGGGTGGTAGGATGATCATTGCAG. The first 24 nucleotides are complementary to
Fusion Protein and Antibody Preparation
A glutathione-S-transferase-IIG4 fusion protein was prepared using the vector pGEX-KG (Guan and Dixon 1991). cDNA clone zIIG4 was cut with AvaII (
The GST-IIG4 fusion protein was prepared by a modification of the method previously described (Girard et al. 1993) in bacterial strain HB101. Cells were harvested by centrifugation, lysed by sonication, and cell debris precipitated in 0.5 M NaCl as described. After centrifugation, the GST-IIG4 protein was found in the pellet rather than in the supernatant as above. The pellet was resuspended in 0.1 M Tris-HCl, pH 8.0, 5 M urea at room temperature (RT), and recentrifuged. The solubilized material was dialyzed overnight against PBS containing 10 mM β-mercaptoethanol and 1% Triton X-100 at 4C. The GST-IIG4 fusion protein, which remained soluble under these conditions, was collected by binding to glutathine-agarose beads as in previous preparations.
A rabbit anti-GST-IIG4 antiserum was raised against this fusion protein at Pocono Farms (Canadensis, PA) according to their standard fusion protein protocol (see Girard et al. 1993). The antiserum was affinity-purified using GST-IIG4 protein coupled to Reacti-Gel beads (Pierce; Rockford, IL). The beads were prepared as follows. GST-IIG4 protein was dialyzed into 0.1 M sodium borate, pH 8.5 (coupling buffer), and coupled to the Reacti-Gel beads overnight at 4C. The beads were then blocked in 1 M ethanolamine HCl, pH 8.0, for 3 hr at RT and washed successively with 1 M NaCl, deionized water, 50 mM glycine-150 mM NaCl, pH 2.5, and PBS.
Anti-GST-IIG4 antibodies were isolated by incubation of rabbit antiserum with the prepared Reacti-Gel beads overnight at 4C. The beads were washed with 120 mM NaCl, 50 mM Tris-HCl, pH 8.0, 0.5% NP 40 (Buffer B), followed by 1 M LiCl, 50 mM Tris-HCl, pH 8.0, 0.5% NP40, again with Buffer B, and finally with PBS. Bound antibodies were eluted with with 50 mM glycine, 150 mM NaCl, pH 2.5, and fractions were immediately neutralized with 1 M Tris-HCl. Fractions containing antibody were identified by SDS-PAGE and brought to 10 μg/ml with RIA grade BSA.
SDS-PAGE and Western Blot Analysis
SDS-PAGE was performed according to the method of Laemmli (1970) using 13% acrylamide resolving gels and 3.5% acrylamide stacking gels. Silver staining was performed by a modification of the method of Morrissey (1981). After electrophoresis, proteins were electroblotted to PVDF membrane (ProBlott; Applied Biosystems, Foster City, CA) by the method of Towbin et al. (1979). Membranes were blocked overnight in TBS (20 mM Tris-HCl, pH 7.5, 0.15 M NaCl) containing 5% nonfat dry milk. Membranes were incubated for 2 hr at RT with affinity-purified anti-GST-IIG4 at a 1:200 dilution, washed with TBST (TBS plus 0.05% Tween 20), then incubated for 1 hr at RT in TBS plus 5% nonfat dry milk containing horseradish peroxidase-conjugated goat anti-rabbit IgG (Jackson Immunoresearch; West Chester, PA) at a 1:20,000 dilution. Proteins were detected with the Renaissance chemiluminescence reagent (New England Nuclear).
PNGase F was obtained from New England Biolabs, and digestion of secretion product was performed overnight at 37C as directed by the manufacturer. Digestions contained 9 μg total secretion product and 1000 U of PNGase F in a final volume of 60 μl. Controls were prepared identically to PNGase F-treated samples but without addition of enzyme.
Immunogold-Silver Staining
For light microscopic immunocytochemistry, developing and adult salivary glands were collected from anesthetized rats and fixed in cold 4% paraformaldehyde in 0.1 M sodium cacodylate buffer, pH 7.4, for 16–24 hr. The tissues were dehydrated in cold methanol solutions, embedded in LR Gold resin (London Resin; Basingstoke, UK), and polymerized under
Results
To identify cDNA clones encoding protein(s) expressed in both neonatal and adult submandibular glands, an adult submandibular gland cDNA library was screened with first-strand cDNA synthesized using poly(A)+ RNA from 1-day-old rat submandibular gland as a template. From this very low complexity library (495 clones), several clones that hybridized to the first-strand cDNA were selected. Clones were tested by use as probes for Northern blot analysis of RNA from neonatal and adult submandibular gland. Of the clones identified in this screen, four corresponded to low-abundance transcripts present in neonatal and adult gland and were thought unlikely to encode major secretory products. One clone encoded neonatal submandibular gland protein CSP-1 (Girard et al. 1993), also expressed in submandibular glands of adult male rats after puberty. A single clone, gIIG4, hybridized to transcripts that were moderately abundant throughout the neonatal period as well as in adult male and female submandibular glands.
IIG4 Is Homologous to Human PIP/GCDFP-15/EP-GP/SABP/gp 17
The nucleotide and deduced amino acid sequences determined from adult submandibular gland clone gIIG4 are presented in Figure 1A, nucleotides 9–558. Comparison of the 146 amino acid protein encoded by this clone with the National Center for Biotechnology Information protein sequence databases revealed that the IIG4 protein is the rat homologue of the human prolactin-inducible protein (PIP) (also identified as gross cystic disease fluid protein-15, secretory actin binding protein, gp 17, and extraparotid glycoprotein). The mouse homologue (Windass et al. 1984; Myal et al. 1994) is also 146 amino acids in length and contains 80% overall sequence similarity (56% amino acid identity) with the gIIG4 protein. The calculated molecular weight of full length rat PIP is 16.4 kD, although the secreted form would be expected to be approximately 14 kD. Alignment of the rat sequence with its mouse and human counterparts is shown in Figure 1B. Sequence similarity is maintained throughout the three proteins. For consistency with human and mouse genomic nomenclature, the rat gene and protein have been named “prolactin-inducible protein” (Pip/PIP). However, Pip is not prolactin-inducible in rat (Myal et al. 1994).
The identity of the neonatal submandibular gland transcripts that hybridize to clone gIIG4 was confirmed by isolating a PIP cDNA from a 5-day-old rat submandibular gland cDNA library. DNA sequence analysis demonstrated that this clone (zIIG4) was identical to gIIG4 but was 20 nucleotides shorter at the 5′ end. To attempt to determine further 5′ untranslated sequence, the 5′ ends of PIP transcripts were amplified as described in Materials and Methods. A total of 18 5′ RACE products, derived from 5-day submandibular, adult submandibular, and adult parotid glands, was sequenced. An additional eight nucleotides (Figure 1A,

Rat PIP and its relationship to the mouse and human homologues. (
Expression Patterns of PIP in Developing Salivary Glands
A Northern blot in which the gIIG4 cDNA was hybridized to RNA from submandibular glands of neonatal rats aged 0–30 days and from those adult male and female rats is shown in Figure 2A. In all samples, the probe hybridized to an approximately 650-
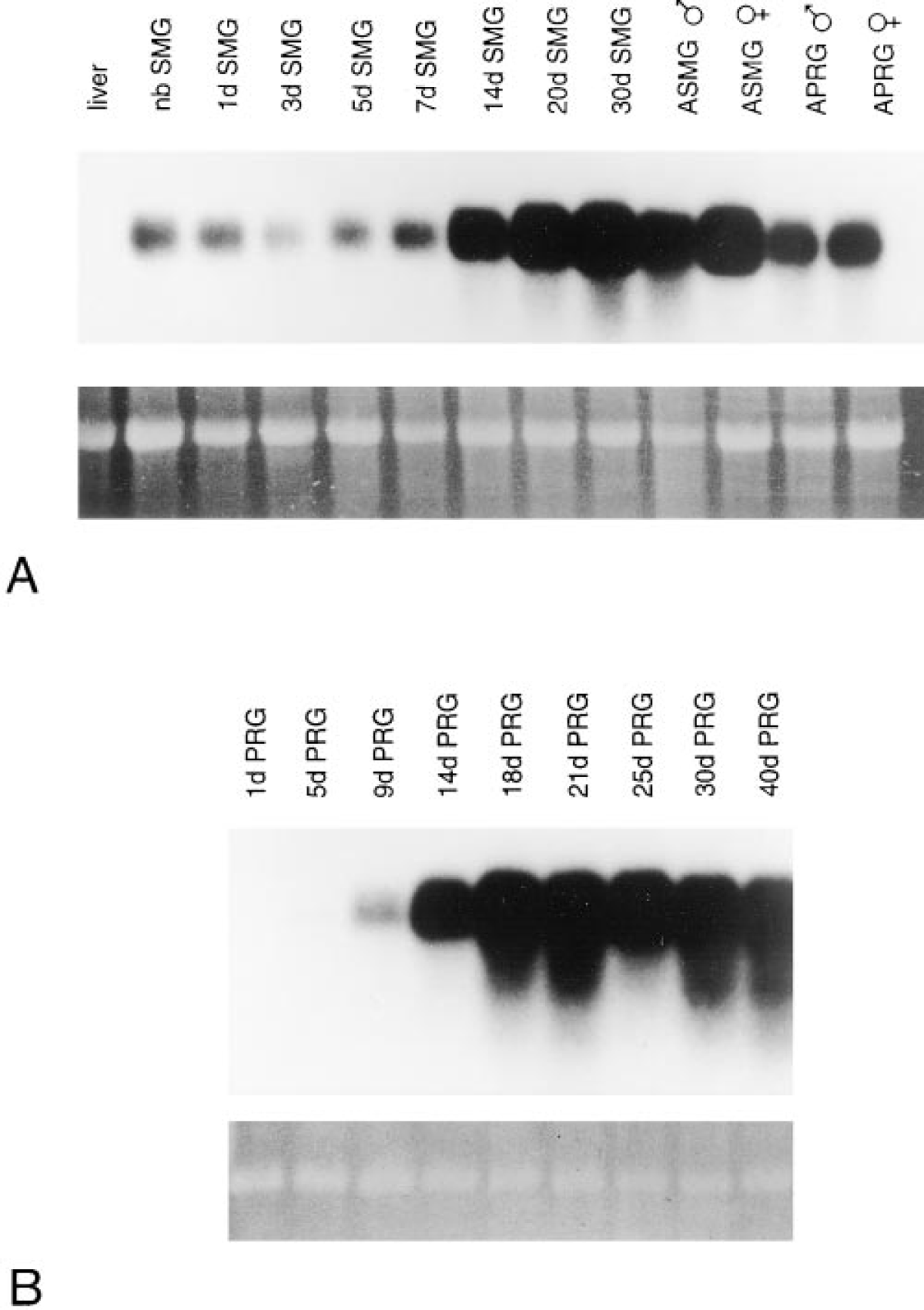
PIP is expressed in neonatal and adult submandibular and parotid glands. (
PIP is an Mr 17,000 secretory protein which is partially N-glycosylated in submandibular and parotid glands. (
Northern blot analysis of RNA from sublingual gland gave inconclusive results. Hybridization was observed at low levels in some samples from two developmental series. It was not possible with this method to distinguish between the presence of low levels of genuine sublingual PIP transcripts and minor contamination of some sublingual gland dissections with submandibular gland. The synthesis of PIP by sublingual gland is addressed below using immunocytochemistry.
PIP Protein in Salivary Secretions
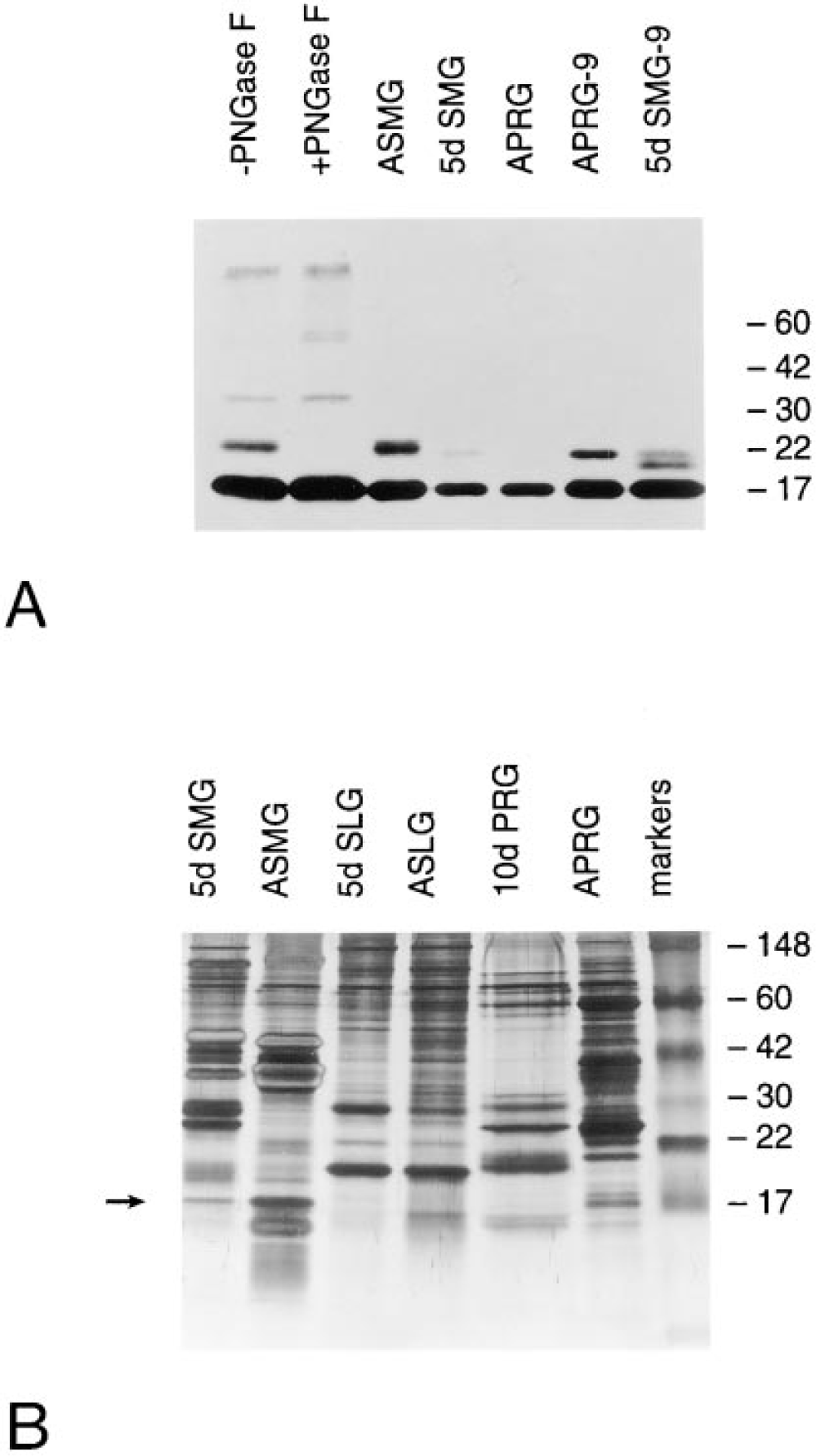
PIP protein was visualized in salivary secretions by Western blot using a rabbit antiserum raised against a glutathione-S-transferase-PIP fusion protein (Figure 3A). The middle lanes (ASMG, 5d SMG, and APRG) contained 1.5 μg of salivary secretory protein. In each case, the anti-PIP antiserum reacted primarily with an Mr 17,000 protein, which was at highest relative levels in adult submandibular gland. The Mr of this protein was slightly larger than the 14 kD predicted from the deduced amino acid sequence of PIP. Adult submandibular gland also contained an approximately Mr 22,000 protein(s) reactive with the anti-PIP antiserum (Figure 3A). A similar minor higher Mr band(s) was observed in parotid and neonatal submandibular gland secretion (APRG-9 and 5d SMG-9) when a sixfold greater amount of secretory protein was examined. No reactive bands were observed in sublingual secretion at either protein concentration (data not shown). The appearance and relative proportion of the PIP protein in salivary secretions is shown in the silver-stained gel in Figure 3B.
The PIP sequence has a single N-glycosylation consensus site (NET, amino acids 29–31; Figure 1A). Lanes 1 and 2 of Figure 3A contain adult submandibular secretion product incubated overnight in digestion buffer with or without addition of PNGase F to remove N-linked oligosaccharides. The relative proportion of Mr 17,000 to Mr 22,000 protein in the -PNGase F sample was similar to that in adult secretion (ASMG). In the +pNGase F sample, however, the Mr 22,000 protein was undetectable, demonstrating that the higher molecular mass band represents N-glycosylated PIP. Incubation of adult submandibular secretion overnight under the conditions of PNGase F digestion with or without enzyme resulted in the appearance of additional immunoreactive species at approximate Mr 33,000 and 60,000, as well as at the stacking/resolving gel interface. These may represent aggregation of PIP monomers and/or complexing with other salivary molecules.
Immunolocalization of PIP in Neonatal and Adult Salivary Glands
Immunogold silver staining of 1-μm plastic sections showed that PIP was present in the fetal submandibular gland as early as 20 days of gestation (Figure 4A). Reactivity was restricted to the Type III (proacinar) cells of the perinatal acini; no labeling of the Type I (terminal tubule) cells was observed. At 5 days after birth, the secretory granules present in the apical cytoplasm of Type III cells (Cutler and Chaudhry 1974; Ball et al. 1988) were heavily labeled (Figure 4B). In addition, Type III cells that were undergoing differentiation to adult seromucous acinar cells (Moreira et al. 1990,1991) were labeled with anti-PIP. In the adult submandibular gland, reactivity was seen only in the seromucous acinar cells (Figure 4C). No labeling could be discerned in any cells of the duct system. In sections incubated in the absence of the primary antibody or with normal rabbit IgG substituted for the primary antibody, no labeling was observed (Figure 4D). In the parotid gland, secretory cell differentiation is evident at 1 day after birth and is well under way at 5 days of age (Redman and Sreebny 1971; Sivakumar et al. submitted for publication). Labeling with anti-PIP was first detected in secretory granules of developing acinar cells of 5-day postnatal animals (Figure 4E) and was increased in 9-day postnatal acinar cells (Figure 4F). In the adult, reactivity remained restricted to the acinar cells (Figure 4G). No labeling was observed in the intercalated or striated duct cells.
In the sublingual gland, differentiation of the mucous acinar cells and serous demilune cells occurs before birth (Redman and Ball 1978). Reactivity with anti-PIP was present in the secretory granules of demilune cells of animals at birth (Figure 4H) and at 5 postnatal days (Figure 4I). Adult sublingual gland demilune cells, although still labeled with anti-PIP (Figure 4J), appeared less reactive than the neonatal glands. No reactivity was seen in the mucous acinar cells at any age. Therefore, PIP is a low-abundance product of sublingual serous demilunes and may be more highly expressed in the neonatal than in the adult gland.
Discussion
In this report we have described the sequence and cell specificity of PIP in rat salivary glands. As visualized by SDS-PAGE, rat PIP is an Mr 17,000 protein which is a relatively minor component of submandibular and parotid salivas. The presence of PIP in adult rat submandibular gland acinar cells was observed by Myal et al. (1994). However, the expression of this protein in neonatal submandibular gland and in parotid and sublingual gland was not previously described. The expression pattern of PIP in neonatal and adult submandibular, parotid, and sublingual glands is similar to that of salivary peroxidase. Like PIP, salivary peroxidase is present in secretory granules of acinar cell progenitors of the neonatal (Yamashina and Barka 1974) and in acinar cells of adult (Bodner et al. 1983; Kruse et al. 1998) submandibular gland. Both PIP and salivary peroxidase are produced by neonatal and adult parotid acinar cells (Redman and Field 1993; Moriguchi et al. 1995; Redman et al. 1998). PIP and peroxidase are also produced by serous demilune cells in the neonatal sublingual gland and are either significantly reduced in amount (PIP) or absent (peroxidase) in the adult (Moriguchi et al. 1995; Kruse et al. 1998).
The presence of PIP in both neonatal and adult submandibular gland acinar cells contrasts with the distribution of the previously described major secretory products (PSP, CSP-1, SMGB1, and SMGB2) of neonatal submandibular gland acinar cell progenitors. None of these proteins is present in adult submandibular acinar cells. In parotid gland, PSP, like PIP, is consistently an acinar cell product and is at sustained peak levels after weaning (Shaw et al. 1986; Sivakumar et al. submitted for publication). Conversely, SMGB and CSP1 are produced at relatively highest levels in neonatal parotid acinar cells but are restricted to intercalated ducts in the adult (Sivakumar et al. submitted for publication). In both neonatal and adult sublingual gland serous demilune cells, SMGB and CSP1 are predominant secretory proteins, whereas PSP and PIP are low-abundance products (Ball et al. 1988; Girard et al. 1993; MS Wolff and ARH, unpublished observations).
Human PIP is present only at exceedingly low levels in parotid gland and saliva (Haagensen 1981; Mazoujian et al. 1983; Rathman et al. 1989). PIP was detected at a similar concentration in homogenates of human sublingual and submandibular gland (Haagensen 1981), suggesting that the sublingual gland contributes substantially to the concentration of PIP in human submandibular/ sublingual saliva. This is opposite to the expression of PIP in rat parotid and sublingual gland. However, the cells that synthesize PIP in human salivary glands [serous cells of submandibular and parotid gland, serous demilunes of sublingual gland (Mazoujian et al. 1983; Rathman et al. 1990; Swanson et al. 1991)] are analogous to the PIP-producing cells in rat. The overall concentration of PIP in human and rat salivary secretions may be similar as well. PIP has been estimated to constitute 1% of total protein in whole saliva (Rathman et al. 1989; Schenkels et al. 1997); Figure 3B shows that PIP is also a relatively minor component of rat salivary secretions.
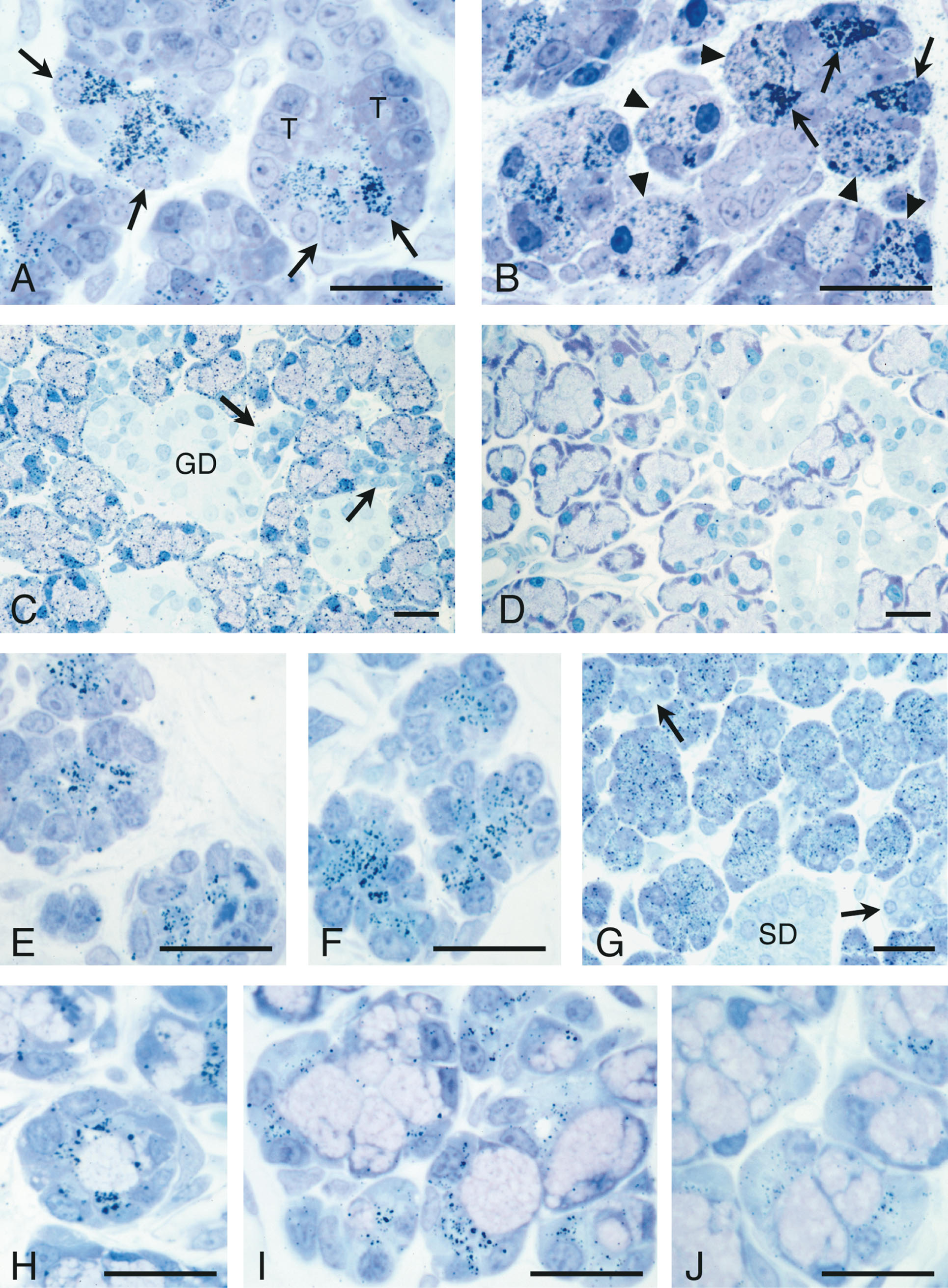
Immunogold-silver staining of 1-μm LR Gold sections of developing and adult submandibular, parotid, and sublingual glands using anti-PIP antibody. (
The conservation of PIP sequence and abundance between rodent and human salivas implies that the function of these proteins is conserved. Rat PIP is predominantly not glycosylated, although Mr 22,000 N-glycosylated forms are secreted by the parotid and submandibular glands. In human and T-47D cells, the majority of PIP is N-glycosylated, whereas mouse PIP does not contain an N-glycosylation consensus sequence (Myal et al. 1994). Together, these findings suggest that the carbohydrate portion of PIP is probably not critical to the function of these otherwise well-conserved molecules. The previously demonstrated ability of PIP to bind to bacteria, keratin, and CD4, as well as the regulation of PIP gene expression (in human breast carcinoma cell lines) by interleukins, suggests that this protein may play a role in mucosal immunity or in nonimmune mucosal defense. This function(s) would be consistent with the presence of PIP in saliva, tears, submucosal glands of the bronchi, and apocrine glands of the skin. The expression and distribution of salivary peroxidase, which protects the oral mucosa by inhibiting bacterial growth and by detoxifying hydrogen peroxide, closely parallel those of PIP. Future studies could address the role of PIP in saliva by targeted disruption of the mouse PIP gene.
Footnotes
Acknowledgements
Supported by National Institutes of Health grant DE09428.
We thank Abigail J. Miranda and Mary W. Goss for expert technical assistance, and Don Rio for providing protocols and advice on affinity chromatography of the anti-PIP antibodies.
