Abstract
Histatins are a family of salivary proteins with bactericidal and fungicidal activities that contribute to the innate defense of the oral cavity. Histatins are present in the serous granules of the parotid and submandibular glands. The important role of histatins in saliva, and the limited information on their cellular and subcellular distribution, prompted us to further define the localization of histatins in the major salivary glands. Immunogold-silver staining of 1-μm sections of plastic-embedded tissue with anti-histatin antibody revealed histatin immunoreactivity in the serous acinar cells of the parotid and submandibular glands, the serous demilune cells of the submandibular and sublingual glands, and in occasional intercalated duct cells. No reactivity was seen in mucous cells or in striated or excretory duct cells. Electron microscopic observations of thin sections labeled with anti-histatin and gold-labeled secondary antibodies revealed immunoreactivity associated with the rough endoplasmic reticulum and Golgi complex and in secretory granules of serous acinar and demilune cells. The granules of parotid acinar cells exhibited relatively uniform labeling of their content, whereas the granules of serous cells in the submandibular and sublingual glands showed variable labeling of the dense and light regions of their content. A few intercalated duct cells adjacent to the acinar cells also exhibited labeled granules. These results suggest that the serous cells of the major glands are the main source of histatins in human saliva. They are also consistent with several previous studies demonstrating the variable distribution of different proteins within the granule content.
Keywords
H
Histatins are derived from two gene products, histatin 1 and histatin 3. Histatin 1, which is 38 residues in length, is encoded for by the HIS1 gene, whereas histatin 3, consisting of 32 residues, is encoded for by the HIS2 gene (Sabatini and Azen 1989). Histatin 2 derives from histatin 1 through tryptic-like activity between R11 and R12, and histatins 4–12 arise from tryptic-and chymotryptic-like activities on histatin 3 (Troxler et al. 1990). Other smaller histatin fragments have been identified in saliva (Perinpanayagam et al. 1995), indicating the high susceptibility of these proteins to proteolysis by salivary enzymes. Histatins can be found only in salivary secretions of humans and Old World monkeys, such as Macaca fascicularis (Xu et al. 1990), but not in those of New World monkeys or other primates. On this basis, it has been suggested that histatins evolved after the continents drifted apart approximately 30 million years ago (Sabatini et al. 1993), but this primate specificity at the DNA level has been questioned in recent genomic studies on the casein gene cluster (Rijnkels et al. 2003). Histatins appear to be secreted specifically by salivary glands because they are not detectable in human tear fluid, nasal, bronchial, or cervical mucus, seminal fluid, sweat, or blood plasma (Schenkels et al. 1995).
Several studies have demonstrated significant changes in salivary histatin concentrations under different conditions. In normal healthy individuals, histatin levels decrease with age (Johnson et al. 2000). In contrast, the concentrations of other salivary proteins show little change during aging in healthy, unmedicated people (Fox et al. 1987; Wu et al. 1993). Histatin levels are increased in otherwise healthy individuals who have experienced recurrent oral Candida infections (Bercier et al. 1999) and in HIV-positive patients (Atkinson et al. 1990). In contrast, two studies of immunocompromised patients with secondary Candida infections reported decreases in histatin concentrations (Lal et al. 1992; Mandel et al. 1992). The mechanisms underlying these changes are unknown but may involve specific factors present in infected individuals that act on salivary gland cells. It also is possible that infection of salivary epithelial cells by HIV alters the normal pattern of protein production and secretion. A more recent study of HIV-infected patients demonstrated reduced salivary anticandidal activity but no significant change in histatin concentrations (Lin et al. 2001). These contradictory findings emphasize the limited state of knowledge of the cell biology of these interesting and important salivary constituents.
The main sources of histatins in human saliva are the parotid and submandibular glands. Stimulation of saliva secretion by mechanical or gustatory stimuli results in increased secretion of histatins by both glands (Jensen et al. 1994; Johnson et al. 2000). However, the exact distribution of histatins in the salivary glands is not completely known. Immunocytochemical studies with anti-histatin antibodies have shown that histatins are present in the granules of serous secretory cells of the parotid and submandibular glands (Takano et al. 1993). Histatin mRNA is present in the minor lingual serous (von Ebner's) glands of primates, as shown by in situ hybridization (Azen et al. 1990). In view of the importance of histatins in regulating the oral microflora, as well as the limited information on their cellular distribution, we undertook the present study to describe, using light and electron microscopic immunocytochemistry, the localization of histatins in the major salivary glands of humans.
Materials and Methods
Samples of normal parotid, sublingual, and submandibular glands were obtained from consenting male and female patients, aged 44–69 years, undergoing surgery at the Otorhino-laryngology Clinic, University of Cagliari, Cagliari, Italy. One sample of normal parotid gland was provided by Drs. Britta and Firoz Rahemtulla (School of Dentistry, University of Alabama, Birmingham, AL). All procedures were approved by the respective institutional committees on human experimentation at the University of Cagliari and University of Alabama.
For light microscopic studies the glands were fixed overnight in either 4% paraformaldehyde or for 1–4 hr in 3% paraformaldehyde-0.1% glutaraldehyde (Polysciences; Warrington, PA) in 0.1 M sodium cacodylate buffer, pH 7.2, then stored in 1% paraformaldehyde in cacodylate buffer at 4C. The samples were rinsed in 0.1 M cacodylate buffer, dehydrated in cold methanol, embedded in LR Gold resin (Polysciences), or Lowicryl K4M resin (Polysciences) and polymerized under UV light (365 nm) at −20C. One-micrometer sections were collected on SuperFrost Plus slides (Fisher Scientific, Pittsburgh, PA) and incubated for 90 min at room temperature (RT) with a goat polyclonal antibody to human histatin (Atkinson et al. 1990) diluted in 1% bovine serum albumin (BSA) in PBS or in a mixture of fish gelatin, ovalbumin, and Tween-20 in PBS. Sections incubated with medium devoid of primary antibody or containing normal goat serum (Sigma Chemical; St Louis, MO) were used as controls. After incubation the sections were rinsed with PBS and incubated for 60 min at RT with rabbit anti-goat IgG labeled with 5-nm gold particles (British BioCell International; Cardiff, UK) diluted 1:50 in 1% BSA-PBS. After rinsing with PBS and distilled water, silver enhancement (British BioCell International) was performed to visualize the bound gold particles. The sections were lightly stained with 1% methylene blue-1% azure II and observed and photographed in a Leitz Orthoplan microscope.
Electron microscope processing employed fixation of small pieces (1 mm3) of tissue from the same patients in either 1% glutaraldehyde or 3% paraformaldehyde-0.1% glutaraldehyde in 0.1 M cacodylate buffer for 1–2 hr, after which the glands were stored at 4C in either 0.1 M cacodylate buffer or 1% paraformaldehyde in cacodylate buffer. After rinsing in buffer the tissues were dehydrated in cold methanol, embedded in either LR Gold or Lowicryl K4M resin, and polymerized as described above. In addition, some of the glutaraldehyde-fixed samples were dehydrated in ethanol, embedded in Polybed epoxy resin (Polysciences), and polymerized at 60C. Ultrathin sections were collected on uncoated or Formvar-coated nickel grids and immunogold labeling was carried out as described previously (Hand 1995). The sections were treated with either 1% BSA-1% instant milk in PBS or fish gelatin, ovalbumin, and Tween-20 in PBS to block nonspecific binding. The sections were then incubated with the anti-histatin antibody diluted in 1% BSA in PBS for 60 min at RT, or incubated overnight at 4C with the anti-histatin antibody diluted in fish gelatin, ovalbumin, and Tween-20 in PBS. Omission of the primary antibody or incubation with normal goat serum was used as control. After rinsing with PBS the grids were incubated for 60 min at RT with rabbit anti-goat IgG labeled with 10-nm gold particles (British BioCell International). The grids were washed with PBS and distilled water, stained with uranyl acetate and lead citrate, and observed and photographed in a JEOL 100CX TEM.
Results
Light Microscopic Immunogold-Silver Staining
Labeling of 1-μm sections of LR Gold- or Lowicryl-embedded salivary glands with the anti-histatin anti-body revealed histatin reactivity in serous secretory cells of all three major glands. In the submandibular gland, the supranuclear and apical regions of serous acinar cells were labeled, indicating the presence of histatin in secretory granules (Figures 1A and 1B). The intensity of the labeling varied from cell to cell. In general, the intercalated ducts were unreactive (Figures 1A and 1B). However, occasional intercalated duct cells adjacent to the serous acini exhibited some labeling over the apical cytoplasm (Figure 1C). No labeling was observed over the cells of the striated and excretory ducts.
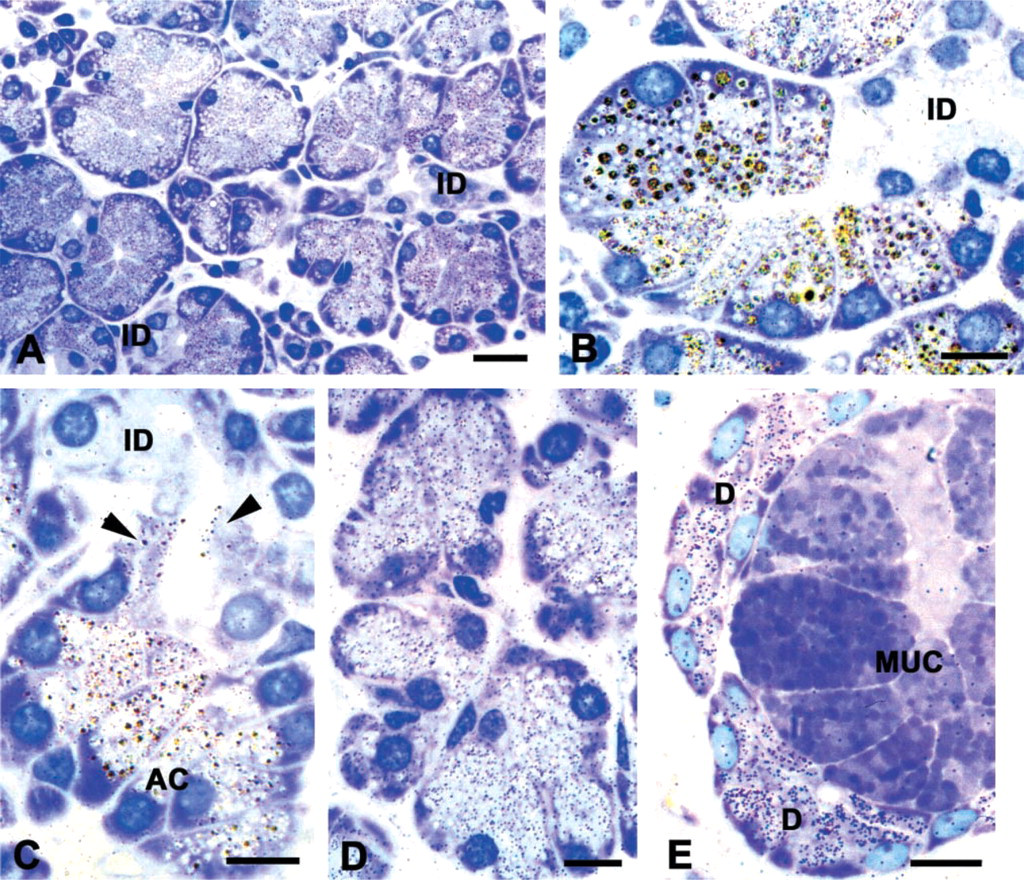
Immunogold-silver stain of histatin localization in human salivary glands. (
Distribution of histatin labeling in human major salivary glands
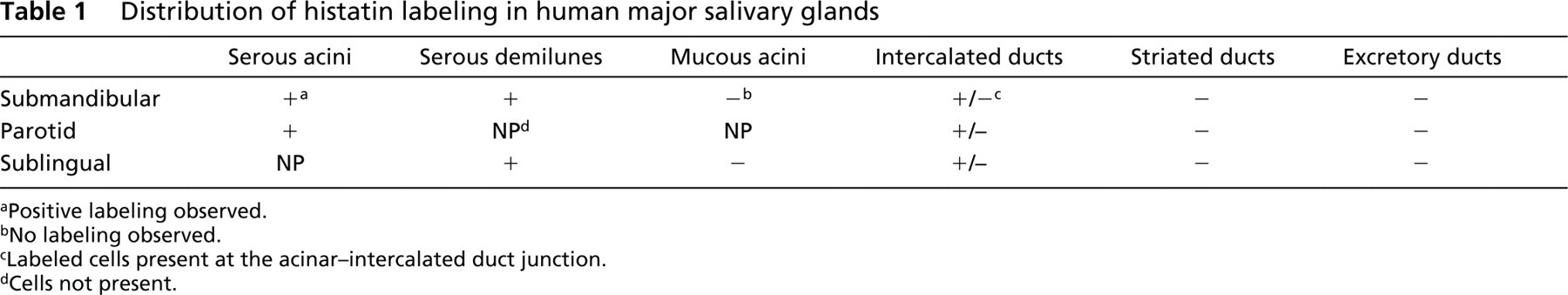
aPositive labeling observed.
bNo labeling observed.
cLabeled cells present at the acinar-intercalated duct junction.
dCells not present.
The serous acinar cells of the parotid gland also were labeled by the anti-histatin antibody (Figure 1D). The labeling of the acinar cells was more uniform in the parotid than in the submandibular gland. In the sub-lingual gland, the serous demilune cells were reactive but no labeling was found in the mucous secretory cells (Figure 1E).
No labeling or only low levels of nonspecific background were observed when the primary antibody was omitted from the incubation medium or when normal goat serum was substituted for the primary antibody (data not shown).
The distribution of histatin labeling in human salivary glands is summarized in Table 1.
Electron Microscopic Immunogold Labeling
In sections of serous acinar cells of the submandibular (Figure 2) and parotid (Figure 3) glands and in serous demilune cells of the sublingual gland (Figure 4), the secretory granules typically exhibited a bi- or tripartite structure and frequently contained a dense core or spherule (Figures 2A, 2B, 3A, 4A, and 4B), as described previously for human salivary gland serous cells (Tandler and Erlandson 1972; Riva et al. 1974,1988; Tandler and Phillips 1993). In sections incubated with the anti-histatin antibody, many granules appeared to be uniformly labeled, i.e., the components of different electron density had similar labeling densities (Figures 2A and 3A). In the parotid gland, the labeling density of the granules was relatively uniform from cell to cell and appeared to be greater than the labeling density of granules in the other glands. In some granules of the submandibular and sublingual glands, the dense cores were labeled to a greater extent than the lighter matrix (Figures 2B and 4B), whereas in others the opposite pattern was observed.
Many gold particles were associated with cisternae of rough endoplasmic reticulum (rER) in the serous cells (Figures 2A and 2C). Labeling was present over the lumen of the cisternae and over the cisternal membranes and attached ribosomes. Although relatively few particles were present over the Golgi complex (Figures 2A and 4A), saccules along the trans side of the Golgi complex and small forming secretory granules occasionally were labeled (Figure 4A, inset).
Some intercalated duct cells close to the acini contained apical secretory granules similar to those described by Lantini et al. (1988). These granules also were labeled with the anti-histatin antibody (Figures 2D and 3B). Like the granules of serous acinar cells, the intercalated duct cell granules exhibited variable labeling density. In some granules gold particles were concentrated over the dense cores (Figure 2D), whereas in others both the lighter matrix and the dense core were labeled (Figure 3B).
The mucous secretory cells of the submandibular and sublingual glands were unlabeled (Figure 4C). No labeling was seen in mitochondria or over nuclei of serous cells. Few gold particles were present in control sections incubated in the absence of the primary antibody (Figure 3C) or with normal goat serum instead of the primary antibody. Occasionally, small nonspecific clusters of gold particles were observed over a few secretory granules, but the majority of granules were free of gold particles. No differences were observed in the cellular distribution of histatin immunoreactivity in sections of tissues embedded in LR Gold, Lowicryl K4M, or Polybed resin. However, la beling of the rER and Golgi complex was very low or undetectable in Polybed sections.
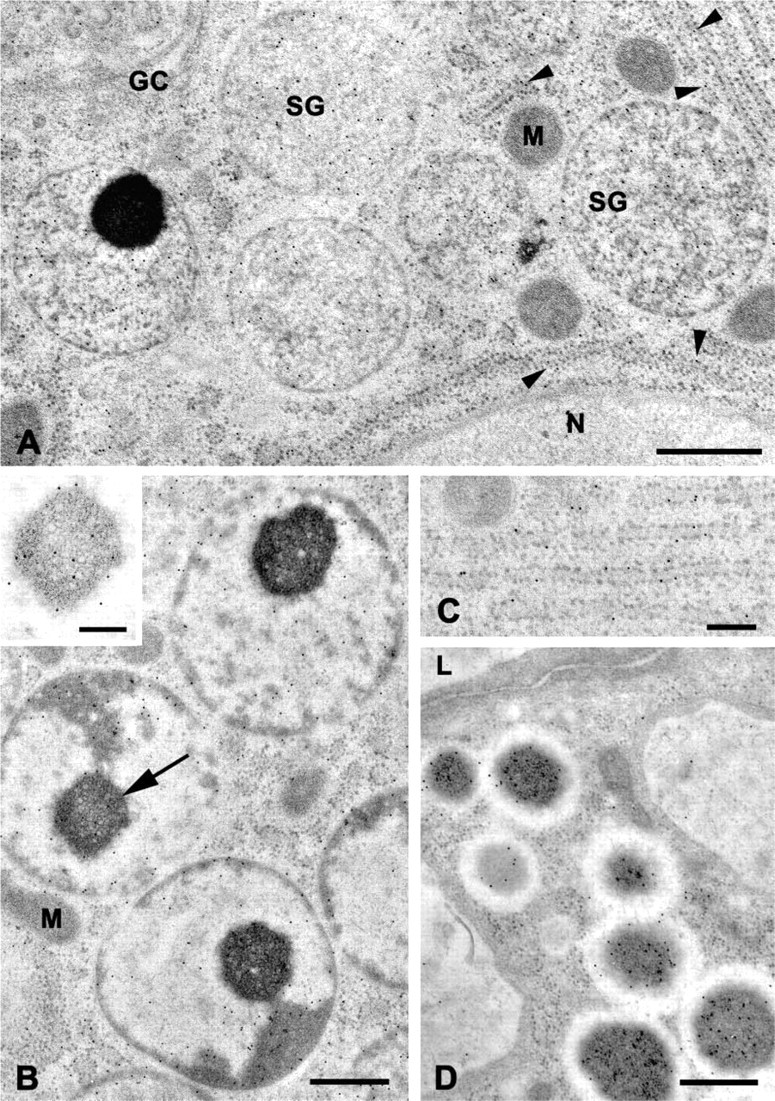
Electron micrographs of immunogold-labeled thin sections of human submandibular gland. (
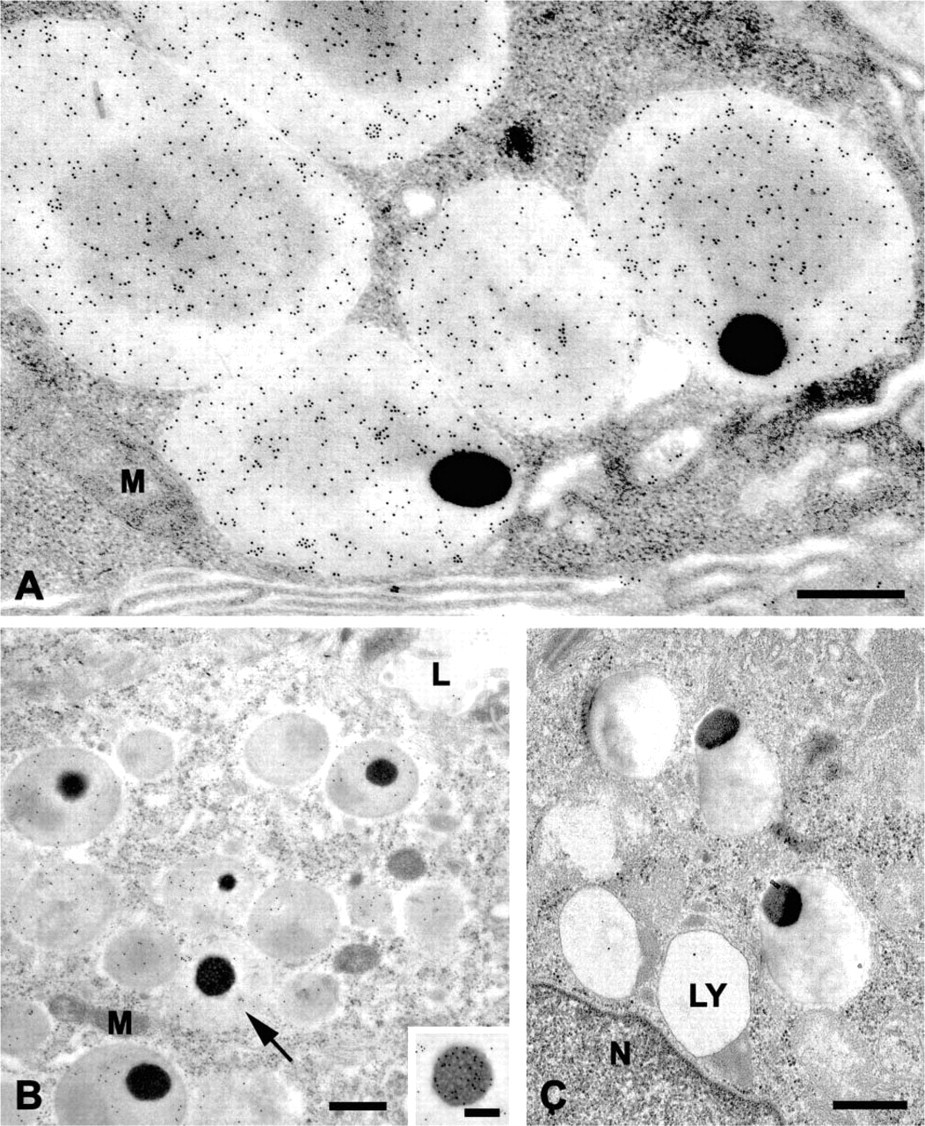
Electron micrographs of immunogold labeled thin sections of human parotid gland. (
Discussion
The results of the immunolabeling studies confirm and extend the observations of Takano et al. (1993), which demonstrated the presence of histatins in the granules of the serous cells of human parotid and submandibular glands. The present results show that histatins also are present in the serous demilune cells of the sublingual gland. Although direct evidence for the synthesis of histatins by the serous cells was not obtained, the presence of gold particles associated with the rER and Golgi complex of the serous cells is consistent with this assumption. Histatins are stored in the secretory granules and presumably are released by exocytosis at the luminal cell surface. A number of other human salivary proteins have a similar distribution. Amylase and salivary agglutinin [recently demonstrated to be identical to the lung scavenger receptor cysteine-rich protein gp-340 (Prakobphol et al. 2000)] are present in the rER, Golgi complex, and in forming and mature granules of parotid and submandibular serous cells (Takano et al. 1991,1993). Acidic proline-rich proteins and the proline-rich polypeptide P-C also are present in the secretory granules of these cells (Takano et al. 1993; Kimura et al. 1998), and lysozyme and cyclic AMP receptor proteins are present in the serous granules of all three major salivary glands (Machino et al. 1986; Miyazaki et al. 2001; Piludu et al. 2002).
In many species, including humans, the secretory granules of serous salivary gland cells display a variety of substructures (reviewed in Tandler and Phillips 1993). Studies employing cryofixation and freeze-substitution of glands of experimental animals have confirmed that the substructures observed in chemically fixed glands are indicative of the true distribution of granule contents (Ichikawa et al. 1980; Ichikawa and Ichikawa 1987). Previous studies of human glands have shown that other secretory proteins, such as amylase, agglutinin, and lysozyme, are differentially distributed within serous secretory granules (Machino et al. 1986; Takano et al. 1991). In an earlier study, we reported that histatins and proline-rich proteins were distributed relatively uniformly within parotid and submandibular serous granules (Takano et al. 1993). In the present study, the distribution of gold particles over the secretory granules indicated some variability in the intragranular localization of histatins. In the parotid gland, histatins appeared to be uniformly distributed within the granules. In the sub-mandibular and sublingual serous cells, however, the labeling density of the dense cores was sometimes greater than that of the lighter portions of the granule content. Sometimes the opposite pattern was observed. The reasons underlying this variability are unknown. It may result from slight differences in the fixation and processing of the tissue samples. On the other hand, differences in intracellular protein sorting and packaging mechanisms, or the association of histatins with different proteins, could account for the observed variability. Histatins are known to form heterotypic complexes with other proteins in saliva (Iontcheva et al. 1997), and some of these complexes initially may form within the granules.
The presence of histatin immunoreactivity in the serous cells of the major salivary glands suggests that these cells are the main source of histatins in saliva. One previous study (Johnson et al. 2000) indicated that the concentration of histatins is greatest in unstimulated parotid saliva, in apparent agreement with the higher immunogold labeling density observed for histatins in parotid secretory granules. However, Atkinson et al. (1990) reported that unstimulated submandibular/sublingual saliva had a 10-fold greater concentration of histatins than parotid saliva. The reason(s) for the difference between these two studies is(are) unclear. After gustatory stimulation, histatin concentrations in submandibular/sublingual gland saliva are equal to or greater than those in parotid saliva (Lal et al. 1992; Johnson et al. 2000). The lack of labeling of the mucous cells of the submandibular and sublingual glands in the present study suggests that these cells are not involved in the synthesis and secretion of histatins. Because most minor salivary glands are predominantly mucous in nature, the results also suggest that most of the minor glands, except for those with a substantial population of serous demilune cells, as well as the lingual serous gland noted earlier, probably contribute little histatin to saliva. However, this must be verified by specific analyses of the minor glands and their secretions.
Histatin immunoreactivity also was found in the granules of some intercalated duct cells. Previous studies have shown that other secretory proteins of serous and mucous cells may be present in intercalated duct cells (Korsrud and Brandtzaeg 1982; Miyazaki et al. 2001; Piludu et al. 2002,2003). Presumably, these proteins are secreted into saliva by exocytosis of the granules. Compared to the acinar cells, the contribution of the duct cells to the protein content of saliva is very small, and therefore the significance of this dual expression pattern for certain proteins is unclear. One possible interpretation is that the expression of acinar proteins by intercalated duct cells may indicate the differentiation of duct cells into acinar cells, or vice versa. For example, treatment of rats with the β-adrenergic agonist isoproterenol initiates morphological changes and induces synthesis of acinar cell proteins by intercalated duct cells (Hand and Ho 1985; Vugman and Hand 1995). On the other hand, during development of the rat parotid gland, acinar cells of the postnatal acini may give rise to some cells of the intercalated ducts (Redman and Field 1993; Sivakumar et al. 1998). Experimental studies in rodents also suggest that, in some glands, intercalated ducts may house stem cells that can differentiate into either acinar cells or other duct cells (reviewed in Denny et al. 1997).
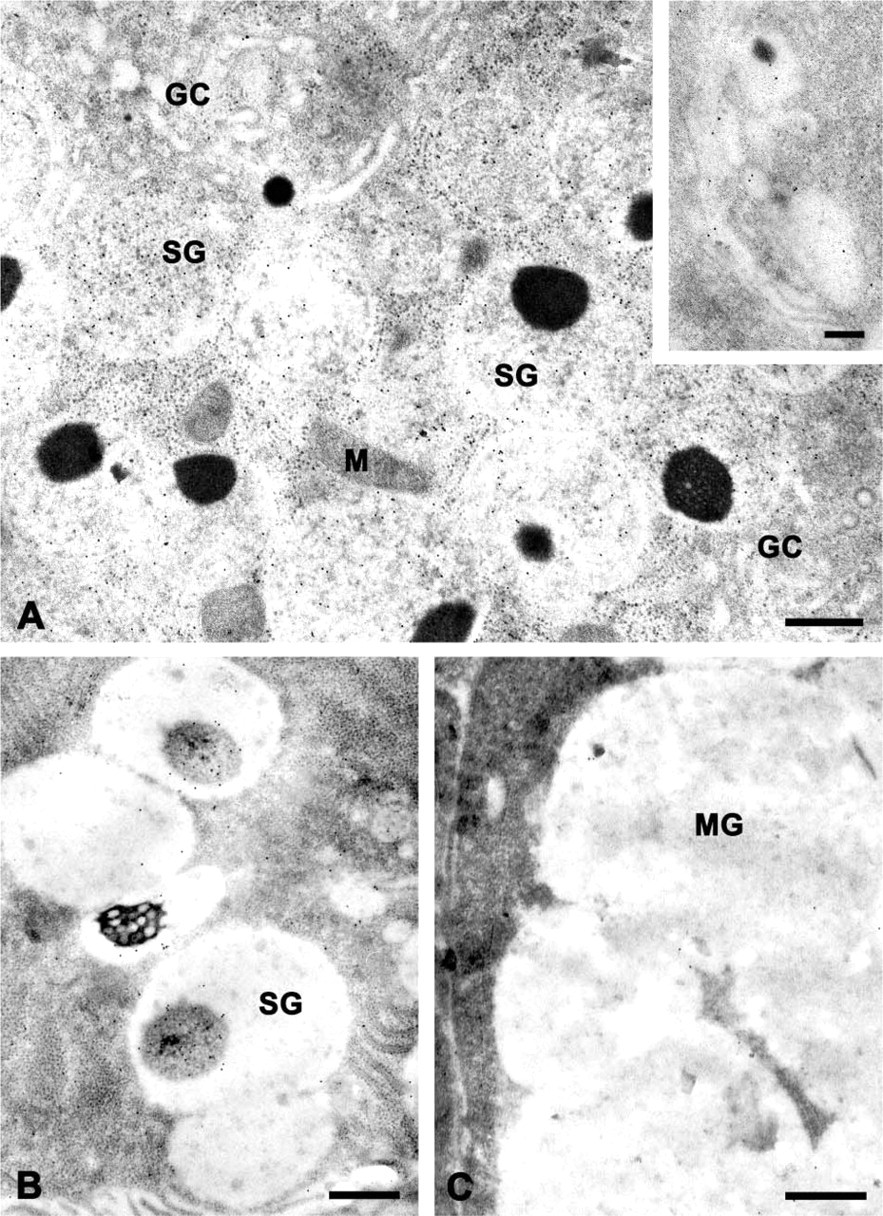
Electron micrographs of immunogold-labeled thin sections of human sublingual gland. (
The present results indicate that the histatins, like several other salivary proteins involved in innate defense of the oral cavity, are synthesized and secreted predominantly by the serous cells of the major glands. Also like other salivary proteins, they exhibit a variable distribution within the secretory granules. Important remaining questions about the cell biology of histatins include the factors that regulate their synthesis and the site and mechanisms of the proteolytic cleavages that produce the various family members.
Footnotes
Acknowledgements
Supported in part by a fellowship from the Universita Degli Studi di Cagliari (MP), the University of Connecticut Health Center, and NIH/NIDCR grants DE 05672, DE 07652, and DE 14950 (FGO).
