Abstract
Keywords
S
However, the exact sites of AT2 mRNA expression within the adult rat ovary have not yet been determined. In this context, it would be interesting to rule out whether specific AT2 mRNA expression could be confined to characeristic stages of follicular development, including atretic degeneration, or to the formation of corpora lutea. The aim of this study was to identify the cell types that express AT2 mRNA during the estrous cycle in the rat ovary. By using nonradioactive in situ hybridization, this report provides evidence that AT2 mRNA is exclusively expressed in granulosa cells of follicles undergoing atretic degeneration.
Materials and Methods
Animals
Female adult Sprague-Dawley rats (70-100 days) were kept under standard laboratory conditions in the Animal Care Facility in Mannheim. Animals were maintained under controlled conditions of temperature (22C) and lighting (lights on from 0600-1800 hr) and had free access to rat chow, containing 19% protein, and tapwater. All experiments were conducted in accord with the NIH Guide for the Care and Use of Laboratory Animals. The stage of the estrous cycle was verified daily by examining vaginal smears stained with hematoxylin and eosin. Groups of three animals per stage (proestrus, estrus, metestrus, diestrus) were subjected to subsequent perfusion-fixation of the ovaries. Before sacrifice, blood samples were obtained under general anesthesia (see below) from the retro-orbital vein to assess plasma estradiol and progesterone levels. Together with the analysis of the smears and the histological examination of the obtained ovaries, definite assessment of the respective estrous cycle stage was thus achieved.
Perfusion-Fixation
Rats were deeply anesthetized by
Preparation of Riboprobes
Full-length rat AT2 cDNA (2.8
Nonradioactive In Situ Hybridization
The in situ hybridization procedure was carried out as described previously (Obermüller et al. 1998). Cryostat sections (5-7 μm thick) were transferrred onto silane-coated glass slides. Sections were postfixed in 4% paraformaldehyde (in PBS, pH 7.4) for 20 min, rinsed three times in PBS, and washed in diethyl pyrocarbonate (DEPC)-treated bidistilled water for 10 min. To improve permeabilization, a mild deproteinization step was performed by immersing the slides in 0.1 M HCl for 10 min, followed by two short rinses (5 min each) in PBS. To reduce background, slides were acetylated for 15 min in 0.1 M triethanolamine, pH 8.0, containing 0.25% acetic anhydride, added immediately before starting this step and again after 7 min and 30 sec. After rinsing in PBS, slides were dehydrated for 5 min each in 70%, 80%, and 96% ethanol and air-dried for 20 min. A prehybridization step was carried out by incubating the sections with prehybridization solution (50% deionized formamide, 50 mM Tris-HCl, pH 7.6, 25 mM EDTA, pH 8.0, 20 mM NaCl, 0.1% SDS, 0.25 mg/ml tRNA from yeast, 2.5 × Denhardt's solution). Sections were placed in a moist chamber and incubated at 42C for 2 hr. Meanwhile, hydrolyzed riboprobes were diluted in deionized formamide, boiled for 2 min, and chilled on ice. Subsequently, the individual components of the hybridization mixture were added and mixed vigorously (final concentrations were 50% deionized formamide, 20 mM Tris-HCl, pH 7.6, 1 mM EDTA, pH 8.0, 0.33 M NaCl, 0.2 M DTT, 0.5 mg/ml tRNA, 0.1 mg/ml sonicated, denatured DNA from fish sperm, 1 × Denhardt's solution, 10% dextran sulfate). Concentration of labeled anti-sense or sense probe was 5-10 ng per μl hybridization mixture. After removal of the prehybridization solution, 25 μl of hybridization mixture was applied to each section and carefully covered with a siliconized coverslip. Hybridization was performed at 42C in a moist chamber for 16 hr. Washing procedures included a first washing step in 2 × SSC (1 × SSC = 150 mM NaCl, 15 mM sodium citrate, pH 7.0) at room temperature (RT) for 20 min, followed by two stringent washes (1 hr each) in 1 × SSC and 0.5 × SSC containing 50% formamide at 49C. Slides were then rinsed at RT in 0.5 × SSC for 15 min, again rinsed in 0.2 × SSC for 10 min, and equilibrated twice for 5 min in Buffer I (100 mM Tris-HCl, 150 mM NaCl, pH 7.4).
Subsequently, sections were covered with blocking medium [Buffer I containing 1% blocking reagent (Boehringer Mannheim) and 0.5% bovine serum albumin (BSA)] and incubated for 30 min at RT in a moist chamber. Blocking solution was then drained from the slides and a polyclonal alkaline phosphatase-coupled sheep anti-digoxigenin antibody (Boehringer Mannheim), diluted 1:500 in blocking medium, was applied to the sections. After an incubation of 2 hr at RT the sections were incubated overnight at 4C. The next morning, sections were washed twice for 15 min in Buffer I and equilibrated for 2 min in Buffer II (100 mM Tris-HCl, pH 9.5, 100 mM NaCl, 50 mM MgCl2). For signal development, a color solution [Buffer II containing 0.4 mM nitroblue tetrazolium chloride, NBT (predissolved in 70% dimethylformamide), 0.4 mM 5-bromo-4-chloro-3-indolyl-phosphate, 4-toluidine salt, BCIP or X-phosphate (predissolved in 100% dimethylformamide), and 1 mM levamisole (to inhibit remaining endogenous alkaline phosphatase activity)] was freshly prepared. After covering sections with large amounts of substrate solution, slides were kept at 4C in a moist chamber in the dark. Color reaction was controlled under the microscope and terminated (usually after 12-48 hr) by immersing the slides twice for 5 min in Buffer III (100 mM Tris-HCl, 1 mM EDTA, pH 8.0). After rinsing in PBS, sections were mounted in bicarbonate-buffered glycerol, pH 8.6.
For a comprehensive analysis, some sections adjacent to those subjected to the in situ hybridization procedure were stained with hematoxylin and eosin.
Controls
The specifity of the obtained in situ hybridization signal was verified by parallel incubation with anti-sense and sense riboprobes on alternate sections. Throughout all experiments the sense probe did not produce any detectable signal. As further negative controls, some sections were hybridized without anti-sense probe, and others were processed by omission of the anti-DIG antibody. Both controls yielded completely negative results.
Results
An intense in situ hybridization signal for AT2 mRNA could be detected in enlarged antral follicles displaying an advanced stage of follicular development (Figures 1A and 1B). On closer examination, it could be shown that specific labeling of the AT2 RNA probe in a tertiary follicle was exclusively attributable to the granulosa cell layers, whereas theca cells were unreactive for the AT2 RNA probe (Figure 2). However, strong labeling was confined to many but not to all granulosa cells of the respective follicle. Detailed analysis demonstrated that a specific population of follicles contains AT2 mRNA-expressing granulosa cells (Figures 1A, 1B, and 3). Whereas follicles with multiple layers of well-delineated multilaminar cells were devoid of a specific hybridization signal, adjacent tertiary follicles contained AT2 mRNA-expressing granulosa cells. The above-mentioned follicles already showed signs of beginning atresia, including a reduction in the number of granulosa cells, cell shrinking, and nuclear pyknosis in scattered granulosa cells, as shown in Figures 3A and 3B. Moreover, individual collapsed atretic follicles also showed labeling for the AT2 riboprobe in granulosa cells. This is illustrated in an atretic follicle in Figures 3C and 3D, where AT2 mRNA-positve granulosa cells are surrounded by thickened theca cell layers. Extensive hypertrophy of theca cells is a characteristic event during follicle atresia in the rat (Erickson et al. 1985). In general, as ruled out in proestrous and estrous ovaries, strong expression of AT2 mRNA was already noted in early antral follicles but was more constantly detected in large antral follicles, including those in the preovulatory stage. At any stage of the estrous cycle, no hybridization signal could be noted in granulosa cells of earlier stages of follicle formation or in other ovarian structures, such as corpora lutea or stromal cells.
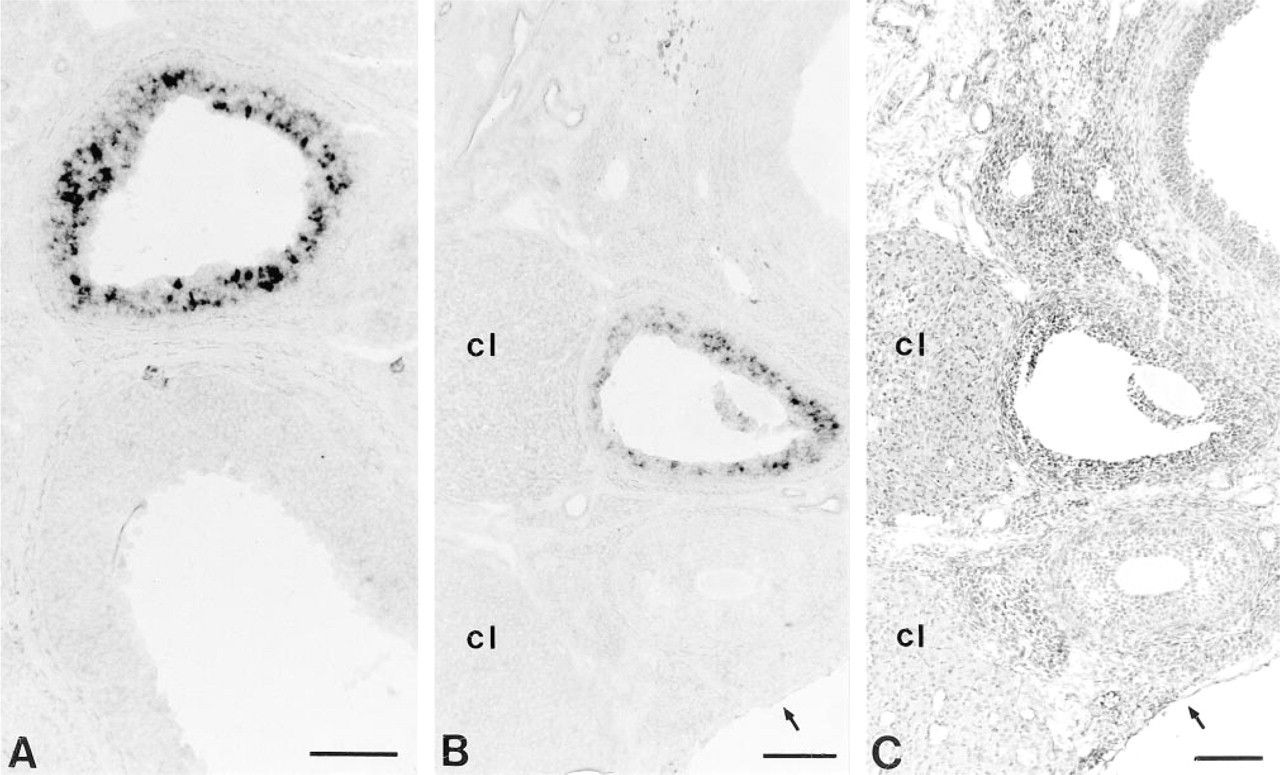
In situ hybridization for AT2 mRNA in rat ovaries fom late proestrus (

High-power magnification shows that the in situ hybridization signal for AT2 mRNA can be found only in the granulosa cell layers and not in theca cells (antral follicle at proestrus). Note that the granulosa cell layers in this follicle tend to lose the well-delineated, undamaged, multilaminar structure of a healthy follicle. Arrowheads mark the border between granulosa and theca cell layers. Bar = 25 μm.
Discussion
This report documents the expression of the AT2 mRNA in ovarian granulosa cells of sexually mature, normally cycling rats, as shown by nonradioactive in situ hybridization. The finding that granulosa cells are responsible for AT2 mRNA expression in the rat ovary is in agreement with previous studies showing AT2 binding sites on rat granulosa cells (Pucell et al. 1991; Tanaka et al. 1995). Further evidence for the presence of the AT2 receptor in granulosa cells comes from an investigation in the rabbit ovary, in which autoradiographic studies demonstrated the AT2 receptor in follicular granulosa cell layers (Yoshimura et al. 1996). In the bovine ovary, however, binding studies demonstrated that AT2 receptors are predominantly expressed on theca cells (Brunswig-Spickenheier and Mukhopadhyay 1992). Remarkably, we found that the mRNA for the AT2 receptor is strongly expressed by granulosa cells of different antral follicles, including those in early antral and in the preovulatory stage. However, we cannot exclude AT2 mRNA expression in granulosa cells of secondary follicles.
Using morphological criteria for describing the antral follicles that contained AT2 mRNA-positive granulosa cells, it became evident that those follicles exhibited signs of atretic degeneration. Obviously, the granulosa cells that exhibited staining for the AT2 RNA probe in an atretic antral follicle thus classified probably reflected different stages of atretic degeneration. This is likely because AT2 mRNA expression was often seen in a much greater number of granulosa cells in a given follicle than was the number of cells demonstrating typical pycnotic nuclei or apoptotic bodies (for a classification of atresia in the rat see Hsueh et al. 1994). In addition, AT2 mRNA expression could also be demonstrated in follicles featuring a more advanced stage of atresia with typically hypertrophied theca cell layers. It is well known that the vast majority of follicles undergo atresia during follicle development. In our experiments we saw that many of the antral follicles in the proestrous and estrous stages present on a given section showed expression of AT2 mRNA. This is in accord with the notion that in the rat about 70% of the antral follicles are atretic (Mandl and Zuckerman 1950). In additon, during the antral stage strict regulation is imposed on follicular development (Hirshfield 1991). If follicles in the penultimate stage of follicle growth, i.e., when granulosa cells reach the seventh generation of follicular development, are not exposed to the periovulatory surge of follicle-stimulating hormone (FSH) that is important for the selection of follicles ready to ovulate in the next cycle, they undergo atresia (Hirshfield 1988; Hsueh et al. 1994). In cell culture experiments, Tanaka et al. (1995) found that the AT2 receptor is abundantly expressed on atretic rat ovarian granulosa cells that were not stimulated with FSH. In another study, however, examining in vitro perfused rabbit ovaries, Yoshimura et al. (1996) reported that administration of angiotensin II induced ovulation and oocyte maturation via the AT2 receptor. Although the exact role of the AT2 receptor has yet to be elucidated, investigations of other groups have related the AT2 receptor to effects leading to antiproliferative actions (Stoll et al. 1995) and apoptosis (Yamada et al. 1996), the latter phenomenon representing a potential mechanism triggering follicular atresia (Hughes and Gorospe 1991).
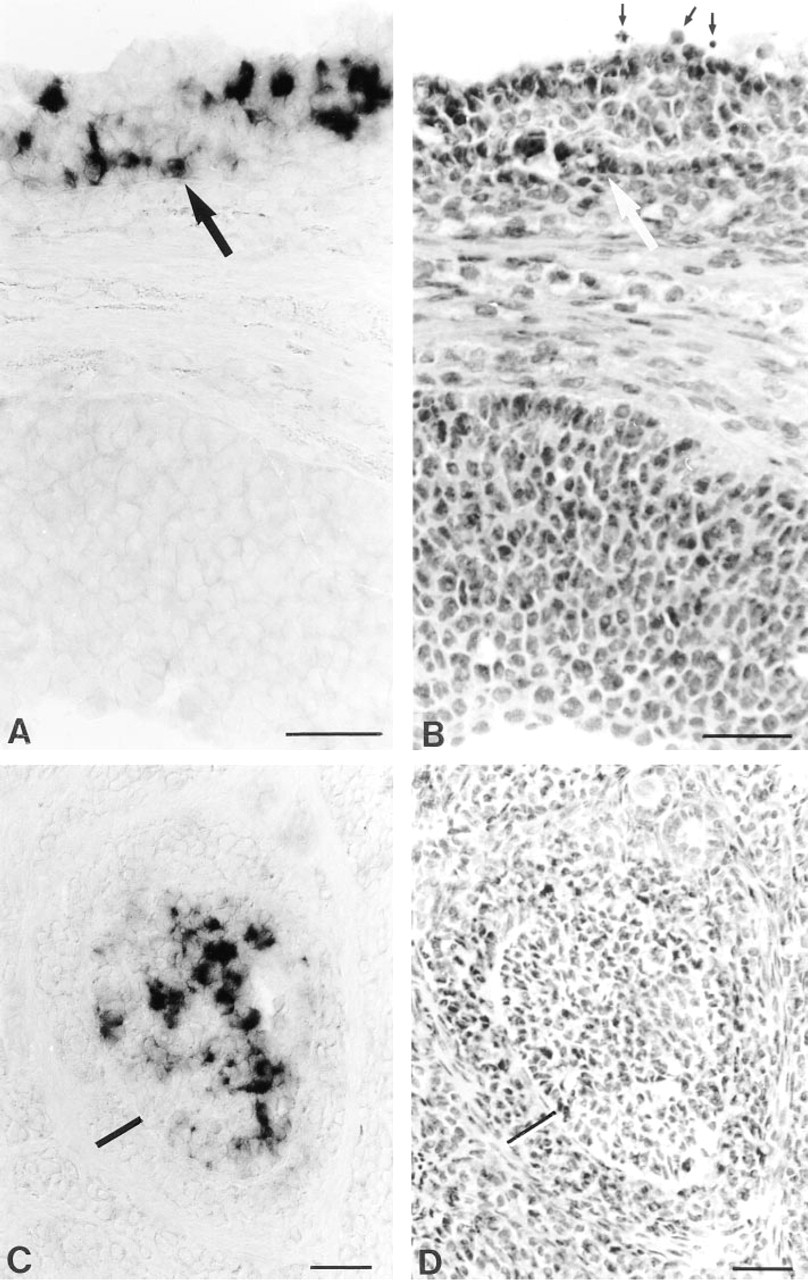
Expression of AT2 mRNA in atretic follicles at late proestrus. In situ hybridization (
Our observations, made by high-resolution in situ hybridization analysis clearly localized the AT2 mRNA to rat ovarian granulosa cells of follicles showing the characteristic of atretic degeneration.
Footnotes
Acknowledgements
Supported by the Forschungsfonds der Fakultät für Klinische Medizin Mannheim der Universität Heidelberg.
We thank Sandra Keller for support in DNA preparation.
