Abstract
Apolipoprotein A-II (apoA-II), an apolipoprotein in serum high-density lipoprotein, is a precursor of mouse senile amyloid fibrils. The liver has been considered to be the primary site of synthesis. However, we performed nonradioactive in situ hybridization analysis in tissue sections from young and old amyloidogenic (R1.P1-Apoa2 c ) and amyloid-resistant (SAMR1) mice and revealed that other tissues in addition to the liver synthesize apoA-II. We found a strong hybridization signal in the basal cells of the squamous epithelium and the chief cells of the fundic gland in the stomach, the crypt cells and a small portion of the absorptive epithelial cells in the small intestine, the basal cells of the tongue mucosa, and the basal cells of the epidermis and hair follicles in the skin in both mouse strains. Expression of apoA-II mRNA in those tissues was also examined by RT-PCR analysis. Immunolocalization of apoA-II protein also indicated the cellular localization of apoA-II. ApoA-II transcription was not observed in the heart. Amyloid deposition was observed around the cells expressing apoA-II mRNA in the old R1.P1-Apoa2 c mice. These results demonstrate that the apoA-II mRNA is transcribed and translated in various extrahepatic tissues and suggest a possible contribution of apoA-II synthesized in these tissues to amyloid deposition.
ApoA-II is the second most abundant apolipoprotein in high-density lipoprotein (HDL) after apoA-I. The main function of lipoprotein is to transport lipids, but the physiological functions of apoA-II are still unclear. Inactivation of the apoA-II gene in mice suggested a complex role of apoA-II in atherosclerosis, with both anti-atherogenic and pro-atherogenic properties (Weng and Breslow 1996). Lipid-free apoA-II mediates the cholesterol efflux from the cell membrane by apolipoprotein–membrane interaction (Yokoyama 1998). In addition, apoA-I and apoA-II have been shown to interact with many cellular systems, including spermatozoa, neutrophils, complements, placental tissue, and bacteria and viruses (Handwerger et al. 1987; Srinivas et al. 1990; Akerlof et al. 1991; Blackburn et al. 1991; Hamilton et al. 1993; Motizuki et al. 1999). In mice, apoA-II is a precursor of senile amyloid fibrils (AApoAII), which were first isolated from senescence-accelerated inbred strain (SAMP1) with severe amyloidosis and were later found to be present universally in mice (Higuchi et al. 1986, 1991b; Yonezu et al. 1986). Whereas liver and intestine contribute equally to production of apoA-I and other apolipoproteins, apoA-II has been reported to be mostly synthesized by the liver (Knott et al. 1985; Kunisada et al. 1986). However, a small amount of apoA-II mRNA has been detected in fetal human intestine by Northern blotting (Hussain and Zannis 1990; Le Beyec et al. 1999), but none in the adult small intestine (Eggerman et al. 1991). To unravel the situation in young and old mice, we performed detailed analysis of apoA-II mRNA expression in specific cell types in extrahepatic tissues using an in situ hybridization technique.
Amyloidosis is defined as a group of biochemically diverse conditions characterized by the extracellular accumulation of fine amyloid fibrils to which normally innocuous soluble proteins polymerize (Sipe 1992; Westermark 1998). One amino acid substitution, proline to glutamine at position 5 in mouse type C apoA-II protein, markedly accelerates age-associated systemic deposition of AApoAII (Higuchi et al. 1991a; Naiki et al. 1993). Type C apoA-II protein polymerizes to amyloid fibril in vitro, but Type B does not (Naiki et al. 1991; Chiba et al. 1999). We developed a congenic strain of mice R1.P1-Apoa2 c that has the apoA-II gene (Apoa2 c ) coding amyloidogenic Type C apoA-II of the SAMP1 strains on the genetic background of the SAMR1 strains (Higuchi et al. 1993, 1995). Histological studies with SAMP1 and R1.P1-Apoa2 c mice indicated that AApoAII is deposited in all tissues except the bone and brain parenchyma in old mice (Takeshita et al. 1982; Higuchi et al. 1999), but that AApoAII deposition was first observed in the papillary layer of the tongue, mucosa, and the submucosa and lamina propria of the small intestine and stomach (Higuchi et al. 1983; Hogenesch et al. 1986). In some systemic amyloidosis, amyloid deposition initiates from particular tissues, e.g., the perifollicular region of the spleen in experimental AA amyloidosis (Hoshii et al. 1997) or the gastrointestinal tract in TTR transgenic mice (Kohno et al. 1997). The fine anatomic structural characteristics of these tissues have been considered responsible for these findings. We hypothesized that apoA-II might be synthesized and polymerize directly to fibrils locally, as has been suggested for AA-amyloid arthropathy in chicken (Ovelgonne et al. 1999) In the present study we found that other tissues in addition to the liver synthesize apoA-II. This finding should be important in elucidating both the mechanism of the lipoprotein metabolism and the pathogenesis of amyloidosis.
Materials and Methods
Animals
R1.P1-Apoa2 c has the amyloidogenic apoA-II gene (Apoa2 c ) and SAMR1 has the non-amyloidogenic apoA-II gene (Apoa2 b ). Both strains were raised under conventional conditions at 24 ± 2C. A commercial diet (CE-2; Nihon CLEA, Tokyo, Japan) and tapwater were available ad libitum. Nine young (3-months-old) and three old (16–18-months-old) R1.P1-Apoa2 c mice and three young (3-months-old) SAMR1 mice were used. All mice were anesthetized with ethyl ether and sacrificed by dislocation of the cervical vertebra. The liver, tongue, stomach, small intestine, heart, skin, spleen, adrenal glands, testis, brain, and kidneys were removed immediately. Half of each organ was fixed in 4% paraformaldehyde with diethyl pyrocarbonate-treated PBS (pH 7.4) at 4C overnight, embedded in paraffin, and cut into 6-μm sections for ISH or into 4-μm sections for immunohistochemical staining as well as hematoxylin and eosin staining. RNA was isolated immediately for RT-PCR analysis from the other half of the organs.
Preparation of apoA-II RNA Probe
A mouse apoA-II cDNA isolated from a SAMP1 mouse liver cDNA library (Kunisada et al. 1986) was used to prepare the apoA-II RNA probe. An apoA-II cDNA containing the whole coding sequence was sub-cloned into the PstI site of pGEM-3Zf(+) vector and the resultant vector was used as a template for construction of the RNA probe. A digoxigenin (DIG)-labeled antisense RNA probe was obtained using a HindIII-cut template and T7 DNA polymerase with a DIG RNA labeling kit (Boehringer Mannheim; Indianapolis, IN). Similarly, a sense probe was prepared for use as the negative control by using an EcoRI-cut template and SP6 RNA polymerase with the same kit.
In Situ Hybridization of apoA-II Transcripts
Tissue sections were subjected to ISH to detect apoA-II mRNA as described previously (Kawakami and Nakayama 1997). In each experiment, both antisense and sense apoA-II DIG–RNA probes were routinely used in serial sections processed in each tissue specimen as positive and negative controls, respectively. After the tissue sections were deparaffinized in Hemo-De (Fujisawa Pharmaceutical Industries; Osaka, Japan), hydrated slides were immersed in 0.2 N HCl for 20 min and then digested with 20 mg/ml proteinase K (Amresco; Solon, OH) at 37C for 20 min, followed by post-fixation with 4% paraformaldehyde. These slides were washed with 0.2% glycine for 10 min and then acetylated for 15 min in freshly prepared 0.25% acetic anhydride in 0.1 M triethanolamine (pH 8.0). The hydrated slides were then dehydrated with ethanol, defatted with chloroform, and air-dried. After prehybridization with 50% deionized formamide and 2 × SSC (1 × SSC = 150 mM sodium chloride, 15 mM sodium citrate, pH 7.0) overnight at 45C, the slides were hybridized with 0.6 μg/ml antisense or sense probe in 50% deionized formamide, 2.5 mM ethylenediaminetetra-acetic acid (EDTA, pH 8.0), 300 mM NaCl, 1 × Denhardt's solution, 10% dextran sulfate, and 1 mg/ml brewer's yeast tRNA at 45C overnight. After hybridization, the slides were washed in 50% deionized formamide and 2 × SSC for 1 hr at 45C with shaking and digested with 10 mg/ml RNase A at 37C for 30 min. After being washed again in 50% deionized formamide and 2 × SSC for 1 hr at 45C, 1 × SSC, and 50% deionized formamide at 45C for 1 hr, the sections were subjected to immunohistochemistry for detection of the hybridized probes using an alkaline phosphatase-conjugated anti-DIG antibody (Boehringer Mannheim). The alkaline phosphatase reaction was visualized with 5-bromo-4-chloro-3-indolyl phosphate and nitroblue tetrazolium.
Immunohistochemistry for apoA-II
Immunohistochemical staining for apoA-II protein was carried out on serial sections from the same organ by the avidin–biotinylated horseradish peroxidase complex (ABC) method with specific antiserum (1:4000 diluted) against guanidine-denatured AApoAII, which reacts with both serum apoA-II and AApoAII amyloid fibrils (Higuchi et al. 1983). In all tissues of old mice, the intensity of the AApoAII deposition was graded 0 to 4 (0, no deposition; 1, slight; 2, moderate; 3, severe; and 4, most severe) in sections stained immunohistochemically. The amyloid index, the average degree of AApoAII deposition in five major organs (heart, liver, spleen, stomach and skin), in three old R1.P1-Apoa2 c mice used here was 3.60 ± 0.40 (mean ± SD).
RT-PCR Analysis
Total RNAs were extracted from the liver, tongue, stomach, small intestine, heart, skin, spleen, adrenal gland, testis, brain, and kidney (RNAeasy Mini Kit; Qiagen, Hilden, Germany). First-strand cDNA was synthesized from 1 μg total RNA of each tissue (First-strand cDNA Synthesis Kit; Amersham Pharmacia Biotech, Piscataway, NJ) and subjected to PCR amplification with Taq DNA polymerase (Promega; Madison, WI). The specific primers for PCR amplification of apoA-II mRNA were a p oA-II-3 (5′-GGAGCTTTGGTTAAGAGACAGG-3′) and aoA-II-4 (5′-TCACTTAGCCGCAGGAGCCG-3′). The RT-PCR amplification was carried out in a 50-μl reaction mixture containing 200 μM each dNTP, 1 × buffer containing 1.5 mM MgCl2, 0.1 μM each primer, and 1.25 U of Taq DNA polymerase. The cycling parameters for RT-PCR were initial denaturation of 1 minute at 94C followed by 30 cycles of 30 sec at 94C, 30 sec at 55C, and 1 min at 72C. A 5-μl aliquot of the PCR product was subjected to 1% agarose (Takara; Tokyo, Japan) gel electrophoresis.
Western Blotting Analysis
Two-month-old R1.P1-Apoa2 c mice were anesthetized with pentobarbital, perfused intracardially with PBS (pH 7.4), and the liver, small intestine, stomach, tongue, skin, and heart were removed. Tissues were sonicated in the lysis buffer containing 20 mM Tris-HCl (pH 7.0), 1 mM EDTA (pH 8.0), 1 mM EGTA, 10 μg/ml leupeptin, and 10 μg/ml pepstatin A on ice and centrifuged at 16,000 × g for 30 min at 4C. Supernatants were collected as cytosol fractions. Cytosol fractions (50 μg protein) were separated by 16.5% SDS-PAGE with a Tris-Tricine buffer system using 8 M urea instead of glycerol (Schagger and von Jagow 1987), electrotransferred to PVDF membrane (Bio-Rad; Hercules, CA) and immunodetected by anti-AApoAII antiserum (1:4000 diluted) as described previously (Higuchi et al. 1997).
In Vitro Amyloid Fibril Extension Assay
The Type C apoA-II monomer was purified from crude AApoAII as described previously (Naiki et al. 1989). HDL particles with Type B and Type C apoA-II were prepared from serum of 3–6-month-old SAMR1 and R1.P1-Apoa2 c mouse serum, respectively, by preparative ultracentrifugation as described previously. Type B apoA-II monomer was purified from HDL of SAMR1 (Higuchi et al. 1999). AApoAII amyloid fibril extension was assayed by the fluorometric method, using the fluorescent dye thioflavin T (ThT). The reaction mixture was prepared on ice and contained 50 ng/μl of sonicated AApoAII and 0.68 μg/μl apoA-II protein, or 2.03 μg/μl HDL containing 0.7 μg/μl apoA-II in 25 mM phosphate buffer (pH 7.5), 300 mM urea. The reaction mixture (40 μl) was put into Eppendorf tubes and the reaction was initiated at the same time in a heat block set at 37C. At each designated incubation time (0–48 hr), the reaction was stopped by cooling on ice and the preparation was subjected to fluorescence spectroscopy, using a spectrophotometer RF-1500 (Shimadzu; Tokyo, Japan). The assay volume was 1.0 ml with excitation at 450 nm and emission at 482 nm (Naiki et al. 1989). The reaction mixture contained 250 nM ThT and 50 mM glycine–NaOH buffer (pH 9.0).
Results
Distribution of apoA-II mRNA-positive Cells in Tissues
Using the antisense apoA-II probes, apoA-II transcripts were clearly observed in the cytoplasm of positive cells, and a negative control study using the sense probe showed no specific reactivity. In the liver, we found positive hybridization signals in all hepatic parenchymal cells (Figure 1A) in young R1.P1-Apoa2 c and SAMR1 mice. However, the intensity of signals for apoA-II was markedly lower in hepatocytes of the old mice (Figure 1B) than in those of the young mice. In general, a strong hybridization signal was observed in specific cells in the stomach, small intestine, tongue, and skin (Table 1) in all mice examined (12 young and three old mice). In the stomach, apoA-II transcripts were restricted to the basal cells of the squamous epithelium and the chief cells of the fundic gland (Figure 1E). In the small intestine, apoA-II transcripts were restricted to the crypt cells and a small portion of the absorptive epithelial cells of intestinal mucosa (Figure 1H). In the tongue, cells with apoA-II transcripts were clearly observed in the basal cells of stratified squamous epithelium (Figure 1K). In the skin, apoA-II transcripts were also noted in the basal cells of the epidermis and hair follicles (Figure 1N). A a weak apoA-II mRNA hybridization signal could be observed in the brain neurons, the lymphocytes in the white pulp of the spleen, and the corona cells of gut-associated lymphoid tissues in the small intestine, in the cortex of adrenal glands, and in the spermatogenic cells, except for spermatozoa in the testis (data not shown). On the other hand, no apoA-II transcripts were seen in the heart (Figure 1Q). The apoA-II-expressing cells and the intensity of the signals in the cells in each tissue were not markedly different among the young R1.P1-Apoa2 c , young SAMR1, and old R1.P1-Apoa2 c mice, except for the liver (Table 1).
Immunohistochemistry for apoA-II Expression
In the young mice of both strains in which no AApoAII amyloid deposition was observed, the localization of ISH signal was entirely consistent with the pattern of the localization of immunoreactive apoA-II protein in serial sections. However, in the old R1.P1-Apoa2 c mice, extensive AApoAII deposition was observed in the extracellular spaces.
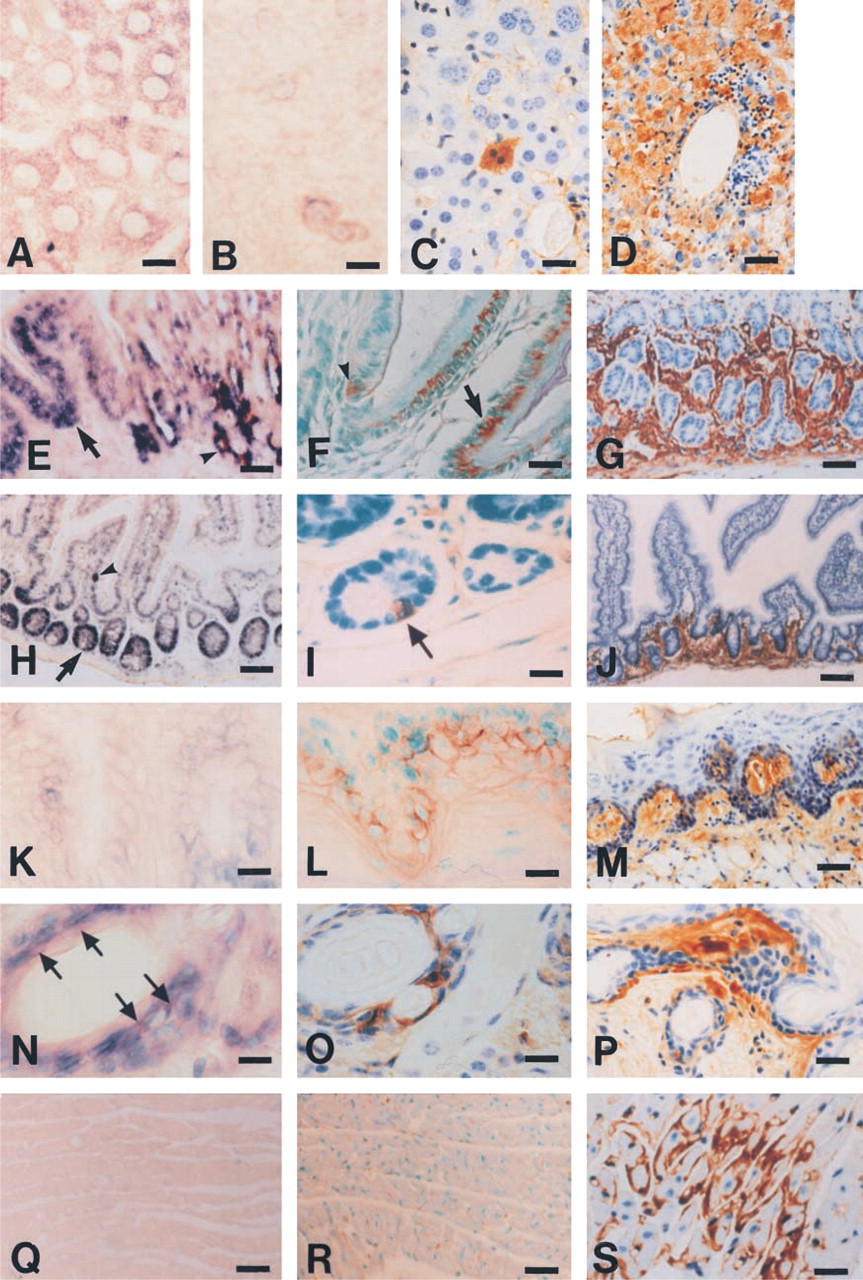
ISH and IHC for apoA-II synthesis in the tissues of young R1.P1-Apoa2c
and SAMR1 mice and old R1.P1-Apoa2c
mice. (
Summary of ISH, immunohistochemistry, and RT-PCR for apoA-II
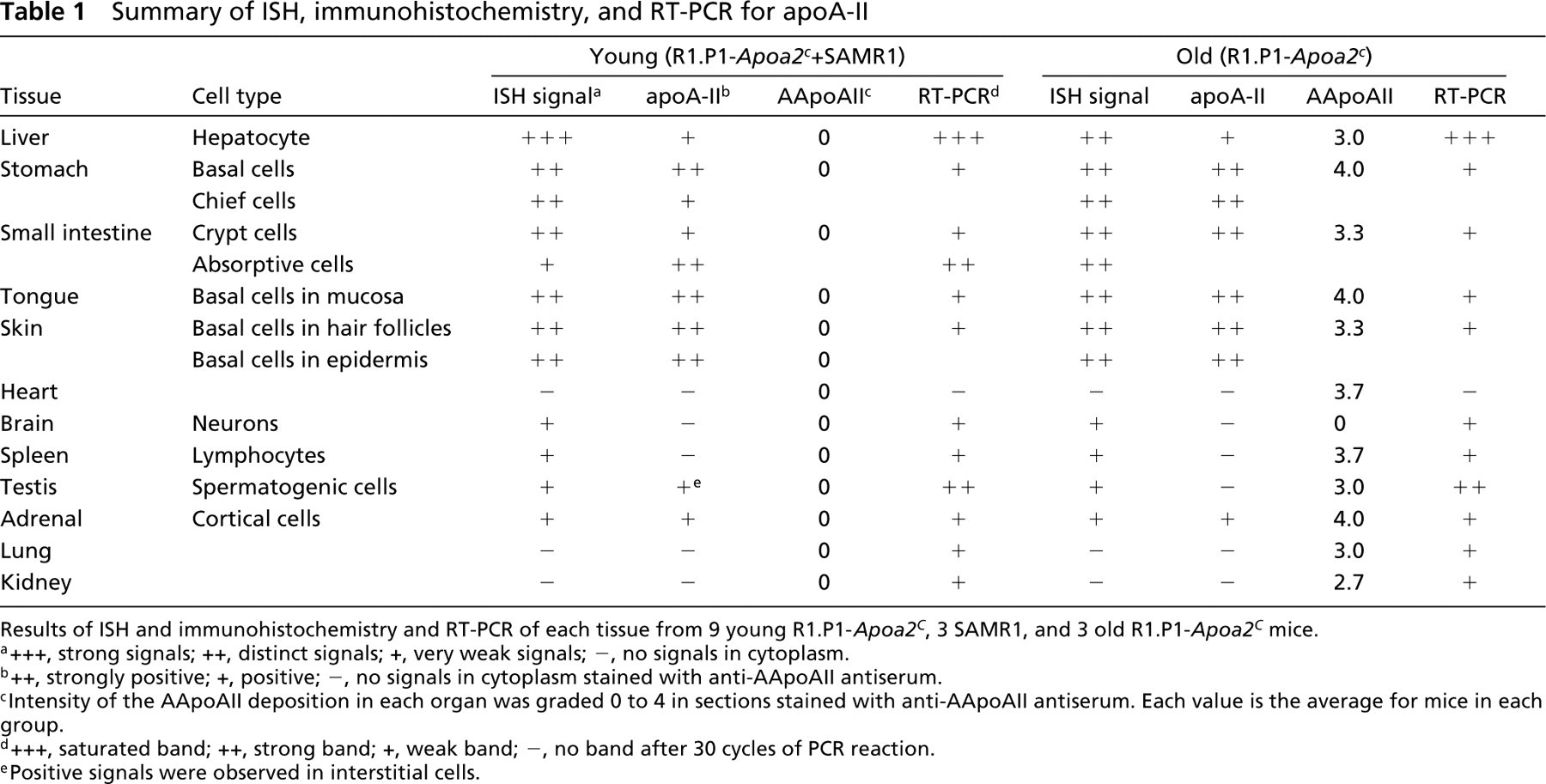
Results of ISH and immunohistochemistry and RT-PCR of each tissue from 9 young R1.P1-Apoa2c , 3 SAMR1, and 3 old R1.P1-Apoa2c mice.
a+++, strong signals; ++, distinct signals; +, very weak signals; -, no signals in cytoplasm.
b++, strongly positive; +, positive; -, no signals in cytoplasm stained with anti-AApoAII antiserum.
cIntensity of the AApoAII deposition in each organ was graded 0 to 4 in sections stained with anti-AApoAII antiserum. Each value is the average for mice in each group.
d+++, saturated band; ++, strong band; +, weak band; –, no band after 30 cycles of PCR reaction.
ePositive signals were observed in interstitial cells.
In the liver, tiny immunoreactive vesicles appeared diffusely in the cytoplasm in the young mice (Figure 1C). AApoAII was deposited around the central vein, extending gradually into the hepatic lobule (Figure 1D) in the old R1.P1-Apoa2 c mice (Table 1). In the stomach, immunoreactive vesicles were weaker in the chief cells than in the basal cells in the young mice (Figure 1F). AApoAII was deposited in the lamina propria and submucosa of the stomach in the old mice (Figure 1G). Similarly in the intestines, weak immunoreactive vesicles were observed in the cells expressing apoA-II mRNA (Figure 1I) in the young mice and AApoAII was deposited in the lamina propria and submucosa (Figure 1J) in the old mice. In the tongue, strong immunoreactive vesicles were seen in the basal cells expressing apoA-II mRNA in the young mice (Figure 1L). AApoAII was deposited in almost all of the papillae of the tongue in the old mice (Figure 1M). In the skin, immunoreactive vesicles were strong in the cells expressing apoA-II mRNA (Figure 1O) and AApoAII was deposited in the papillary layer of dermis just beneath the basement membrane and interstitial tissue around the hair follicles in the skin in the old mice (Figure 1P). Apparent evidence of cellular apoA-II protein immunolocalization was not observed in the heart in the young mice (Figure 1R), but AApoAII deposition was observed in the interstitial tissues between the myocardium and around capillaries in the old mice (Figure 1S). Neither immunoreactive vesicles nor AApoAII deposition was observed in the brain of the young and old mice (data not shown). Although immunoreactive vesicles were not observed in the spleen, testis, and kidney in the young mice (data not shown), extensive AApoAII deposition in these tissues was observed in the old mice (data not shown). In the testis, weakly immunoreactive vesicles were observed in the stromal cells in which apoA-II mRNA was not detected in the young mice (data not shown). In the cytoplasm of adrenal cortex cells, weak immunoreactivity was observed (Table 1).
RT-PCR Amplification
As shown in Figure 2, the RT-PCR products of the expected sizes (320 bp) for apoA-II mRNA were obtained in the liver, stomach, small intestine, tongue, skin, spleen, adrenal gland, testis, brain, and kidney. No RT-PCR products for apoA-II mRNA were obtained in the heart samples.
Table 1 summarizes the results of ISH, IHC, and RT-PCR for apoA-II synthesis in the young and old mice.
Detection of apoA-II Protein in Tissues by Western Blotting
Western blotting analysis after Tris–tricine–urea/SDS-PAGE of the cytosol fractions revealed three anti-apoA-II-reactive bands with molecular weights of 6.8, 14, and 30 kD in the cytosol fractions of the liver, small intestine, and stomach, respectively (Figure 3). These molecular weights corresponded to the apoA-II monomeric, dimeric, and tetrameric forms, respectively. In the tongue, only apoA-II monomer was detected. A weak apoA-II monomer was detected in the heart. This small amount of apoA-II might be in the blood, which perfusion could not wash away completely.
AApoAII Amyloid Fibril Extension with Free apoA-II and HDL In Vitro
When AApoAII amyloid fibrils were incubated with free Type C apoA-II, a rapid increase in fluorescence was observed by 12 h (Figure 4). However, no increase was observed during incubation of AApoAII with HDL isolated from R1.P1-Apoa2 c and SAMR1 mice until 48 hr. When AApoAII was incubated with Type B apoA-II, a slight decrease in fluorescence was observed.
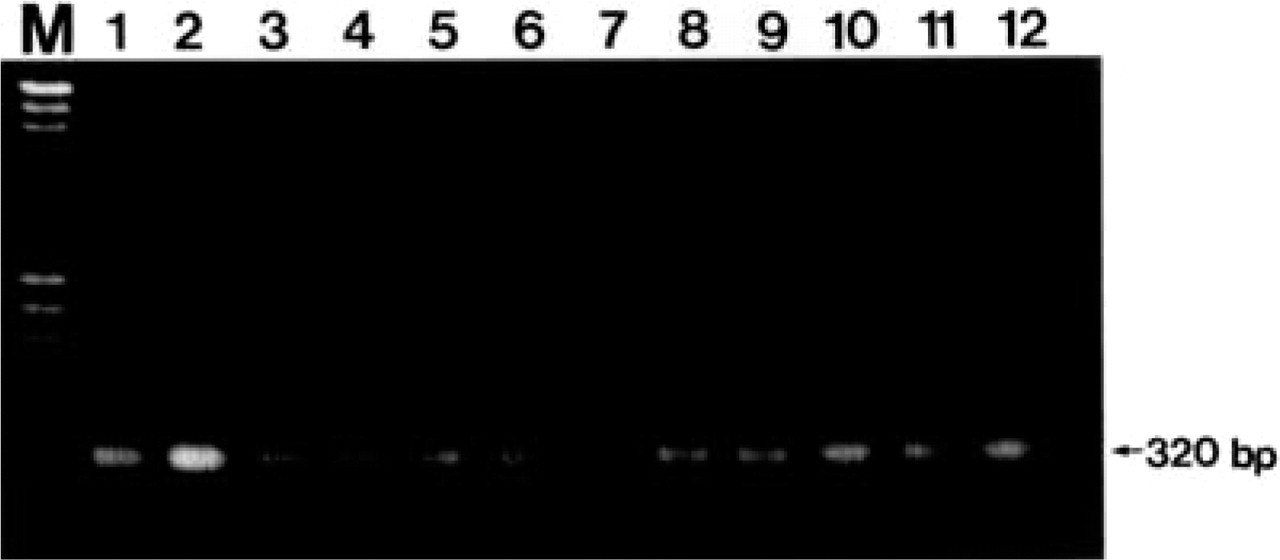
RT-PCR analysis for apoA-II mRNA from various tissues of a 3-month-old R1.P1-Apoa2 c mouse. The PCR products of the expected sizes (320 bp) for apoA-II mRNA were obtained from all tissues examined except the heart. Lane M, marker (λ/HindIII +φχ174/HaeIII); Lane 1, positive control (plasmid pSPA2–16 containing apoA-II cDNA sequence); Lane 2, liver; Lane 3, stomach; Lane 4, small intestine; Lane 5, tongue; Lane 6, skin; Lane 7, heart; Lane 8, spleen; Lane 9, adrenal gland; Lane 10, testis; Lane 11, brain; Lane 12, kidney.
Discussion
We successfully demonstrated apoA-II transcripts in several cell types in various tissues fixed with 4% paraformaldehyde and embedded in paraffin. The apoA-II mRNA was transcribed not only in the liver but also in the stomach, small intestine, tongue, and skin. This is a new finding, and we have been careful to exclude other possible reasons for these observations. No hybridization signal was visualized on the serial sections from the same tissue with the sense probe (data not shown). Furthermore, we verified the presence of apoA-II mRNA in the tissues of the same mouse by RT-PCR analysis using apoA-II-specific primers. These results support the conclusion that the positive hybridization signals in the cells were specific for the presence of apoA-II mRNA. In addition, the presence of apoA-II cDNA sequences in the human and mouse database of expressed sequence tags (dbEST: http://www.ncbi.nsh.gov/dEST) of the liver, spleen, lungs, testis, kidney, placenta, gallbladder, activated T-cells, and multiple sclerosis verify the extrahepatic apoA-II mRNA expression. Both apoA-II mRNA and apoA-II immunoreactivities were clearly seen in the hepatocytes in the liver, the basal cells and the chief cells in the stomach, the basal cells of the squamous epithelum in the tongue, the crypt cells in the small intestine, and the basal cells of epidermis and hair follicles in the skin (Table 1). Although the apoA-II immunolocalization was weak in the hepatocytes, the chief cells in the stomach, and the crypt cells in the small intestine, we considered that these cells might secrete apoA-II immediately after synthesis in the cells. These findings revealed that apoA-II was transcribed and synthesized in these extrahepatic tissues.
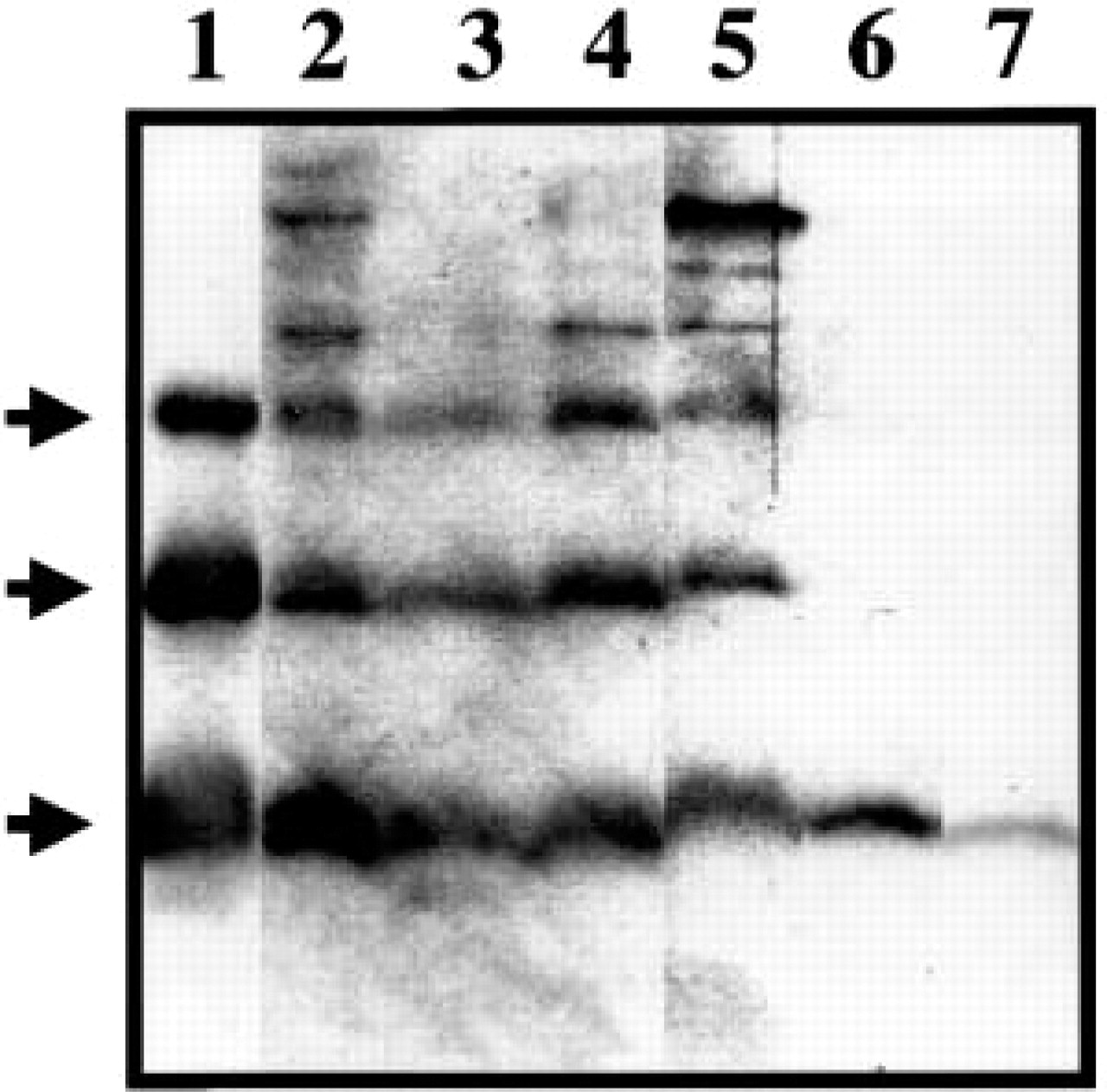
Western blotting analysis for apoA-II protein in various tissues of a 3-month-old R1.P1-Apoa2 c mouse. Lane 1, 0.5 μg AApoAII amyloid fibrils. Cytosol fractions were isolated from the liver (Lane 2), small intestine (Lane 3), skin (Lane 4), stomach (Lane 5), tongue (Lane 6), and heart (Lane 7). Monomeric, dimeric and tetrameric apoA-II (arrows) were detected in all tissues examined except for the tongue and heart.
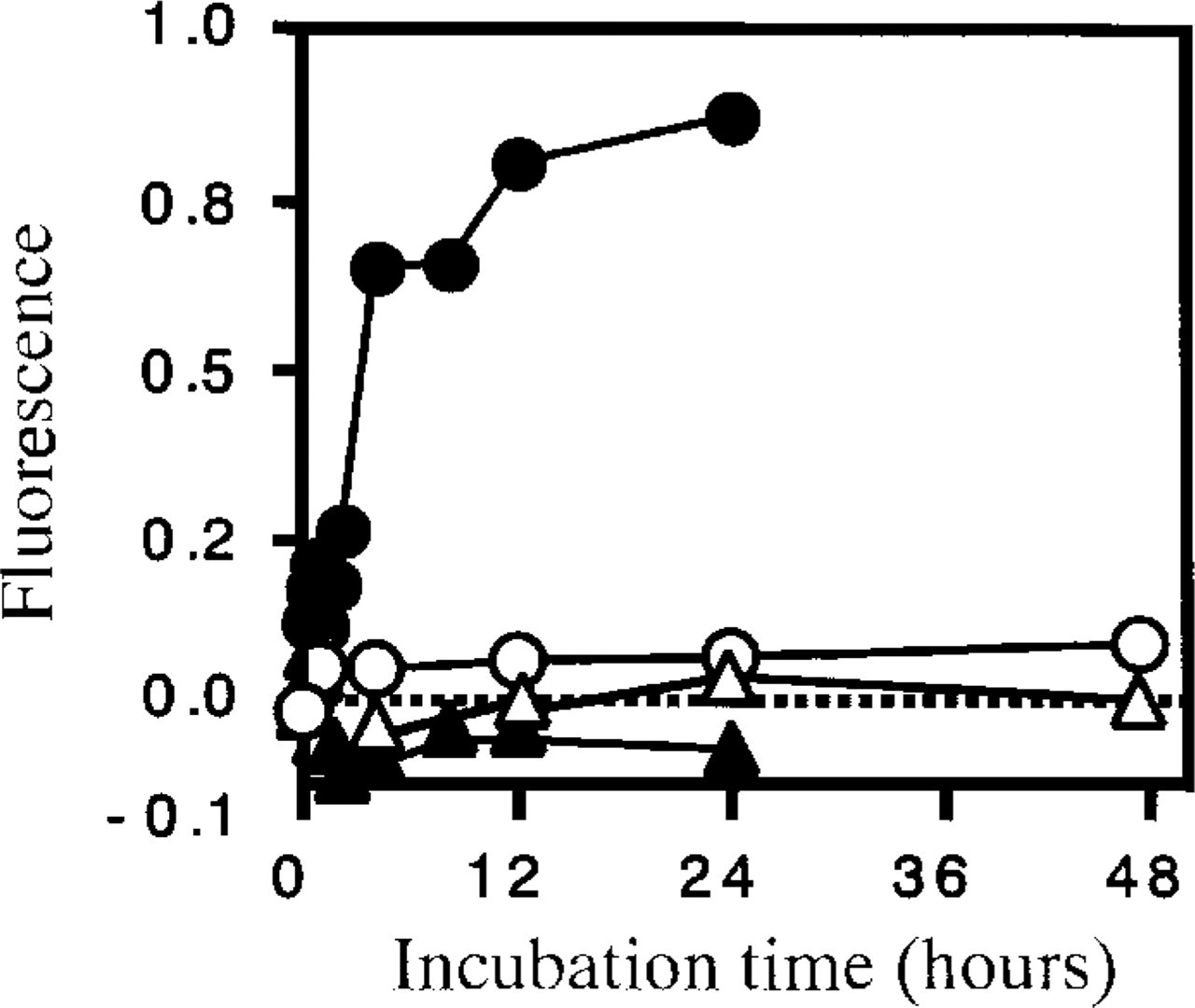
In vitro AApoAII fibril extension kinetics with apoA-II and HDL. Time course of the fluorescence after the initiation of the polymerization reaction. The reaction mixture contained 50 ng/μl of AApoAII fibrils and Type C apoA-II (•), Type B apoA-II (▴), HDL of R1.P1-Apoa2 c (○), and HDL of SAMR1 (△) in 25 mM phosphate buffer, pH 7.5, 300 mM urea.
It remains unclear whether apoA-II proteins synthesized in cells other than hepatocytes have specific roles in cholesterol metabolism and amyloidogenesis. It is noteworthy that the earliest spontaneous AApoAII deposition was observed in the tissues close to the apoA-II-synthesizing cells in the stomach, small intestine, tongue and skin (Higuchi et al. 1998, 1999). Nucleation-dependent polymerization is postulated to be a model that explains well the kinetics of fibril formation in amyloidosis (Jarrett and Lansbury 1993; Kisilevsky et al. 1995; Horwich and Weissman 1997). This model consists of two phases, i.e., the nucleation and extension phases. Because the nucleation phase is thermodynamically unfavorable, it is the rate-limiting step in amyloid fibril formation. The mechanism of nucleation has not yet been elucidated, but it should need highly concentrated monomers. Western blotting analysis revealed a considerable amount of apoA-II in the extrahepatic cells. Once the nucleus has been formed, further addition of apoA-II monomer results in rapid extension of amyloid fibrils. The finding that organs with heavy amyloid deposition trapped more radiolabeled apoA-II in reconstituted HDL suggested that circulating apoA-II binds and extends pre-existing amyloid fibrils (Naiki et al. 1988). Here, we compared the fibril formation ability of free apoA-II with apoA-II in HDL. Free apoA-II (Type C) extended amyloid fibril rapidly but HDL did not (Figure 4). These results suggest that apoA-II proteins synthesized locally may contribute, at least in part, to nucleation and fibril extension in those tissues. We need further studies to elucidate how much the local tissue synthesis of apoA-II contributies to local amyloid deposition.
Type C apoA-II protein has profound effects on cholesterol metabolism and age-related amyloidosis in mice. The mouse strains that have Type C apoA-II protein have low concentrations of plasma cholesterol and early-onset severe senile amyloidosis. In the liver tissue, the levels of mRNA, translational efficiency, and protein synthesis of apoA-II were no different between mouse strains having Type C and Type B apoA-II (Kitagawa et al. 1994; Wang et al. 1997). In this experiment, the non-hepatic cells expressing apoA-II mRNA were the same, and we observed no clear difference in the intensity of hybridization and immunohistochemical signals in any cell types of R1.P1-Apoa2 c and SAMR1 mice (Table 1). The intensity of positive staining in hepatocytes for apoA-II mRNA was markedly decreased in the old mice compared to the young mice. This is consistent with a previous report (Kitagawa et al. 1994), but the mechanism is not yet clear.
In our study, a weak hybridization signal and PCR products for apoA-II mRNA were obtained in the brain, spleen, lung, and testis, but we could not find cellular immunolocalization of apoA-II except in the stromal cells of the testis (Table 1). Because the stromal cells of the testis secrete steroid hormones, the uptake of HDL particles containing apoA-II from the circulation for hormonopoiesis may be responsible for this finding. Additional studies are needed to determine whether apoA-II is synthesized in these tissues. Neither apoA-II mRNA nor immunolocalization was seen in the heart, which suggests that apoA-II is not synthesized in this tissue. AApoAII deposited around the myocardium should come from circulating apoA-II.
In the present study, expression of apoA-II could be detected in the cells with potential proliferation or differentiation activity. The basal cells of squamous epithelium in the stomach, skin, and tongue can proliferate. Hair follicle cells in the skin can also proliferate. The chief cells of the fundic gland can differentiate. In the small intestine, the cells of the bottom of the intestinal crypt are called undifferentiated cells because they move to the top of the intestinal villus with differentiation to the absorptive epithelium. A small part of the absorptive epithelial cells expressed apoA-II mRNA weakly in the present study. This finding is consistent with our previous immunohistochemical findings (Takeshita et al. 1985). Disappearance of the transcriptional activity of an apoA-II promoter associated with the differentiation of Caco-2, the human colon adenocarcinoma cell, has been reported (Le Beyec et al. 1999). These results, taken together, suggest that the expression of apoA-II may be closely associated with the ability of the cells to proliferate or differentiate.
In conclusion, the present in situ hybridization and immunohistochemical localization studies have demonstrated that apoA-II is transcribed and translated not only in the liver but also in extrahepatic tissues, including the stomach, small intestine, tongue, and skin. These findings may be important for elucidating the apolipoprotein-mediated cholesterol metabolism and pathogenesis of mouse senile amyloidosis. A negative relation between the expression of apoA-II gene and the process of cell differentiation was also suggested.
Footnotes
Acknowledgements
Supported by Grants-in-Aid on Scientific Research on Priority Areas (09276209) and Scientific Research (B) (114700596) from the Ministry of Education, Science, Sports and Culture of Japan and by a grant from the Ministry of Health and Welfare of Japan.
