Abstract
Anhidrotic ectodermal dysplasia (EDA) is characterized by defects in the development of teeth, hair, and sweat glands. To study the expression of the human gene defective in EDA in human fetal development (Weeks 6–23 of gestational age) and in adult tissues, in situ hybridization and immunohistochemistry were used. First signs of expression were detected at Week 8 in epidermis and in neuroectodermal cells. Starting at Week 12, osteoblasts and thymus were positive for EDA mRNA. Hair follicles expressed EDA mRNA from 18 weeks. The presence of the EDA protein coincided with mRNA expression in the tissues examined. The expression pattern of the EDA gene is consistent with typical involvement of the skin in the syndrome. However, the expression is not limited to the ectodermal tissues and many sites of expression are not obviously reflected in the clinical features of the syndrome.
X-
Recently, we isolated an X chromosomal gene with mutations in EDA patients (Kere et al. 1996). Expression of the gene was detected in adult scalp kerati-nocytes, hair follicles, and skin glands. Taken together, our evidence strongly suggested that this gene causes EDA when mutated and is required for normal epithelial development.
To gain knowledge of the role of the EDA gene in the developing human fetus, we have studied its expression patterns by mRNA in situ hybridization and immunohistochemistry. We report that the expression of the EDA gene is not restricted to tissues affected in the syndrome. Its expression starts as early as Week 8 of gestation in some tissues and continues through fetal life to some adult cell types.
Materials and Methods
Tissues
Formalin-fixed, paraffin-embedded archival specimens were obtained from the Departments of Pathology of the Universities of Oulu and Helsinki, Finland. The study was approved by the ethics committees of the Departments of Medical Genetics and Dermatology. The following subgroups of histological sections were examined: (a) complete embryos and fetuses at gestational ages of 6, 7, 8, 9, 10, and 12 weeks. All material originated from medical abortions. Fetal age was estimated by menstrual age and morphological criteria; (b) selected tissues from older fetuses: skin biopsies from fetal scalp and trunk at 15, 16, 18, 21, and 23 weeks of gestation; kidney, testis, and gastric and colon mucosa at Weeks 15, 19, and 20; liver at Weeks 15 and 20; pancreas at Weeks 15 and 19; and esophagus at Weeks 19 and 20; and (c) histologically normal adult organ specimens: liver (
Summary of tissues showing expression of EDA and mRNA (for detailed descriptions of cell types and timing, see Results)a
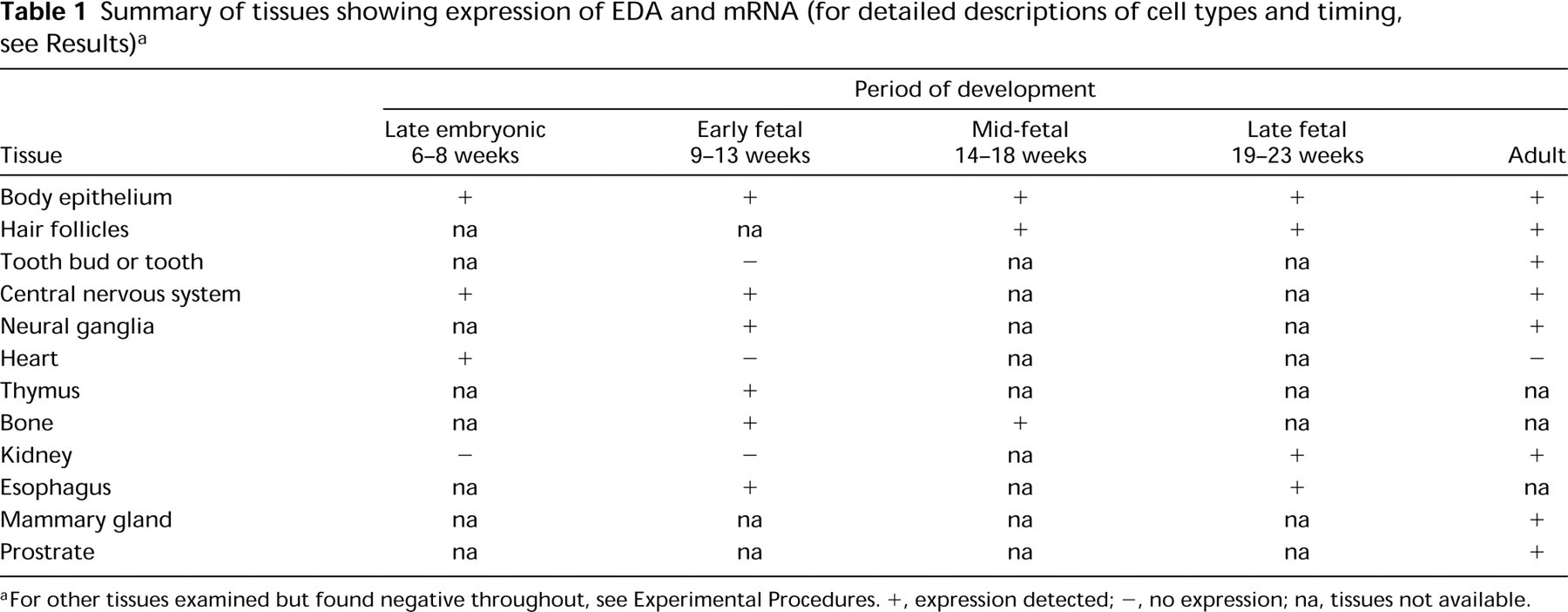
For other tissues examined but found negative throughout, see Experimental Procedures. +, expression detected; -, no expression; na, tissues not available.
In Situ Hybridization
The sequence and specificity of the 551-bp EDA anti-sense cRNA probe have been described (Kere et al. 1996). As a control for nonspecific hybridization, tissue sections in each experiment were hybridized with a sense cRNA probe transcribed from the same DNA. In situ hybridization was performed on 5-μm sections as described (Prosser et al. 1989; Saarialho–Kere et al. 1993). All samples were pretreated with various concentrations of proteinase K, depending on the tissue. Sections were covered with 30–50 μl of hybridization buffer containing 3.5–5 X 104 cpm/μl of 33P- or 35S-labeled anti-sense or sense cRNA probe. Sections were incubated at 50C for 18 hr in a humidified chamber. After hybridization, the slides were washed under stringent conditions [0.1 X standard saline citrate (SSC) at 60C], treated with RNase A (20 μg/ml at 37C), and processed for autoradiography. After exposure for 7–35 days, the photographic emulsion was developed and the slides were stained with hematoxylin and eosin. All slides were included in at least two separate hybridization experiments and were scored independently by at least two investigators.
Immunohistochemistry
Polyclonal antibodies against the EDA protein expressed in
Results
A summary of tissues showing EDA mRNA expression by in situ hybridization is shown in Table 1. More detailed descriptions of different tissues and timing of expression are described below according to organ groups.
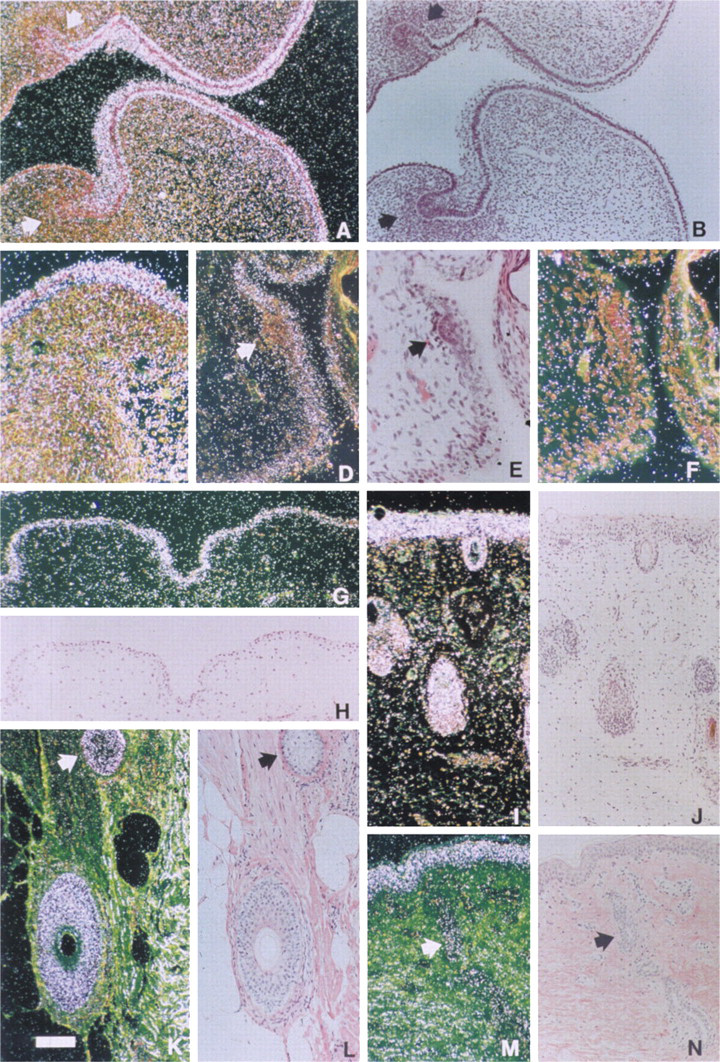
Expression of EDA in skin and hair. Sections were processed for in situ hybridization with a 33P- or 35S-labeled anti-sense RNA probe (see Materials and Methods). (
Expression of EDA mRNA in Epidermis and Its Appendages
The first signs of expression were detected in Week 8 of gestation, when mRNA for EDA was seen in epidermis covering the lower jaw. At Week 9, epidermis of the developing limb was positive and by Week 10, epidermis of fingers, upper and lower jaw, acoustic canal, and scalp displayed EDA mRNA (Figures 1A–1C,1G,1H). No expression was seen in a tooth germ at an early stage of development at Week 10. The epidermis of the trunk was consistently positive beginning at Week 16, whereas the condensed mesenchymal cells in the hair papilla were negative (Figures 1D–1F). EDA mRNA was consistently expressed in the outer root sheath of scalp hair follicles from Week 18 of gestation (Figures 1I and 1J). In contrast, appendages in their early stages of development, including developing hair pegs, were consistently negative. In addition to hair follicles, eccrine sweat glands and sebaceous glands were positive in adult skin (Figures 1K–1N).
Expression in Neuroectodermal Cells
From Week 8, neuroectoderm surrounding the cerebral vesicles and the spinal canal was positive for EDA mRNA (Figures 2A–2H). The expression continued consistently in all brain sections that were studied (Table 1). From Week 10, EDA mRNA was detected in dorsal root ganglia and anterior horn cells of the spinal cord (Figures 2I and 2J). EDA-positive neurons were also seen in adult sympathetic and spinal ganglia as well as hypothalamus (Figures 2K–2M).
Osteoblasts, but not Chondrocytes, Express EDA mRNA
At Week 12 of gestation, EDA mRNA was detected in osteoblasts of ribs (Figures 3A and 3B). In contrast, chondrocytes and hypertrophic chondrocytes of developing bones showed no signal (Figure 3A). Osteo-blasts lining the newly deposited bone matrix also showed intense expression in the calvarial bones at Week 16 (Figures 3C and 3D).
EDA mRNA Expression in Internal Organs
Mesenchymal cells in most organs were negative for EDA mRNA. Bronchial epithelium and lungs were negative in two specimens studied (Figure 3G). Scattered cells in the developing heart showed abundant signal for EDA mRNA at Week 8, and the developing thymus showed strong signal for EDA at Week 12 (Figures 3E and 3F). The esophageal surface epithelium was positive at least at Weeks 12 and 19 (Figures 3J and 3K). At Week 20, the renal collecting tubules and pelvic epithelium were positive (Figures 3L and 3M).
Patterns of Expression in Adult Tissues Other than Skin
EDA mRNA was detected in the duct epithelium of adult prostate (Figures 3O and 3P), mammary gland, kidney, sympathetic ganglia, and hypothalamus (Figures 2L–2N). Pancreas, liver, colon mucosa, endo-metrium, and bronchial epithelium showed no signal. In the one adult tooth section available for study, mesenchymal cells around the tooth were positive for EDA mRNA (Figures 3H and 3I).
EDA Protein Co-localizes with Sites of mRNA Expression
To study whether EDA protein was translated at sites of mRNA expression, immunohistochemistry using a polyclonal antibody was performed on several of the specimens. EDA protein co-localized with mRNA in tissues examined, including the neuroectoderm of brain vesicles (Figure 2H), the dorsal root ganglia (Figure 2K), thymus (Figure 3G), kidney tubules (Figure 3N), esophageal epithelium, and developing bone.
Discussion
Cloning of the EDA gene revealed a predicted trans-membrane protein with no homologies to known proteins that would have suggested a functional role (Kere et al. 1996). Northern analyses suggested a role for the EDA gene in development because of the expression of the EDA mRNA in several other tissues during the fetal period.
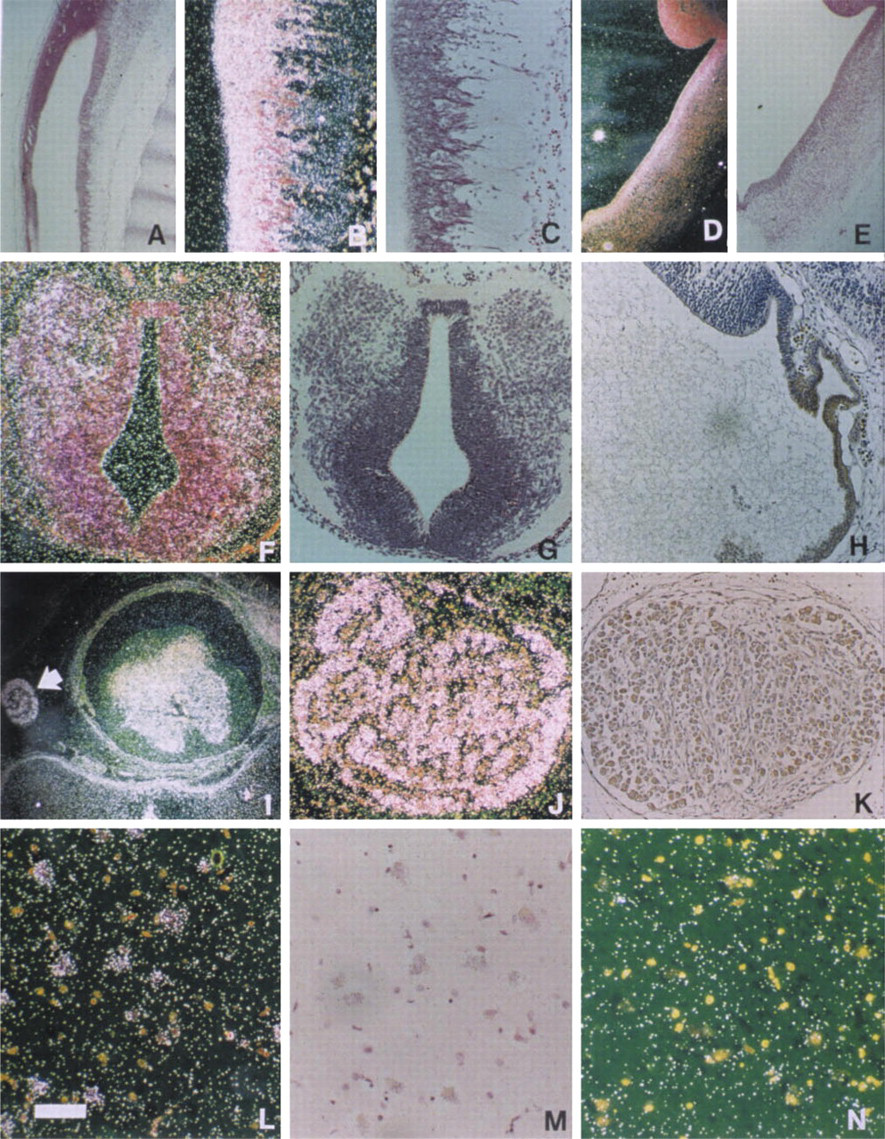
EDA gene is expressed in neural cells. (
A more detailed study of the expression of the EDA gene using both in situ hybridization and immunohis-tochemistry is motivated by several points. First, expression in a wide range of fetal and adult tissues (as suggested by Northern analyses) impelled further study and the identification of cell types expressing EDA mRNA. Second, data on the timing of expression could aid in defining the role of the EDA gene in the chain of morphogenetic events. Third, information on the expression pattern in various organs and cell types might offer clues to the phenotypic features of EDA. Finally, it is useful to know whether transcription of the gene in various tissues and cell types coincides with translation and the presence of the protein product.
Expression of EDA in Fetal Tissues
Northern analyses had suggested that many fetal and adult organs consisting of different cell types express EDA mRNA (Kere et al. 1996). Our results clarify the picture by showing that, in fetal tissues, EDA mRNA was produced by a variety of epithelial and neuroectodermal cells and also by mesenchymal cells such as thymus, bone-forming osteoblasts, and some cardiac muscle cells. In addition to squamous epithelium in various sites of the skin, esophagus and epithelial cells in the kidney tubules, but not bronchi, showed expression. Neuroectodermal cells lining the brain vesicles and the spinal canal and in dorsal root ganglia and anterior horn were positive.
The strong expression of EDA mRNA in osteoblasts contrasts with the absence of EDA mRNA in chondro-cytes, fibroblasts, and dermal papillae in the skin and in mesenchymal cells in many other organs. These results indicate that EDA expression is not restricted to epithelial cells and suggest the presence of specific regulatory mechanisms for controlling EDA gene expression.
Timing of Expression Suggests Multiple Roles
Further support for the specificity of regulation came from the staged timing pattern of EDA mRNA expression during fetal development. Expression was first detected in the epithelial and neuroectodermal cells at Week 8. Within the next few weeks, expression spread to other tissues, and by Week 18 was observed in mature appendages throughout the skin. In general, some tissues showed early expression that was later turned off, whereas in other tissues mRNA first appeared later (Table 1). These observations suggest a double role for the EDA gene: first, a developmental role that is necessary for morphogenesis to be completed and, second, a maintenance role, e.g., in adult skin, supported by continued expression.
Phenotypic Correlations
Expression of EDA mRNA in the developing and adult skin, including consistent expression in hair follicles, is in good agreement with hypotrichosis, one of the prominent phenotypic features in EDA. Hypodon-tia is another major feature, but teeth other than those in early germ stages in fetal sections from Week 10 and one adult section were not available for study. The absence of expression from the early developmental stages in hair and around tooth buds, but expression in mesenchymal cells around one adult tooth, warrants further study.
On the basis of the symptoms in EDA patients, it is conceivable that the function of the EDA protein is associated with the regulation of early morphogenetic events during the development of hair follicles, sweat glands, and teeth. These organs share striking morphological similarities during the initiation of development and during early morphogenesis. Our observation of the lack of EDA expression during early hair follicle morphogenesis and in tooth buds was unexpected but is not incompatible with a developmental role, because expression was seen even earlier (Week 8) throughout the epidermis. A possible interpretation is that an intact EDA gene product may be needed for normal morphogenesis very early in the epithelium, and again in more mature forms of hair follicles, sweat and sebaceous glands, and mesenchyme around teeth.
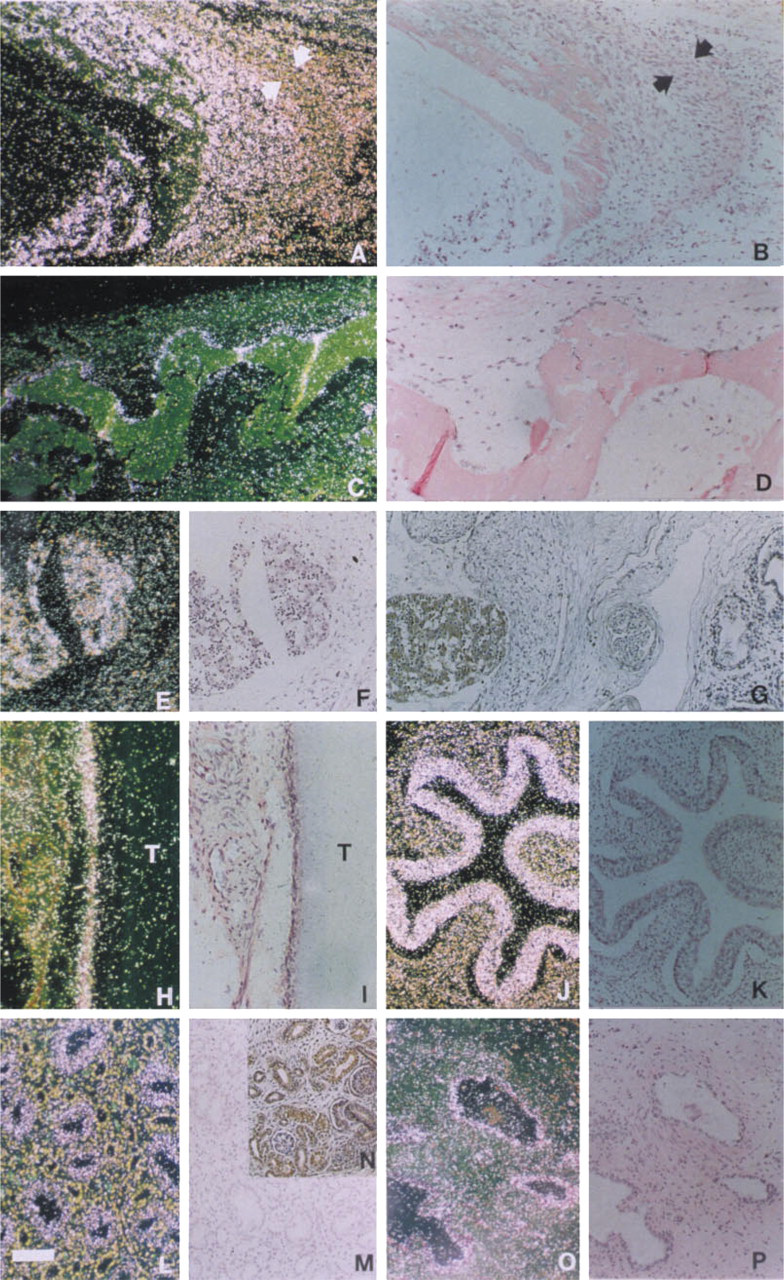
EDA mRNA is expressed in developing bones and internal organs. (
Consistent symptoms and phenotypic features in EDA include mild dysmorphic features of the skull and facial bones and an increased tendency to infections, such as bronchitis (Reed et al. 1970). The finding of EDA mRNA expression in the neuroectoderm and developing bone may, at least in part, explain the slight although distinct dysmorphic features of the skull and facial bones observed in EDA. Such a small effect may indicate a functional redundancy of the EDA protein, i.e., the ability of other cellular factors to replace its function in part.
The airway symptoms might have suggested that the EDA gene was involved in bronchial gland development. Surprisingly, we did not observe EDA signal in bronchial epithelium in fetal development (two samples) or in the adult. The absence of signal may be artifactual and these negative results should be viewed with caution. If the EDA gene is actually not involved in the development of the bronchial epithelium, then bronchial oversensitivity to infections in EDA might result from an indirect mechanism, possibly involving the immune system. Our observation that the fetal thymus specifically expressed EDA provides evidence that the gene product may be associated with the maturation or function of the immune system.
Translation of EDA Protein
The presence of EDA mRNA in a cell type does not necessarily imply translation and expression of the protein. To study whether the corresponding protein antigens could be correlated to mRNA expression, adjacent tissue sections were stained by a specific antibody. We conclude that the sites of transcription and occurrence of protein were in good agreement, suggesting that mRNA in situ hybridization predicts sites of protein expression.
Clues to Molecular Interactions
The overall spatiotemporal pattern of EDA expression in embryonic and adult tissues bears similarities to, but to our knowledge is different from, that of any other known molecule studied thus far in hair and teeth (Holbrook et al. 1993). Analysis of molecular regulation of hair and tooth morphogenesis indicates that there are more similarities than differences in the patterns of molecules involved in the regulation of early development. Furthermore, the composition of extracellular matrix and cell surface receptors shares many similarities in teeth and hair follicles (Chuong et al. 1993; Thesleff et al. 1995).
The expression of EDA in some epithelia resembles EGFR expression (Nanney et al. 1984; du Cros 1993). A possible functional association of EDA with EGF signaling is supported by previous findings in which symptoms of the mouse mutation Tabby were cured by injections of EGF (Blecher et al. 1990). The expression of EGFR was recently found to be reduced in fi-broblasts of EDA patients and Tabby mice (Vargas et al. 1996). Interestingly, osteoblasts also express EGFR, and EGF has been shown to affect osteoblast function (Hata et al. 1984; Davideau et al. 1995). Therefore, it is possible that EDA is involved in the regulation of bone development and functions in the same pathway as EGF. EDA and EGFR are, however, differentially expressed in early hair follicles and tooth buds, in which EGFR is intensely expressed (Thesleff et al. 1995; Partanen et al., manuscript in preparation). One interesting possibility is that EDA could function downstream to the transcription factor LEF-1 (Travis et al. 1991). It is one of the few molecules thus far shown to be necessary for both tooth and hair development (van Genderen et al. 1994; Zhou et al. 1995), and our analysis of the EDA promoter indicated the presence of a potential LEF-1 binding site (Kere et al. 1996). It is an intriguing possibility that EDA might be part of one of these conserved signaling pathways regulating epithelial morphogenesis.
Footnotes
Acknowledgements
Supported by the Sigrid Juselius Foundation, Finska Läka-resällskapet, Academy of Finland, and Folkhälsan Institute of Genetics.
We thank Johanna Pispa for discussions and Alli Tall-qvist and Liisa Sund for excellent technical assistance.
