Abstract
The secretory granule protein syncollin was first identified in the exocrine pancreas where a population of the protein is associated with the luminal surface of the zymogen granule membrane. In this study we provide first morphological and biochemical evidence that, in addition to its pancreatic localization, syncollin is also present in neutrophilic granulocytes of rat and human origin. By immunohistological studies, syncollin was detected in neutrophilic granulocytes of the spleen. Furthermore, syncollin is expressed by the promyelocytic HL-60 cells, where it is stored in azurophilic granules and in a vesicular compartment. These findings were confirmed by fractionation experiments and immunoelectron microscopy. Treatment with a phorbol ester triggered the release of syncollin indicating that in HL-60 cells it is a secretory protein that can be mobilized upon stimulation. A putative role for syncollin in host defense is discussed.
P
The small 13-kDa protein syncollin was first identified as a component of zymogen granules derived from exocrine pancreatic acinar cells (Edwardson et al. 1997). A thorough biochemical characterization of pancreatic syncollin revealed several unusual properties. Syncollin can form oligomeric structures, most likely hexamers, and is predominantly associated with the luminal surface of the zymogen granule membrane (An et al. 2000; Hodel et al. 2001; Geisse et al. 2002). In contrast to typical peripheral membrane proteins, it resists salt washing of the granule membrane but is removed under alkaline conditions when it becomes monomerized (An et al. 2000). Cross-linking studies revealed an association of syncollin with GP-2, the major glycoprotein present in the granule membrane. Both proteins are associated with detergent-insoluble, cholesterol-enriched complexes, indicating that they are present in lipid microdomains (Kalus et al. 2002). Interestingly, purified syncollin binds to liposomes in a pH- and cholesterol-dependent manner, and cholesterol can be coimmunoprecipitated with syncollin (Hodel et al. 2001). The notion that syncollin exists as a homo-oligomer in conjunction with its lipid interaction has led to the suggestion that syncollin might have pore-forming properties. In support of this idea, the oligomer has been shown to form a doughnut-shaped, pore-like structure, both when bound to a solid support and in association with a lipid bilayer (Geisse et al. 2002). Furthermore, purified native syncollin was able to permeabilize both liposomes (Geisse et al. 2002) and erythrocytes (Wasle et al. 2004).
Despite this thorough biochemical characterization, the physiological role of syncollin is still a matter of debate. Previous studies of syncollin knockout (KO) mice revealed abnormalities on the secretory pathway in the exocrine pancreas and a higher susceptability for a hormone-induced pancreatitis, but no changes in secretion of digestive enzymes (Antonin et al. 2002). In contrast, Wäsle et al. (2005) found that pancreatic secretion in KO mice was significantly compromised and concluded that syncollin is required for efficient exocytosis in pancreatic acinar cells.
In this study we present evidence that syncollin is also present in neutrophilic granulocytes of rat and human origin. We show that syncollin is expressed and secreted by promyelocytic HL-60 cells where it is found to be partially associated with myeloperoxidase (MPO)-containing secretory granules. An additional function for syncollin in host defense is discussed.
Materials and Methods
Antibodies
Antibodies used were as follows: 87.1, a mouse monoclonal antibody against recombinant syncollin (An et al. 2000); anti-syncollin B and FG, two polyclonal antibodies raised against recombinant syncollin (B) (Hodel and Edwardson 2000) or a C-terminal peptide (FG) (Edwardson et al. 1997) (kindly provided by J.M. Edwardson, Department of Pharmacology, University of Cambridge, Cambridge, UK); rabbit polyclonal (kindly provided by A. Hasilik, University of Marburg, Marburg, Germany), and mouse monoclonal (PeliCluster; Amsterdam, The Netherlands) antibodies against MPO; RP-1, a mouse monoclonal antibody against rat neutrophils (BD Biosciences-Pharmingen; San Diego, CA); and CD63 Ab-1, a mouse monoclonal against CD63 (Lab Vision Corp.; Fremont, CA). Species-specific anti-IgG antibodies conjugated to horseradish peroxidase (HRP) or to the fluorophores TRITC and Alexa Fluor 488 were obtained from BioRad (Richmond, CA) or Molecular Probes Europe (Leiden, The Netherlands).
Animals and Tissues
Animals were handled according to German law for the protection of animals, with the permission of the local authorities. Male Wistar rats (Charles River; Sulzfeld, Germany) weighing 200-230 g were killed by exsanguination. Tissues were dissected from the animal, immediately frozen in liquid nitrogen, and stored at −80C until use.
Cell Culture, Neutrophil Isolation, and Stimulation of Cells
The human promyelocytic HL-60 cell line (kindly provided by A. Hasilik, University of Marburg, Marburg, Germany) was cultured in RPMI medium containing 10% fetal calf serum (FCS). The human mast cell line HMC-I (kindly provided by J.H. Butterfield, Allergic Diseases and Internal Medicine, Mayo Clinic, Rochester, MI) was grown in Iscove's modified Dulbecco's medium containing 10% FCS and 1.2 mM α-thioglycerol. RBL cells (a rat mast cell line kindly provided by U. Lippert, Department of Dermatology, University of Goettingen, Goettingen, Germany) were cultured in DMEM containing 15% FCS and 1.2 mM sodium pyruvate. The mouse macrophage cell line RAW-264.7 (kindly provided by K. Heeg, University of Marburg) was grown in DMEM containing 15% FCS. HepG2 and AR42J cells were cultured in DMEM containing 10% FCS. All media were supplemented with 2 g/liter sodium bicarbonate, 2 mM glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin (all from PAA Laboratories; Cölbe, Germany), and cells were maintained at 37C in a humidified atmosphere containing 5% CO2. Human neutrophils (>80% pure, as verified by light and fluorescence microscopy) were isolated from the blood of healthy volunteers by density gradient centrifugation over Ficoll-Hypaque. Neutrophils were separated from erythrocytes by sedimentation in 1.5% dextran and subsequent hypotonic lysis of the remaining erythrocytes. Bone marrow was isolated from the tibia and femur of Wistar rats. Isolated monocytes from human blood (95% purity) were kindly provided by A. Kaufmann (University of Marburg). For stimulation experiments, HL-60 cells were either incubated in serum-free medium (Panserin 401; PAN Biotech GmbH, Aidenbach, Germany) or resuspended in a Na+-based solution [127 mM NaCl, 1.2 mM KH2PO4, 5.4 mM KCl, 0.8 mM MgSO4, 1.8 mM CaCl2, 5.6 mM glucose, 10 mM N-2-hydroxyethylpiperazine-N-ethanesulfonic acid (HEPES), pH 7.3]. Cells were incubated in the absence or the presence of phorbol 12-myristate 13-acetate (PMA; 100 nM) for different time intervals at 37C. Cells were spun down afterwards, and the pellet and supernatant fractions were analyzed by immunoblotting or by ELISA. Lactate dehydrogenase (LDH) activity was assayed using commercially available test kits (Boehringer Mannheim; Mannheim, Germany).
Enzyme-linked Immunosorbent Assay
Microtiter plates were coated by overnight incubation at 4C with dilutions of experimental media. Plates were washed six times with phosphate-buffered saline (PBS) (116.4 mM NaCl, 4.9 mM Na2HPO4, 1.7 mM KH2PO4, pH 7.3) containing 0.05% Tween 20 and were thereafter blocked with 200 μl/well washing buffer containing 1% bovine serum albumin (BSA) for 2 hr at room temperature (RT). This was followed by incubation with antibodies against syncollin (B) and MPO (pcRb) (100 μl/well, dilution 1:1000) for 1 hr at RT. Bound antibody was detected using the appropriate secondary antibody coupled to HRP (100 μl/well, dilution 1:2000) and a chromogenic substrate solution (100 μg/ml tetramethylbenzidine in 100 mM sodium acetate, pH 6.0, 2 μl/well 30% H2O2). The reaction was stopped after 10-15 min by adding 1 M H2O2. Each incubation step was followed by a washing step.
Indirect Immunofluorescence and Deconvolution
Cryostat sections of rat tissues and cultured cells were fixed with 4% paraformaldehyde in PBS, pH 7.4, or with cold acetone. Non-adherent cells were carefully spun down on poly-
Electron Microscopy
For routine electron microscopy, cultured cells were fixed with Ito-fixative (Ito and Karnovsky 1968) for 30 min at RT. After postfixation with 1% K4Fe(CN)6-reduced osmium tetroxide (1 hr at 4C), samples were stained with 0.3% uranyl acetate. For cytochemical localization of MPO, HL-60 cells were incubated in DAB medium (0.2% 3,3î-diaminobenzidine, 0.15% H2O2 in 0.1 M cacodylate buffer, pH 7.35) for 1 hr at RT followed by postfixation in 1% K4Fe(CN)6-reduced osmium tetroxide and 0.3% uranyl acetate. Samples were dehydrated in a graded series of alcohol and embedded in Epon 812 (Polysciences Ltd.; Eppenheim, Germany). For immunostaining, samples were fixed in 0.1 M cacodylate buffer, pH 7.35, containing 1% paraformaldehyde (Serva; Heidelberg, Germany). The samples were dehydrated in a graded series of alcohol, embedded in Lowicryl K4M (Polysciences Ltd.), and polymerized at −20C and UV light (360 nm) for 48 hr. Thin sections (70 nm) were incubated with the polyclonal antibodies B or FG directed against syncollin in a dilution of 1:200 to 1:500 and visualized using 10-nm protein A-gold solution (kindly provided by Dr. J. Slot, University of Utrecht, The Netherlands) at a dilution of 1:60 or 1:70, both in 0.5% BSA in PBS. Appropriate controls were performed with anti-syncollin antibodies preincubated with isolated syncollin. Sections were stained with uranyl acetate/lead citrate and analyzed using a Zeiss EM 109 electron microscope (Zeiss; Oberkochen, Germany).
Isolation of Zymogen Granules and Purification of Syncollin
Zymogen granules were isolated as described previously (Dartsch et al. 1998) from the pancreas of male Wistar rats (200-230 g) (Charles River) that were fasted overnight. The following buffer was used for homogenization: 0.25 M sucrose, 5 mM 2-N-morpholino-ethanesulfonic acid, pH 6.25, 0.1 mM Mg SO4, 1 mM dithiothreitol, 10 μM Foy-305 (Sanol Schwarz; Monheim, Germany), 2.5 mM Trasylol (Bayer; Leverkusen, Germany), and 0.1 mM phenylmethylsulfonyl fluoride. Granules were resuspended in 50 mM HEPES, pH 8.0, and lysed by freezing and thawing. Granule membranes and content were separated by centrifugation at 100,000 × g for 30 min. Syncollin was purified as described recently (An et al. 2000). Briefly, freshly isolated granule membranes were washed with 0.6 M KI for 30 min at 4C with gentle agitation. The membranes were then recovered by centrifugation at 21,000 × g for 20 min and washed with 0.1 M Na2CO3, again for 30 min at 4C. The membranes were pelleted by centrifugation at 21,000 × g for 30 min. The supernatant was recovered and dialyzed overnight against 50 mM HEPES, pH 7.6. The resulting precipitate was collected by centrifugation at 21,000 × g for 5 min. The pellet was either dissolved in 50-100 μl of 0.5% taurodeoxycholate in 50 mM HEPES, pH 7.6, or resuspended in 50 mM HEPES, pH 8.0.
Subcellular Fractionation
Subcellular fractionation of HL-60 cells was performed by density centrifugation on OptiPrep (Axis-Shield; Oslo, Norway) gradients. Cells were homogenized in disruption buffer (250 mM saccharose, 10 mM HEPES, pH 7.4, 0.3 mM Na-EDTA, 10 μM Foy-305 (Sanol Schwarz), 2.5 mM Trasylol (Bayer), 0.1 mM phenylmethylsulfonyl fluoride) using a Potter S homogenizer (Braun; Melsungen, Germany). Nuclei and unbroken cells were sedimented by centrifugation at 500 × g for 5 min at 4C. The postnuclear supernatant (PNS) was further separated in a flotation gradient to isolate secretory vesicles from plasma membranes (Dahlgren et al. 1995). The PNS (7 ml) was mixed with a heavy OptiPrep solution (7 ml, 1.12 g/ml). The mixture was layered under 14 ml light OptiPrep solution (1.05 g/ml). Five ml heavy OptiPrep solution was applied to the bottom of the tube. Relaxation buffer (5 ml) was applied on top of the gradient. The gradient was centrifuged at 37,000 × g for 35 min at 4C using a fixed-angle Beckman JA-20 rotor (Beckman Instruments, Inc.; Palo Alto, CA). After centrifugation, 1-ml fractions were collected from the bottom of the tube. Proteins were precipitated with 10% (w/v) trichloracetic acid (TCA) and analyzed by gel electrophoresis and immunoblotting.
Gel Electrophoresis and Immunoblotting
Cells were either solubilized in lysis buffer [25 mM Tris-HCl, pH 8.0, 50 mM NaCl, 0.5% deoxycholate, and 0.5% Triton X-100 supplemented with 1 mM phenylmethylsulfonyl fluoride, 10 μM Foy-305 (Sanol Schwarz), and 2.5 mM Trasylol (Bayer)] for 30 min at 4C or were homogenized as described above. Protein samples were boiled in SDS sample buffer containing DTT, separated by SDS-PAGE, transferred to nitrocellulose (Schleicher and Schüll; Dassel, Germany) using a semi-dry apparatus, and analyzed by immunoblotting. INSTA-Blot ready-to-use membranes were obtained from Imgenex (San Diego, CA). A membrane contained 10 μg per lane of different rat tissue lysates, which were separated on 12.5% acrylamide gels and transferred to Immobilon PVDF membranes. Immunoblots were processed using HRP-conjugated secondary antibodies and enhanced chemiluminescence reagents (Amersham; Arlington Heights, IL). For quantification, immunoblots were scanned and processed using Pcbas software (Raytest; Straubenhardt, Germany).
Isolation of RNA, Reverse Transcription, and PCR
RT-PCR was performed to amplify parts of the coding sequence of rat (GenBank accession number NM_139086) (Edwardson et al. 1997), mouse (GenBank accession number NM_026716), and human (GenBank accession number BC039541) syncollin. Total RNA was isolated using the RNeasy Protect Mini Kit (Qiagen; Hilden, Germany), and reverse transcribed using an oligo(dT) primer and AMV reverse transcriptase (Clontech; Mountain View, CA) at 42C. PCR was performed with 100 ng of template using the following primer pairs: 5î-CAC TTG CGC CAA GCT CTA TGA-3î (forward primer hs syncollin.up); 5î-GCA GTA GAG CGC GGA GAT AGC-3î (reverse primer hs syncollin.down); 5î-TCC CCC CGG GAC CAT GGC TTG TCC AGT GCC CGC A-3î (forward primer rat syncollin.up); 5î-AGC AAT AGC ACT TGC AGT AGA-3î (reverse primer rat syncollin. down); 5î-CCC ACT GCT GCT GGC TTT A-3î (forward primer mouse syncollin.up); 5î-TTG CAG TAG AGG GCA GAG ATG-3î (reverse primer mouse syncollin.down). Samples were analyzed by agarose gel electrophoresis.
RNA Quantitation by Northern- and Dot-blot RNase Protection Assay
For Northern blots, 15 μg of total RNA was subjected to denaturing agarose gel electrophoresis followed by overnight capillary blotting. 32P-labeled probes were generated by random primed labeling of syncollin-specific cDNA fragments using a Hexalabel DNA labeling kit (Fermentas; St. Leon-Rot, Germany). The probe was purified via a Microspin G50 column (Amersham) to exclude unbound radioactivity. Blots were prehybridized in ultrasensitive hybridization buffer (Ultrahyb; Ambion, Austin, TX) for 1 hr at 42C. Hybridization was performed at 42C overnight. For quantitative analysis of mRNA, a dot-blot RNase protection assay was used (Zhan et al. 1997). Briefly, 1-5 μg RNA was directly applied in a constant volume onto a nylon membrane (Qiagen; Chatsworth, CA), hybridized with the above probe, and incubated with RNase A (1 μg/ml). The membranes were exposed to autoradiographic film (Biomax; Kodak) and quantified by densitometric analysis. The methylene blue-stained 18S rRNA band was used to normalize the amount of loaded RNA and to correct for minor loading variations.
Statistical Analysis of Data
Significant differences between experimental groups were detected by ANOVA for unpaired variables using Microsoft Excel. Data are presented as means ± SD, with an unpaired t-test used to determine statistical differences; p values <0.05 are considered as significant, and p values <0.01 are considered as highly significant.
Results
Immunohistological Localization of Syncollin
Syncollin was first identified as a secretory granule protein of the rat pancreas (Edwardson et al. 1997) but was also reported to be expressed in the small intestine, colon, spleen, and parotid gland using in situ hybridization or PCR techniques (Tan and Hooi 2000; Imai et al. 2001). To identify the cells expressing syncollin in the spleen, we first performed immunohistological studies using rat tissues (Figure 1). Because the localization of syncollin to the exocrine pancreas has been confirmed in several studies (Edwardson et al. 1997; An et al. 2000; Hodel et al. 2001; Antonin et al. 2002; Kalus et al. 2002; Wasle et al. 2005), rat pancreas served as a positive control. A specific granular staining of the apical region of the acinar cells was obtained when the polyclonal antibodies B and FG specific for rat syncollin were used (Figures 1A and 1B). The endocrine pancreas with the islets of Langerhans, or interlobular septa, blood vessels, and connective tissues showed no labeling. We observed that staining of cryosections with syncollin antibody B required paraformaldehyde fixation, whereas staining with antibody FG was improved by acetone fixation. In the spleen, single cells or small groups of cells were stained by the syncollin antibodies showing a fine granulovesicular staining pattern (Figures 1C and 1D). Interestingly, the syncollin-positive cells all had lobulated nuclei indicating that they were neutrophilic polymorphonuclear leukocytes. This notion was confirmed by costaining of the cells with a monoclonal antibody specific for rat neutrophilic granulocytes (Figures 1E and 1F). Other leukocytes such as B- or T-lymphocytes of the white pulp were not positive for syncollin. In controls where the immunoreactivity of the syncollin antibodies was blocked by preincubation with isolated syncollin, no labeling of subcellular structures was observed.
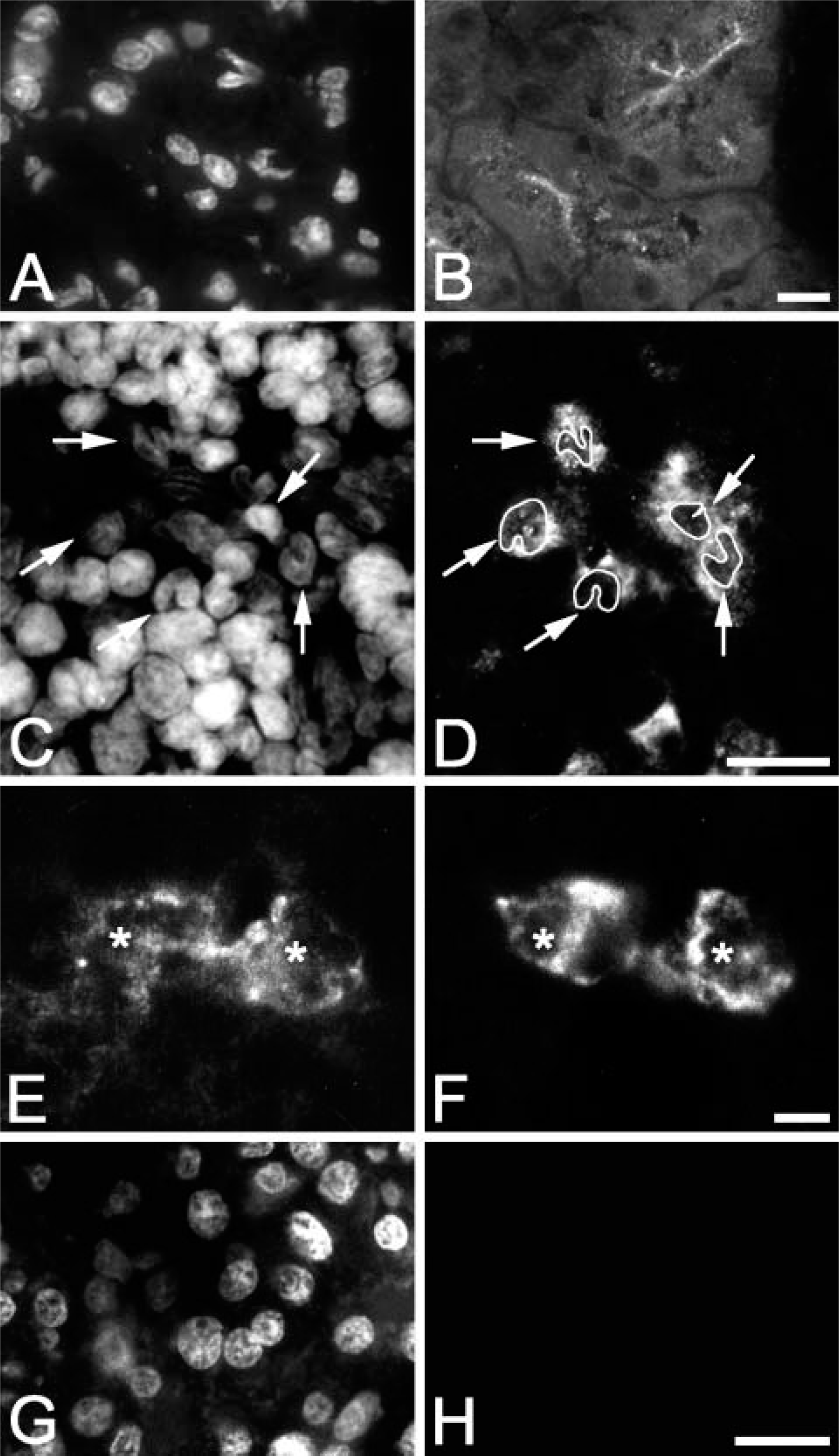
Immunohistological localization of syncollin. Cryosections of rat pancreas (
Tissue-specific Distribution and Expression of Syncollin in the Rat
To verify the morphological data, we analyzed the tissue-specific distribution of syncollin in the rat. Lysates of different tissues were separated on 12.5% acrylamide gels, and immunoblots were performed with a specific monoclonal antibody against syncollin (87.1) (Figure 2A). The syncollin-specific antibody detected a single band of the expected size (~16 kDa) in the lysates of the pancreas, spleen, small intestine, and lung (Figure 2A). The most prominent band was observed in the pancreas lysate. A less prominent band was also observed in the heart and the liver lysate, where an additional band of ~30 kDa was detected under our experimental conditions. Syncollin was absent from brain, kidney, skeletal muscle, stomach, and testis lysates (Figure 2A). To confirm the immunoblot results, RT-PCR was performed with total RNA isolated from different rat tissues and pancreatic AR42J cells followed by agarose gel electrophoresis of the PCR products (Figure 2B). A band with the expected size of 359 bp was found in samples from the pancreas and from stimulated rat AR42J cells, a pancreatic adenocarcinoma, which served as positive controls. A 359-bp PCR product was also detected in samples from the spleen, colon, and a bone marrow preparation when primers specific for rat syncollin were used. Skeletal muscle served as a negative control. With RNA samples prepared from rat liver or from rat hepatoma cells (Fao, WIFB-9), a specific PCR product was not obtained (not shown). A negative result was also obtained with commercially available cDNAs from rat liver and rat heart (not shown). Furthermore, hepatocytes were not labeled by immunofluorescence using syncollin antibodies (not shown). Thus, the immunoblot detection of syncollin in the lung, liver, and heart might be due to contamination with neutrophils from the tissues or the blood or might point to different syncollin isoforms in these organs that are not recognized by our primers. It should be mentioned that to our knowledge such putative syncollin isoforms have not yet been described. However, our findings clearly indicate that syncollin is specific to the epithelial cells of the pancreas, intestine, and spleen. This is in agreement with findings from Tan and Hooi (2000), who reported on the expression of syncollin in the duodenum and colon. Because syncollin was also found to be expressed in the spleen and in a bone marrow preparation, the morphological localization to neutrophils appears to be reasonable.
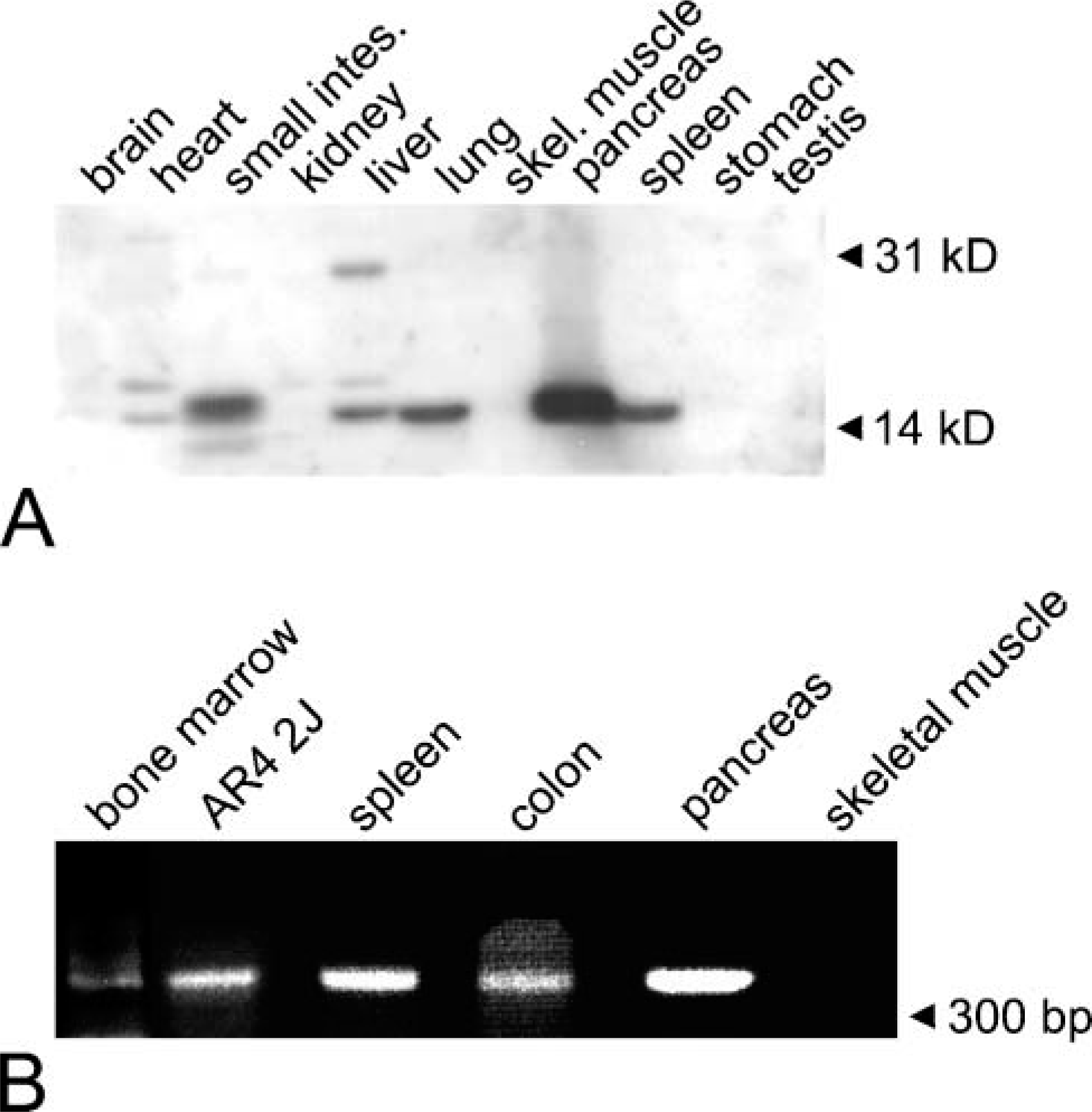
Tissue-specific distribution and expression of syncollin in the rat. (
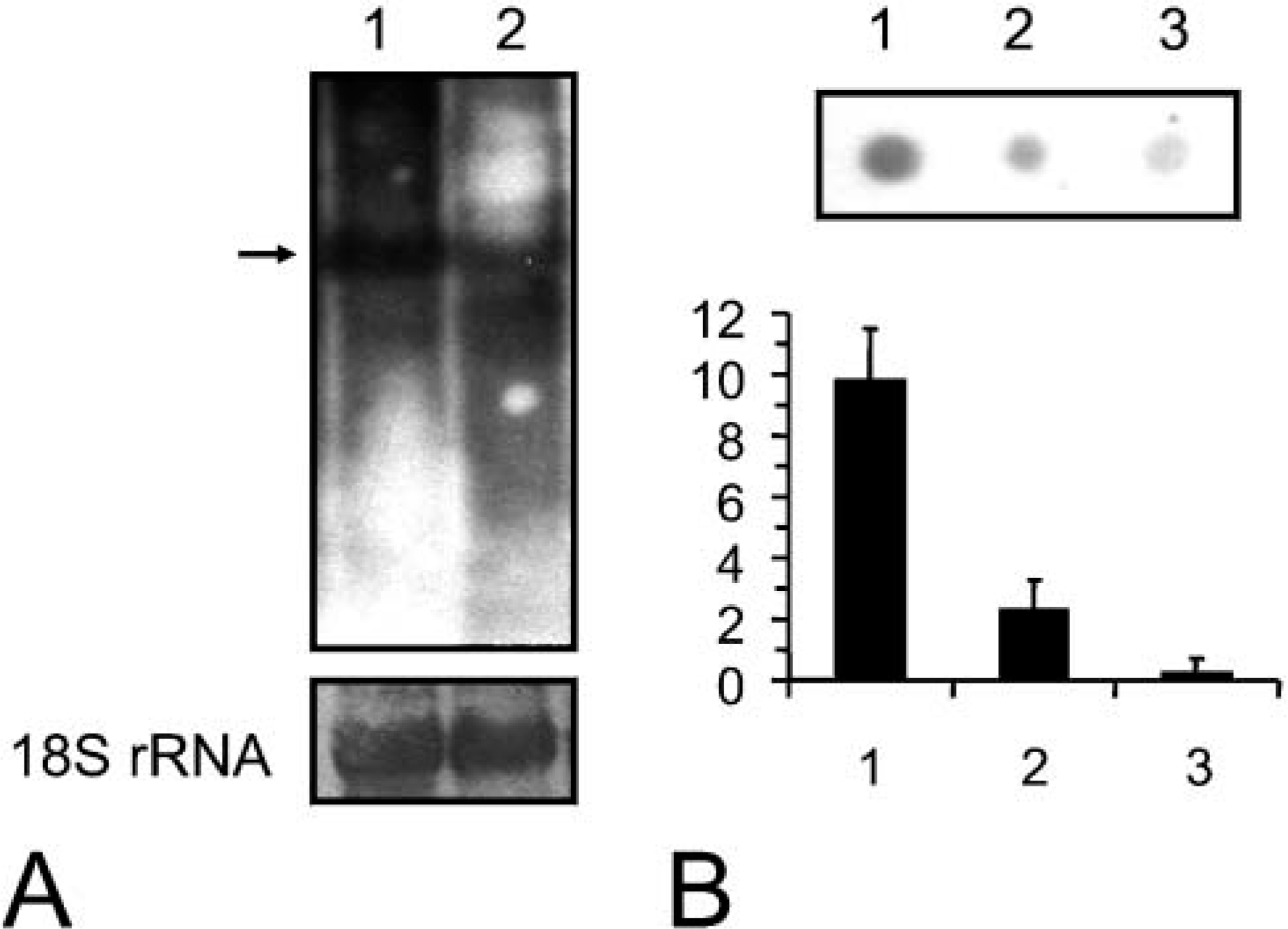
Northern- and dot-blot RNase protection assay for syncollin mRNA. (
To confirm further the expression of syncollin in neutrophils, we next performed Northern analysis of mRNAs from bone marrow (Figure 3A). These experiments revealed the presence of syncollin mRNA in a rat pancreas sample, which was used as a positive control with high expression level, and in a sample from rat bone marrow where expression was significantly lower. Similar loading was confirmed by examination of the 18S rRNA. For quantitative analysis, a dot-blot RNase protection assay was performed (Zhan et al. 1997), which revealed a 5- to 6-fold difference in the relative signal densities (Figure 3B).
Expression of Syncollin in Human Neutrophils
Next we investigated whether syncollin was also expressed in neutrophils of human origin and in other cells involved in immune defense. For this purpose, immunoblots were performed with a postnuclear supernatant from HL-60 cells, a promyelocytic leukemia cell line, and isolated neutrophilic granulocytes from human blood (Figures 4A and 4B). In both homogenates, a protein band of ~16 kDa running at the same size of isolated syncollin from rat pancreas was detected. Furthermore, additional bands with a higher molecular weight were observed, which might represent oligomeric forms of syncollin (Figure 4) (Hodel et al. 2001). For unknown reasons syncollin appeared to be more prominent in HL-60 cells than in neutrophils isolated from human blood. We therefore performed the following experiments with HL-60 cells. The immunoblot results were confirmed by RT-PCR. A band with the expected size of 286 bp was detected in samples from HL-60 cells when primer pairs specific for human syncollin were used. Human pancreas served as a positive control. Syncollin was not detected in HepG2 cells, a well-differentiated hepatoblastoma cell line of human origin, suggesting that it is presumably not expressed by hepatocytes (see above). Furthermore, a variety of cells involved in immune defense were tested negative for syncollin by RT-PCR (Figure 4C) and immunoblotting of cell homogenates or lysates (not shown). Syncollin was absent from monocytes isolated from human blood and from the human mast cell line HMC-I. In addition, syncollin was not detected in RBL cells, a basophilic granulocyte cell line from rat, and in RAW-264.7 cells, a macrophage cell line from mouse (Figure 4C). Although dendritic cells and natural killer cells have not been tested, among the cells responsible for immune defense, syncollin appears to be expressed solely by neutrophilic granulocytes.
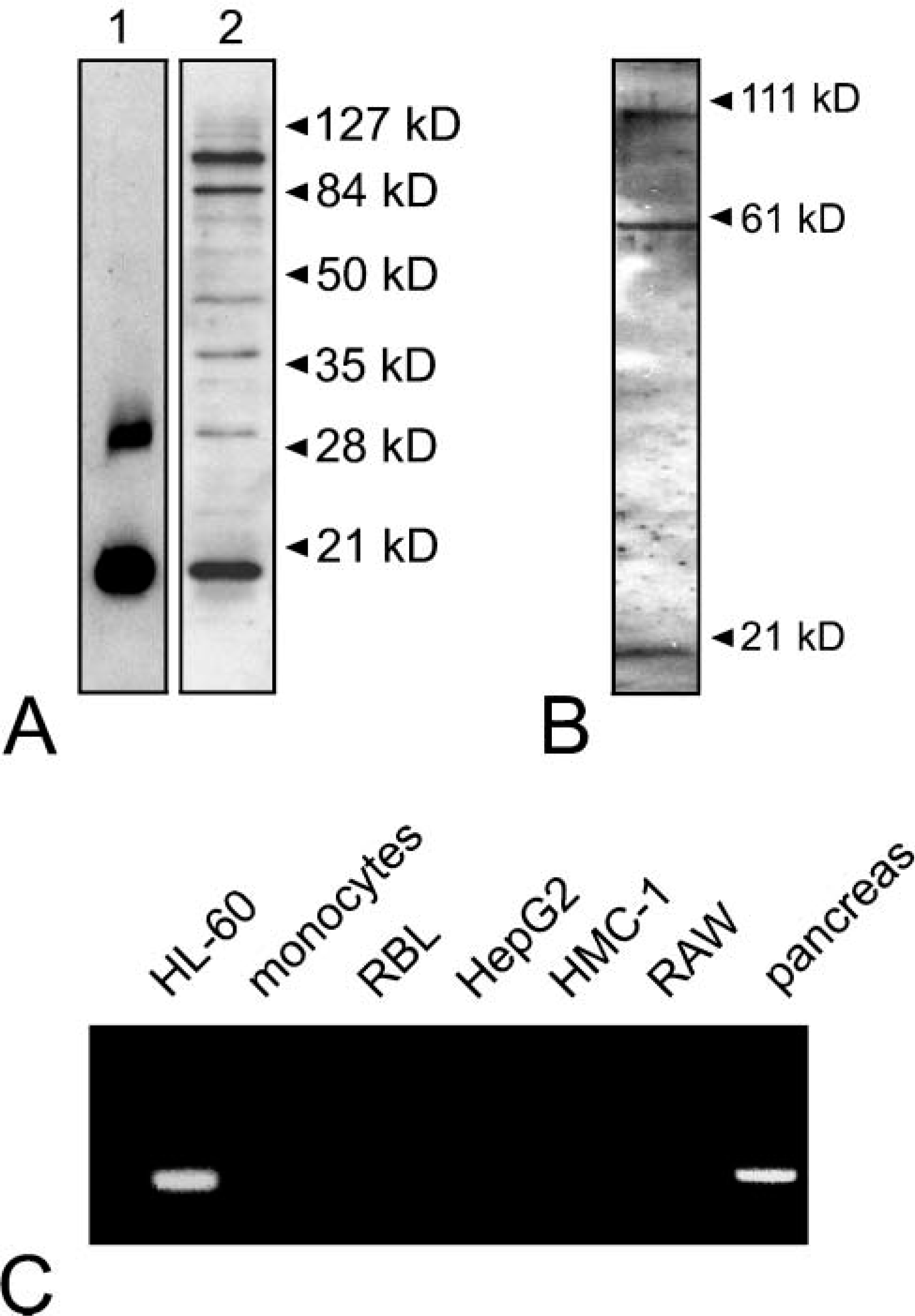
Syncollin is expressed by human neutrophils. Purified syncollin (
Syncollin Is Stored in a Granulovesicular Compartment in HL-60 Cells
To localize syncollin in HL-60 cells, immunofluorescence microscopy was performed. As shown in Figure 5, labeling of HL-60 cells with syncollin antibody B resulted in a punctate intracellular staining pattern. The granulovesicular structures observed were either peripherally located or uniformly distributed within the cell. Image deconvolution revealed partial colocalization ofsyncollin with MPO, a marker for azurophilic granules, in HL-60 cells (Figures 5C-5E). Similar observations were made with isolated neutrophils from human blood, but detection was more difficult than in HL-60 cells (Figures 5F-5H). No staining of granulovesicular structures was observed in controls, where the immunoreactivity of the syncollin antibody was blocked by preincubation with isolated syncollin (not shown).
The presence of azurophilic granules in HL-60 cells was confirmed on the ultrastructural level by electron microscopy and by DAB staining for MPO (Figures 6A and 6B). The azurophilic granules showed some variation in size and in the intensity of the DAB staining for MPO, which reflects differences in the amount of MPO. Immunoelectron microscopy revealed that syncollin is present in some but not all azurophilic granules, thus confirming the immunofluorescence results (Figures 6C and 6E). Gold particles were not observed at the plasma membrane. Although different fixation conditions have been tested, granular labeling of syncollin remained scarce, which might either be due to a reduced accessibility or a low amount of syncollin in those structures. In line with this, we have observed that labeling of pancreatic syncollin was more prominent when the zymogen granules were extracted, indicating that syncollin is more accessible when the granule content is in a decondensed form or partially removed (unpublished observation). However, labeling of granular structures was not observed in controls where the antibody was preincubated with isolated syncollin, indicating that the labeling we obtain is specific.
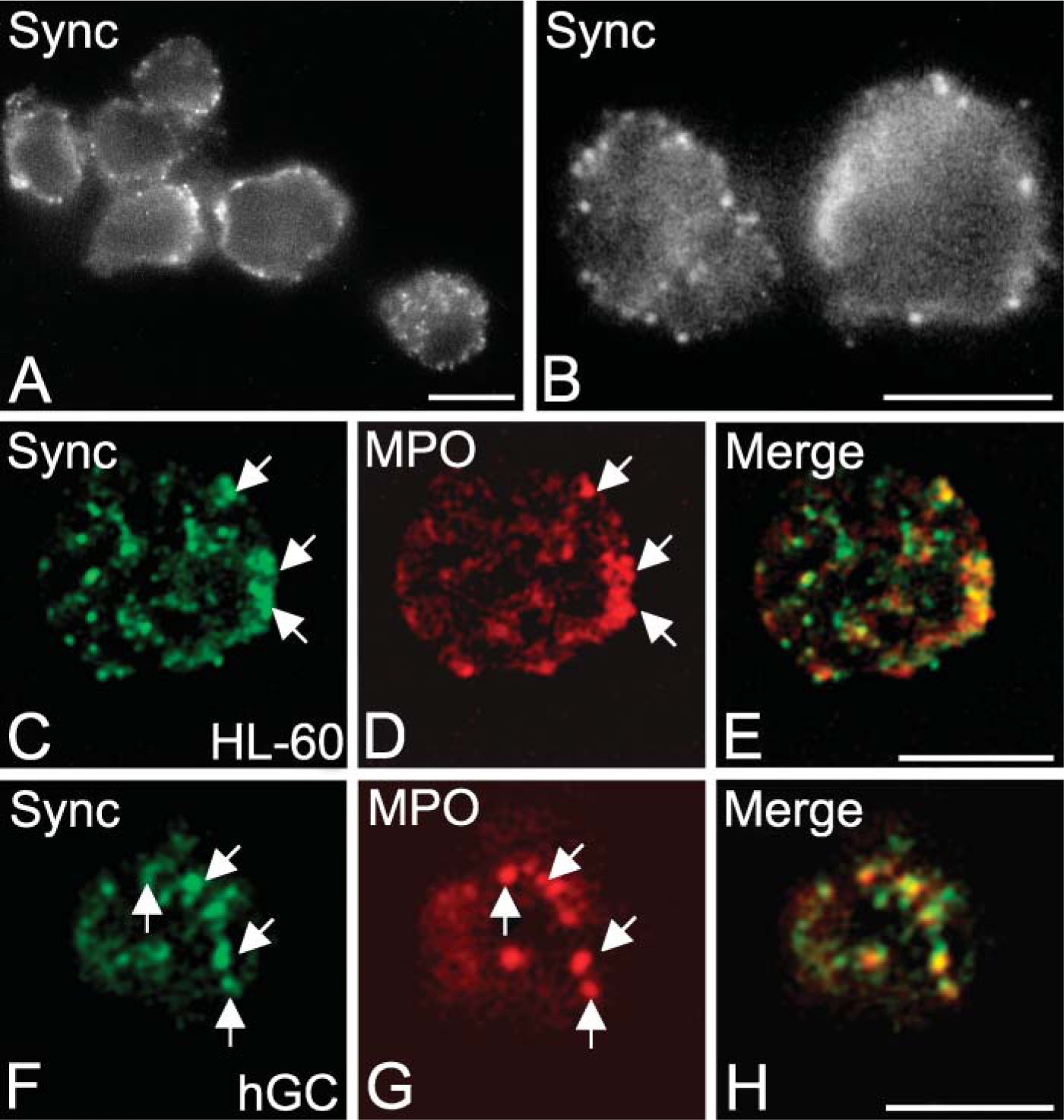
Subcellular localization of syncollin in HL-60 cells. (
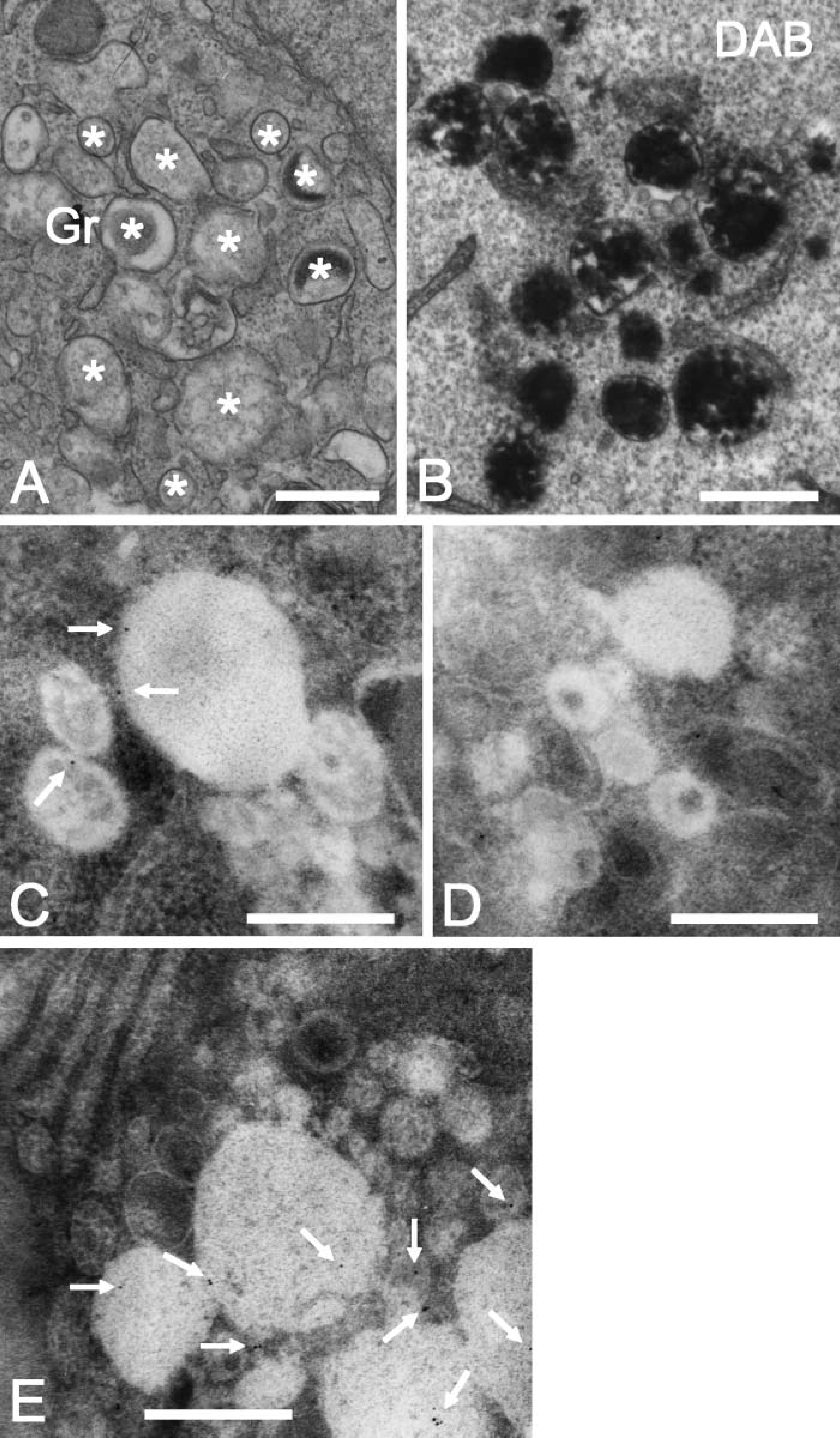
Ultrastructural localization of syncollin in HL-60 cells. (
To further investigate the subcellular localization of syncollin biochemically, HL-60 cells were fractionated by gradient centrifugation on OptiPrep (Axis-Shield) gradients. The cells were carefully homogenized and a PNA was separated on a two-layer OptiPrep gradient. A flotation gradient in which light membranes or vesicles float and separate from heavy secretory granules was used (Dahlgren et al. 1995). The fractions obtained after gradient centrifugation were analyzed by immunoblotting and quantified by densitometry. In Figure 7, the marker profile of a flotation gradient is shown. MPO, a marker for azurophilic granules, is mainly found in fractions 25-34. Staining each fraction with antibodies to syncollin showed a band at ~16 kDa that localized partially in the azurophilic granules. However, syncollin was also localized in the middle fractions (12-18) to which light membranes such as vesicles float. As secretory vesicles have not yet been described in HL-60 cells, which are arrested in maturation at a stage after the formation of azurophilic granules and before the formation of other known granule types, we examined if the light fractions that contain reactivity for syncollin represent the membranes of broken azurophilic granules. Staining with an antibody to CD63 revealed that most of the CD63 was found in fractions positive for MPO. Only a minor amount of CD63 was present in the light fractions containing syncollin. A clear colocalization of these fractions was also not observed with ER marker proteins, which showed a much broader distribution (not shown). Based on the morphological findings it is therefore possible that syncollin localizes to two different compartments in HL-60 cells, namely, azurophilic granules and a yet uncharacterized membrane compartment. However, we cannot rigorously exclude that syncollin in the light fractions does not associate with membrane fragments containing low amounts of CD63 or with other constituents of broken granules (e.g., proteoglycans).
Syncollin Is Secreted from HL-60 Cells upon Stimulation
Pancreatic syncollin has been described as a secretory protein of the regulated secretory pathway (An et al. 2000). To investigate secretion of syncollin from neutrophils, HL-60 cells were treated at 37C with phorbol 12-myristate 13-acetate (PMA) or were incubated without stimulation (Figure 8). In a first set of experiments, HL-60 cells were incubated and stimulated in a serum-free culture medium. After 4 hr, the medium was collected and soluble proteins were precipitated by TCA and processed for immunoblotting. Staining with antibodies to syncollin showed a single band at ~16 kDa in the PMA-treated samples, whereas syncollin was barely detectable in unstimulated controls (Figure 8A). Immunoblotting with an antibody to MPO, a marker for release of a genuine granule constituent, confirmed stimulation. Measurement of LDH activity, a marker for cell breakage, revealed no differences between controls and treated cells (1.15 ± 0.23-fold over control), thus confirming the intactness of the cells during the experiment. In another set of experiments, HL-60 cells were stimulated with PMA in a Na+-based solution, and the release of syncollin and MPO was determined in an indirect ELISA. In a series of experiments, HL-60 cells were treated at 37C with PMA for 30 min or were incubated for 30 min at 37C or 4C without stimulation. As shown in Figure 8B, stimulation of HL-60 cells with PMA triggered the release of syncollin and MPO, whereas temperature elevation did not. These findings indicate that syncollin in HL-60 cells is a secretory protein and can be mobilized upon stimulation.
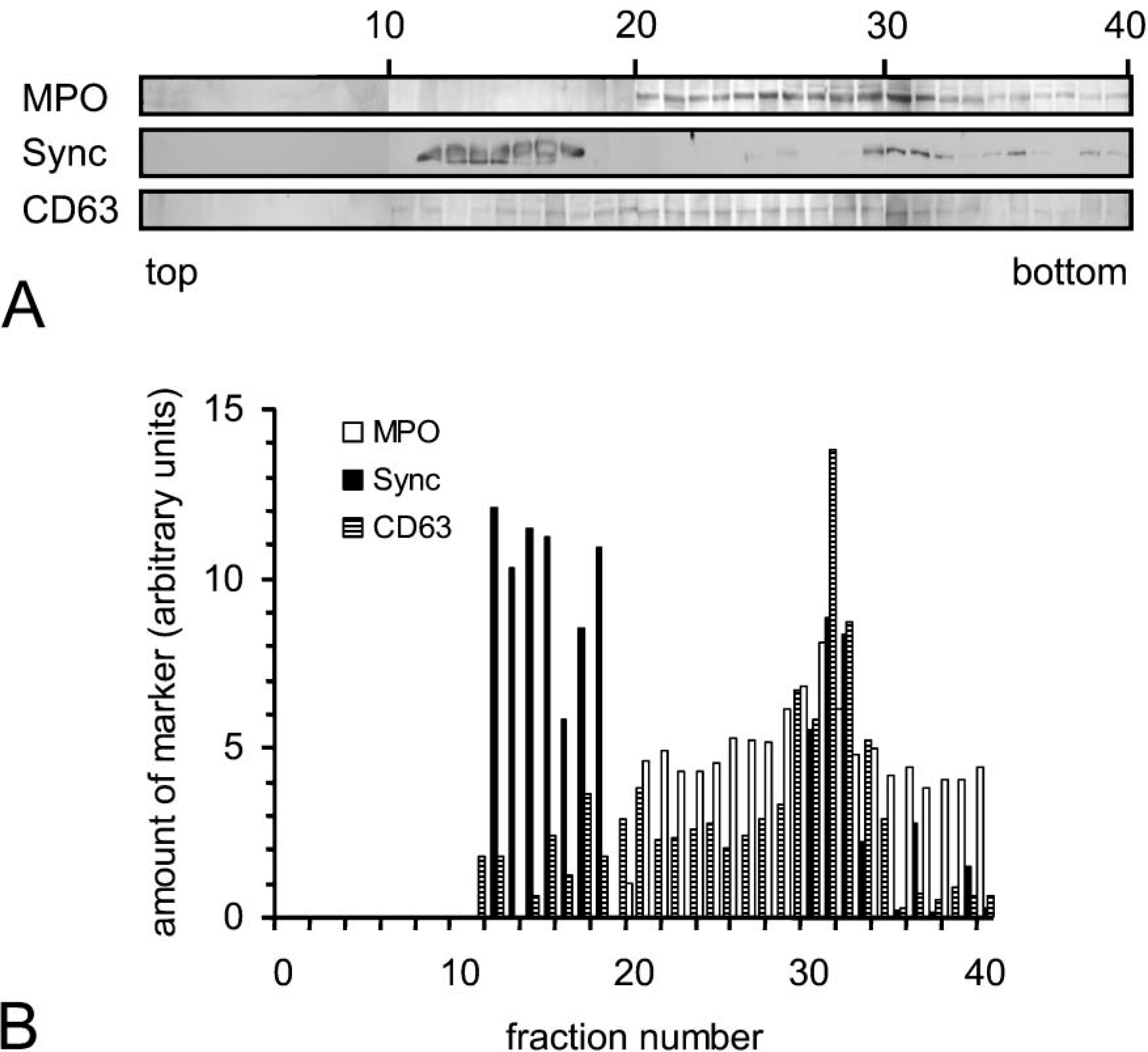
Localization of syncollin in subcellular fractions from HL-60 cells. (
Discussion
In this study, we present evidence that the secretory granule protein syncollin is expressed by neutrophils of rat and human origin. Our findings are supported by the use of different mono- and polyclonal antibodies specific for syncollin in morphological and biochemical studies, as well as by PCR and Northern blotting. By morphological studies and fractionation experiments we show that syncollin is found in intracellular membrane compartments in HL-60 cells. It partially localizes to azurophilic granules and is secreted upon stimulation. We have no indication that syncollin is bound to the plasma membrane in HL-60 cells and assume that it is almost exclusively stored in intracellular organelles.
A recent study has presented the proteome of granules isolated from human polymorphonuclear neutrophils from peripheral blood (Lominadze et al. 2005). Unfortunately, syncollin has so far not been detected by this group. This might be due to the low amount of syncollin present in azurophilic granules from human neutrophils. As indicated, we have chosen rat tissues and HL-60 cells for our studies because syncollin appeared to be less prominent in neutrophils isolated from human blood. It is currently not clear if and how the expression of syncollin in neutrophils is regulated and if such a regulation shows variations in rodents and humans. It is possible that the level of syncollin is upregulated when the neutrophils leave the circulation and enter the lymphatic organs or during the course of an infection. Furthermore, the detection of cationic syncollin on 2D gels is hampered by the fact that MPO, which is a very prominent cationic protein of azurophilic granules, forms a huge spot on 2D gels that covers most of the basic region.
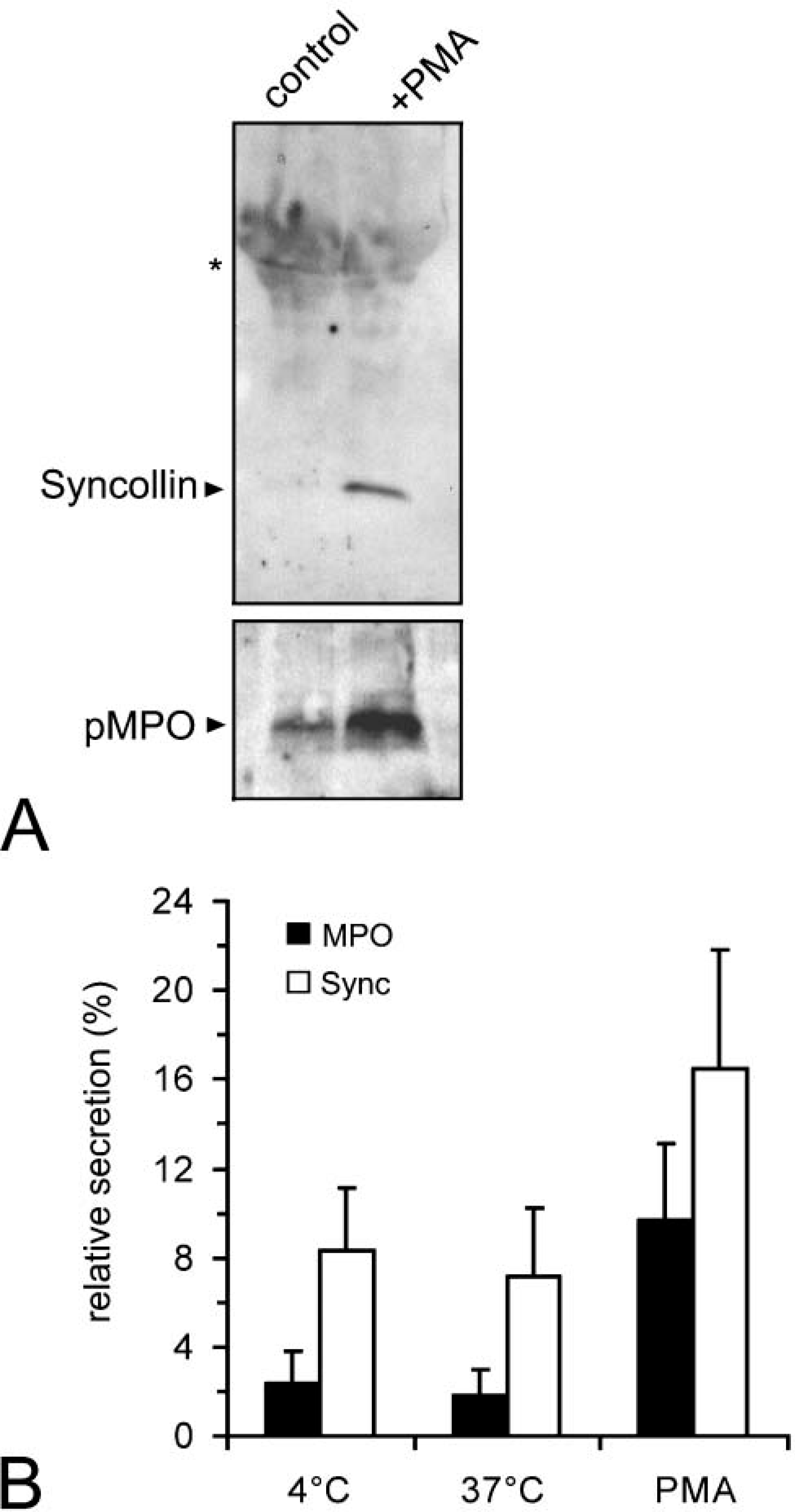
Release of syncollin from HL-60 cells. (
Our data confirm that syncollin is not exclusively expressed by pancreatic acinar cells. In addition to neutrophils, it appears to be present (although in lower abundance relative to rat pancreas) in the duodenum and colon (Tan and Hooi 2000), in the parotid gland (Imai et al. 2001), and according to a recent report in lacrimal acinar cells of the rabbit (Jerdeva et al. 2003). In most cases, the secretory products of these cell types are packaged in secretory granules or vesicles and are released in a regulated manner.
The physiological role of syncollin is still unclear. However, evidence has been presented that it is required for efficient exocytosis in the pancreatic acinar cell, particularly in compound exocytosis (Wasle et al. 2005). As azurophilic granules in neutrophils are regarded as regulated, secretetory organelles (Cieutat et al. 1998), syncollin might also play a role in granule exocytosis of neutrophils. However, syncollin is a secretory protein and does not localize to all but only to a subset of azurophilic granules in HL-60 cells arguing somewhat against a prominent function in neutrophil exocytosis. An interesting, alternative function might be a role for syncollin in host defense. Neutrophils, and especially azurophilic granules, contain several biologically active molecules. Microbicidal polypeptides, such as defensins, lysozyme, bactericidal permeability-increasing protein, and MPO are mostly cationic at physiological pH and thus are able to bind to the negatively charged surface of bacteria. Interestingly, syncollin is also a cationic protein with a theoretical isoelectric point of 8.6. Furthermore, pancreatic syncollin has been shown to interact with lipids and to form oligomeric, doughnut-shaped structures that might have pore-forming properties (Geisse et al. 2002). Purified native syncollin from rat pancreas was able to permeabilize both liposomes (Geisse et al. 2002) and erythrocytes (Wasle et al. 2004). Based on the biochemical properties and the expression pattern of syncollin, it is tempting to speculate that it might fulfill different functions before and after secretion. As shown recently by proteomic studies (Gronborg et al. 2004), syncollin is also a component of the pancreatic juice. In the pancreatic acinar cell where it is best studied, it might be involved in the exocytosis of zymogen granules (Wasle et al. 2005) and afterwards might fulfill a protective or defensive function within the pancreatic duct and the intestinal lumen. This hypothesis would nicely fit the observation that syncollin is expressed by pancreatic, parotic, and lacrimal acinar cells as well as by intestinal epithelial cells.
To examine a possible microbicidal role of syncollin, we have studied the effect of isolated native syncollin from rat pancreas on the growth of different aerobic (Staphylococcus aureus, Enterococcus faecalis, Escherichia coli, Pseudomonas aeruginosa) and anaerobic (Bacteroides fragilis, Prevotella buccalis, Fusobacterium nucleatum, Veillonella sp.) bacteria usually found in the intestinal tract using minimal inhibitory concentration and 3-(4,5-dimethyl thiazol-2-yl)-2,5-diphenyl tetrazolium bromide assays. In these preliminary experiments we could not detect a microbicidal effect of syncollin (unpublished observation). Although isolated syncollin was able to permeabilize liposomes and erythrocytes (Geisse et al. 2002; Wasle et al. 2004), we cannot exclude that additional factors are required for efficient permeabilization of bacterial membranes that are not/no longer present in our syncollin preparation. It is likely that potential pore-forming properties of syncollin will be regulated. A potential binding partner of syncollin in the pancreas is GP-2, the major glycosylphosphatidylinositol (GPI)-anchored glycoprotein within zymogen granule membranes (Kalus et al. 2002). The physiological role of GP-2 is also a matter of debate (Scheele et al. 1994; Schmidt et al. 2001), but based on studies with GP-2 KO mice an extracellular function has been suggested (Yu et al. 2004). Interestingly, uromodulin/THP, a homolog of GP-2 in the kidney, which is also secreted after cleavage of its GPI-anchor, is suggested to function in host cell defense against infection by binding to E. coli and preventing bacterial adherence to the host cell (Bates et al. 2004; Mo et al. 2004). However, an additional function in the regulation of transporters has recently been proposed (Bachmann et al. 2005). Without doubt, further work is required to elucidate the functions and the importance of syncollin during and after acinar and neutrophil secretion.
Footnotes
Acknowledgements
This study was partially supported by the Jürgen Manchot foundation (Düsseldorf, Germany) (MS,HB).
The authors thank all the researchers mentioned in Materials and Methods who have kindly provided antibodies, cell lines, or other reagents. We are grateful to K. Heeg and S. Zimmermann (Department of Medical Microbiology, University of Marburg, Marburg, Germany) for examining the putative microbicidal role of syncollin, and we thank G. Schneider, B. Agricola, and V. Kramer for excellent technical assistance.
