Abstract
Insulin-producing β cells within the vertebrate fetal pancreas acquire their fate in a step-wise manner. Whereas the intrinsic factors dictating the transcriptional or epigenetic status of pancreatic lineages have been intensely examined, less is known about cell–cell interactions that might constitute a niche for the developing β cell lineage. It is becoming increasingly clear that understanding and recapitulating these steps may instruct in vitro differentiation of embryonic stem cells and/or therapeutic regeneration. Indeed, directed differentiation techniques have improved since transitioning from 2D to 3D cultures, suggesting that the 3D microenvironment in which β cells are born is critical. However, to date, it remains unknown whether the changing architecture of the pancreatic epithelium impacts the fate of cells therein. An emerging challenge in the field is to elucidate how progenitors are allocated during key events, such as the stratification and subsequent resolution of the pre-pancreatic epithelium, as well as the formation of lumens and branches. Here, we assess the progenitor epithelium and examine how it might influence the emergence of pancreatic multipotent progenitors (MPCs), which give rise to β cells and other pancreatic lineages.
Introduction
Is it possible to replace the cells of the body when needed to alleviate disease or replace damaged tissues? This question represents a central challenge in the nascent field of regenerative medicine, and is likely to require a fundamental understanding of the processes by which cells acquire their fates and manifest their full functionality. Niches that support cell renewal and differentiation have been characterized in continuously regenerating tissues, such as skin, blood and intestine. Consequently, the dynamics of stem cell compartments in these tissues are well studied and understood. This has led to the successful recapitulation of the normal processes for replacement therapies (Lane et al. 2014). Other tissues, by contrast, have slower turnover rates and no identifiable resident stem cells. The adult heart, kidney and pancreas fall into this latter category. As an alternative approach, research has aimed to elucidate the generation of differentiated cell types during development in the hopes of recapitulating the step-wise generation of tissue cells in the laboratory. Therefore, elucidating the cell and molecular underpinnings of organ ontogeny is likely to instruct either stem cell or induced pluripotent stem cell approaches aimed at coaxing cells to take on desired fates.
In this review, we examine the emergence of pancreatic lineages, including endocrine β cells, within a common embryonic progenitor epithelium. We ask when and where progenitor cells can be identified, and discuss some of the “black boxes” in our understanding of the early morphogenesis and differentiation of this organ. Many other reviews have provided excellent assessments of later pancreas development and β-cell differentiation, covering areas from transcriptional control of β-cell fate, to bud morphogenesis, to cell fate plasticity (Collombat et al. 2010; Pan and Wright 2011; Shih et al. 2013; Stanger and Hebrok 2013). Here, we focus on the earliest progenitor epithelium, about which little is known, and assess how its three dimensional (3D) architecture may provide important clues as to the required developmental events. Ultimately, elucidating the microenvironment that may provide a niche for pancreas progenitors will forward our understanding of β-cell ontogeny initiation and instruct us in recapitulating it for therapeutic approaches.
The Adult Pancreas
The adult pancreas is a complex organ composed primarily of three cell populations with distinct functions: acinar, ductal and endocrine cells. Acinar and ductal cells comprise the exocrine portion of the pancreas. Acinar cells produce digestive enzymes that break down carbohydrates, proteins and lipids (Shih et al. 2013; Slack 1995). Ducts secrete sodium bicarbonate and mucins, and transport digestive enzymes to the duodenum. The exocrine compartment encompasses over 95% of the total mass of the adult pancreas, whereas the endocrine compartment represents only 5%. The endocrine compartment consists of islets that lie scattered within the vastly more abundant exocrine pancreas, and these islets are responsible for maintaining metabolic homeostasis through hormone production and secretion. Endocrine islets contain at least five different cell types, namely glucagon-producing α cells, δ cells that secrete somatostatin, PP cells that produce pancreatic polypeptide, ghrelin-secreting ε cells, and β cells that produce insulin (Orci and Unger 1975). β cells are by far the most abundant endocrine cell type, comprising between 60%-90% of the islet mass. Of great interest is how these cell types arise during embryogenesis (Herrera et al. 1991; Pan and Wright 2011; Pictet and Rutter 1972); also, how such diversity is generated from the progenitor epithelium is an area of active investigation.
Origin of Pancreatic Lineages in the Progenitor Epithelium
It has long been known that, during embryonic development, the three main functional pancreatic lineages arise from the small, common progenitor epithelium of the pancreatic bud. Lineage tracing using transgenic reporter lines for hepatocyte nuclear factor 1 homeobox β (Hnf1β), SRY-box 9 (Sox9), Neurogenin3 (Ngn3), pancreas transcription factor 1a (Ptf1a), and pancreatic duodenal homeobox1 (Pdx1) demonstrate that the pool of cells co-expressing these genes in the early bud gives rise to all lineages (Gu et al. 2002; Kopinke et al. 2011; Kopp et al. 2011; Solar et al. 2009). However, when reporters for the same genes are used to inducibly label cells later during pancreas development (after embryonic day 14.5, or E14.5), Hnf1β and Sox9 expression display restriction to the ductal lineage, Carboxypeptidase A1 (CPA1) and Ptf1a to the acinar lineage, and Ngn3 and Pdx1 to the endocrine lineage. Unlike embryonic liver progenitors, which can expand even after birth facilitating regeneration, pancreatic progenitors appear to be set aside in the early bud epithelium (prior to E12.5) and to be constrained by an intrinsic program that is not subject to growth compensation. Indeed, the early elimination of progenitor cells results in a greatly reduced pancreas by birth, implying that setting their early number is critical to organ size (Stanger et al. 2007)
Progenitors have been further shown, at slightly later stages (E11–E14), to reside in epithelial ‘tips’ or protrusions that bud from the peripheral layer of pancreatic bud cells (Zhou et al. 2007). These tip cells co-express the genes Cpa1, c-Myc and Ptf1a, and were designated as ‘multipotent progenitor cells’ (MPCs). These were shown to be multipotential between E9.5–E13.5 but became restricted to the acinar lineage in the midgestation pancreas after E14.0.
Although these studies have pointed to the early epithelium as containing ‘tip’ MPCs, where they come from and how they ultimately contribute to the pancreatic epithelium, either early or late, remains unexplored. In addition, it is still unknown whether the progenitor population in the early bud epithelium is a homogeneous group of cells that express all the aforementioned markers, or whether it is actually heterogeneous, composed of cells expressing different levels and/or combinations of these genes. Understanding pancreatic progenitors and the step-wise series of fate restrictions they undergo requires a better understanding of the early progenitor epithelium. A better understanding is also needed with regards to how cells within the pancreatic bud acquire their cell fates and whether this either drives, or is influenced by, their morphogenetic reorganization during development.
Overview of Early Pancreas Development
In both mouse and human embryos, the pancreas originates from two separate domains of the foregut endoderm, one dorsal and one ventral. Pancreatic placodes, or epithelial thickenings of the pre-pancreatic epithelium, become apparent by E8.5 in the mouse and are characterized by the expression of Pdx1 and Ngn3 (Villasenor et al. 2008). As mentioned above, and of great interest, is the fact that the functional compartments of pancreas—endocrine, acinar and ductal—all originate from the relatively mysterious, single-layered and polarized epithelium at this time, when the bud begins to emerge and is full of potential. As shown by Jorgensen et al. (2007), the pancreatic epithelium evaginates around E9.0, and by E9.5, distinct dorsal and ventral buds are observable as ‘fin-like’, midline protrusions from the gut tube. During these events, epithelial cells within the placodes columnarize (Fig. 1A, 1A’). This initial morphogenesis is soon followed by rapid and transient stratification into a ‘fist-like’ bud, which has been recently shown to constitute a ‘bag’ of unpolarized epithelial cells (Fig. 1B, 1B’) (Hick et al. 2009; Kesavan et al. 2009; Villasenor et al. 2010). Almost as soon as the stratified layers of cells build up in the budding pancreas, dramatic rearrangements begin to occur. Within layers of epithelial cells, cell polarity is reacquired and microlumens form, thereby progressively resolving the stratified epithelium back into stacked single layers that remodel into branches (Fig. 1C–1D’). However, the dynamic architecture of these rearrangements remains elusive. The bud nonetheless continues to expand, remodel and generate branches via a process of continuous budding of epithelial ‘tips’. Around E11.5–E12.5, rotation of the gut brings the two now elongated pancreatic buds together at their proximal stalks, which fuse and continue to develop into the single organ that will give rise to the adult pancreas (Oliver-Krasinski and Stoffers 2008; Pan and Wright 2011; Pictet et al. 1972). By this time, the pancreatic epithelium is rapidly proliferating and expanding, generating the ramifying, tree-like glandular morphology of the mature organ. Strikingly, to date, the intrinsic geometry and molecular underpinnings of these dramatic early events remain almost completely unknown.
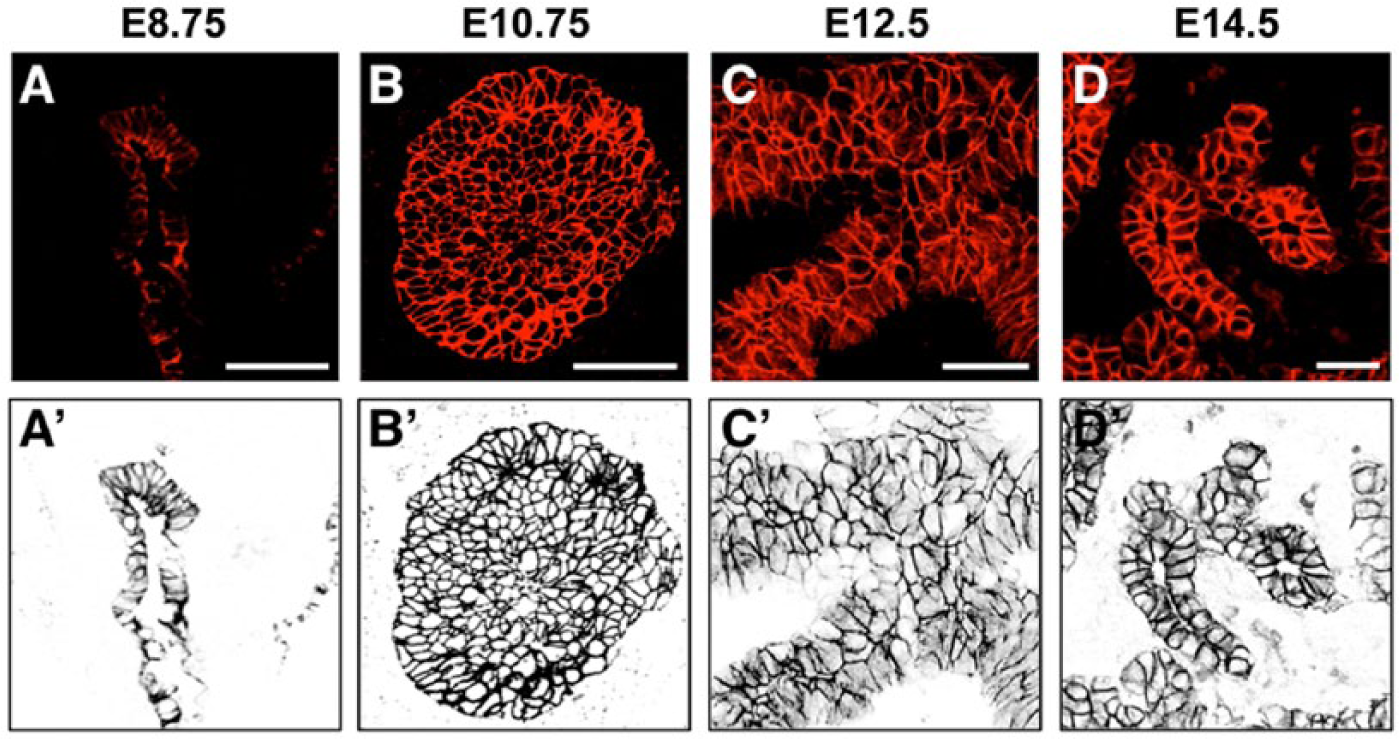
Architectural dynamics of the pre-pancreas progenitor epithelium. The budding pancreatic epithelium undergoes a transient stratification from the initially monolayered gut endoderm, which peaks around embryonic day 10 of development (E10.0) and later resolves back to a monolayer as it forms the highly complex set of tubular branches that comprise the pancreatic gland. The pancreatic epithelium begins as a single-layered epithelial structure (A, A’). By E10.75, the epithelium is highly stratified and begins the process of resolution. By this stage, epithelial cells have reorganized and opened microlumens (B, B’). By unknown cellular mechanisms, the cells rearrange, branches become recognizable, and the epithelium largely resolves back down to a single layer by E14.5, forming a ramifying tree (D, D’). (A–D) Immunofluorescence staining for E-cadherin in red. (A’–D’) False color reversed image of E-cadherin in black. Scale (A–B), 25 µm; (C–D), 50 µm. Scale bars in panels A–D apply to their false color reversed images in panels A’–D’.
In mice, pancreas development is generally divided into two sequential waves, referred to as the ‘first transition’ and the ‘second transition’. The first occurs as the bud forms, up to E12.5, and is characterized by dramatic morphogenesis of the epithelium, as well as emergence of early, primitive endocrine cells. The second transition begins after E12.5 and constitutes massive coordinated growth and differentiation of the three main pancreatic lineages. This review will primarily examine the often overlooked and little understood events of the first transition epithelium.
Pancreas Morphogenesis
Here, we will examine in greater detail the landmarks of embryonic pancreas development. Covering key events in the morphogenesis of the pancreas provides a context for discussing the origins of the different lineages, including β cells. Since a majority of studies have focused on the later features of the fetal pancreas, we will work our way backwards in developmental time. We will cover the range of events from the late gestation pancreas, where features are easily observable and therefore better understood, to those early stages that are still relatively unclear, as they have been more difficult to observe and study.
Pancreatic Branching
Investigation of pancreas morphogenesis has largely focused on the more tractable later gestational stages, much of which involves expansion of this tree-like gland. The mature pancreas consists of a ramifying set of ductal branches connected to a ‘trunk-like’ central duct proximally, and studded with acini at the distal tip of each fine branch. How does the pancreas come to form all of these branches?
In vitro studies have identified different aspects of branching in many organ systems (Affolter et al. 2009; Bellusci et al. 1997; Karner et al. 2009; Metzger et al. 2008; Puri and Hebrok 2007; Varner and Nelson 2014). Tissue explants in 3D matrices, such as Matrigel or fibronectin, allow observation of morphogenesis and growth of ‘organoids’ derived from embryonic tissues, yielding insights into cellular movement when using live imaging in conjunction with lineage reporters (Davies 2005; Packard et al. 2013). Although much of this work has assessed the developing mammalian kidney and lung buds in culture, a key study of pancreatic development used Pdx1-GFP tissue to visualize epithelial growth in culture after isolation of E9.5 and E10.5 pancreatic rudiments (Puri and Hebrok 2007). This report used live-imaging to demonstrate that the pancreas exhibits a “1–3” lateral branching pattern similar to the well-characterized events that occur during early lung formation. However, in clear contrast to lung branching, each branch did not reiteratively lengthen, extend and branch again but, instead, the overall branch epithelium expanded as more tips were added.
A number of in vivo studies have examined the onset of branching of the pancreatic epithelium, which occurs within its sheath of mesenchyme (Hick et al. 2009; Kesavan et al. 2009; Villasenor et al. 2010). Although these studies were limited to static imaging of sectioned and whole-mount tissues, they nonetheless provide insights into branching events at different stages. They reveal how branches emerge from a stratified epithelium full of microlumens (discussed in detail below), while later, during embryogenesis, they show the broccoli-like array of branches that emerge. These studies underscore that, unlike the stereotyped branching patterns of lung and kidney epithelial tubules, pancreatic branching exhibits more variability at the level of individual branches, while still displaying some predictable trends in overall morphology (Villasenor et al. 2010). In addition, in vivo analyses have confirmed that pancreatic branch growth consists of increasing numbers of epithelial tips that fuel overall growth of new branches.
Endocrine Delamination
A critical event that results in the generation of the endocrine compartment of the pancreas is the delamination of islet precursor cells from the pancreatic epithelium. Unlike early endocrine cells of the first transition, which express primarily glucagon or co-express multiple hormones and emerge primarily via ‘cluster-budding’, the bulk of the embryonic endocrine mass is generated via classical delamination following the secondary transition (Gouzi et al. 2011; Pan and Wright 2011; Pictet and Rutter 1972). Indeed, the onset of branching is followed closely by the emergence of endocrine precursors from bipotent trunk progenitors. These commit to their fate, then delaminate from the ductal epithelium and then coalesce into islets. This delamination is characterized by an initial expression of the transcription factor Ngn3, leading to a repression of E-cadherin expression through Snail2, which is required for the endocrine population to leave the trunk epithelium and ultimately differentiate (Gouzi et al. 2011). As pancreatic development progresses in the mouse, endocrine precursors mature and differentiate, leading to the formation of islets composed of a β−cell core that is surrounded by an α-, δ- and PP-cell mantle (Pan and Wright 2011). Delamination of the endocrine precursors has been shown to be a critical step in the differentiation of β cells and the other endocrine fates along with overall pancreatic morphogenesis, as loss of Ngn3 in the pancreas results in the absence of fully differentiated endocrine cells, as well as an aberrantly expanded ductal population and abrogation of the acinar tip compartment (Magenheim et al. 2011). How is a cell within the epithelium determined to delaminate and then how does it find other endocrine cells to coalesce with and form islets? These questions remain open. However, findings suggest that cell fate is intimately tied to tissue architecture, and that proper morphogenesis is critical to the specification and/or commitment of pancreatic cell fates, including that of β cells.
The Pancreatic Ductal Plexus
A better understanding of pancreatic morphogenesis and branching has surprisingly been gleaned from studies not of the epithelium itself or its outward appearance but of its luminal network. In the last few years, it has become clear that, similar to other glandular tubes, the pancreatic ductal network arises via the fusion and interconnection of microlumens into an internal network (Hick et al. 2009; Villasenor et al. 2010). Strikingly, this 3D multi-lumen array of tubes appears prior to outward evidence of branching and continues to expand along with the growing pancreas. Somehow, the internal network of lumens and the outwardly evident branches co-align and a monolayered, tree-like gland results. By E14.5, the mouse pancreas displays a highly complex and dense arrangement of lumens and branches. As these events occur, over time, remodeling of the epithelium must involve some lumens regressing and disconnecting, while others extend and branch further. The ultimate result is a ramifying hierarchical tree with single-lumen branches that feed into a main central duct. Although the dynamics or molecular underpinnings of epithelial remodeling have not been well examined, a few reports note that genetic ablation of key transcription factors, such as Pdx1 and nuclear receptor subfamily 5 group A member 2, Nr5a2 (also known as liver receptor homolog 1, Lrh1), result in severe architectural defects in the early pancreas. Specifically, loss of Pdx1 results in total pancreatic agenesis and a block of endocrine specification (Offield et al. 1996), and loss of Nr5a2 leads to retention of inappropriate plexus-like connections at later stages to the skewing of lineages (Hale et al. 2014). As the field turns a more exacting eye on pancreas morphogenesis in available and newly generated mouse models, it is likely that additional critical factors will be reported. To date, the geometry of how epithelial cells must rearrange for morphogenesis to faithfully occur, as well as the forces and mechanisms driving these events, remain completely unknown. Understanding pancreatic epithelial ontogeny, microlumen formation, ductal remodeling and the epithelial reorganization that subsequently occurs is likely to yield important insights into pancreas development, the fate of its lineages, and the context within which these fates are acquired.
Epithelial Bud Reorganization and Progenitors
A putative niche in which pancreatic progenitors emerge has not yet been described (for an overview of pancreas development, see Fig. 2). In addition, little is known about how cell fate is established in the early pancreas epithelium as it undergoes remodeling and lineages are sorted out. We do know that shortly after transient stratification and microlumen formation, tip cells and MPCs can be identified (Stanger et al. 2007; Zhou et al. 2007). Although it is unclear how and when MPCs arise as multi-lineage progenitors, it is intriguing that they are located in ‘tip’ domains on the pancreatic bud. Markers for tip cells include Cpa1, Pft1a and Nr5a2; however, we note that these factors are also expressed in more centrally located body cells at earlier stages (Fig. 3). Other factors, such as Sox9 and Hnf1β, segregate instead to those central cells over time, eventually becoming restricted to mature ducts (Kopp et al. 2011; Solar et al. 2009). Interestingly, prior to identifiable MPC bearing tips, cells that lie in a peripheral layer around the epithelial bud display particular characteristics. Namely, they are semi-polarized, relatively columnar, and display basally localized nuclei and express basal markers like laminin but not apical markers like atypical protein kinase C (aPKC) or zonula occludens -1 (ZO-1) (Villasenor et al. 2010). We termed these outer cells ‘cap cells’, borrowing terminology from terminal end buds at the tips of mammary gland branches, which have similarly stratified buds surrounded by a peripheral layer of semi-polarized cells (Gjorevski and Nelson 2011; Villasenor et al. 2010). Similarly, we termed the inner unpolarized stratified cells ‘body cells’; although these have also been referred to as ‘trunk’ cells (Shih et al. 2013).
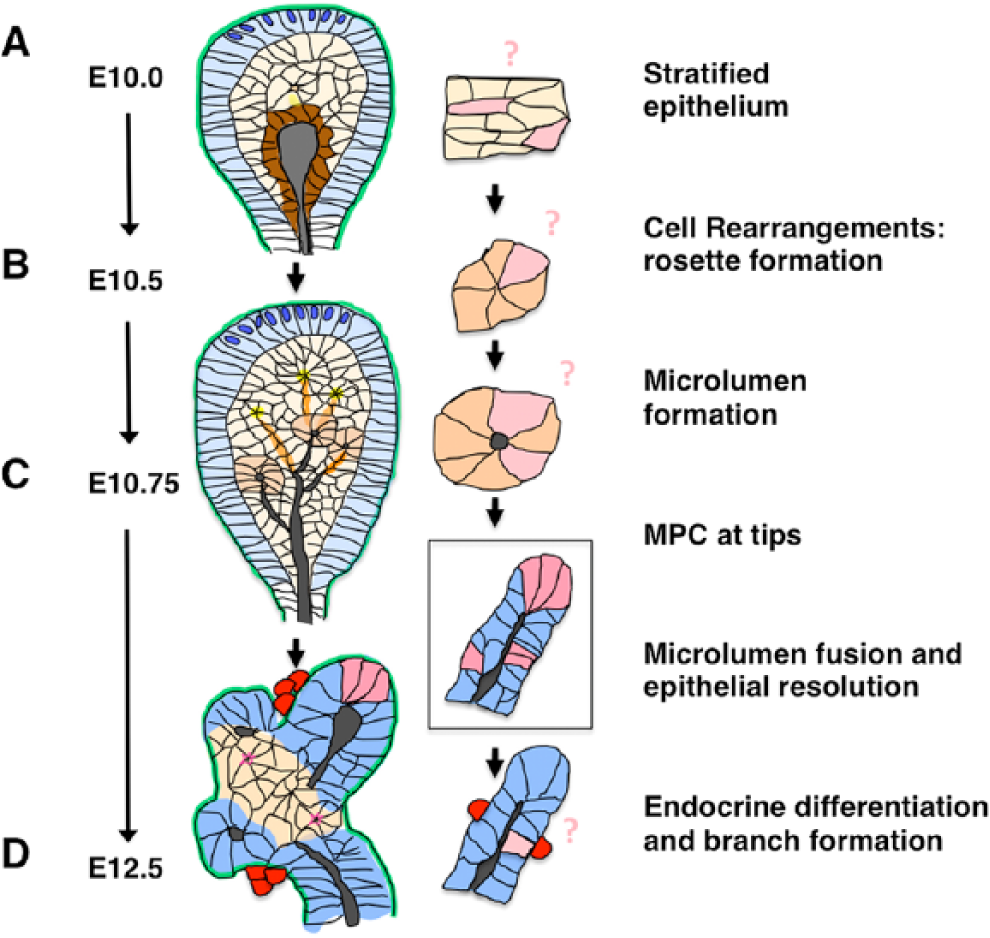
Relationship of early pancreatic epithelial architecture and pancreatic progenitors. (A) Stratified epithelium in the E10 pancreatic bud displays few features other than outer cap cells, which display basal polarity (blue), and the inner central lumen lining cells, which display apical polarity (brown). A few nascent rosettes and microlumens can also be identified at this stage. First transition endocrine cells (red) emerge within the early bud. The location of the cells that give rise to MPCs (light pink) within this epithelium is unknown. (B) Cells within the bud epithelium will reorganize, change shape and apically constrict (yellow); they then form rosettes (orange), ultimately leading to opening of microlumens (grey) (C). These will connect, forming an inner 3D ductal plexus. Again, the location of progenitors for endocrine, acinar and ductal lineages within the epithelium at this stage is unknown. (D) By E12.5, tips (dark pink) contain progenitors with the potential to give rise to all three of these lineages. This multipotency, however, becomes restricted shortly thereafter (by E13.5). Subsequently, endocrine cells (red) continue to delaminate from the more central bi-potential proto-differentiated epithelium to give rise to new endocrine cells. (Blue are cap cells, orange are rosettes, pink are progenitors, yellow indicates the center of a rosette and an opening microlumen, grey represents established lumens, red are delaminated endocrine cells, including β cells).
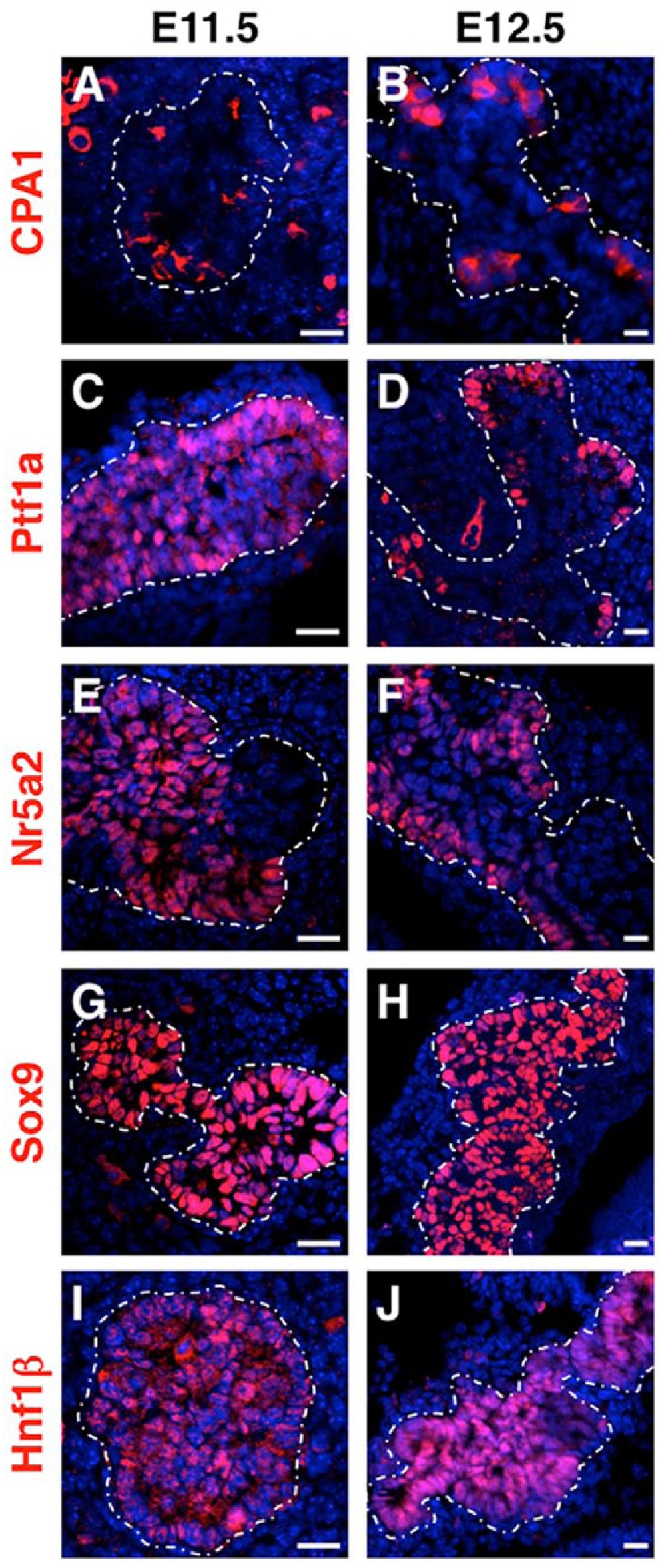
Heterogeneity of pancreatic progenitor epithelium during tip formation. MPC markers within the early stratified epithelium are expressed in a heterogeneous manner. Whereas these factors overlap initially, they label distinct populations of progenitor cells over time. By E12.5, tip enrichment of markers (A, B) carboxypeptidase A1 (CPA1), (C, D) Ptf1a (pancreas transcription factor 1a) and (E, F) nuclear receptor subfamily 5 group A member 2 (Nr5a2) can be observed, whereas (G, H) SRY-box 9 (Sox9) and (I, J) hepatocyte nuclear factor 1 homeobox β (Hnf1β) are still observed throughout the pancreatic epithelium. White dotted outline indicates the periphery of the epithelium. (Staining outside of the outline is non-specific autofluorescence of erythrocytes). Scale, 25 µm.
Cell Biology of the Pancreatic Epithelium
In this section, we will discuss the cellular and molecular underpinnings associated with the development of the pancreatic epithelium, from its interactions with the mesenchyme and extracellular matrix (ECM) to the establishment of apical–basal polarity. We are only beginning to understand the ontogeny of the early pancreatic epithelium and recent studies are increasingly providing new insights. Here, we assess both classical and more recent studies evaluating fundamental behaviors of the cells within the early pancreatic epithelium and we compare mechanisms observed in the pancreas with those that occur in other branching organs.
Epithelio-Mesenchymal Crosstalk
Branching organs, including the prostate, salivary and mammary glands, as well as the pancreas, require active communication between the branching epithelium and the surrounding mesenchyme. Indeed, pancreas development has been shown to exquisitely depend on epithelio-mesenchymal crosstalk for both growth and survival (Ahlgren et al. 1996; Golosow and Grobstein 1962). Classical embryological experiments demonstrate that isolated wild-type epithelium fails to expand in culture in the absence of mesenchyme (Golosow and Grobstein 1962). These experiments were more recently reproduced using elegant genetic approaches to ablate the mesenchyme using Cre-mediated expression of Diphteria Toxin (DT) (Landsman et al. 2011). A host of secreted factors have been identified as critical mediators of crosstalk, including fibroblast growth factor 10 (Fgf10), bone morphogenetic proteins (BMPs), Follistatin and others (Ahnfelt-Ronne et al. 2010; Bhushan et al. 2001; Miralles et al. 1998). Interestingly, pancreatic epithelium does not require interaction with its own particular mesenchyme, as salivary and kidney mesenchyme can also support pancreatic branching and differentiation in culture (Golosow and Grobstein 1962). Of note, although the pancreas requires mesenchymal support to grow, the converse is not true and pancreatic mesenchyme can develop normally without the pancreatic epithelium. An example is the embryonic Pdx1−/− pancreas, where expansion of the epithelium is arrested shortly after the onset of budding while the mesenchyme continues to expand, eventually forming a “hollow pocket” that occupies its normal position adjacent to the stomach and pancreas (Ahlgren et al. 1996).
Mesenchymal Cues Drive Pancreatic Branching
Secreted mesenchymal factors have been shown to regulate pancreatic branching, as well as cell fate specification. Stromal cell-derived factor 1 (SDF1), for instance, has been shown to be an important factor expressed by pancreatic mesenchyme. Reduction of SDF1 by chemical inhibition using AMD3100 in culture or by genetic ablation of either SDF1 or its receptor C-X-C chemokine receptor type 4 (CXCR4) suppresses pancreatic branching and remodeling (Hick et al. 2009). Similar to the lung and kidney, Fgf10 has been shown to be a “branch generator” during pancreas development. Fgf10 signaling from the pancreatic mesenchyme to the epithelium is required not only for growth and branching morphogenesis, but also for proliferation and subsequent differentiation of the pancreatic progenitor cells (Bhushan et al. 2001). Therefore, the mesenchyme is positioned to both initiate and sustain proper epithelial morphology. Understanding the cues that influence the architecture of the progenitor epithelium that gives rise to β cells has important implications for future directed differentiation efforts.
Impact of Mesenchymal Extracellular Matrix (ECM)
The basement membrane and ECM proteins have also been shown to guide branching in glandular organs (Varner and Nelson 2014). Indeed, specific ECM proteins have been identified as driving the development and branching of different glands. For example, the prostate gland exhibits strong dependence on α6β1-integrin and on laminin-1 (but not collagen or fibronectin) for establishing and maintaining cell polarization (discussed below), as well as for epithelial branching and acinar morphogenesis (Bello-DeOcampo et al. 2001). Antibody blocking of laminin signaling in tissue culture suppresses branching in the developing prostate (Bello-DeOcampo et al. 2001). In the submandibular salivary gland, fibrils of fibronectin are deposited at the sites of new clefts in the growing epithelium, which allows for branching to occur (Larsen et al. 2006; Sakai et al. 2003). Inhibition of fibronectin, either through siRNA or by the addition of inhibiting antibodies, leads to abrogation of cleft formation and suppression of subsequent submandibular gland branching. In the mammary gland, type I collagen fibers are present within the stroma and nascent epithelial branches use these fibrils as guidance cues for their elongation and extension (Brownfield et al. 2013). Genetic manipulation of matrix metalloproteinase 11 (MMP11) in mice results in a decrease in collagen within the mammary stroma and leads to defects in mammary gland branching (Tan et al. 2014). Recent work underscores the requirement for laminin and integrins for lung branching, as the loss of either leads to abrogated lung branching and defects in alveolarization (Nguyen and Senior 2006; Nicolaou et al. 2012; Plosa et al. 2014). The loss of β1-integrin in endothelial cells disrupts both cellular polarity and lumenogenesis in developing arterioles (Zovein et al. 2010). Together, these data indicate that the ECM plays a central role in the branching of epithelium, as well as endothelium, in diverse organs, and could very well play a role in the developing pancreatic epithelium.
Establishment and Control of Cell Polarity In Vivo
ECM and basement membrane components have also been shown to influence basic epithelial cell properties, such as cell polarity establishment. This has been observed in branching organs, such as the lung and the vasculature. Indeed, acquisition of cell polarity is frequently initiated by ECM interactions mediated through receptors such as integrins, dystroglycans and proteoglycans (Bryant and Mostov 2008; O’Brien et al. 2001; Yu et al. 2005). Such interactions allow epithelial cells to sense their environment and respond to changes in the chemical or mechanical properties by changing cell polarity and/or shape. This has been modeled in vitro using the Madin-Darby canine kidney (MDCK) 3D culture system, as well as in vivo in different organ systems (Kass et al. 2007; Maller et al. 2010; Paszek et al. 2005; Provenzano et al. 2009; Yu et al. 2008). For example, the mouse mammary gland epithelium displays a strict dependence on communication with the ECM through integrin receptors for establishment of apicobasal polarity, as well as for signaling pathways that drive cell proliferation. Loss of ECM receptors, or changes in either ECM stiffness or composition, can all lead to disruption or even complete inversion of apical–basal polarity. These disruptions are often associated with onset of tumorigenesis.
At the time of this review, a role for ECM and basement membrane proteins during pancreas development has not been rigorously explored using genetic models. Ex vivo studies, however, suggest that at least one ECM component, laminin-1, supports the differentiation of pancreatic lineages, including both ductal and acinar (Crisera et al. 2000). Laminin-1 was also linked to both pancreatic morphogenesis and proper allocation of cell fate in a mouse model that disrupted epithelial polarity (Kesavan et al. 2009). Do additional ECM components help expand or instruct endocrine fate? Are there particular basement membrane proteins that drive interactions between the pancreatic mesenchyme and the underlying epithelium? Do matrix-mediated signals drive basic events in the pancreatic epithelium, such as the acquisition of cell polarity and/or subsequent branching? These questions, like so many others in pancreas development, remain to be answered.
Epithelial Cell Polarity
Control of cell polarity is fundamental to proper morphogenesis and differentiation of all epithelial organs, including the pancreas. Ablation of the classical cell polarity determinant Cdc42, a Rho family small GTPase, in the pancreatic epithelium leads to profound defects in its development and bud branching (Kesavan et al. 2009). Importantly, disruption of normal architecture of the progenitor epithelium is associated with prevention of proper differentiation of its lineages, including β cells. Here, we explore how control of cell polarity may drive early events in pancreas formation.
Epithelia display two types of polarity, planar cell polarity (PCP) and apical–basal polarity. PCP refers to the orientation of cells along the axis of the tissue (perpendicular to the apical–basal axis) and has been shown to direct various cellular processes, including directed migration, convergent extension and oriented cell division (Devenport 2014; Karner et al. 2009). Apical–basal polarity involves the establishment of different ‘sides’ of an epithelial sheet, each domain displaying a distinct molecular composition—as the plasma membrane has a different set of proteins targeted towards each domain—and functionality. Generally, the basal domain of an epithelium forms the adhesive surface that interacts with the ECM, whereas the apical surface of epithelial cells is non-adhesive, often facing a lumen or cavity (Nelson 2009, 2003; Zhou et al. 2011). Apical and basal domains are usually separated along the lateral sides by tight and adherens junctions (TJs and AJs, respectively), which act as a “molecular fence” that prevents diffusion of the contents of one domain into the other (Nelson 2009). Cellular polarity allows for spatial functional specialization in many types of epithelium. This can be observed in epithelial cells of the eye called photoreceptor cells, where the apical surface is capable of sensing light while the basal side communicates with underlying neurons (Koike et al. 2005; Walther and Pichaud 2010). Another well-characterized example of a polarized epithelium is the mammalian intestine, which has a specialized apical surface with microvilli that maximize the absorptive functions of this organ (Saotome et al. 2004). Since the PCP pathway is well described in other reviews, yet few studies address its role in pancreas development (Barrow 2006; Cortijo et al. 2012; Devenport 2014; Karner et al. 2006), we will focus here on the establishment of apical–basal polarity in the pre-pancreatic epithelium.
Core Polarity Determinants
Core evolutionarily conserved polarity complexes have been identified across diverse phyla using in vivo studies and these studies have illustrated the importance of establishment and maintenance of epithelial cell polarity. Studies in Drosophila have shown that loss of the Scribble or Crumbs complexes result in basal and apical polarity defects, respectively, which can be rescued by a decrease in the expression of proteins that determine the opposite polarity (Bilder et al. 2003; Tanentzapf and Tepass 2003). Ablation of Crumbs homologues in Drosophila photoreceptors and Malphigian tubes, as well as in zebrafish neurons and nephrons, leads to disruption of these tissues, including cyst formation (Muschalik and Knust 2011; Omori and Malicki 2006). Deletion of the apical polarity determinant Crumbs3 in the mouse has been shown to cause disruption in the lung, kidney and intestinal epithelia, and deletion of Crumbs1 leads to defects in the retinal epithelium (den Hollander et al. 1999; Whiteman et al. 2014). The partition-defective (Par) complex (or aPKC-Par3-Par6), which interacts with the Rho family small GTPase Cdc42, has also been shown to be critical to epithelial polarity. Cdc42 has been identified as required for the recruitment of cell–cell junction molecules such as cadherins (discussed below) and the subsequent establishment of cell polarity in the development of diverse tissue types, ranging from the mouse mammary gland and epicardium, to the C. elegans embryo at the one-cell stage and at gastrulation, to Drosophila neurons (Hirose et al. 2006; Macara 2004; Ohno 2001). Many studies show that maintaining a balance in the crosstalk between these protein complexes is necessary for epithelial cell polarization. However, the contribution of these polarity cues remains unexamined in the developing pancreas. Could mutation or tissue-specific deletion of components of the Crumbs, Par and Scribble complexes lead to defects in polarity in the early development of the pancreas or contribute to the formation of cysts later, as observed in cases of pancreatitis? These are straight-forward questions that remain to be answered.
Polarity Acquisition
Epithelial cell polarity acquisition transforms the stratified pancreatic epithelium and represents a central characteristic that must be properly regulated for faithful organ and tissue morphogenesis. To date, a powerful reductionist approach for addressing basic questions of epithelial cell polarity has been in vitro systems. MDCK cells are often used as a model to study the molecular basis for epithelial polarization and lumen formation (Martin-Belmonte and Mostov 2008). MDCK cells in 3D matrices are tractable as they spontaneously polarize and form monolayered cysts that resemble the in vivo organization of epithelia. Interestingly, external cues that arise from surrounding ECM have been found to be required for apical–basal polarity initiation in the MDCK 3D culture system (O’Brien et al. 2001). For instance, interaction of β1-integrin with collagen has been shown to lead to the activation of the small GTPase Rac1, as well as the assembly of a peripheral laminin network (Yu et al. 2005). Laminin then allows for the rapid induction of apical–basal polarity, and, in MDCK systems, drives epithelial lumen formation via a ‘hollowing’ rather than ‘cavitation’ (apoptosis) mechanism, permitting cysts to form lumens by separation of cell membranes (Martin-Belmonte et al. 2008). Recently, ECM signals were shown to be mediated by a β1-integrin/FAK/p190RhoGAP cascade to downregulate Rho signaling and induce transcytosis of critical apical determinants, thereby driving the establishment of cell polarity (Bryant et al. 2014). Therefore, polarity acquisition appears to be triggered by a signal emanating from the basal aspect of epithelial cells. In the case of the pancreatic epithelium, such a signal would have to involve either the surrounding mesenchyme that encases the growing epithelial tree or the basement proteins at the interface between the two tissues.
MDCK assays have been used to further delineate the cascade of events involved in apical–basal polarity acquisition, including localization of specialized membrane lipids. In fact, an early event during polarity initiation involves the segregation of phosphatidylinositides (PtdIns) to either the apical or basal aspects of epithelial cells. These lipids become asymmetrically distributed along the cell plasma membrane and act as recruitment signals for the assembly of molecular complexes. In vitro studies have shown that PtdIns(3,4,5)P3 is directed to the basolateral membrane, whereas PtdIns(3,4)P2 is localized to the apical surface (Martin-Belmonte et al. 2007). After PtdIns are in place, the apical localization of Cdc42 is mediated by Annexin2. Cdc42 then targets the polarity determinant aPKC to the apical plasma membrane, which allows for building of the apical PAR protein complex (Martin-Belmonte et al. 2007). Other polarity complexes also begin to be differentially distributed in the cells, such as the apical determinant Crumbs complex and the basolaterally located Scribble complex (Nelson 2009). Whether apical–basal membrane lipid segregation will be found to play a role during pancreas development is an open question that will require further examination.
Subsequent to cell polarity initiation, vesicle trafficking is used to build the apical surface of the cells and initiate other events, such as lumen formation (discussed further below under “Tubulogenesis”). Directed vesicular transport is thought to contribute new membrane to the apical surface via processes mediated by the small GTPases, Cdc42 and a host of Rab proteins (Paszek et al. 2005). The nascent apical surface between adjacent unpolarized cells is therefore primarily thought to arise as a result of trafficking and exocytosis of the new membrane and associated proteins. Mostov and colleagues have elucidated many of the key steps during this process, including formation of the apical membrane initiation site (AMIS) and the pre-apical patch (PAP), where new membrane and other apical molecules, such as podocalyxin, are deposited in a directed manner (Datta et al. 2011). Despite tremendous progress in understanding polarity acquisition, the mechanisms driving the targeting of these complexes in vivo remain poorly understood in developing tissues, and particularly so in pancreas formation.
Architectural Ontogeny of the Progenitor Epithelium
Epithelial Cell–Cell Adhesion
A fundamental characteristic of epithelial cells involves their close cell–cell associations, which are mediated in part by TJs and AJs. One important class of molecules is the AJ cadherins, which have been implicated in pancreas development. E-cadherin is one example, as tight spatio-temporal regulation has been observed during endocrine delamination and clustering into islets (Dahl et al. 1996; Gouzi et al. 2011). By contrast, N-cadherin was shown instead to play a critical role in the formation of the pancreatic mesenchyme and in the release of insulin granules from β-cells (Edsbagge et al. 2005). In addition, R-cadherin is expressed throughout the developing pancreas epithelium and early endocrine cells, only later becoming restricted to the exocrine compartment (Sjodin et al. 1995). Of note, cadherins may cross-regulate each other’s expression within the same epithelium. For instance, cadherin-6 and E-cadherin are both expressed in MDCK cells, yet they display opposing roles during morphogenesis, with cadherin-6 normally inhibiting the formation of lumens and E-cadherin being required for lumen formation (Jia et al. 2011). By contrast, in blood vessel endothelial cells, N-cadherin positively regulates the expression and localization of VE-cadherin at cell–cell junctions (Luo and Radice 2005). At this point, the role for cadherins in pancreas epithelium has not been explored. Cross-regulation of E- and R-cadherin in pancreatic epithelium is possible but this has not been examined. Investigations into the structural molecules that mediate the integrity of the pancreatic progenitor and developing epithelium are needed.
Apical Constriction
Fundamental to the formation of the pancreatic tree—from which islets are born—is the generation of a ductal plexus. This event requires initial opening of microlumens by epithelial cells within the stratified epithelium of the early bud. Apical constriction has been suggested as a driving event in microlumen formation (Villasenor et al. 2010). Apical constriction, as its name implies, is the process whereby the apical membrane surface of epithelial cells undergoes a reduction in size, leading to cell shape changes. If a cell acquires a constricted apical end, it takes on a ‘bottle cell’ morphology. If this occurs in a coordinated manner by a group of neighboring cells, this results in localized morphogenesis of new tissue (or rosettes, for instance, discussed below). Reduction in the area of the apical membrane of cells is often dependent on cytoskeletal rearrangements, specifically driven by actomyosin contractility (Sawyer et al. 2010) involving complexes of molecules, including MyosinIIA (composed of Non-Muscle Myosin II, NMHCIIA/B, and phosphorylated Myosin Light Chain, pMLC). An important requirement allowing for apical constriction is that polarity must already be established within the given population of cells undergoing tubulogenesis. Of the known apical determinants, the Par complex has been shown to play an important role in directing apical constriction. When Par3/6 are absent in C. elegans gastrulation, myosin is incapable of gathering apically to mediate constriction (Putzke and Rothman 2003). This same requirement for polarization is also observed in chick neural tube formation (Harding et al. 2014). However, apical–basal polarity is unaffected in epithelial cells where downstream components of the apical constriction machinery have been ablated (Ernst et al. 2012; Harding and Nechiporuk 2012). These findings demonstrate that initiation of apical–basal polarity is critical for apical constriction to occur.
Rosette Formation
Coordinated apical constriction of epithelial cells drives morphogenetic changes in embryonic tissues. When groups of epithelial cells undergo apical constriction perpendicular to the plane of the epithelium, for instance, this can lead to the bending of a monolayer of cells. This has been observed in the Drosophila blastoderm during invagination events that drive gastrulation, as well as in the ectoderm during vertebrate neurulation (Lecuit and Lenne 2007; Sawyer et al. 2010). By contrast, coordinated apical constriction within the plane of the epithelium, amongst a small group of adjacent cells, has been shown to result in the formation of multicellular ‘rosettes’. These rosettes are usually formed from a collection of five or more cells that meet at a central point, and have been shown to be required for the coordinated movement of cells within diverse epithelia (Karner et al. 2006; Trichas et al. 2012). The disruption of PCP signaling alters rosette formation and ultimately epithelial organization in a variety of tissues, such as the Drosophila salivary gland, the mouse visceral endoderm and developing kidney (Blankenship et al. 2006; Karner et al. 2009; Trichas et al. 2012). In the mammalian pancreas, where microlumens form within the heart of the stratified epithelium, 3D rosettes have been observed using reconstruction of confocal Z-stacks (Villasenor et al. 2010). How rosettes coordinate to allow fusion of microlumens into a fine plexus, however, is unknown.
Tubulogenesis
Coordinated changes in cell shape often constitute the foundation for tissue morphogenesis, including the formation of epithelial tubules in many organs, such as lung, kidney and pancreas. Rosettes of apically constricted cells often form transiently and drive the opening of a small lumen, or microlumen, at their center. The zebrafish lateral line is a well-characterized model for epithelial tube formation, where many steps in this process have been analyzed, from the onset of organ patterning, involving apical constriction-based rosette formation, to tube formation or ‘tubulogenesis’ (Harding et al. 2014). Rosette formation in the lateral line is dependent on coordinated apical constriction that occurs within groups of approximately 20 cells with established apical–basal polarity (Gompel et al. 2001; Harding and Nechiporuk 2012). Apical constriction requires the localization of activated myosin-II to the apical surface of the cell (Ernst et al. 2012). A similar process has also been observed in the development of the zebrafish intestine, where apically located actin drives the formation of microlumens between cells (Alvers et al. 2014). These microlumens expand and eventually fuse into a single, continuous lumen, thereby generating the fish intestine (Horne-Badovinac et al. 2001).
Microlumen opening following rosette formation is also observed in the transiently stratified epithelium of the pancreatic bud (Villasenor et al. 2010). Unpolarized body cells within the center of the bud coordinately undergo apical constriction in 3D and open small isolated lumens that rapidly interconnect. Peripheral cap and underlying body cells also coordinate to form rosettes at their interface (Fig. 4). Microlumens are also observed in other glandular organs, such as the salivary, prostate and mammary glands, where groups of unpolarized cells coordinate the formation of small isolated lumens within the developing tissue (Hogan and Kolodziej 2002). Cells change their shape, acquiring a ‘bottle cell’ morphology, and aggregate their constricted ends between them, forming epithelial rosettes and opening up microlumens. This is evident as they coordinately orient their ZO-1-expressing apical ends to the location where the new emerging lumen will ultimately form. We have proposed this occurs via apical constriction, as pMLC is enriched at these constricted ends; however, functional actomyosin constriction has not yet been demonstrated. When microlumens open at the center of rosettes, other apical polarity markers begin to accumulate, such as ezrin and aPKC (Villasenor et al. 2010). These scattered lumens later fuse with each other, as polarity is reacquired by neighboring cells (Kesavan et al. 2009), generating a complex network of fine interconnected lumens.
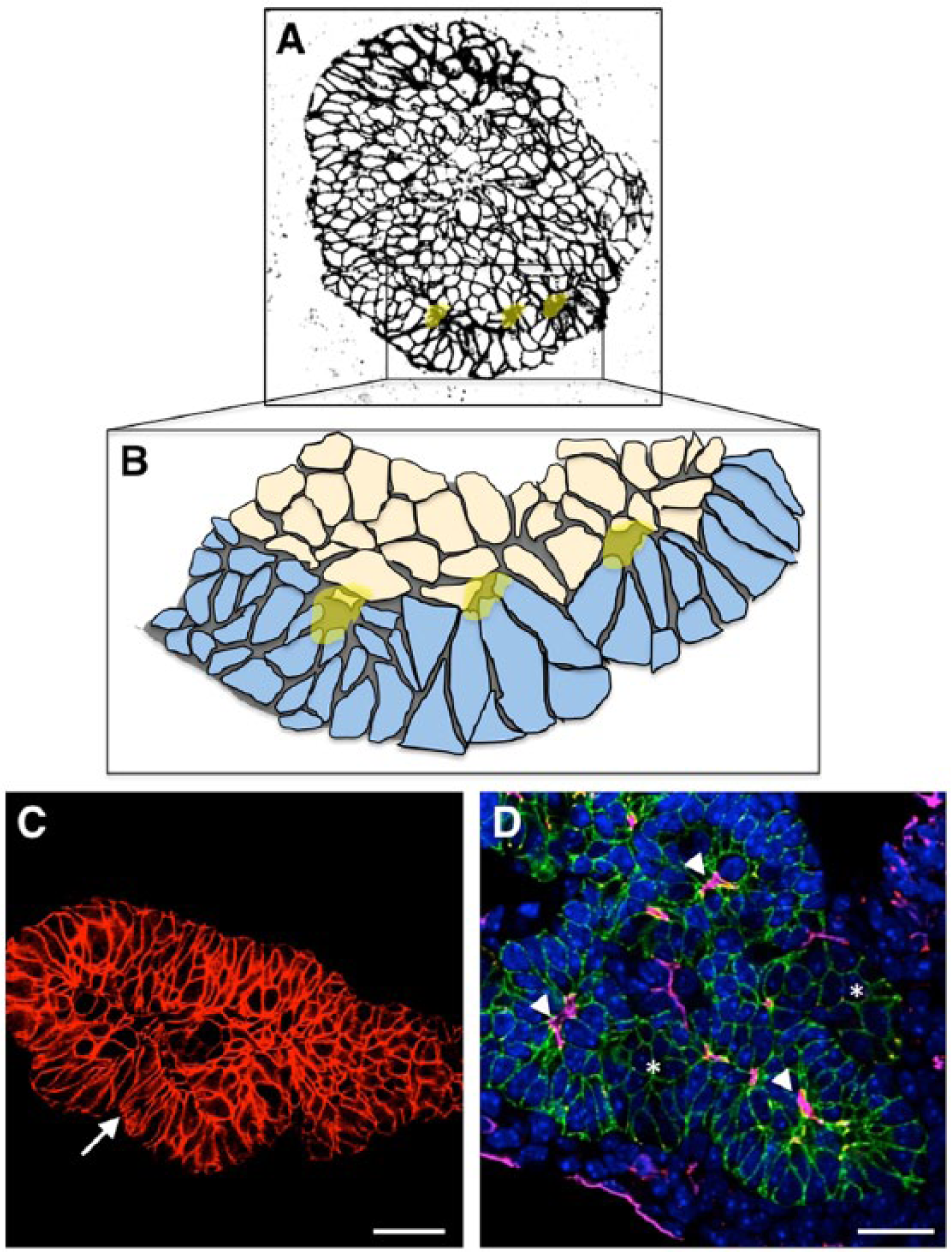
Microlumen formation and resolution of the stratified epithelium during morphogenesis. Peripheral cap cells are columnar and inner body cells more cuboidal. These internal unpolarized cells undergo cell shape changes that result in microlumen formation (lumens highlighted in yellow). (A) Stratified epithelium in the E10 pancreatic bud (black color is E-cadherin staining). (B) Cap cells (in blue) give rise to tips and participate in opening microlumens (represented in yellow) along with underlying body cells (in beige). These constitute the initial stages of reorganization of the progenitor epithelium, which will remodel and give rise to a growing ramifying tubular tree. (C) Resolving epithelium of the E11.5 bud (red color shows E-cadherin staining). Note columnarization of peripheral cap/tip cells (arrow). (D) Microlumen formation (arrowheads) is coordinated with resolution and branch formation. Immunofluorescence for E-cadherin was used to outline epithelial cells (in green), lumens were stained with podocalyxin (shown in pink) and ZO-1 (in red), and nuclei (in blue) were stained with DAPI. Scale, 25 µm.
Mutations in polarity determinants have been shown to profoundly impact organogenesis. Examples include aPKC, Par3 and integrins, which, when disrupted, affect lumen coherency in the zebrafish gut (Bagnat et al. 2007; Horne-Badovinac et al. 2001), in mammalian arterioles (Zovein et al. 2010), and in the Drosophila tracheal system (Levi et al. 2006), resulting in tubes with multiple, occluded or discontinuous lumens. These findings indicate that the acquisition and control of apico-basal polarity play a role in the initiation and maintenance of lumens in different tissue systems. However, although dynamic control of apical polarity clearly occurs during formation of the pancreatic ductal plexus (Villasenor et al. 2010), the only apical–basal polarity determinant identified to date as having a significant role in pancreas tissue architecture and lumen formation is Cdc42 (Kesavan et al. 2009). Studies that have carried out ablation of other polarity determinants, including Crumbs3 and Ezrin (Saotome et al. 2004; Whiteman et al. 2014), do not report defects in pancreatic epithelium; although, it is likely this tissue was not included in the analysis. Epithelial architecture and lineage allocation is affected upon deletion of many other factors, including Pdx1, Stard13 (discussed below), and the PCP determinants Celsr2 and -3 (Cortijo et al. 2012). However, future studies focusing on the developing pancreas will be needed to fully investigate the requirement for polarity regulators for morphogenesis and cell fate.
Tip Cell Allocation
Sorting of MPC-bearing tip cells from the progenitor epithelium and how this process interfaces with bud morphogenesis or cell fate represent the next frontier in cracking open the ‘black boxes’ in early pancreas development. An elegant recent study showed that spatiotemporal regulation of Rho activity during remodeling of the pancreatic epithelium was crucial for normal morphogenesis and pancreatic cell fate (Petzold et al. 2013). Specifically, Spagnoli and colleagues identified the RhoGAP Stard13 as an essential regulator of tip domain organization (Petzold et al. 2013). Genetic ablation of Stard13 using Pdx1-Cre resulted in branching abrogation, a reduction in epithelial proliferation, and organ hypoplasia, which is strikingly preceded by a loss of apical constriction and rosette formation within the stratified epithelium. Further, the authors showed that Stard13 acts by regulating Rho signaling and mitogen-activated protein kinase (MAPK) in tip cell domains. Interestingly, taking into account the smaller size of the mutant pancreas, it was found that relative numbers of endocrine cells were unchanged; this puts forth the argument that, by the time Stard13 was deleted with this Cre driver line, lineages were already specified. The question remains, however, whether an earlier disruption of the progenitor epithelium might impact specification of endocrine, acinar or ductal lineages. This study represents one of the first thorough attempts to molecularly delineate the events in the early pancreatic progenitor epithelium to assess how disruption of normal architecture impacts the ontogeny of the lineages therein.
Cap/tip cells and body/trunk cells of the early pancreatic bud become molecularly distinguishable shortly after the onset of epithelial stratification. For instance, Hnf1β expression, which is initially expressed throughout the early bud, increases in body cells as cap cells form tips and becomes extinguished in these peripheral cells; by contrast, Ptf1a expression becomes extinguished within the body cells but increases in cap/tip cells (Fig. 3) (Solar et al. 2009). The transcription factors Nk6 homeobox 1 and 2 (Nkx6.1 and Nkx6.2) are initially co-expressed throughout the early bud epithelium with Ptf1a; however, cross-repression between Nkx6.1/6.2 and Ptf1a is thought to allow for tip and trunk compartmentalization by promoting segregation (Schaffer et al. 2010). Notch signaling has also been shown to contribute to the expansion of the tip and trunk compartments, as Notch prevents tip formation by repressing Ptf1a expression and promoting expansion of the trunk domain through the activation of Nkx6.1 (Afelik et al. 2012; Kopinke et al. 2011; Kopp et al. 2011). Together, these signaling events result in tips becoming restricted to an acinar cell fate, while centrally located cells (body/trunk) give rise to endocrine and ductal cell lineages.
Perspectives on Cell Fate and Unknown Niches
Dynamic Progenitor Niche
Where do pancreatic progenitors reside in the stratified epithelium prior to tip formation (Fig. 2)? Is the progenitor epithelium homogeneous or heterogeneous? Do cells of the stratified epithelium contribute to, or influence non-cell-autonomously, peripheral tip MPCs? Does the 3D context in which they arise matter? The fact that we cannot currently answer such basic questions underscores our lack of understanding of the ontogeny of these critical cell types. Given how 3D culture techniques have proven to be a critical turning point for improving the efficiency of directed differentiation protocols (Schulz et al. 2012), the impetus for clearly defining early niches for multipotent pancreas progenitors is compelling. In other contexts, such as the developing neural tube, studies of tissue architecture and cell fate specification have identified new mechanisms by which progenitors and their descendants are regulated. These studies have had a profound impact yielding insights into areas from adult neurogenesis to regeneration. A deeper understanding of those progenitors that give rise to the three main pancreatic lineages is likely to similarly instruct our approaches to pancreatic disease and replacement efforts.
New Areas for Investigation
How can we delve into the question of early niches and sorting of pancreatic lineages? Understanding the dynamic context progenitors experience over developmental time is likely to reveal much about the sequential inductions they might experience. The challenge becomes how to address this question. Developmental biology has long relied on visualization techniques such as the analysis of fixed tissue specimens by immunohistochemistry and immunofluorescence and, although this analysis has advanced our understanding of cell rearrangements in the pancreas, it is still limited to comparing static time points in the development of an organ. Many studies in other systems have successfully used live imaging to explore the contribution of cell movement and elucidate the cellular step-wise events during epithelial morphogenesis. Examples include neuronal precursors in the zebrafish lateral line (Lecaudey and Gilmour 2006), border cell migration in Drosophila (Bianco et al. 2007; Prasad and Montell 2007), and mammary gland and kidney development in mouse (Ewald et al. 2008; Packard et al. 2013). However, this technique has been used sparingly in the developing pancreas, with there being only a few studies that focus on late branching and the clustering of islet endocrine cells (Puri and Hebrok 2007). There is a need for careful analysis of where progenitors arise in the early bud, where they go, what inductive influences they experience, and how lineages sort out as the bud expands and branches. Live imaging of different pancreatic reporter lines would be of particular interest. It would allow characterization of how cells divide to contribute to the stratification of the early bud, how bud epithelial cells organize themselves within the stratified bud, and could be used later to understand epithelial resolution from multi-layered to single-layered tubes. Current efforts in this arena are likely to yield important insights.
Together, the discussion in this review has aimed to cover what is known and what is unknown with regards to the earliest pancreatic progenitor cells. Much attention in work published to date has been given to the deletion of transcriptional regulators and cell intrinsic regulators, assessing the impact on later pancreas development, and the field has largely shied away from simple characterizations of the earliest progenitor epithelium and its niche. A better description is clearly needed and would help reveal important non-autonomous events, such as potential extrinsic inductions between epithelial cells. Understanding the early events in the pancreatic bud is likely to reveal how cells become specified, committed and ultimately differentiated into the different pancreatic lineages—ductal, acinar and endocrine —and to set the stage for more informed development of methods for the replacement of β cells in individuals with diabetes.
Footnotes
Acknowledgements
Our apologies to those works we were not able to cite due to space restrictions. We are grateful to Caitlin Braitsch and Berfin Azizoglu for critical reading of the review manuscript.
Competing Interests
The authors declared no potential competing interests with respect to the research, authorship, and/or publication of this article.
Author Contributions
LMS performed the immunofluorescence staining. LMS and OC prepared figures and drafted the manuscript together. All authors have read and approved the final manuscript, and are accountable for all aspects of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by F31 DK092098-03 to LMS, and NIH R01 grant DK079862 to OC.
