Abstract
The expression and distribution of deoxyribonuclease I (DNase I) in human duodenum, jejunum and ileum were examined by DNase I activity assay and the reverse transcriptase-polymerase chain reaction (RT-PCR), immunofluorescence, in situ hybridization, and immunocytochemical ultrastructural analyses. High levels of DNase I were detected in the cytoplasm of Paneth cells in human small intestine. A tissue homogenate fraction rich in Paneth cells showed strong DNase I-specific enzymatic activity. Immunofluorescence analysis using several specific anti-human DNase I antibodies showed very strong immunoreactivity in the cytoplasm of every Paneth cell. In situ hybridization demonstrated high levels of DNase I mRNA in Paneth cells. Immunogold electron microscopy revealed gold particles localized along the secretory pathway, with the exocrine secretory granules mostly labeled. Our findings strongly suggest that Paneth cells synthesize and secrete DNase I into the intestinal lumen.
Keywords
D
Although Paneth cells in intestinal crypts have been studied extensively since their discovery by Schwalbe (1872), their function has not been elucidated. Morphologically, Paneth cells have a distinctively staining cytoplasm and their ultrastructural features are common with those of other exocrine cells. Paneth cells resemble zymogenic cells, such as pancreatic acinar exocrine cells, and it has been suggested that they may participate in food digestion, acting as a diffuse exocrine gland scattered along the intestine (Trier 1963; Satoh et al. 1990). They have also been suggested to be involved in heavy metal elimination (Kodama et al. 1993; Sawada et al. 1994) and regulation of crypt cell growth (Cheng and Leblond 1974; Mathan et al. 1987). Furthermore, Paneth cells are believed to be involved in the regulation of the intestinal flora because they are able to phagocytose microorganisms (Masty and Stradley 1991; Leis et al. 1997). The histochemical localization of IgG and IgA in Paneth cells was interpreted as probably reflecting an opsonization phenomenon (Rodning et al. 1976; Coutinho et al. 1996). The hypothesis that Paneth cells are involved in immunological defense has been supported by the detection of tumor necrosis factor (TNF) (Schmauder-Chock et al. 1994) and cryptdin (a member of the corticostatin/defensin family) gene transcripts in tissue sections (Bry et al. 1994; Harwig et al. 1995; Ouellette and Selsted 1996). Therefore, these cells have been hypothesized to participate in host defenses in the small intestine (Selsted et al. 1992; Schmauder-Chock et al. 1994; Mallow et al. 1996).
In this study we used five different polyclonal antibodies and one monoclonal antibody (MAb) specific for human DNase I, raised by immunizing rabbits, chickens, or mice repeatedly with specific antigens. All six antibodies used yielded similar results and immunohistochemical, enzymological, and PCR analyses confirmed the presence of significant amounts of DNase I in Paneth cells.
Materials and Methods
Tissue Preparation
Tissue samples were obtained from male human bodies during 10 postmortem examinations with the consent of the bereaved families. The patients, aged 37–66 years, died within 2 weeks after having suffered a myocardial infarction (n = 6), angina (n = 1), a subarachonoidal hemorrhage (n = 2), and anaphylactic shock (n = 2). The biopsy specimens were obtained from two of the authors, a healthy man (OS) and a healthy woman (HT-S), during gastrointestinal and colon endoscopic medical examinations.
Antibodies
We prepared five different polyclonal antibodies and one MAb specific for human DNase I. The characteristics of some of these antibodies have already been reported (Yasuda et al. 1989,1994). The following antibodies were used: (a) an antibody obtained from a rabbit immunized with DNase I purified from human urine; (b) two different antibodies obtained from rabbits immunized separately with one of two synthetic peptides corresponding to the N- and C-terminal residues of DNase I; (c) two different antibodies from chickens immunized separately with these peptides; and (d) an MAb raised against DNase I purified from human urine.
Immunofluorescence and In Situ Hybridization
As described previously (Shimada et al. 1993), the tissues were immersed in a fixative composed of 1% paraformaldehyde and 0.1% picric acid in 0.01 M PBS for 24 hr at 4C, washed with PBS, immersed in 30% sucrose in PBS overnight at 4C, and then frozen rapidly in isopentane cooled by liquid nitrogen. Then 5-μm-thick sections were cut with a cryostat at −20C and mounted on glass slides that had been coated with poly-L-lysine. These cryostat sections were treated with 1% normal goat serum for 1 hr and then with the required monoclonal (1:50 dilution) or polyclonal antibody (1:500 dilution) with PBS containing 1% BSA for 24 hr at 4C. After washing, the sections were incubated with purified FITC-conjugated anti-rabbit, anti-chicken, or antimouse immunoglobulin (1:500 dilution) for 30 min at room temperature (RT), washed, and mounted in 50% glycerol in PBS (pH 8.6) containing 50 mg/ml 1,4-diazabicyclo-octane (DABCO; Aldrich Chemical, Milwaukee, WI), an anti-bleaching agent.
For in situ hybridization, the oligonucleotide probe (5′-CTGAAGATCGCAGCCTTCAACATC-3′), corresponding to the N-terminal region of human DNase I (Yasuda et al. 1994), was labeled with biotin using a biotinylation kit (American Qualex International; San Clemente, CA). Cryostat sections, prepared as described above, were treated with absolute methanol, dried, and then permeabilized for 20 min at 37C with proteinase K (1 μg/ml) in 100 mM Tris-HCL, pH 8.0, 50 mM EDTA, according to the method of Lacasse and Martin (1992). After hybridization, sections were washed well three times with this buffer, treated with FITC-labeled streptavidin for 15 min at RT, washed, mounted in 50% glycerol containing 5% DABCO in PBS, and then examined with a fluorescence microscope.
Immunogold Electron Microscopy
For histological analysis, small pieces of tissue were fixed with 1% paraformaldehyde, 0.125% glutaraldehyde, and 0.1% picric acid in 0.01 M PBS, pH 7.2, for 24 hr at 4C. We found that the routinely used fixatives, particularly glutaraldehyde and osmium tetroxide, dramatically reduced the amounts of DNase I detected by all the antibodies; therefore, only weak fixation was feasible. After fixation the tissues were dehydrated with a graded ethanol series at 0C, embedded in Lowicryl K4M, and then cured for 3 days at −35C.
As described previously (Shimada and Tosaka-Shimada 1989), ultrathin sections were treated with 3% hydrogen peroxide for 10 min and 1% normal goat serum for 1 hr, then incubated with the required anti-human DNase I antibody (1:500 dilution) with PBS, pH 7.4, containing 1% BSA. After rinsing with PBS, the sections were incubated for 1 hr with 10-nm colloidal gold-labeled anti-rabbit, anti-chicken, or goat anti-mouse immunoglobulin (1:200 dilution), followed by rinsing with PBS and then distilled water. Finally, the sections were stained with uranyl acetate and lead citrate and examined with an electron microscope (Hitachi H-800; Hitachi, Tokyo, Japan). The specificity of each immunoreaction was confirmed by a preabsorption test involving addition of excess human DNase I to the first antibody.
Assay of Tissue DNase I Activity
DNase I activity was determined using the single radial enzyme diffusion (SRED) method (Nadano et al. 1993). Tissue samples of human duodenum, ileum, and jejunum were obtained within 24 hr after death. The tissues were cut, notched, and then mounted with the mucosal surface on square silicone plates (5 × 5 mm) previously coated with poly-L-lysine. The corners of the patched tissue were fixed by small pins. Under a binocular microscope, the tissue was gently grated with a handmade silicone grater. The first grated material consisted mainly of the upper part of the villi, including villous tips. However, blood cells and sometimes Paneth cells were also present. The third or fourth grated material consisted mainly of the lower part of the villi, including the crypts of Lieberkuhn and some residual material that was composed mainly of smooth muscle tissue debris. The residual part (muscle tissue) was hard, so the grated materials possessed little contamination. The fragments were washed well with physiological saline, centrifuged, and the pellets were collected and frozen in liquid nitrogen. In this experiment, the fragment rich in Paneth cells was used for biochemical analysis as the Paneth cell-rich fragment. Likewise, the first grated fragment was called the upper part of the villous fragment and the residual fragment was called the smooth muscle fragment of the small intestine. These fractions were homogenized in 0.5-1.0 ml 50 mM Tris-HCl, pH 7.5, containing 1.0 mM PMSF, then centrifuged at 10,000 × g for 20 min. The supernatants obtained were used for assay.
Deoxyribonuclease I (DNase I) activity in human small intestine (U/mg protein)

p<0.05; ∗∗p<0.01; ∗∗∗p<0.001. Significantly different from the DNase I activities of “smooth muscle fragment” by Welch t-test.
We determined the protein contents of the samples using a Bio-Rad protein assay kit (Bio-Rad; Hercules, CA) with BSA as the standard.
Detection of DNase I Gene Transcripts by PCR
The procedure used was previously described in detail (Yasuda et al. 1994,1995). In brief, total RNAs were extracted from human tissues and 1 μg each RNA was transcribed with reverse transcriptase. A set of primers, 5′-CTGAAGATCGCAGCCTTCAACATC-3′ and 5′-CAGTTGGTCACTCAGGCCATAG-3′, corresponding to the N-terminal and C-terminal portions, respectively, of human DNase I protein was synthesized and used for the PCR. After heat denaturation at 90C for 5 min, a 1-μl aliquot of each sample was subjected to PCR analysis. Each DNase I gene transcript was PCR-amplified. The amplified products were subjected to electrophoresis on a 2% agarose gel and the bands were visualized by ethidium bromide staining.
Immunoblot Analysis
Each tissue specimen was homogenized in 100 times its volume of sample buffer containing 4% sodium dodecyl sulfate (SDS) and 10% 2-mercaptoethanol, incubated for 5 min at 90C, condensed several times using Centricell (Polyscience; Warrington, PA), and then subjected to SDS-polyacrylamide gel electrophoresis (PAGE) on a 10% acrylamide gradient gel. The proteins were transferred to a nitrocellulose sheet and incubated with an antibody (1:1000 dilution). Finally, the antigens were detected with alkaline phosphatase-conjugated anti-rabbit, anti-chicken, or anti-mouse immunoglobulin (1:1000 dilution).
Results
DNase I Activity in the Small Intestine
The specific DNase I activities in various parts (Paneth cell-rich, upper part of villi, smooth muscle fragments, and whole tissue) of the duodenum, jejunum, and ileum were determined by the SRED method and the results are summarized in Table 1. The specific activities in the extracts from the Paneth cell-rich fragments of every portion of the human small intestine were considerably higher than those in the other samples tested. The DNase I activities in smooth muscle fragments of the small intestine were very low. The detectable enzymatic activities were inhibited by 1 mM EDTA or monomeric (G) actin, and, most notably, were abolished by the anti-human DNase I antibodies.
Distribution of 31-kD Protein in the Small Intestine
Immunoblot analysis showed high levels of DNase I-like immunoreactivity in the Paneth cell-rich fragments of the small intestine (Figure 1), whereas little or no DNase I immunoreactivity was detected in the smooth muscle fragments. We obtained similar results using other antibodies.
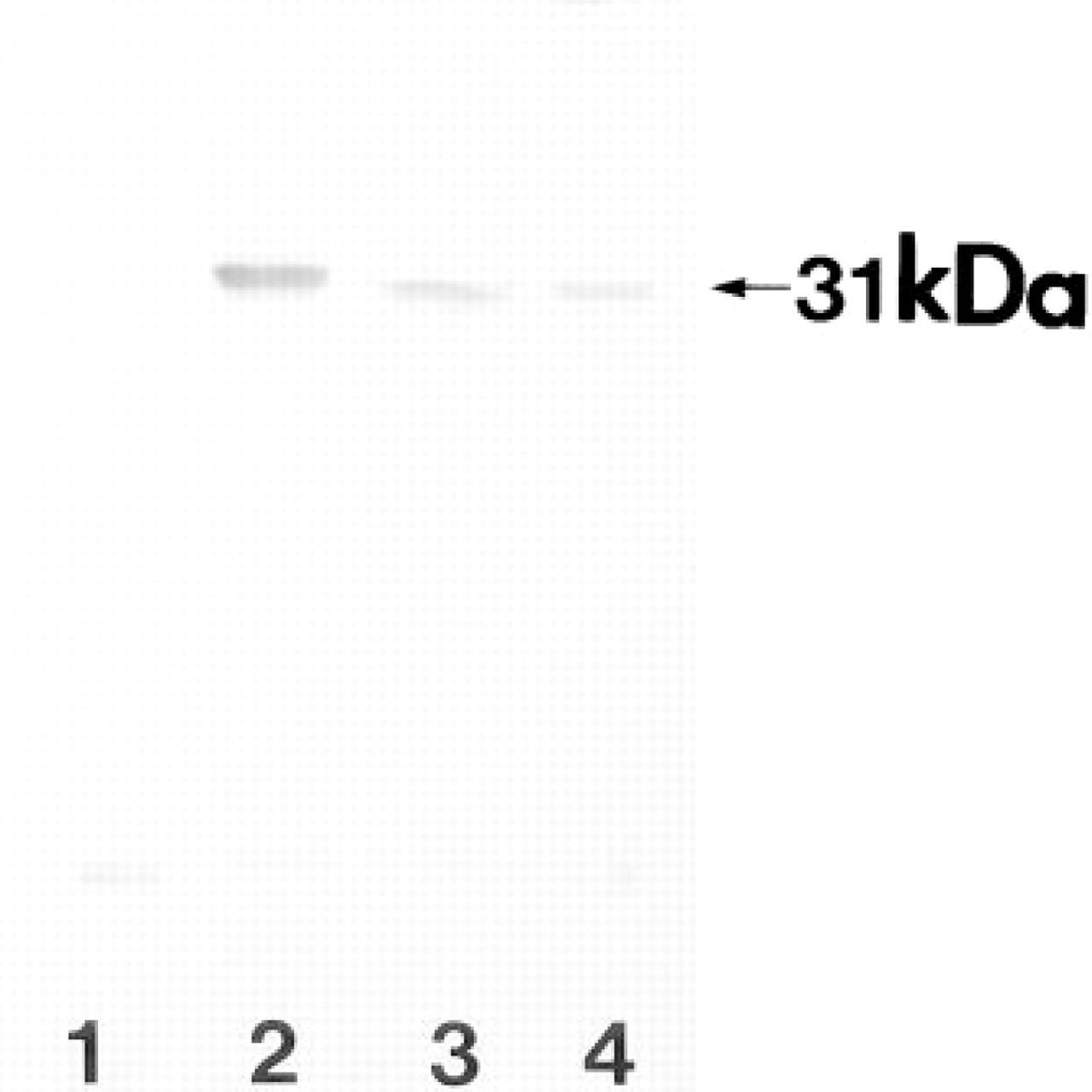
Immunoblot analysis using a specific MAb against human DNase I of various fragments of the ileum. Aliquots (20 ng) of each tissue fragment were subjected to SDS-PAGE on a 10% acrylamide gradient gel. Lane 1, smooth muscle fragment of human ileum. Lane 2, Paneth cell-rich fragment. Lane 3, upper part of villous fragment. Lane 4, 5 pg of DNase I purified from human urine. A single 31-kD band is detected in every lane except Lane 1.
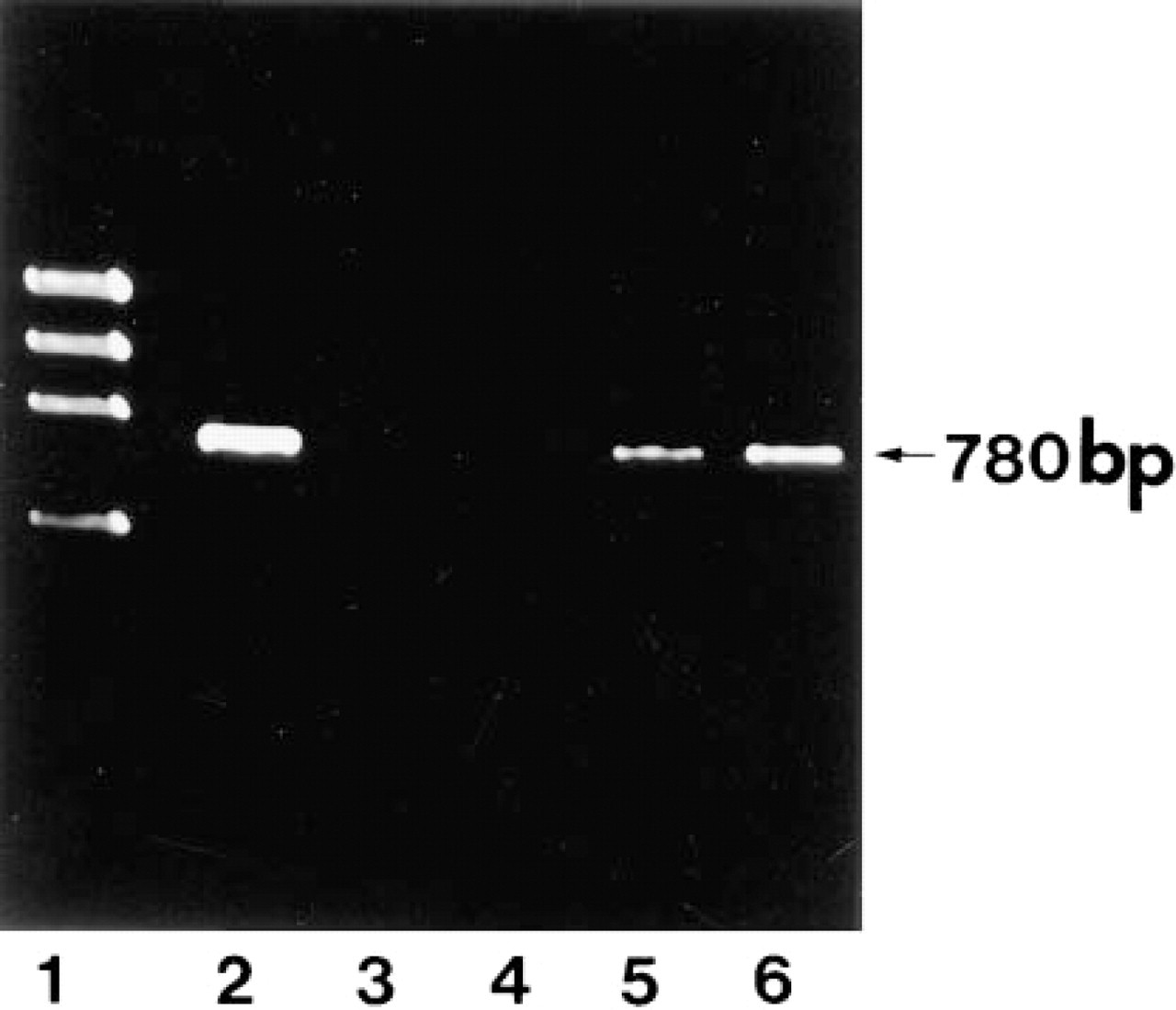
PCR analysis of the reverse-transcribed total RNA from human tissues. The total RNA isolated from each tissue was transcribed and PCR-amplified using a specific primer. A unique 780-BP fragment was amplified from the total RNA isolated from pancreas (Lane 2), brain (Lane 3), smooth muscle fragment (Lane 4), upper part of villous fragment (Lane 5), and Paneth cell-rich fragment (Lane 6) of ileum. Lane 1, ϕX174 DNA/HaeIII-digested DNA markers (GIBCO BRL; Gaithersburg, MD).
DNase I Gene Transcripts by PCR
The presence of DNase I-specific mRNA was verified by PCR analysis of the total RNAs extracted from several human tissues. A unique 780 base pair (BP) fragment, corresponding to the region encoding the mature enzyme, could be amplified from the total RNAs of the human pancreas and the Paneth cell-rich fragment of the small intestine (Figure 2). No amplified products were obtained from the brain or the smooth muscle fragments.
Immunofluorescence and In Situ Hybridization
Immunofluorescence revealed very strong immunoreactivity in the cytoplasm of Paneth cells at the bases of the crypts of Lieberkühn in all regions of the small intestine, including the duodenum, jejunum, and ileum (Figure 3). The immunoreactivity was widely distributed in the Paneth cell cytoplasm but the staining intensity was heterogeneous, with particularly strong immunoreactivity observed in the cellular apical regions. Interestingly, little or no immunoreactivity was found in the nuclear regions of Paneth cells. The staining intensities differed according to the antibodies we used, but the distribution patterns were basically the same with all six antibodies, confirming that human DNase I itself is concentrated in the cytoplasm of human Paneth cells.
In situ hybridization showed high levels of specific human DNase I mRNA in the Paneth cells of duodenum, jejunum, and ileum (Figure 4). No fluorescence was observed in a control section. No fluorescence was observed in the control sections without a probe (Figure 7A).
Immunogold Electron Microscopy
At the fine-structural level, immunogold electron microscopy demonstrated DNase I distributed along the exocrine secretory pathway of the Paneth cells (Figures 5 and 6). The secretory granules were heavily labeled with anti-human DNase I, especially in their electronlucent halo regions, whereas the electron-dense inner domains (the granular core regions) showed less intense DNase I labeling. The Golgi apparatus and rough endoplasmic reticulum (rER) appeared to be labeled with fewer but still considerable numbers of gold particles. The exocytotic granules in Paneth cells were also DNase I-immunopositive. We obtained similar results with other antibodies. No significant gold particles were detected in a control section in which excess human DNase I was added to the first antibody (Figure 7B).
Discussion
The general structure of Paneth cells was almost identical to that described previously (Trier 1963; Satoh et al. 1990). Paneth cells at the bottom of the crypts of Lieberkühn in the small intestine have their nuclei located in the basal regions. For our experiments, we prepared six different anti-DNase I antibodies and compared the results obtained with them. Very similar results with all six antibodies were obtained for small intestinal Paneth cells, confirming our observation that DNase I is distributed mainly along the secretory pathway of Paneth cells. These findings suggest that the enzyme is synthesized in the rER, modified in the Golgi apparatus, stored in secretory granules, and secreted into the lumen, probably together with other exocrine secretory materials. In this connection, a previous report by Shak et al. (1990), in which the DNA sequence of DNase I possessed a code for a signal peptide, was very interesting. This article supports our findings that DNase I is synthesized in rER as the starting point of the secretory pathway (Shimada et al. in press). Peitsch et al. (1993) found that the intensity of DNase I immunoreactivity in the rat small intestine increased towards the epithelial villous tips, and concluded that the epithelial cells prepare for their own elimination (apoptosis) by switching on DNase I expression and storing DNase I in their cytoplasm during migration, long before reaching their final location. Such clear deposition of DNase I and preparation for apoptosis was not observed in human small intestine. These different results might be due to species differences. The apoptotic phenomena in the small intestinal epithelium should be further investigated (Moss and Holt 1996; Que and Gores 1996), requiring more studies to elucidate the relationship between DNase I and apoptosis.
Although the epithelial cells in villous tips histologically showed no clear immunoreactivity like that in Paneth cells, the fragment containing the villous tips biochemically showed significant amounts of DNase I and its mRNA. The inconsistency between histological and biochemical results might be explained by sample contamination with blood cells and Paneth cells. The villi, particularly the upper part, were quite soft and easily flexible, so it was impossible to obtain only a selective portion of the villi. Actually, we found a number of blood cells and some Paneth cells contaminating the villous tip fragment under a light microscope. According to a number of previous reports, DNase I is secreted by exocrine glands, such as the pancreas and parotid gland, into the alimentary tract (Laskowski 1971; Rohr and Mannherz 1978; Lacks 1981). Furthermore, DNase I was also found to present in various tissues, including the kidney, lymph node, small intestine, heart, liver, and epididymis. This wide distribution probably reflects a variety of cellular functions of this enzyme. Peitsch et al. (1992) showed that the nuclease activity, which is responsible for internucleosomal DNA degradation during apoptosis, is functionally indistinguishable from DNase I. It has been established that DNase I complexes readily with G-actin, as has been exploited widely in histological and biochemical studies, but the physiological significance of the actin-DNase I interaction is still mysterious (Dabrowska et al. 1949; Mannherz et al. 1977; Hays and Lindberg 1991; Philip et al. 1992).
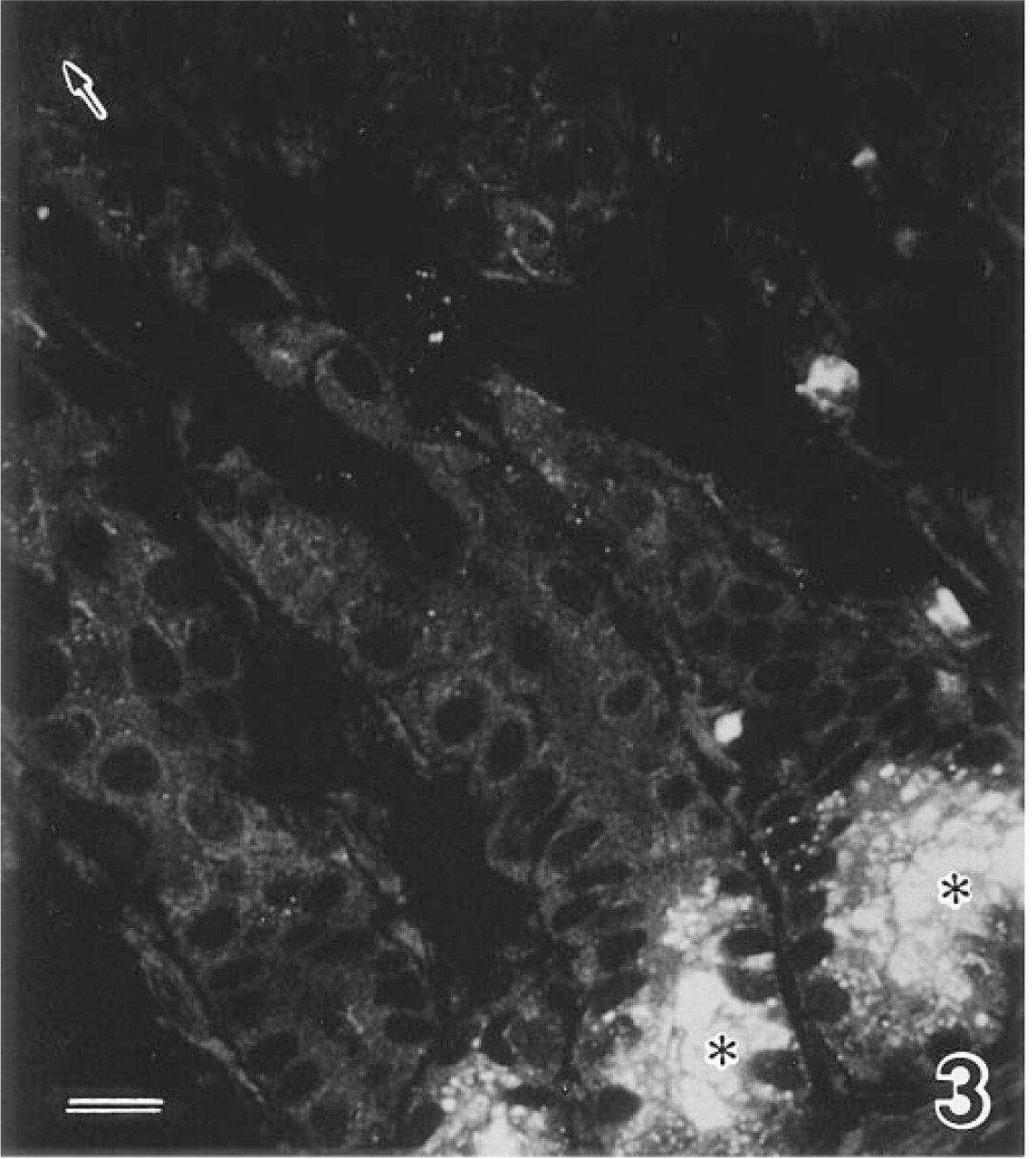
Immunofluorescence of DNase I in the human small intestine (ileum). The cytoplasm (asterisks) of Paneth cells shows strong immunoreactivity. Note that the staining intensity is heterogeneous inside the cells, with stronger immunoreactivity in their apical regions. Such clear immunoreactivity is not observed in the villous tip (arrow). Bar = 20 μm.
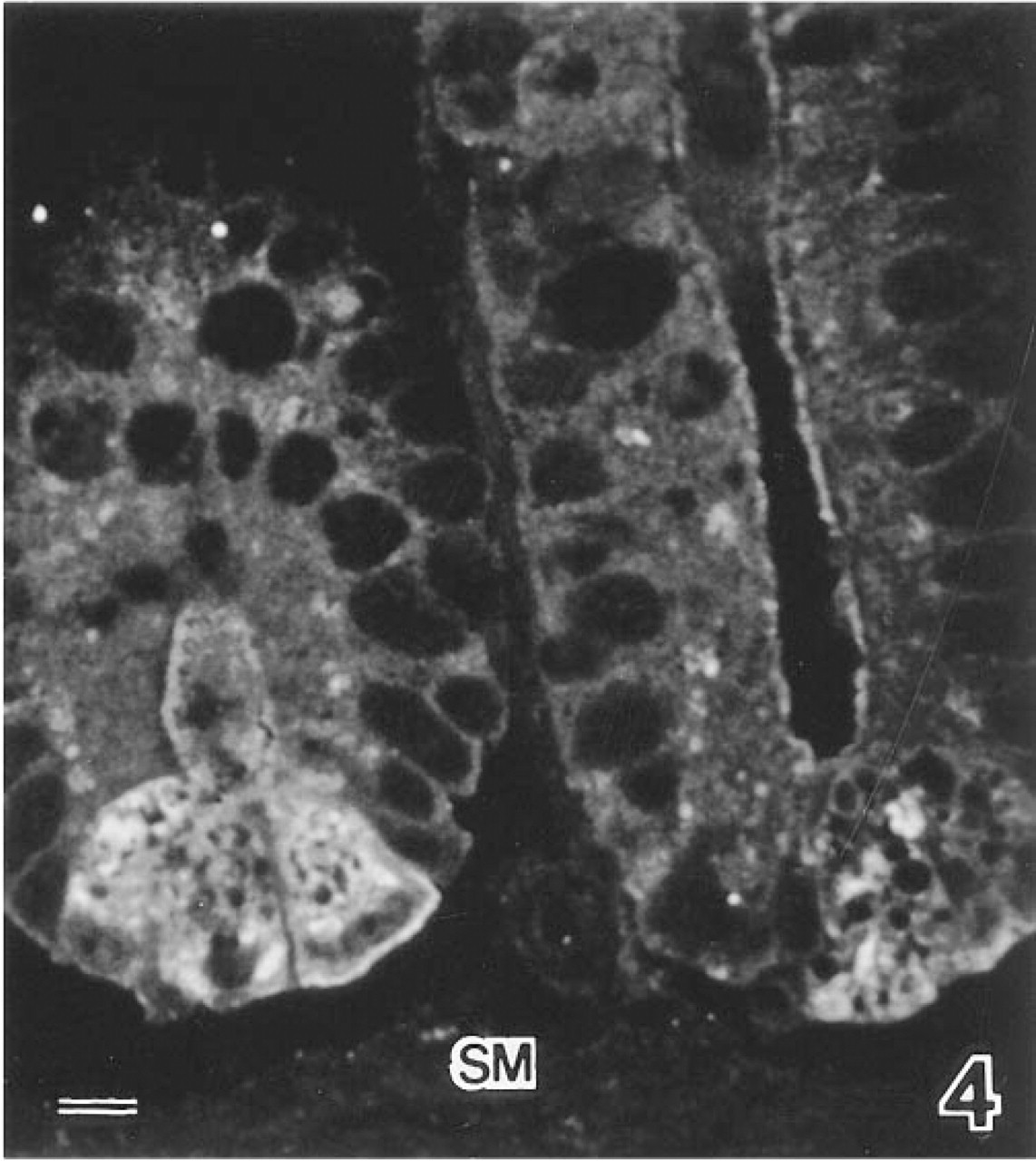
In situ hybridization demonstrating high levels of specific human DNase I mRNA in Paneth cells of the human ileum. SM, smooth muscle. Bar = 10 μm.
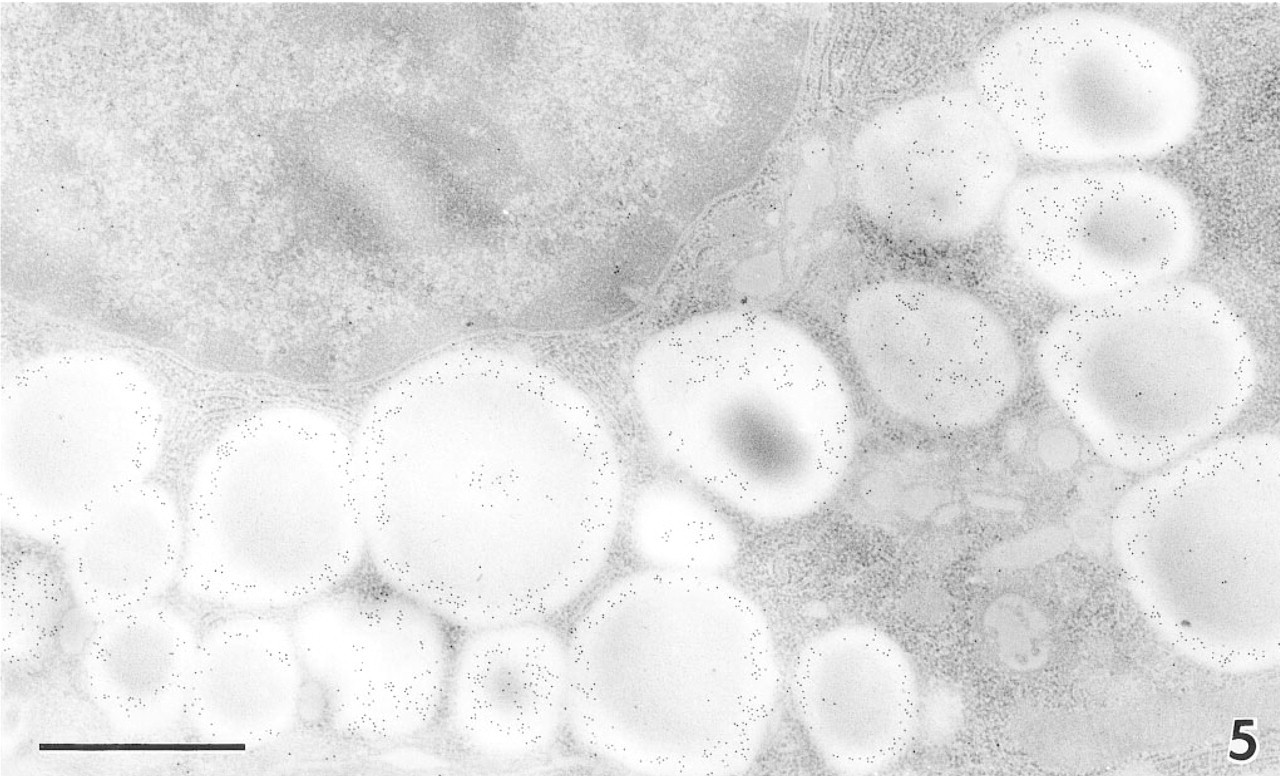
Immunoelectron micrograph of a Paneth cell in the human ileum. Note the many 10-nm gold particles in secretory granules, preferentially distributed in their periphery. Bar = 1 μm.
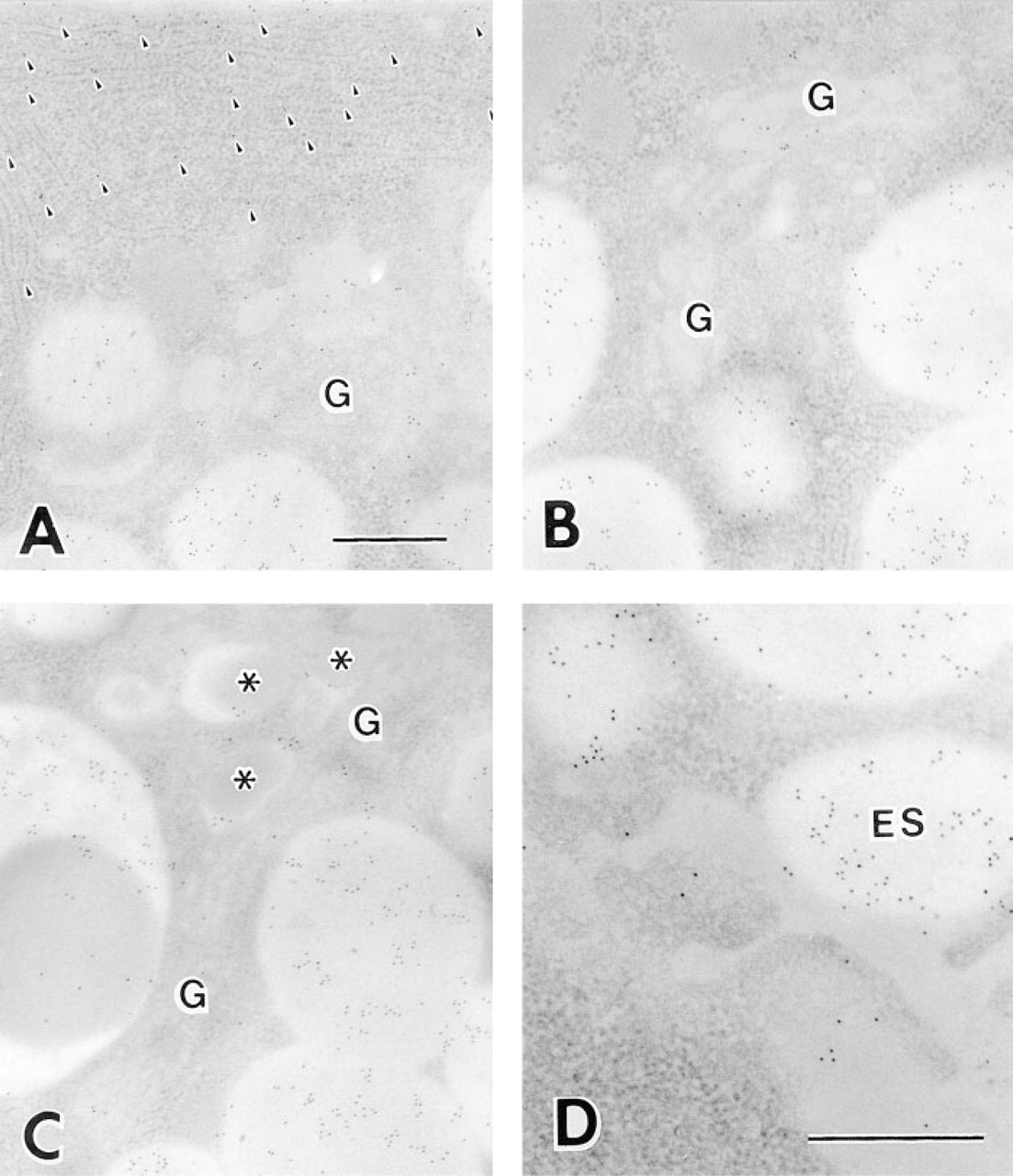
Immunoelectron micrographs of Paneth cells of the human ileum, showing gold particles in each step of the secretory pathway. (
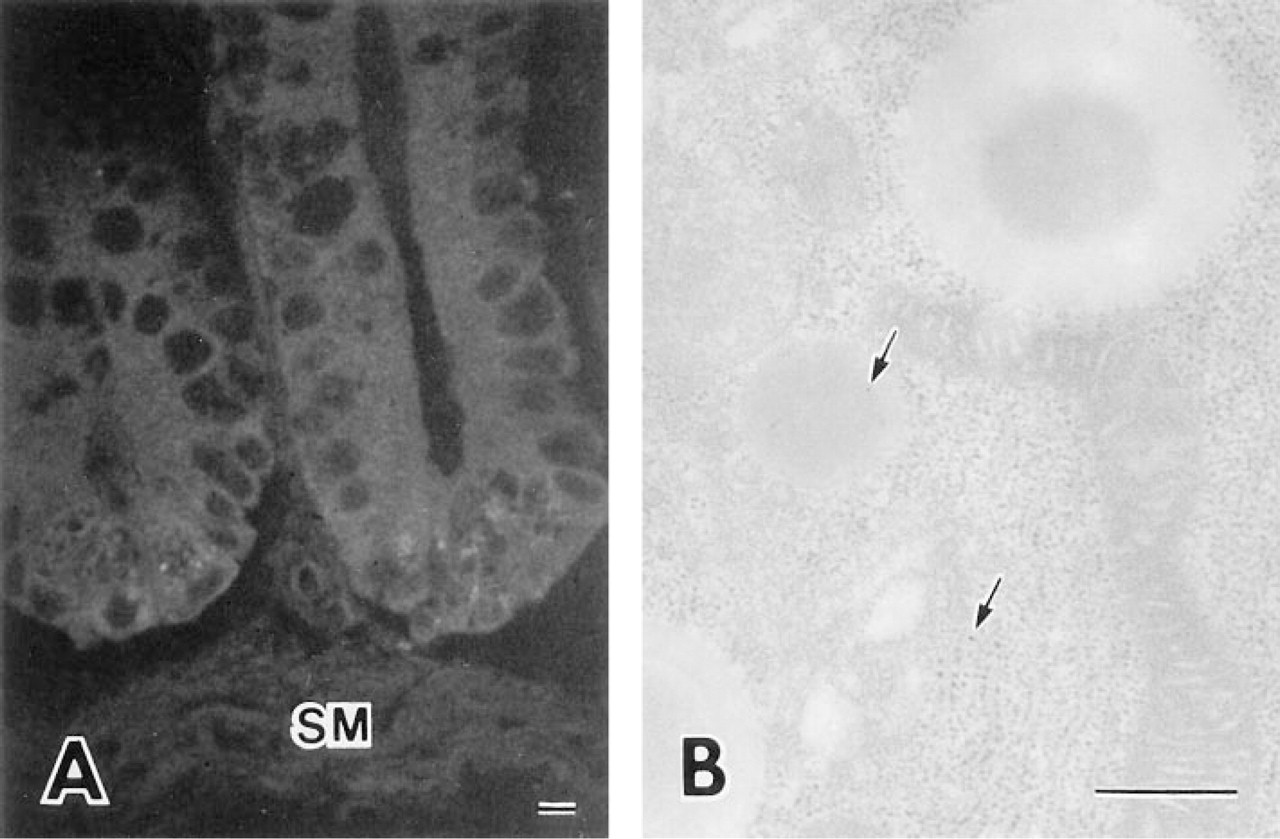
Controls of in situ hybridization and immunogold electron microscopy. (
Paneth cells are generally considered to be involved in the regulation of intestinal flora because of their ability to secrete antimicrobial materials (Rodning et al. 1976; Selsted et al. 1992; Coutinho et al. 1996; Mallow et al. 1996; Leis et al. 1997). The bacterial environment of the gut lumen has a direct effect on Paneth cell secretory activity (Masty and Stradley 1991). Several classes of antimicrobial peptides have also been identified in the mammalian small intestine. Cryptdin is the name given to the peptides of the mouse small intestine belonging to the defensin antimicrobial peptide family (Selsted et al. 1992). Results of in situ hybridization studies demonstrated specific and abundant expression of cryptdins in Paneth cells. Our experiments showed that DNase I was synthesized and secreted from Paneth cells of the human small intestine, suggesting that this enzyme participates in host defense, together with other antibacterial proteins, including cryptdins, immunoglobulins, and TNF. DNase I may also be an important component of the host intestinal immune defense system.
Footnotes
Acknowledgements
We are very grateful to Prof Lee D. Peachey (Department of Biology, University of Pennsylvania, Philadelphia, PA) for revising this manuscript, and also to Drs Tomoyuki Kawada and Yoshiaki Sasazawa (Department of Public Health, Gunma University School of Medicine, Gunma, Japan) for valuable and practical advice.
