Abstract
Interactions between epithelial cells and the extracellular matrix through integrins play a key role in the development of the lung by modulating branching morphogenesis, epithelial cell polarization, and differentiation. To determine the role of integrins during the different stages of lung development, we investigated the distribution of eight integrin subunits in the trachea and lung from human fetuses. In distal airways, during the early pseudoglandular stage of development, the α2-, α5-, α6-, αv-, and β1-subunits were detected in all epithelial cell plasma membranes, and polarized but undifferentiated tracheal epithelial cells expressed α3-, α6-, and β1-subunits in the plasma membrane of the cells facing the basement membrane. The α6- and β4-chains were detected along the basal plasma membrane of the basal cells in differentiated tracheal epithelia. The α4-subunit was detected in all respiratory cells throughout fetal development. In the submucosal glands, myoepithelial cells expressed the integrin subunits found in the undifferentiated cells of the developing airways, whereas the secretory cells expressed only α2-, α3-, α4-, α6-, and β1-subunits. These results demonstrate differential expression of integrins during lung development and suggest that integrins may play multiple roles in organogenesis and maturation of respiratory surface epithelium and glands.
I
Materials and Methods
Materials
Twelve normal human embryos and fetuses ranging from the pseudoglandular (n = 4) to the canalicular (n = 5) and the alveolar stage of development (n = 3) were studied. These fetuses were obtained from collaborating obstetricians and were the products of spontaneous or medically interrupted pregnancies. Fresh lungs and tracheas were dissected and maintained in RPMI 1640 culture medium (Seromed; Biochrom, Berlin, Germany) containing penicillin (100 U/ml) and streptomycin (100 μg/ml) before cryofixation. Tissues were then embedded in OCT (Tissue Tek; Miles, IN) and frozen in liquid nitrogen. Samples were stored at −80C.
Antibodies
Primary antibodies used for single or double immunostaining were mouse monoclonal antibodies (MAbs) P1E6 against α2 (dilution 1:500), P1B5 against α3 (1:500), P4G9 against α4 (1:10), and 3E1 against β4 (1:500) human integrin subunits, purchased from GIBCO BRL (Gaithersburg, MD), mouse MAb SAM1 against VLA5 (α5) (1:50) and rat MAb GoH3 against VLA6 (α6) (1:100) from Immunotech (Marseille, France). The integrin subunits αv and β1 were detected with mouse MAb [nondiluted supernatants of H5P10 (Boylan et al. 1995) and P5D2 (Dittel et al. 1993) clones, respectively], kindly provided by Dr. D. Sheppard (University of California, San Francisco, CA). Human α-actin was used to identify the myoepithelial cells of the glands (Gugliotta et al. 1988) and was recognized by a mouse MAb (clone asm-1) (1:50) purchased from Boehringer Mannheim Biochemica (Mannheim, Germany). Negative controls were performed by omitting the incubation with primary antibodies or by replacing the primary antibodies with murine nonimmune IgG2ακ fractions (Sigma Immunochemicals; St Louis, MO). All antibodies were diluted in 0.1 M PBS, pH 7.2, containing 1% bovine serum albumin (BSA).
Single or Double Immunofluorescence
To detect the localization of the integrin subunits in fetal lungs, single immunostainings were carried out: 5 μm frozen sections were fixed in precooled methanol (-20C). The sections were preincubated with PBS containing 3% BSA. They were first exposed to the primary MAb for 90 min, incubated with PBS-BSA 3%, and then exposed to the secondary MAb (1:50 in PBS-BSA 1%): biotinylated goat anti-mouse or biotinylated goat anti-rat IgG (Boehringer Mannheim), depending on the primary MAb, for 60 min. After incubation with PBS-BSA 3%, the complexes formed were detected by streptavidin-coupled fluorescein isothiocyanate (1:50 in PBS-BSA 1%) (streptavidin-FITC) (Amersham Life Sciences; Poole, UK).
Double immunostainings using antibodies directed against integrin subunits and α-actin were carried out to analyze the localization of integrin subunits in the fetal glands. For all integrin subunits except α6, after fixation in methanol, frozen sections were first incubated with the MAb directed against integrin (90 min). These MAbs were recognized by antimouse F(ab')2 fractions coupled with digoxigenin (1:50 for 60 min) (Boehringer Mannheim), which were then detected by anti-digoxigenin Fab fractions coupled with FITC (1:50 for 30 min) (Boehringer Mannheim). For the α6-subunit, we used biotinylated goat anti-rat IgG as a secondary antibody and streptavidin-FITC as a detection system. For the second immunostaining, we used the technique described for single immunofluorescence. Sections were incubated with primary antibodies directed against α-actin, which were recognized by biotinylated goat anti-mouse IgG. The complexes formed were then detected by streptavidin-Texas Red (1:50 in PBS-BSA 1%) (Amersham Life Sciences).
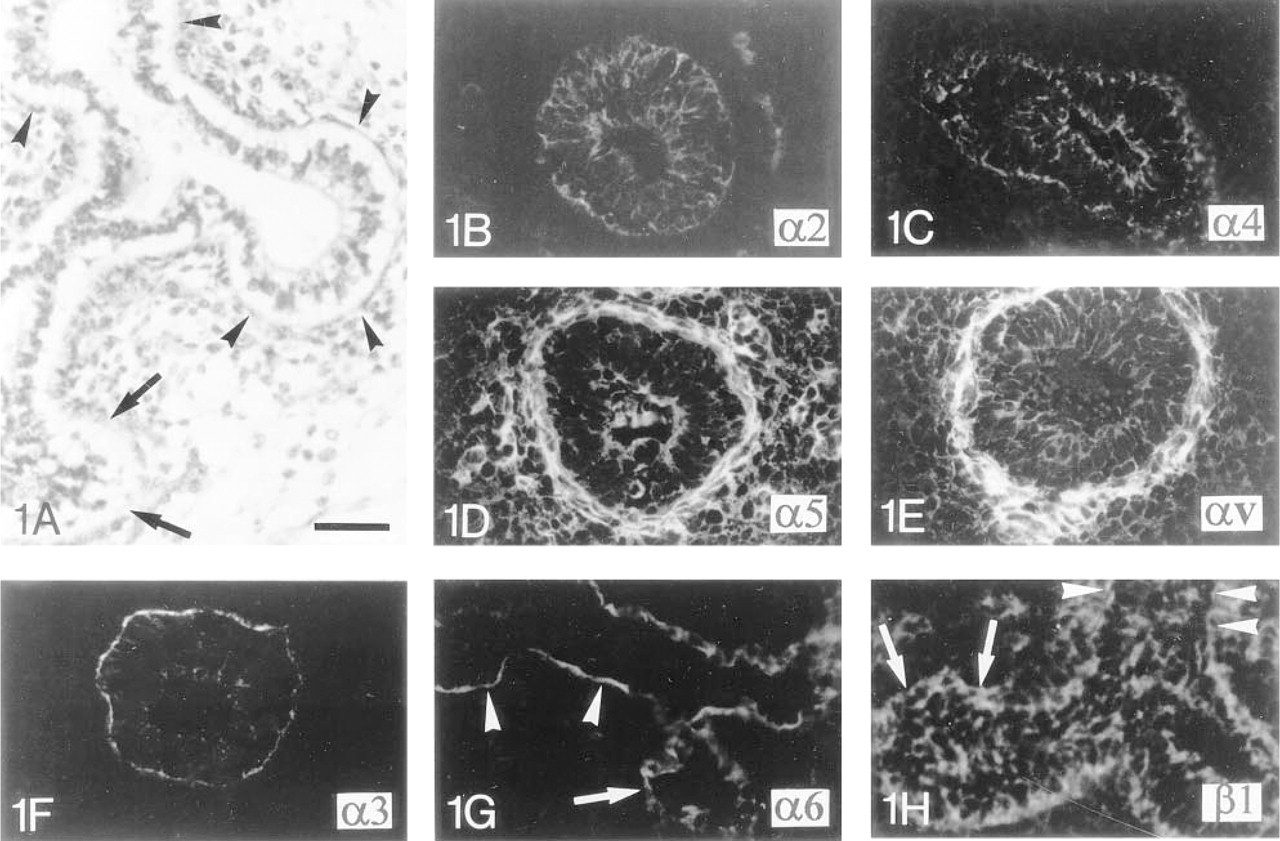
Immunocytochemical localization of integrin subunits in human fetal lung during the early pseudoglandular stage of development.
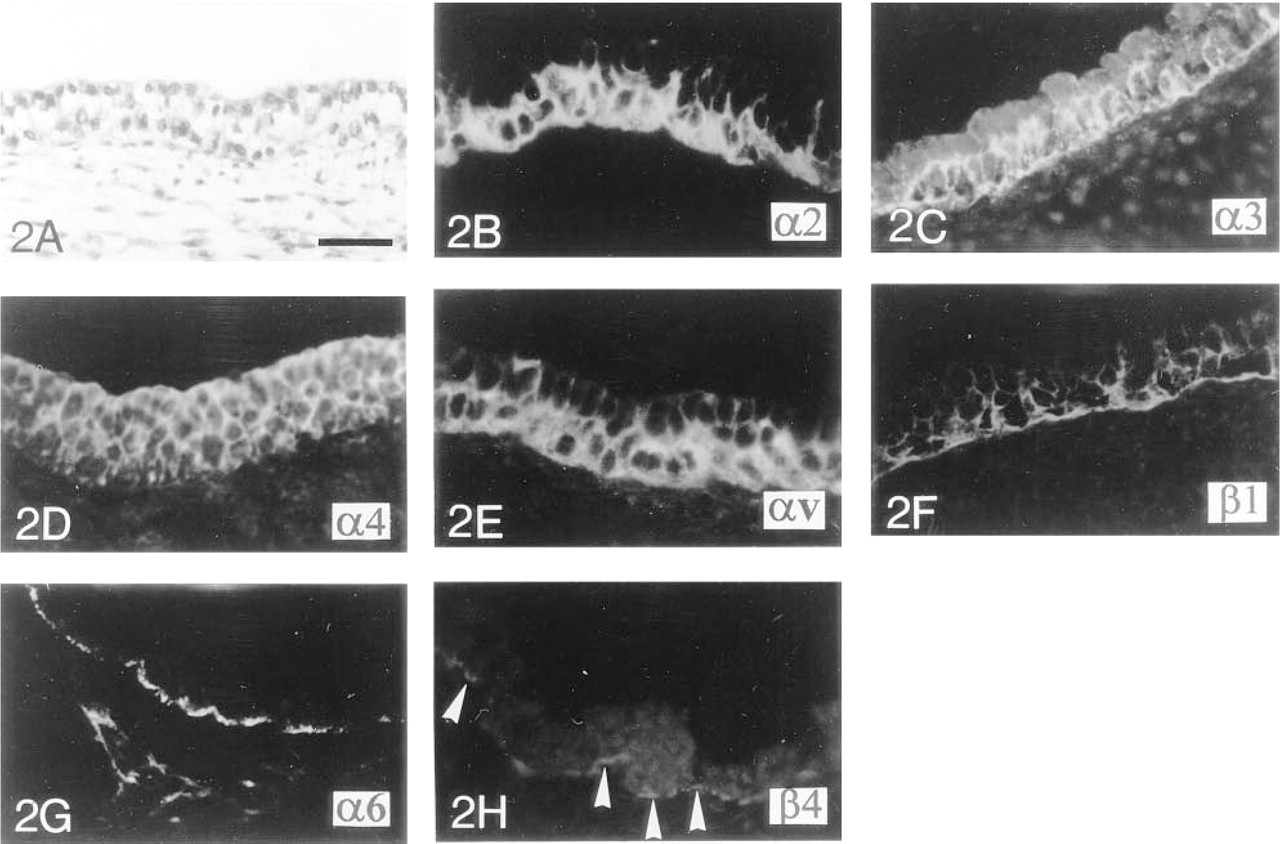
Immunocytochemical localization of integrin subunits in human fetal tracheas during the early pseudoglandular stage of development.
The observations were made under an Axiophot microscope (Zeiss; Oberkochen, Germany) using epifluorescence and Nomarski differential interference illumination.
Results
Localization of Integrin Subunits During the Early Pseudoglandular Stage of Development
Integrin Subunits in the Lung During Distal Branching Morphogenesis. During the pseudoglandular stage of development between 7 and 16 WD, the proximal respiratory tubules are lined by columnar epithelial cells containing apical nuclei. The cells are undifferentiated, as indicated by the absence of ciliated and secretory cells (Figure 1A). At this stage of development, the α2 integrin subunit is distributed in the pericellular plasma membrane and the α4-chain in the apical and basal sides of the plasma membrane of all undifferentiated epithelial cells in both growing pulmonary buds and tubules (Figures 1B and 1C). The α5 and the αv integrin subunits are detected in the pericellular plasma membrane of epithelial cells of the tubules (Figures 1D and 1E). The α3-chain is detected in the cytoplasm of epithelial cells of the growing buds, but a higher intensity of fluorescence is observed along the basal plasma membrane of the epithelial cells of the respiratory tubules (Figure 1F). α6 and β1 integrin subunits are detected in the cytoplasm and on the pericellular plasma membrane of epithelial cells growing at the branch tips. However, in more proximal respiratory tubules, the expression of these α6- and β1-subunits is restricted to the basal plasma membrane of the epithelial cells (Figures 1G and 1H). At this pseudoglandular stage, β4 integrin staining is either absent or exhibits a focal and punctate distribution along the basal plasma membrane of a few polarized cells facing the BM of respiratory tubules (data not shown).
Integrin Subunits in Polarized and Undifferentiated Tracheal Epithelium. Differentiation of the airways follows a cephalo-caudal direction. During the early pseudoglandular stage (between 7 and 11 WD), the surface epithelium of the trachea appears to be more mature than the epithelium of the distal pulmonary buds or tubules, as indicated by the presence of a bilayered surface epithelium containing columnar and polarized cells with apical nuclei. However, tracheal epithelial cells remain undifferentiated, without any ciliated or secretory cells (Figure 2A). The most basal cells show a cytoplasmic and pericellular plasma membrane staining for the α2 integrin subunit (Figure 2B). The cytoplasm of all epithelial cells stains positively for the α3-subunit. However, the cells lining the tracheal BM exhibit stronger pericellular plasma membrane staining (Figure 2C). As shown in Figures 2D-2F, both layers of tracheal epithelial cells stain positively for the α4, αv, and β1 integrin chains within the cytoplasm and pericellular plasma membrane. The level of labeling of the αv- (Figure 2E) and β1- (Figure 2F) subunits is lower in the apical layer of the surface tracheal epithelium. As previously shown in the pulmonary buds and tubules, the α6-subunit is localized close to the BM, along the basal plasma membrane of the most basal cells (Figure 2G), whereas the β4-chain only shows a very punctate distribution in the same area (Figure 2H). The α5 integrin cannot be detected within the tracheal epithelium (data not shown).
Localization of Integrin Subunits in the Differentiated Trachea During the Canalicular and Alveolar Stages of Development
During the end of the pseudoglandular stage of development as well as during the canalicular and alveolar stages (between 12 and 32 WD), the surface tracheal epithelium is progressively differentiated and pseudostratified. At 24 WD, the three major types of cells (ciliated cells, secretory cells, and basal cells) found in mature respiratory proximal airways are observed (Figure 3A). Furthermore, by 17 WD the glandular network is already developed, containing many tubules and acini which are surrounded by myoepithelial cells. The different types of integrin subunits expressed during the canalicular and alveolar stages of development are similar. The α2, α3, α4, and β1 integrin subunits are localized to the cytoplasm and pericellular plasma membrane of all tracheal surface epithelial cells (Figures 3B-3E). However the β1 integrin is preferentially localized to the basal plasma membrane of the cells lining the BM (Figure 3E). During the canalicular stage of development, α6- and β4- chains are detected at the basal plasma membrane of tracheal basal cells in close proximity to the BM (Figures 3F and 3G). The α5- and αv-chains are not detected in the tracheal epithelium (data not shown). In the glandular ducts during the canalicular and alveolar stages of development, the α2, α3, α4, and β1 integrin grin subunits are detected in the pericellular plasma membrane of the most basal cells (Figures 3B, 3C, and 3E). The α5- and αv-chains are not detected in the glandular ducts (data not shown). The localization of the α6- and β4-subunits is restricted to the basal plasma membrane of the cells in the proximal part of ducts and is detected in the cytoplasm and on the pericellular plasma membrane of the epithelial cells in the distal part of ducts (Figures 3F and 3G). In the glands, myoepithelial cells, identified by smooth muscle α-actin, show positive staining for α2, α3, α4, α5, α6, αv, β1, and β4 integrin subunits (Figures 3H-3O), whereas the cytoplasm of the secretory cells stains positively for the α2-, α3-, α4-, α6-, and β1-subunits (Figures 3H-3J, 3L, and 3N) but not for α5-, αv-, and β4-chains (Figures 3K, 3M, and 3O).
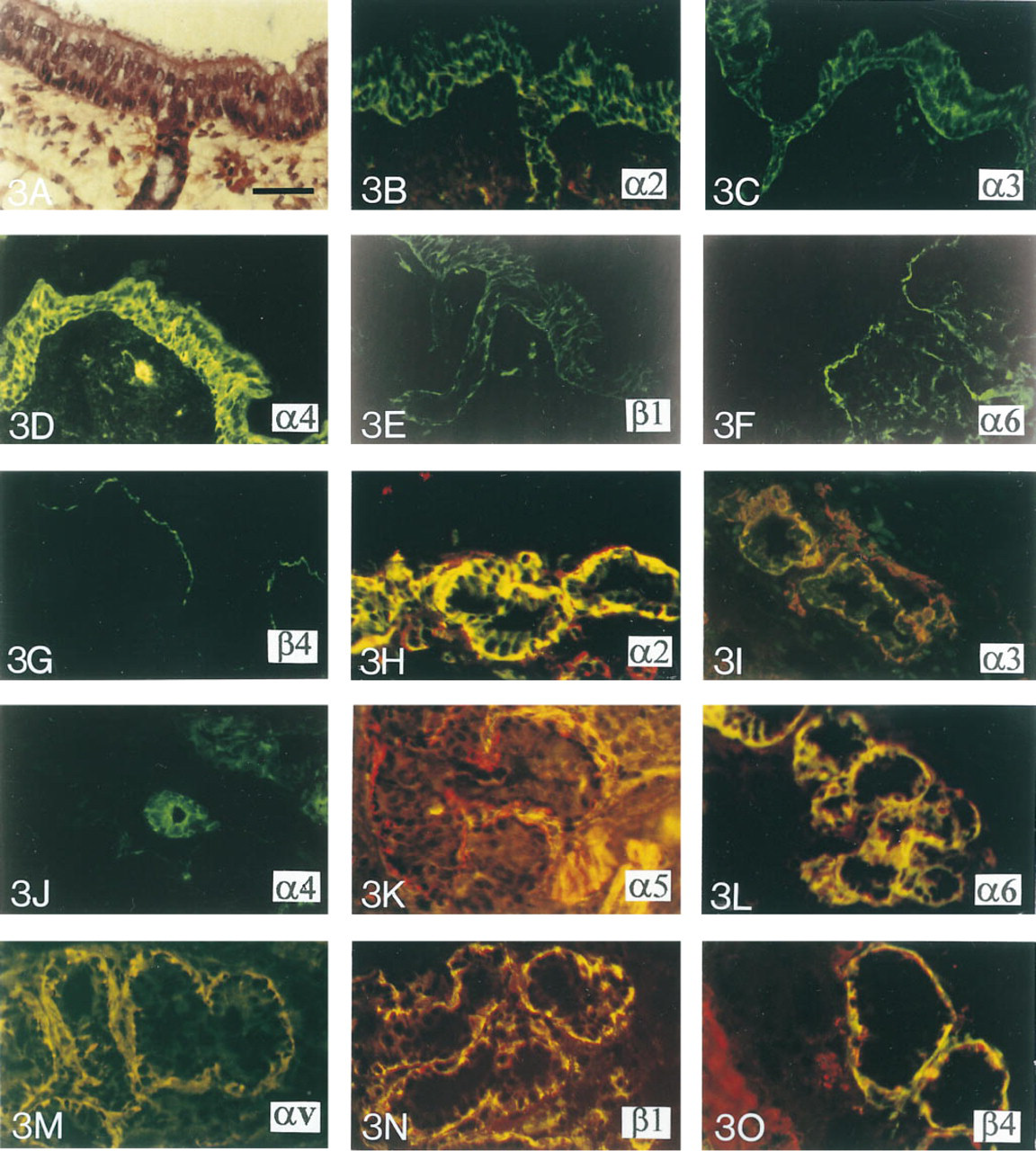
Immunocytochemical localization of integrin subunits (FITC) and α-actin (Texas Red) in human fetal trachea during the canalicular and alveolar stages of development.
Discussion
We have studied the temporal and spatial distribution of integrin subunits in developing human lungs. The results suggest a potential role for each of these receptors during the development of human airways.
Integrins and Branching Morphogenesis
Our results suggest that α2, α5, α6, αv, and β1 integrins play a role in the process of branching morphogenesis that leads to the formation of the airways. These receptors were localized to the pericellular plasma membrane of the cells in the developing respiratory buds, suggesting that they may play a role in the process of migration or proliferation that is responsible for branching morphogenesis during the pseudoglandular stage of development. α2 integrin, associated with the β1 integrin subunit, was already detected in branch tips of developing mouse lung (Wu and Santoro 1996). The role of the α2 integrin subunit in branching morphogenesis was demonstrated using MDCK cells (Saelman et al. 1995) or mammary epithelial cells (Berdichevsky et al. 1994). Roman et al. (1991) also demonstrated a reduction in the number of branch tips of mouse lung in vitro by incubating rudiments with a hexapeptide containing the RGD sequence which binds to α3, α5, and αv integrin subunits, thus suggesting a role for these integrins in the process of branching morphogenesis. Mutant mice for the α3 integrin gene showed an aberrant branching morphogenesis of airways (Kreidberg et al. 1996). The α5 integrin subunit was not detected during mouse fetal tracheal development (Roman et al. 1991), although it was reported in human migratory cells during wound healing after epithelial injury (Herard et al. 1996). Taken together, these studies suggest that, in the human, the α5 integrin subunit may act during branching morphogenesis by modulating cell migration. Because the α6 integrin subunit is demonstrated only in association with β1- or β4-subunits (Niessen et al. 1994), our results suggest that the α6-subunit is probably associated with the β1-subunit because the β4-subunit was not detected in early development. Some authors, using anti-α6-subunit antibodies, were also able to demonstrate inhibition of branching morphogenesis in mouse explanted submandibular glands and kidney (Kadoya et al. 1995; Falk et al. 1996). Nevertheless, Georges-Labouesse et al. (1996) reported that gene knock-out mice for α6 integrin show normal branching morphogenesis of the kidney.
Integrins and Cell Polarization
Our investigations have demonstrated a redistribution of the α3, α6, and β1 integrin subunits during the pseudoglandular stage of development from the pericellular plasma membrane of all the respiratory cells localized in the distal growing buds to the basal plasma membrane (for α6- and β1-subunits) or to the pericellular plasma membrane of the most basal cells (for the α3-subunit) in the most proximal respiratory tubules. Our observations, like those of Wu and Santoro (1996) during mouse lung development, suggest that these integrins may play a role in cell polarization and attatchment. The membrane redistribution of the α3 and α6 integrin subunits has also been reported during development and maturation of renal tubules (Korhonen et al. 1990), in which recognition of the laminin E8 site by an integrin containing the α6-subunit is essential for epithelial polarization (Sorokin et al. 1990).
Integrins and Surface Epithelial Cell Differentiation
In this study we have demonstrated that polarized and poorly differentiated tracheal epithelial cells started to express the β4 integrin during the late pseudoglandular stage of gestation. Furthermore, similar to the α6 integrin subunit, the β4-chain was localized to the basal plasma membrane of mature tracheal epithelial cells lining the BM during the canalicular and alveolar stages of development. Although the α6 integrin subunit may be associated with the β1-chain during early stages of development along growing epithelial buds, it is possible that during polarization and differentiation of epithelial cells this integrin may also associate with the β4-chain. Because it was shown that this integrin is implicated, along with laminin-5, in hemidesmosome formation (Stepp et al. 1990), the hemidesmosomes connecting the BM proteins with the keratin filament network of the basal cells via the integrins, our results suggest that the α6β4 heterodimeric complex could play a role in cell differentiation, especially in basal cell differentiation. In the present study, except during early development, no av integrin subunit was detectable in the surface respiratory epithelium, as observed by Yamada et al. (1995) during mouse lung embryogenesis. However, av integrin was reported in the basal part of normal adult human respiratory epithelium (Damjanovich et al. 1992).
Our results also showed that respiratory epithelial cells stained positively for the α4 integrin subunit whatever their degree of maturation. Recently, Caniggia et al. (1996) identified α4β1 integrin as a fibronectin receptor in fetal rat lung cells in vitro. However, investigations in normal or malignant adult lung tissues were unable to detect the α4 integrin subunit (Damjanovich et al. 1992). The identification of the α4 integrin subunit in fetal lungs both in vivo and in vitro suggests that α4β1 integrin could be a marker of fetal respiratory epithelial cells, although its role remains yet to be determined.
Integrins and Gland Differentiation
Submucosal glands start their development by multiplication of the most basal cells of the surface epithelium, forming small buds that invade the mesenchyme (Thurlbeck et al. 1961). The cells of these buds later differentiate into secretory and myoepithelial cells (de Haller 1969). Myoepithelial cells are located at the periphery of the glandular acini and tubules and are believed to be the basal cells of the mature breast glands (Dulbecco et al. 1986). In the present study, myoepithelial cells and secretory cells were found to express a different distribution of integrins during gland development. We showed that myoepithelial cells expressed all the integrin markers found in undifferentiated respiratory cells of the growing lung buds during branching morphogenesis, particularly the α5 and the αv integrin subunits, which were found neither in the developing surface epithelial cells nor in the secretory cells of the glands. According to Dulbecco et al. (1986), some of the myoepithelial cells are pluripotent and may be involved as stem cells for mammary gland development. Such potentiality has yet to be demonstrated in tracheal glands.
In conclusion, our results demonstrate differential expression of integrins during lung development. It will be of major interest to further investigate the roles of these integrins and their relationships with the ECM proteins during airway and lung development.
Footnotes
Acknowledgements
Supported in part by EC-Network no. BIO-CT 95-0284 and by the Association Française de Lutte contre La Mucoviscidose (AFLM).
We are grateful to André Guidez for his excellent photographs.
