Abstract
Peroxisome proliferator-activated receptors (PPARs) are transcription factors belonging to the nuclear receptor superfamily and have been initially described as molecular targets for compounds that cause peroxisome proliferation (for review see SØrensen et al. 1998). To date, three isotypes of PPAR have been described in humans: α (Sher et al. 1993); NUC1, also called β or δ (Schmidt et al. 1992); and α (Elbrecht et al. 1996; Fajas et al. 1997). There are three subtypes of PPARα mRNA, transcribed from three different promoters, which give rise to two proteins, PPARα1 and α2, as the protein encoded by PPARα3 mRNA is indistinguishable from PPARα1 (Fajas et al. 1998).
In humans, PPARα is present mainly in liver, heart, and kidney, whose tissues exhibit high fatty acid metabolism and high peroxisome-dependent activity. PPARβ is ubiquitously expressed in all tissues tested, whereas PPARα predominates in adipose tissue, large intestine, and macrophages and monocytes (Mukherjee et al. 1994; Auboeuf et al. 1997; Marx et al. 1998; Ricote et al. 1998; Spiegelman 1998). Consistent with their localization, the PPAR subtypes play different roles. After binding to specific elements (PPREs), PPARα regulates the transcription of several target genes involved in lipid metabolism and homeostasis (Desvergne and Wahli 1995; Wahli et al. 1995; Lemberger et al. 1996; SØrensen et al. 1998). PPARα controls adipocyte (Chawla et al. 1994; Tontonoz et al. 1995; Schoonjans et al. 1996; Spiegelman et al. 1997) and monocyte/macrophage (Marx et al. 1998; Ricote et al. 1998; Tontonoz et al. 1998) differentiation. Until now, the precise role of PPARβ has not yet been elucidated.
Detailed descriptions of the morphological and functional changes occurring during the development of the human gastrointestinal tract are available (MéAnard and Calvert 1991; MéAnard 1994, 1995; Montgomery et al. 1999). Several lines of evidence suggest an early role of the fetal digestive tract in fat digestion. We have reported that peroxisomes with fatty acid β-oxidation capacity are present in the fetal human gut (DauçLa et al. 1996). The latter exhibits functional mechanisms to synthesize all the lipid classes and to secrete them in the form of lipoproteins (Levy et al. 1992; Basque et al. 1998). Thus far, no studies have been devoted to the expression of PPARs in the developing human fetal digestive tract. Because the localization of these receptors may help to clarify the physiological roles of PPARs, this analysis was undertaken by immunohistochemistry in esophagus, stomach, small intestine, and colon of human fetuses from Week 7 to Week 23 of development (WD). In this study we demonstrated a spatial and temporal distribution of PPARs in developing digestive tract that suggests a differential role for these receptors during morphogenesis and cell differentiation.
Materials and Methods
Tissue Specimens
Samples of esophagus, stomach, intestine, and colon from 23 fetuses ranging from 7 to 22 weeks of age were obtained from normal elective pregnancy terminations. The project was performed in accordance with the requirements of the Institutional Human Subject Review Board (University of Sherbrooke) for the use of human tissues. The latter were embedded in Polyfreeze Tissue Freezing Medium (Polysciences; Warrington, PA) and frozen in liquid nitrogen.
Production of Anti-PPAR Antibodies
As shown in Figure 1, the anti-PPARα antibody was raised against the amino acid sequence 45SSGSFGFTEYQY56 of human PPARα (Sher et al. 1993). The sequence 24EGAPE-LNGGPQHAL37 of human NUC1 (Schmidt et al. 1992) was used to produce the anti-PPARβ polyclonal antibody. The anti-PPARα1/α2 antiserum was raised against the amino acid sequence EMPFWPTNFGISSVD common to PPARα1 and PPARα2 [amino acids 5-19 of mouse PPARα1 (Zhu et al. 1993); 35-49 of PPARα2 (Tontonoz et al. 1994)]. The sequence is well conserved in the two human PPARα isoforms (Elbrecht et al. 1996; Fajas et al. 1997). Taking advantage of the fact that the PPARα2 differs from the PPARα1 by an additional specific N-terminal amino acid region, the sequence of the hapten used to produce the anti-human PPARα2 antibody was mapped at that region and corresponded to 2GETLGDSPIDPESDS16 of human PPARα2 (Elbrecht et al. 1996; Fajas et al. 1997). The synthetic peptides were coupled to keyhole limpet hemocyanin as a carrier according to the glutaraldehyde method (Avrameas 1969). Polyclonal antibodies were raised by SC injections into rabbits using standard procedures. In addition, we used a commercial anti-PPARα antiserum (Interchim; MontluçLon, France) directed against the amino acid sequence MMGEDKIKFKHITPL common to PPARα1 and PPARα2 (amino acids 256-270 of human PPARα1, 284-298 of human PPARα2).
Characterization of the Antibodies
The polyclonal antibodies produced were characterized by immunoprecipitation and Western blotting assays. In vitro transcription and translation of mouse PPARα/pSG5, PPARβ/pSG5, PPARα1/pSG5, and human PPARα2/pBluescript IIKS+ plasmids (gift of Prof. W. Wahli; University of Lausanne, Switzerland) were performed using reticulocyte lysate (Promega; CharbonniéGres, France) and L-[35S]-methionine. Translated products were either immunoprecipitated with the antibodies and analyzed by SDS-PAGE, followed by autoradiography, or directly submitted to Western blotting and enhanced chemiluminescence (ECL) in crossreaction assays.
Adult and fetal colon mucosae were homogenized in 25 mM Hepes buffer, pH 7.4, containing 0.4 M KCl, 1 mM EDTA, 2 mM dithiothreitol, and a cocktail of protease inhibitors (Complete; Roche, Mannheim, Germany). The homogenates were centrifuged at 15,000 × g for 20 min (4C). The protein concentration of the supernatant was determined (Bradford 1976). Samples were analyzed by Western blotting and ECL according to the manufacturer's protocol (Boehringer Mannheim Biochemica; Mannheim, Germany) using the different antibodies.
Nuclease Protection Assay
Partial human PPARα2 cDNA corresponding to the 5′UTR sequence (Fajas et al. 1997) was obtained by standard RTPCR using total RNA extracted from human adipocytes (Chomzynski and Sacchi 1987) and primers up (5′-CCCATC-TCTCCCAAATATTT-3′) and down (5′-GGGCCAGAATGCGATCTCTGTG-3′). The resulting fragment (282 bp) was cloned into the pBSIIKS+ plasmid (Stratagene; La Jolla, CA), giving rise to the pBIIKS+/hPPARα2 vector. A pBIIKS+/hG3PDH clone containing a 380-bp DNA fragment of the human G3PDH encoding sequence (Tso et al. 1985) was produced using the same protocol and primers up (5′-CCCATCACCATCTTCCGA-3′) and down (5′-CTACAGGCCACAGTTTCC-3′). Nuclease protection assay was carried out according to Sambrook et al. (1989). Total RNA was extracted from human fetal intestines (14WD) and from 3T3 L1 cells (passage 58) as described above. 3T3 L1 cells were chosen for a positive control because those preadipocytes express PPARα2. Total RNA (5 μg) was hybridized overnight with 32P single-stranded DNA probes (105 cpm/sample) at 60C. After incubation, nonhybridized cDNA was digested by nuclease S1 (50 U/sample) for 60 min at 37C. The DNA/RNA hybrids were resolved by electrophoresis and the gel was exposed to Kodak film for 24 hr.
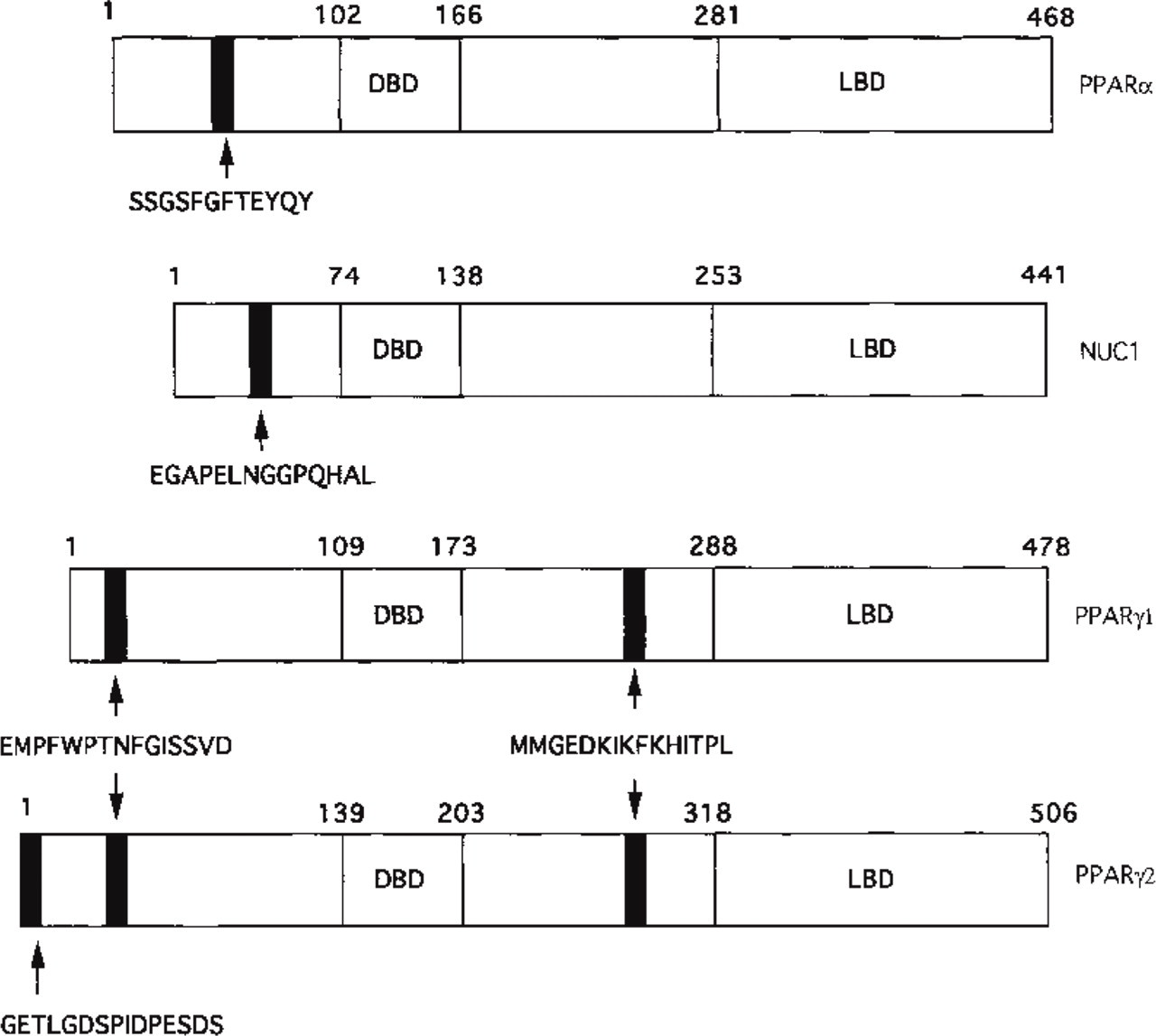
Schematic comparison of the different human PPAR subtypes vs PPARα. The positions of the peptides used to produce the polyclonal antibodies are mapped. The DNA (DBD) and ligand (LBD) binding domains are represented.
Immunohistochemical Analysis
Cryostat sections (3 ± thick) were fixed in 2% formaldehyde in PBS for 45 min at 4C and rinsed in PBS. They were immersed in 100 mM glycine in PBS for 45 min at 4C, then washed in PBS. The sections were preincubated with a blocking solution containing 0.1% fish gelatin, 0.8% bovine serum albumin, and Tween-80 (2 μl/100 ml PBS) for 30 min at room temperature (RT). They were first exposed to the primary antibody (diluted 1:250 in PBS/defatted dry milk 5% w/v) for 60 min at RT. After two washes in PBS, sections were exposed to the secondary antibody (1:50 in PBS-BSA 2%), fluorescein-conjugated goat anti-rabbit IgG (Boehringer Mannheim), for 60 min at RT. Negative controls were performed by replacing the primary antibody with PBS or with preimmune serum. Sections were then mounted in Vectashield medium and photographed with a Reichert-Jung Polyvar microscope (Vienna, Austria).
Results
Antibody Specificity
In vitro-translated mouse PPARα, β, and α1 and human PPARα2 were used for immunoprecipitation assays, taking advantage of the fact that the human peptide sequences chosen for immunization are well-conserved in the corresponding rodent sequences. Figure 2 shows that each antibody recognized the PPAR subtype against which it was raised. When preimmune serum was used as a control, no signal was obtained. Cross-reaction between each anti-PPAR antibody against the other PPAR subtypes was absent or very low, as demonstrated by Western blotting assays (Figure 3). The anti-PPAR antibodies produced were also characterized by Western blotting using cytosolic extracts from human adult and fetal colon mucosae. The presence of the different PPAR subtypes was detected in both samples examined (Figure 4). However, our results indicated a higher expression of PPARα2 in fetal colon compared to adult colon. In addition, the anti-PPARα antiserum provided by Interchim recognizes both human PPARα1 and PPARα2, as attested by the manufacturer.
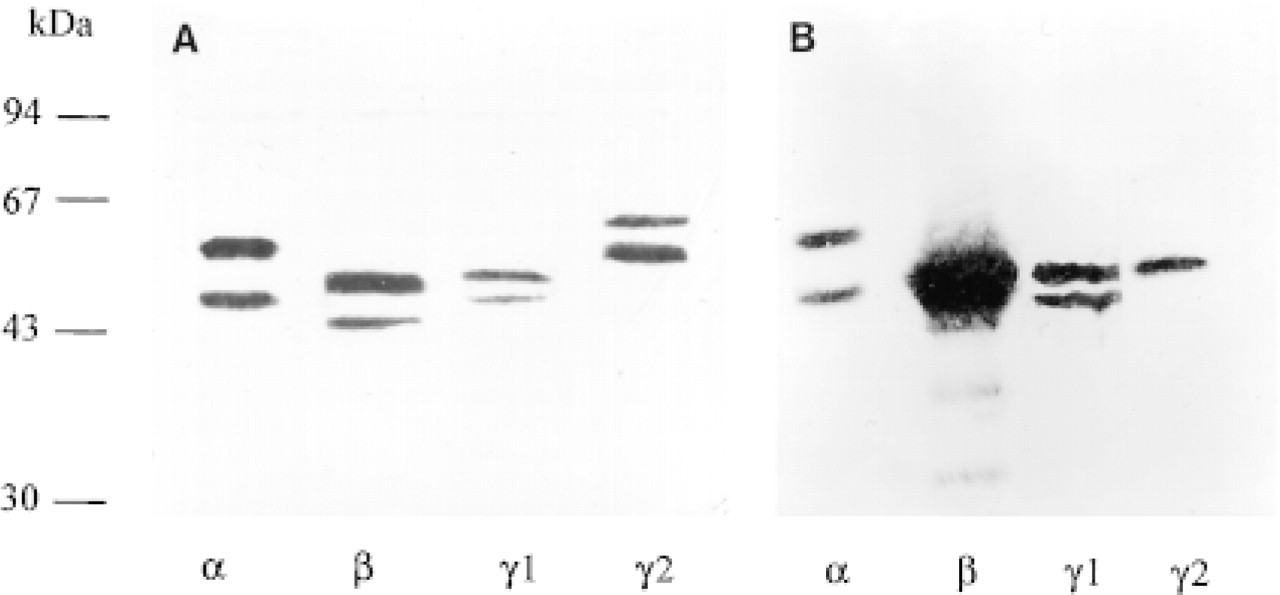
Recognition of the different PPAR subtypes by their respective antibody. (
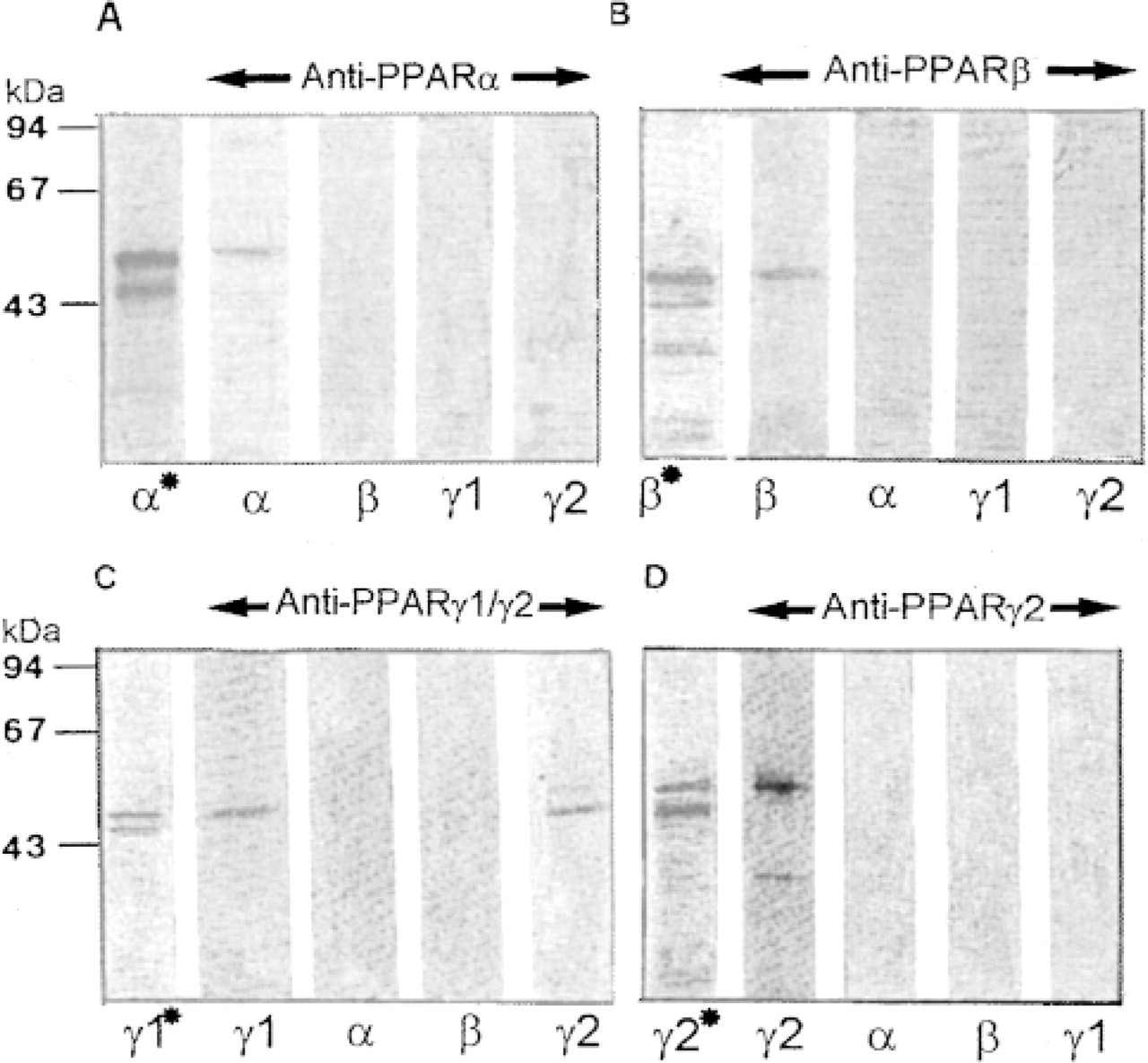
Specificity of anti-PPAR antibodies by Western blotting. Mouse PPARα/pSG5, PPARβ/pSG5, PPARα1/pSG5, and human PPARα2/pBSIIKS+ plasmids were in vitro-translated using reticulocyte lysate and l-[35S]-methionine. Translated products were submitted to SDS-PAGE (10%). The gels were either subjected to autoradiography (∗) or processed by Western blotting and ECL using the anti-PPAR antibody (diluted 1:500) indicated above the different lanes.
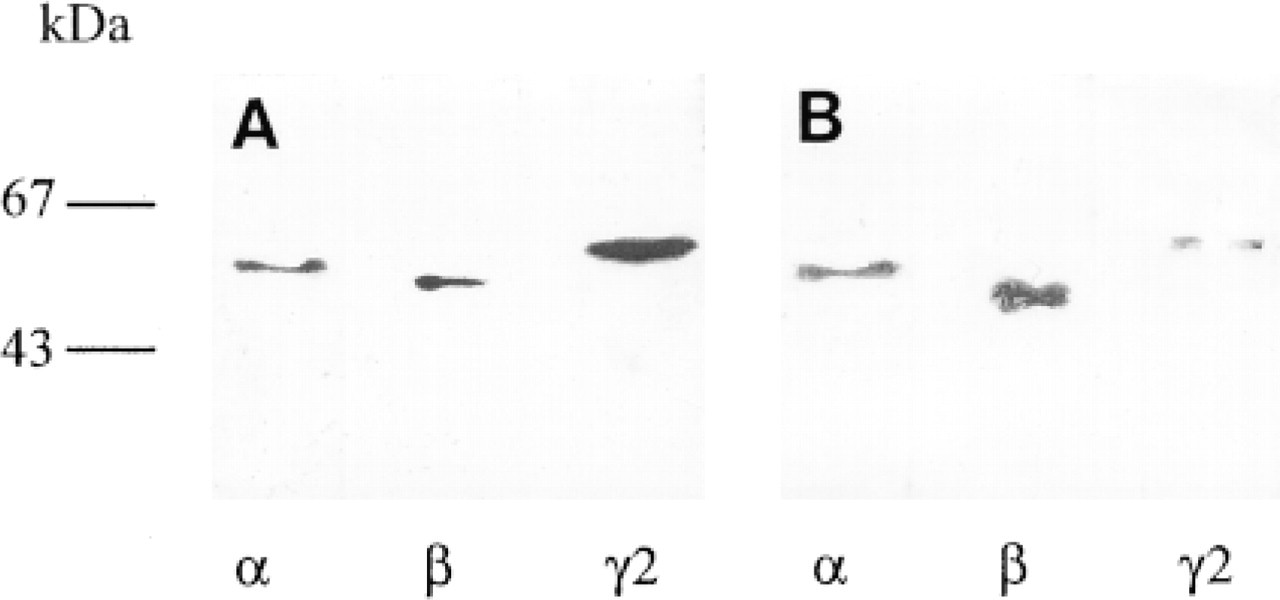
Western blotting analysis of PPARα, β, and α2 expression in human fetal (
PPARα Expression
The average immunohistochemical intensity values, as estimated by two independent investigators in four tissue sections from different fetuses, are summarized in Table 1 for the different PPARs. No immunoreactivity was found in control sections when the primary antibody was omitted or replaced by preimmune serum (not shown).
Figure 5A shows that the PPARα subtype was expressed as early as 7WD in the stratified columnar epithelium. At this stage, the tissue exhibited a cytoplasmic and nuclear staining. At 14WD (Figure 5B), a lower intensity of fluorescence was observed for this tissue. The decrease was much more pronounced at 20WD because the immunoreaction was mainly restricted to the nuclei of epithelial cells (Figure 5C). Faint staining was observed in the nuclei of mesenchymal and muscular cells throughout development of the fetal esophagus.
At 12WD the gastric epithelium showed the highest labeling with the anti-PPARα antibody compared with the intensity of fluorescence in the extra-epithelial layers. The PPARα protein was detected in the surface epithelial cells as well as in the epithelial growing buds (Figure 5D). At 15WD no significant change was noted in PPARα expression (Figure 5E). Four weeks later the staining was barely detectable in the gastric epithelial cells (Figure 5F). PPARα was faintly detected at 7WD in the stratified jejunal epithelium (Figure 5G). At 12WD PPARα expression remained very low in the villous jejunum (Figure 5H) but was higher in ileum (Figure 5J). No immunoreaction was detected at 16WD in the jejunal epithelium (Figure 5I). Meanwhile, PPARα was well expressed in the ileal tissue (Figure 5K). At 22WD (Figure 5L) PPARα expression was high, particularly in nuclei of ileal cells. At 8WD (Figure 5M) the colon was a simple tube with a slitlike lumen composed of stratified epithelium surrounded by mesenchyme. At this stage the PPARα subtype was moderately expressed in the epithelial cells (Figure 5M). Nuclei of mesenchymal cells were also stained by the antibody. At 14WD (Figure 5N) the luminal surface of the colon exhibited well-formed villi in which staining was slightly decreased. As mucous goblet cells differentiated, they produced secretory granules in which the PPARα was not detected. At 20WD (Figure 5O), the villous structures were present in the different segments of the colon. The specialized cells facing the colon lumen were stained. However, the intensity of fluorescence was faint and diffuse.
Differential expression of PPARα, PPARβ, and PPARα2 during fetal development of the human digestive tract a
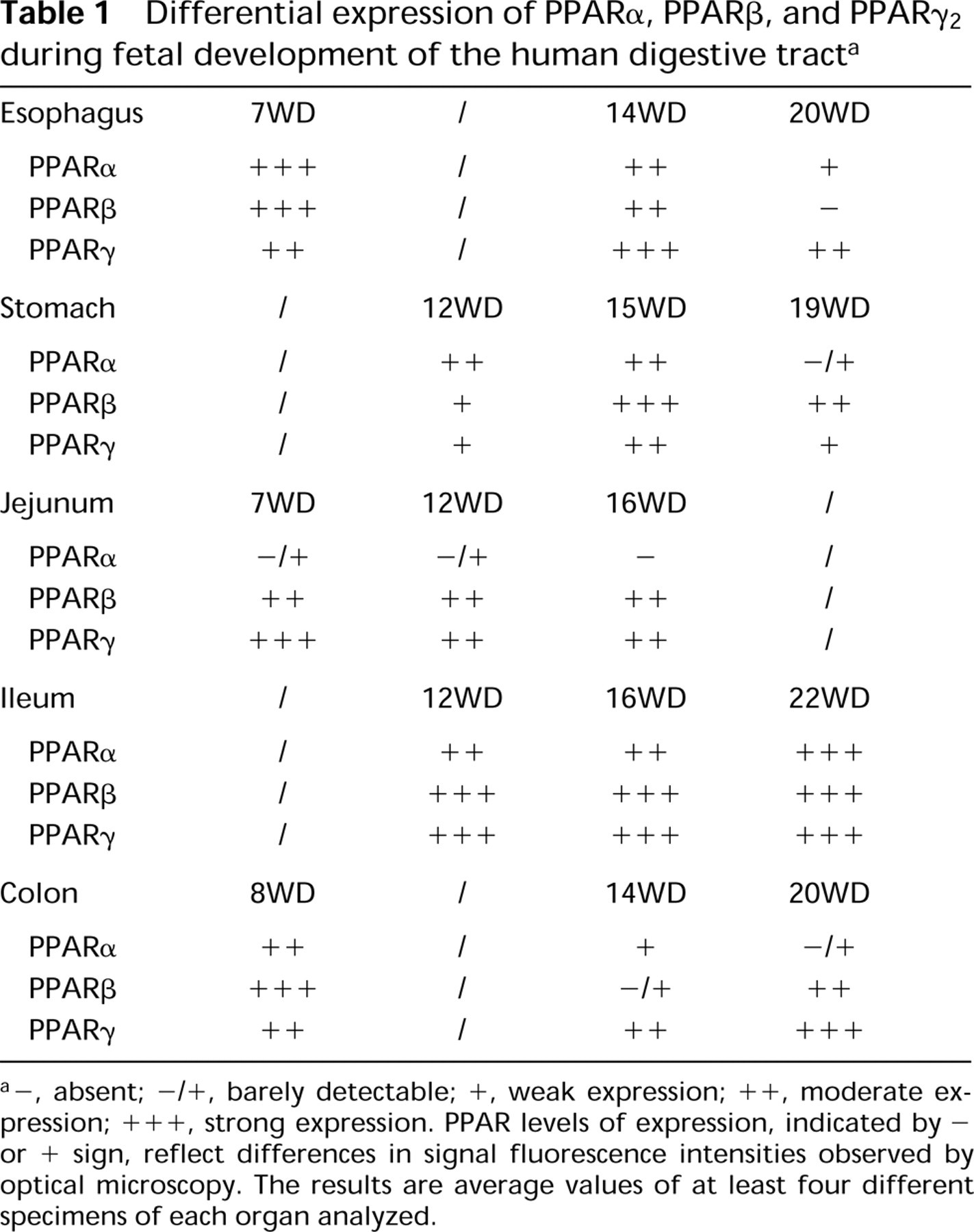
a-, absent; -/+, barely detectable; +, weak expression; ++, moderate expression; +++, strong expression. PPAR levels of expression, indicated by -or + sign, reflect differences in signal fluorescence intensities observed by optical microscopy. The results are average values of at least four different specimens of each organ analyzed.
PPARβ Expression
Between 7WD (Figure 6A) and 14WD (Figure 6B), the PPARβ subtype was substantially expressed in the cytoplasm and nucleus of human esophageal epithelial cells. A marked decrease was observed in the intensity of immunoreaction at 20WD (Figure 6C). Whatever the developmental stage examined, PPARβ was detected overall in the gastric epithelium (Figures 6D-6F). A peak in fluorescence intensity was observed at 15WD (Figure 6E; Table 1). At later stages the staining was more restricted to nuclei (Figure 6F).
In the small intestine, the anti-PPARβ antibody showed stronger staining in the ileum (Figures 6J-6L) than in the jejunum (Figures 6G-6I) at all stages studied. The staining in the epithelial cells remained moderate and high throughout development of the jejunum and ileum, respectively. At 22 WD the intensity of fluorescence was stronger in the ileal crypt epithelial cells than in the differentiated villous cells (Figure 6L).
The PPARβ subtype was well expressed in the different layers of the human fetal colon at 8WD (Figure 6M) and 20WD (Figure 6O). The anti-PPARβ antibody was located mainly in the nuclei of the epithelial and mesenchymal cells. As for the ileum, stronger staining was observed in the crypt regions. However, it was barely detected at 14WD (Figure 6N).
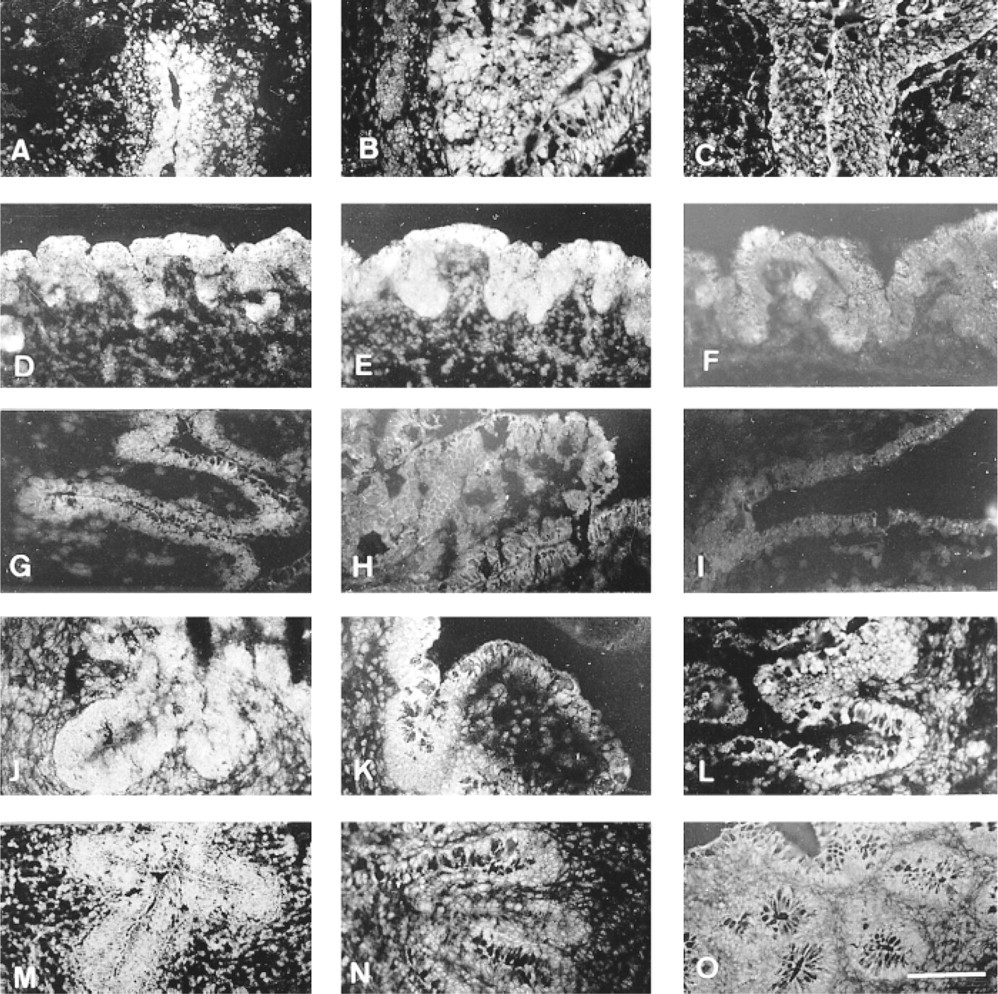
Differential expression of PPARα in developing human fetal digestive tract by immunohistochemistry. (
PPARα Expression
The different antibodies (anti-PPARα, anti-PPARα1/α2, and anti-PPARα2 antisera) used gave similar results for their distribution throughout the development of the human fetal digestive tract. However, the immunoreactivity was always higher with the anti-PPARα2 antibody compared to that observed with the two other antibodies. One can explain this difference by a higher level of immunoglobulins in the anti-PPARα2 antiserum. Therefore, only results obtained with the anti-PPARα2 antiserum are presented here (1). In addition, the presence of mRNA encoding PPARα2 was confirmed in intestinal extracts from human fetuses by nuclease S1 protection assay (Figure 7).
The presence of the PPARα2 protein was detected at 7WD (Figure 8A), 14WD (Figure 8B), and 20WD (Figure 8C) in esophagus. Throughout human fetal esophageal development, the staining was moderate or high and was restricted to nuclei of both epithelial and extra-epithelial cells.
Expression of PPARα2 was lower in the gastric epithelium (Figures 8D-8F) than in the esophageal tissue. A slight increase was noted at 15WD (Figure 8E).
Staining with the anti-PPARα2 antibody was particularly prominent in epithelial cell nuclei of jejunum (Figures 8G-8I) and ileum (Figures 8J-8L). Because nuclei were localized in the basal part of epithelial cells, PPARα2 staining showed a spotted distribution along the basal plasma membrane. Owing to the abundance of nuclei, the intensity of fluorescence appeared higher in the crypt regions compared with the immunoreactivity in the upper villous regions (Figures 8I, 8K, and 8L).
At 8 WD (Figure 8M) and 14WD (Figure 8N), most nuclei of colon epithelial and mesenchymal cells were labeled with the anti-PPARα2 antibody. At 20WD the immunoreaction was higher and was more restricted to nuclei of specialized cells facing the colon lumen (Figure 8O).
Discussion
The results described here establish for the first time the presence and the spatiotemporal distribution of three PPAR subtypes in the developing human fetal digestive tract.
Expression of PPARs in Human Digestive Tract
With the use of polyclonal antibodies specifically directed against PPARα, PPARβ, and PPARα (α1/α2 and α2), our immunohistochemical data show that the different PPAR subtypes are expressed as early as 7WD in human fetal digestive tract and in different cell types of endodermal and mesodermal origin. The presence of PPARα, PPARβ, and PPARα has already been reported in adult rat esophagus, stomach, small intestine and colon by in situ hybridization (Braissant et al. 1996). In another recent study, quantification of PPAR mRNA by ribonuclease protection assay revealed relatively high expression of PPARα and PPARβ in adult mouse colon compared to small intestine. In contrast, PPARα expression was higher in the intestinal mucosa (Mansen et al. 1996). Furthermore, PPARα is reported to be also expressed in human (Brockman et al. 1998; Sarraf et al. 1998) and rodent (Lefebvre et al. 1998; Saez et al. 1998) colon tumor cells. Although both PPARα1 and α2 proteins are detected in fat tissues, it is believed that expression of the PPARα2 isoform remains mainly adipocyte-specific, whereas the PPARα1 isoform expression may be extra-adipocytic. However, in two recent studies the presence of the two PPARα isoforms is obvious in human (Dubois et al. 1998) and rodent (Lefebvre et al. 1998) colon cells as shown by the results obtained in Western blotting using polyclonal antibodies reactive with both PPARα1 and PPARα2. Our data from immunohistochemical, Western blotting, and nuclease S1 protection assays agree with these results. They are somewhat at variance with those of Braissant and Wahli (1998), who detected by in situ hybridization the presence of PPARα and PPARβ mRNA, but not PPARα mRNA, in rat fetal intestine. The discrepancy probably reflects differences in models examined or in timing of functionality of the gut in fetal life, or in the techniques used, as we mostly analyzed protein levels.
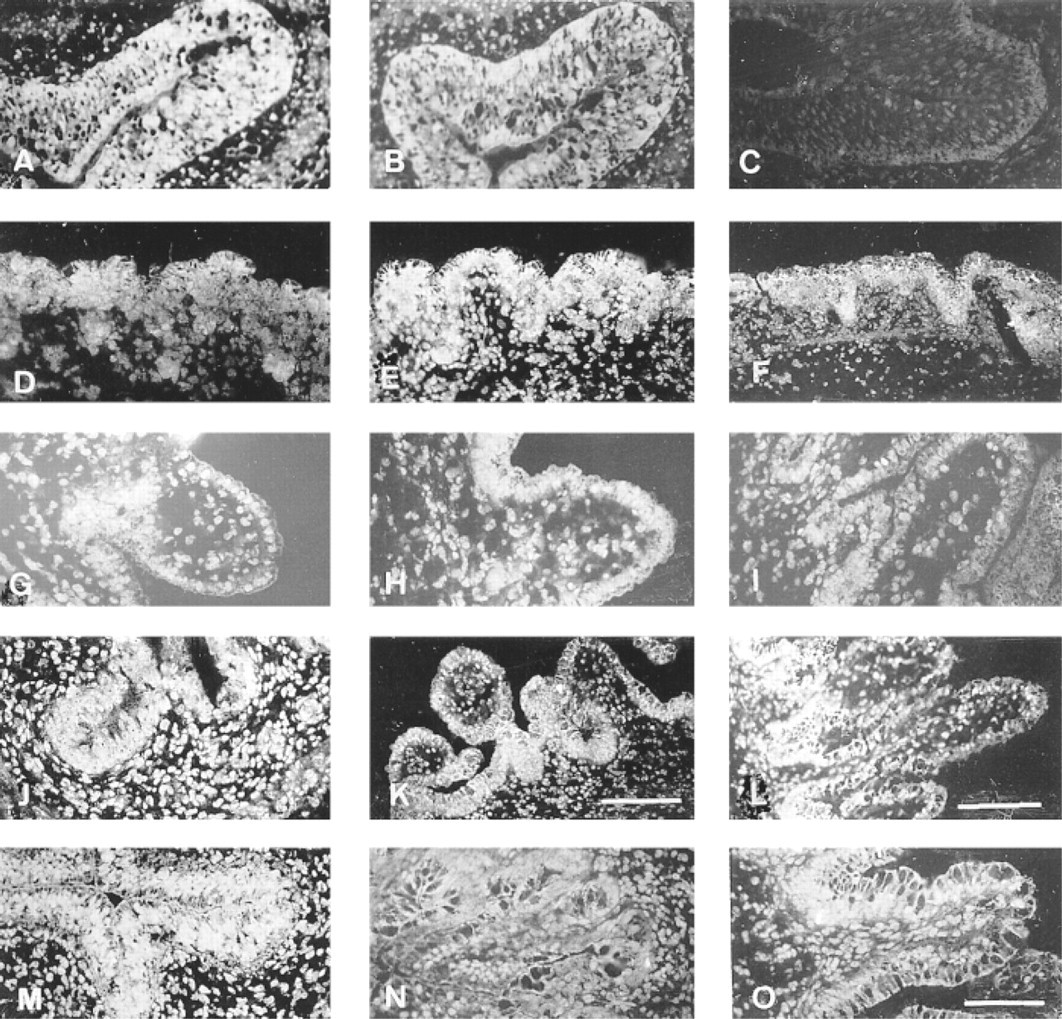
Differential expression of PPARβ in the developing human fetal digestive tract by immunohistochemistry. (
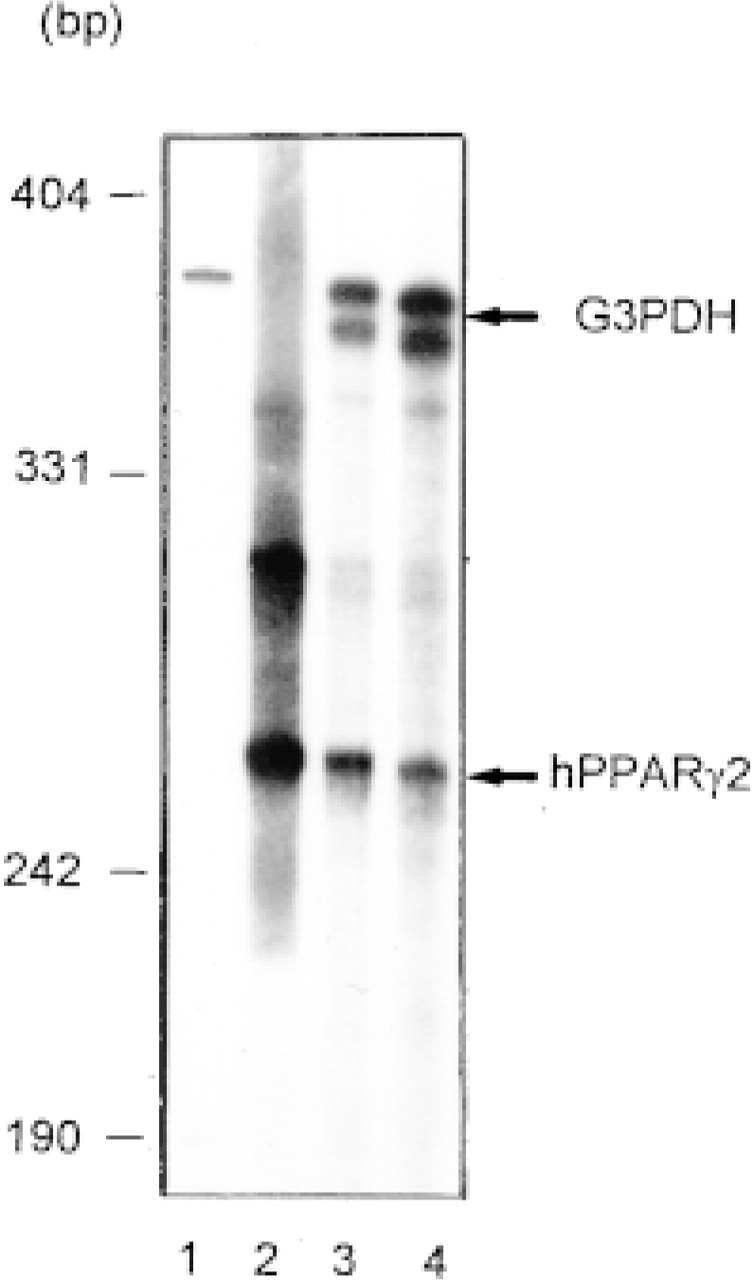
Detection of PPARα2 mRNA in human fetal intestine (14WD) by nuclease protection assay. 32P-labeled DNA probes to detect either G3PDH or PPARα2 mRNA were prepared as described in Materials and Methods. 32P-labeled G3PDH (Lane 1) and PPARα2 (Lane 2) DNA probes (105 cpm) were run on a 5% polyacrylamide gel. Total RNA (5 μg) isolated from 3T3 L1 cells (Lane 3) or from human fetal intestine (14WD) (Lane 4) were hybridized with the two radiolabeled probes. After nuclease S1 digestion, the resulting products were electrophoresed and the gels were autoradiographed. The presence of PPARα2 mRNA is obvious in the two models examined.
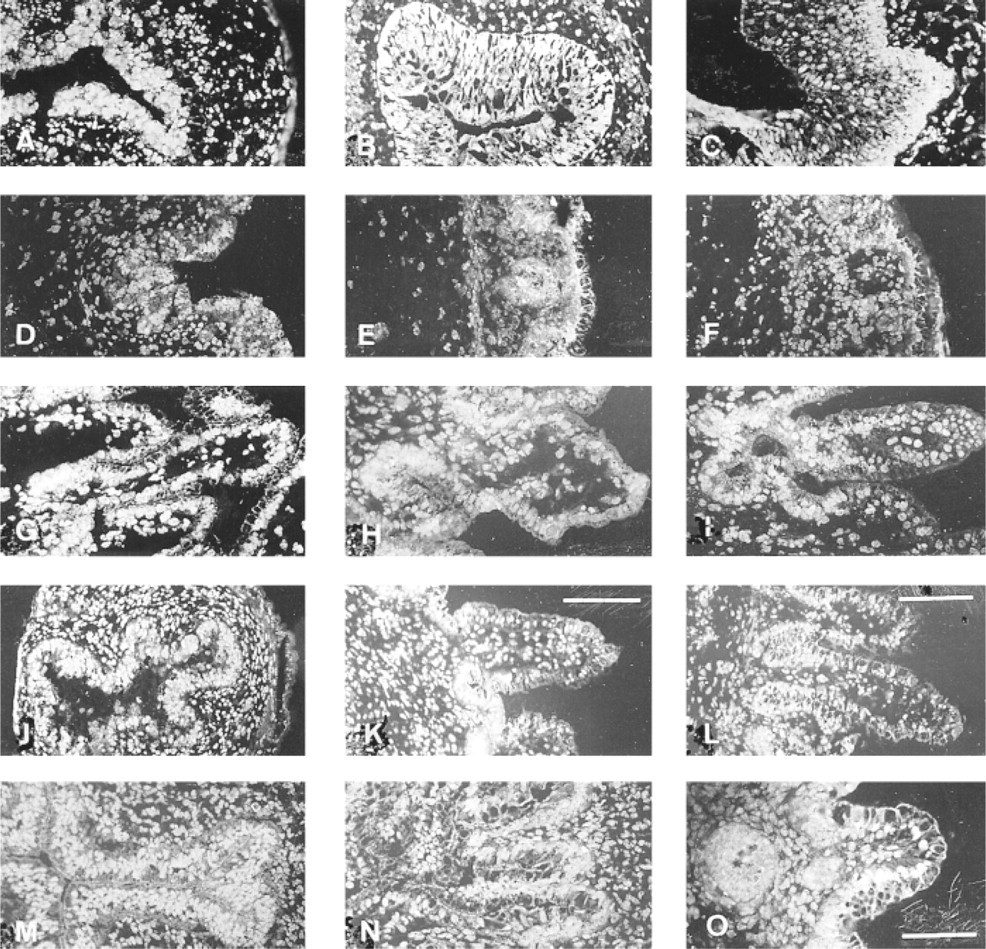
Differential expression of PPARα2 in developing human fetal digestive tract by immunohistochemistry. (
Spatiotemporal Distribution of PPARs
PPARs are expressed at different levels in cell types of endodermal and mesodermal origin during development of the human fetal digestive tract.
At early stages (7-15WD) of esophageal and stomach development, PPARα and PPARβ are more localized in the cytoplasm than in the nucleus, whereas at later stages they become predominantly nuclear. This expression pattern overlaps for the esophagus, with replacement of the columnar ciliated epithelium by adult squamous tissue exhibiting flattened cells with microvillous processes in their apical membrane (Johns 1952). For the stomach, it coincides with the acquisition of adult features, characterized by differentiation of gastric cells appearing at the base of the evolving pits (Salenius 1962), followed by differentiation of enterochromaffin cells and mucous neck cells (De Lomos 1977). PPARα is involved in lipid metabolism and homeostasis (Wahli et al. 1995; Schoonjans et al. 1996). Indeed, the absence of PPARα expression in knockout mice prevents the PP inducibility of genes encoding peroxisomal and microsomal lipid-metabolizing enzymes (Lee et al. 1995). The early onset of PPARα expression in the esophagus and stomach suggests the involvement of this subtype in establishment of epithelial lipid metabolism. The decrease of PPARβ expression in the esophageal epithelium is likely due to a shift in the physiology of this tissue. A role of PPARβ in the onset of gastric cell differentiation is possible because Braissant and Wahli (1998) have found a correlation between the peak of ubiquitous expression of PPARβ during mouse embryogenesis and the period of greatest cell differentiation. Our results also show that PPARα is predominantly nuclear throughout esophageal and stomach development. This subtype is well expressed during esophageal morphogenesis, suggesting a role for PPARα during this process. On the other hand, PPARα expression is low or moderate during stomach formation.
In the intestine as a whole and whatever the fetal stage examined, the different PPAR subtypes are more expressed in ileum and, to a lesser extent, in colon than in duodenum. The different spatiotemporal expression of PPARα, PPARβ, and PPARα during development of the human fetal intestine and their ligand specificity (SØrensen et al. 1998) suggest that these receptors are involved in different intestinal functions. PPARα ligands induce the expression of genes involved in lipid absorption and transport in the rat small intestine (Martin et al. 1997; Motojima et al. 1998). The physiological role of PPARβ in the small and large intestine remains unknown. PPARα ligands have been shown to inhibit proliferation and to induce differentiation of human colon cancer cells (Brockman et al. 1998; Sarraf et al. 1998). On the other hand, the same ligands enhance colon polyp and tumor formation in the min/+ mouse model (Lefebvre et al. 1998; Saez et al. 1998). It is evident from our results that PPARα is expressed along the intestinal crypt-villous region in both proliferating and differentiated cells. At present, it is difficult to speculate about the precise role played by PPARα in intestinal cell life.
In summary, the spatiotemporal distribution of the PPAR subtypes has been described during development of the human fetal digestive tract. The different PPARs are predominantly expressed in epithelial cells, although their presence is also detected in nuclei of cells of mesodermal origin. The three PPAR subtypes exhibit different patterns of expression in relation to the morphogenesis of the digestive tract. They are expressed very early, suggesting that these receptors play major roles in the development and/or the physiology of the digestive tract. Furthermore, the fact that PPARα is expressed at a high level whatever the region considered (except the stomach) and the stage studied argues for a prominent role of this receptor in human digestive tract.
Footnotes
Acknowledgements
Acknowledgments
Supported by the Association de la Recherche contre le Cancer (Contrat ARC no. 9233), the Ligue contre le Cancer (ComitéA de Meuthe et Moselle), the Fondation de la Recherche MéAdicale (ComitéA de Lorraine), and the Conseil de Recherches MéAdicales du Canada.
We are grateful to W. Wahli (University of Lausanne) for the mPPARα/pSG5, mPPARβ/pSG5, mPPARα1/pSG5, and hPPARα2/pBSIIKS+ plasmids, to M. Donner (UPRES 2402, Nancy) for the 3T3 L1 cells, and to A. Stoekel for her skillful assistance.
