Abstract
Monoclonal antibody (MAb) HIK1083, which is obtained by immunizing mice with a preparation of rat gastric mucins, has been shown to bind specifically to α-linked N-acetylglucosamine (α-GlcNAc). We investigated the specificity of MAb HIK1083 by immunostaining normal human organs, mucinous metaplasia of human pancreas, adenocarcinomas of human stomach, pancreas, and colon, and normal rat organs. The specificity was investigated by making comparisons with (a) a stain that labels Class III concanavalin A (ConA)-reactive mucin (Class III mucin), i.e., paradoxical ConA (PCS), and (b) staining with horseradish peroxidase (HRP)-conjugated Griffonia simplicifolia agglutinin II (GSA-II). In normal human and rat organs and in mucinous metaplasia of human pancreas, immunostaining with MAb HIK1083 and PCS showed similar specificities for mucins in glandular mucous cells. In adenocarcinoma of stomach and pancreas, GSA-II showed the most widespread positivity, PCS showed the least, and MAb HIK1083 showed a reactivity between those two extremes. Colon adenocarcinomas were labeled only with GSA-II. These results demonstrate that MAb HIK1083 could be a useful screening tool for Class III mucin in normal, metaplastic, and carcinoma tissues, and that the α-GlcNAc residue is one of the specific sugar residues found in Class III mucin.
C
Class III mucin is an essential component of the gastric surface mucous gel layer and plays an important role in protecting the gastric mucosa (Ota and Katsuyama 1992). Furthermore, the occurrence of Class III mucin in carcinoma tissues is consistently associated with the expression of gastric phenotypes. This is true not only in gastric carcinoma but also in carcinoma of the pancreas, biliary tract, lung, and other sites (Matsuzawa et al. 1992; Ota et al. 1995). Histochemical staining of Class III mucin is therefore indispensable to a proper understanding of the physiological role of the surface mucous gel layer and the phenotypic expression of various carcinomas.
The terminal sugar residue responsible for Class III ConA reactivity was suggested to be GlcNAc after biochemical and histochemical analysis (Hotta et al. 1982). Recently, Ishihara et al. (1996) introduced MAb HIK1083, which was obtained by immunizing mice with mucins prepared from rat gastric mucosa. This MAb reacts with oligosaccharides that have a nonreducing terminal of α-GlcNAc residues.
The present study was undertaken (a) to explore the localization of MAb HIK1083-reactive sites in normal human organs, metaplastic tissues of the pancreas, carcinoma tissues of the stomach, pancreas, and colon, and normal rat organs, and (b) to compare those results with the results of paradoxical ConA staining (PCS) for Class III mucin and the results of staining with GSA-II (which is also known to bind to Class III mucin). The results demonstrated the highly selective nature of the reactivity of HIK1083 with Class III mucin and reinforced the idea that it has potential diagnostic value.
Materials and Methods
Preparation of Tissues
Human tissues (20% formalin-fixed, routinely processed, and paraffin-embedded) were selected from the surgical pathology files of the Department of Laboratory Medicine Shinshu University School of Medicine. The following histologically normal tissues were selected: esophagus (n = 7), gastric cardiac mucosa (n = 7), fundic mucosa (n = 8), pyloric mucosa (n = 6), duodenum (n = 10), liver (n = 2), gallbladder (n = 9), common bile duct (n = 6), pancreas (n = 10), and colon (n = 10). In addition, gastric adenocarcinoma (10 cases of intestinal type, 10 cases of diffuse type), pancreatic duct carcinoma (five cases of papillary type, six cases of tubular type), and colon adenocarcinoma (six cases of well-differentiated type, four cases of moderately differentiated type) were also studied. The criteria used for the histological classification of gastric adenocarcinomas, pancreatic duct carcinomas, and colon adenocarcinomas were based on (a) Lauren's system, (b) the General Rules for the Study of Pancreatic Cancer issued by the Japan Pancreas Society, and (c) WHO guidelines, respectively.
In addition, tissues from male Sprague-Dawley rats (n = 5) weighing approximately 200 g were also examined. Tissues were extracted from these animals under sodium pentobarbital anesthesia (50 mg/kg
Monoclonal Antibody
MAb HIK1083 was generated by immunizing BALB/c mice with purified rat gastric mucins according to a previously reported method (Ishihara et al. 1993). The binding activity of MAb HIK1083 with the oligosaccharides of purified rat gastric mucins was confirmed by ELISA. The epitope sugar structure of such mucins is reported to be characterized by the presence of oligosaccharides containing peripheral α-GlcNAc residues (Ishihara et al. 1996).
Histochemistry
Serial 3-μm-thick sections were stained by the following methods. To produce five degrees of trypsin digestion, formalin-fixed sections of human gastric mucosa were treated with 0.2% trypsin (DIFCO; Detroit, MI) in 0.05 M Tris buffer, pH 7.6, together with 0.1% CaCl2 for 5, 15, 30, 45, or 60 min at 37C. These sections were then stained with MAb HIK1083 (dilution 20%), PCS (ConA was purchased from Sigma, St Louis, MO), or horseradish peroxidase (HRP)-conjugated GSA-II (GSA-II-HRP; EY Laboratories, San Mateo, CA) (dilution 1%). Immunostaining with MAb HIK1083 was performed by the indirect immunoperoxidase method. PCS and staining with GSA-II-HRP followed methods described previously (Katsuyama and Spicer 1978; Katsuyama et al. 1985). Diaminobenzidine was used as the chromogen for these stains. After the same tissues had been subjected to pretreatment (see below), all sections were stained with lectins and MAb HIK1083. A dual-staining method, galactose oxidase-cold thionine-Schiff (GOTS) followed by PCS (Ota et al. 1991), was used to differentiate gastric surface mucous cell-type mucins from gastric gland mucous cell-type mucins in mucinous metaplasia of the pancreas. All lectin stains were preceded by amylase digestion because both ConA and GSA-II react with glycogen.
Interference between GSA-II and MAb HIK1083 was investigated in sections of human fundic, pyloric, and duodenal mucosa. The procedures were as follows: (a) pretreatment with GSA-II and subsequent indirect immunoperoxidase staining with MAb HIK1083, and (b) pretreatment with MAb HIK1083 and subsequent GSA-II-HRP staining.
The effects of various pretreatments on the reactivity of MAb HIK1083, ConA, and GSA-II with human stomach and liver was investigated using the following procedures: (a) no pretreatment; (b) oxidation with 1% aqueous periodic acid (PA) for 10 or 60 min at room temperature (RT); or (c) interposed reduction with 0.2% NaBH4 in 1% Na2HPO4 for 2 min at RT after PA oxidation. The effect of amylase digestion was also evaluated for each protocol.
The reactivity for each stain was graded on the basis of the ratio of positive cells.
Results
Comparative Studies of Fixation Protocols and Trypsinization Protocols
The binding of MAb HIK1083, ConA, and GSA-II was preserved when these tissues were fixed in 20% buffered formalin, 70% ethanol, or Carnoy's solution. The binding of MAb HIK1083 and lectins was best preserved in tissue sections fixed with either 70% ethanol or Carnoy's solution, to judge from the staining intensity, although immunostaining with MAb HIK1083 produced some background staining when these fixation methods were used. Formalin-fixed tissue sections gave satisfactory results whether immunostaining with MAb HIK1083 or lectin staining was performed.
PCS, paradoxical concanavalin A staining; GSA-II, Griffonia simplicifolia agglutinin II; g, Golgi region.
-, tissues were negative; ±, only traces of the tissue were stained; +, less than one third of the tissue was stained; ++ and +++, more than one third or two thirds of the tissue was stained, respectively.
Trypsin digestion of formalin-fixed, paraffin-embedded tissues enhanced the intensity of the reactivity to MAb HIK1083, a 10-min period of digestion providing the most favorable results. The studies detailed below were carried out using materials fixed with 20% buffered formalin and with 10-min trypsin digestion.
HIK1083 and Lectin Binding in Non-neoplastic Tissues
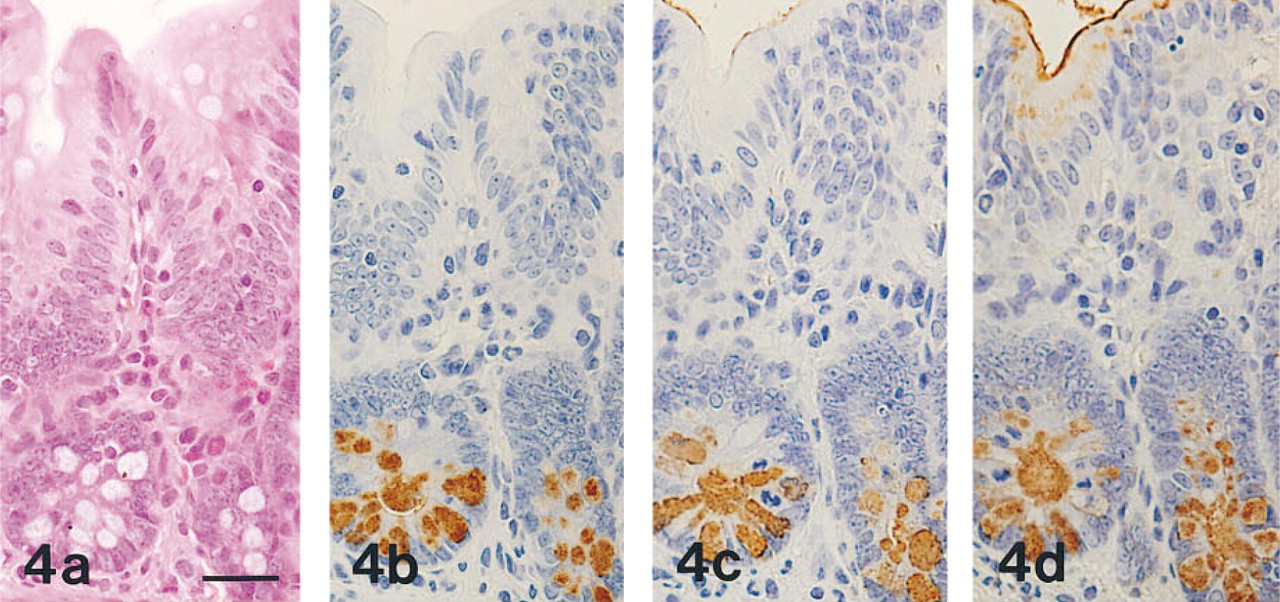
The results obtained in normal human and rat organs are summarized in Tables 1 and 2, respectively. MAb HIK1083-reactive sites were identical to those visualized with PCS. In human and rat organs, MAb HIK1083 and PCS both labeled mucins in gastric gland mucous cells (including cardiac gland cells, mucous neck cells, and pyloric gland cells) and in Brunner's gland cells (Figure 1). In human organs, periductal glands of the common bile duct and the main pancreatic duct contained mucous cells that were both immunostained with MAb HIK1083 and positive for PCS. Mucinous metaplasia of the pancreas and gastric metaplasia of the gallbladder consisted of PCS-positive and GOTS-positive cells. The former (the gastric gland mucous cell type of metaplastic cells) also showed reactivity for MAb HIK1083 and GSA-II (Figure 2) and the latter (the gastric surface mucous cell type of metaplastic cells) also stained with GSA-II (Figure 2).
Staining reactions with HIK1083, PCS and GSA-II in normal rat tissues a
aAbbreviations as in Table 1.
GSA-II, but not HIK 1083 or PCS, also labeled the supranuclear Golgi regions in a variety of epithelial cells, including gastric surface mucous cells (Figure 1), intestinal absorptive cells, and goblet cells (Figure 3).
In rat colon, basal secretory cells were positive with MAb HIK1083, PCS, and GSA-II (Figure 4).
The following tissues were negative with PCS, MAb HIK1083, and GSA-II: human and rat esophagus, rat salivary gland, and rat kidney.
HIK1083 and Lectin Binding in Carcinoma Tissues
All 20 cases of gastric carcinoma were positive for MAb HIK1083 and GSA-II, and 19 cases (95%) were positive for PCS. Of the 11 cases of pancreatic ductal carcinoma, nine cases (82%), eight cases (73%), and all 11 cases were positive for MAb HIK1083, PCS, and GSA-II, respectively. The two cases of pancreatic ductal carcinoma that were negative for MAb HIK1083 were also negative for PCS.
In HIK1083-positive and PCS-positive carcinoma tissues, the apical cytoplasm of the tumor cells and the extracellular mucins were both stained. The intracytoplasmic reaction took the form of coarse or fine droplets. HIK1083-reactive mucins appeared to be some-what more abundant than those stained by PCS (Figures 5 and 6). GSA-II also labeled these regions and, in addition, the paranuclear region of the tumor cells. Staining with HIK1083, PCS, and GSA-II differed quantitatively from case to case. GSA-II provided the most widespread staining, and the GSA-II-positive regions included the HIK1083- and PCS-reactive regions.
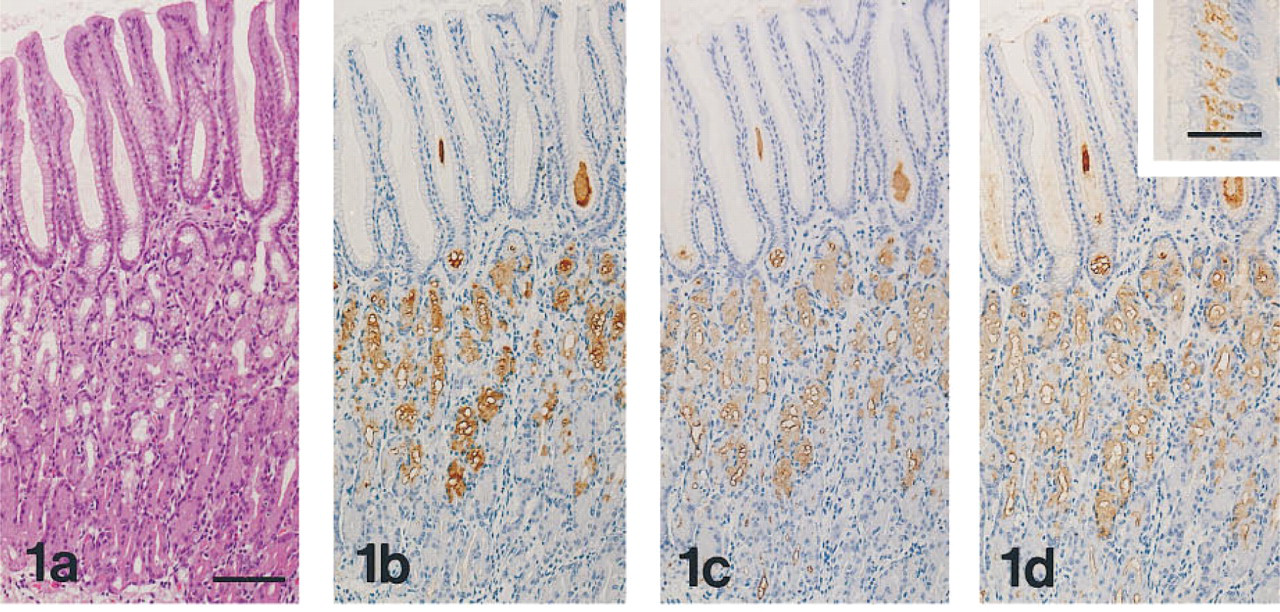
(a-d) Fundic mucosa of the human stomach prepared from serial sections. Sections are stained with H&E
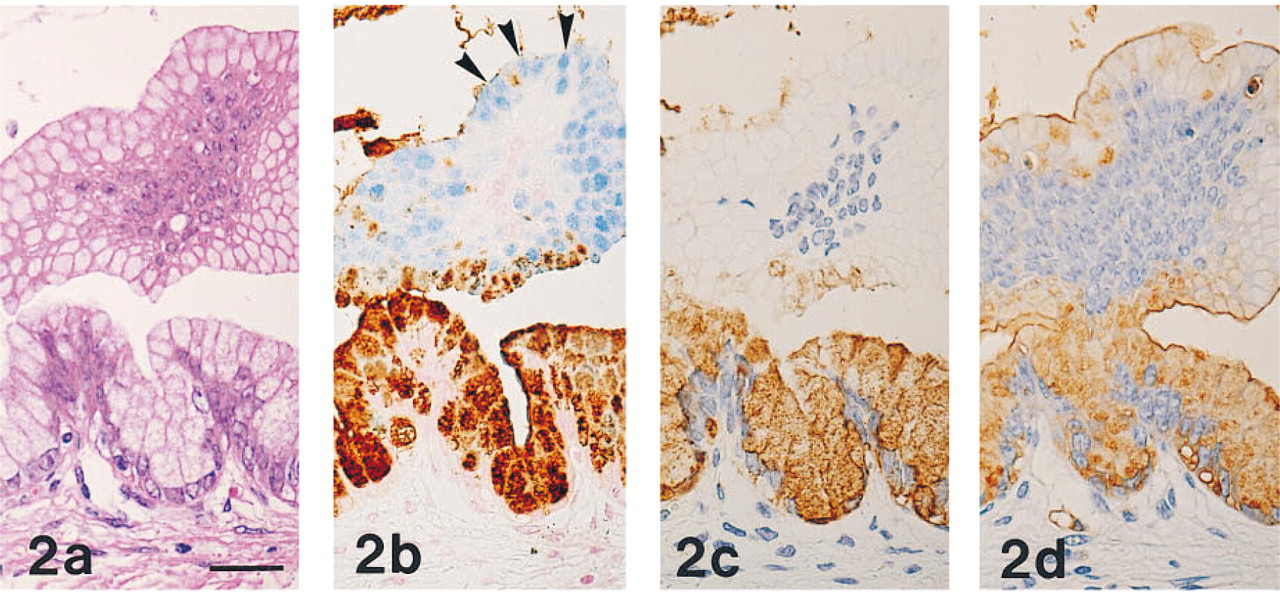
(a-d) Mucinous metaplasia of the human pancreatic duct prepared from serial sections.
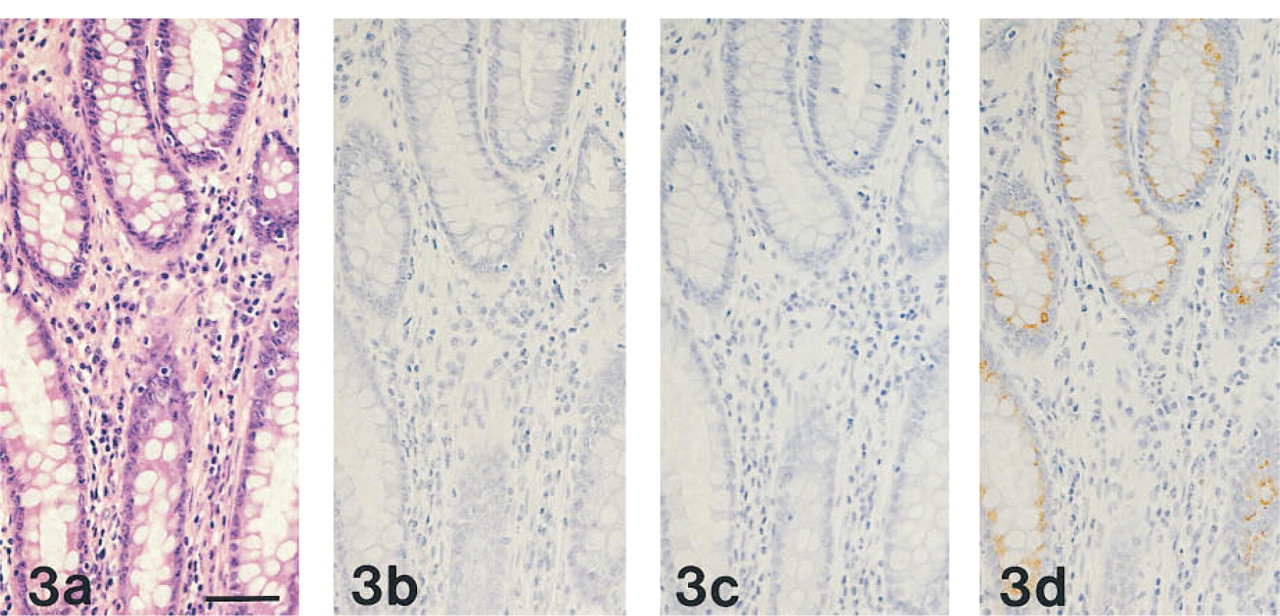
(a-d) Normal human colon, prepared from serial sections stained as in Figures 1a-1d, respectively. PCS-
All 20 cases of colon adenocarcinoma consistently exhibited reactivity for GSA-II but lacked reactivity for MAb HIK1083 and PCS (Figure 7).
Interference Between GSA-II and MAb HIK1083
Pretreatment with GSA-II prevented the binding of MAb HIK1083 to human mucous neck cells and to pyloric and Brunner's glands (Figures 8a and 8b), but pretreatment with MAb HIK 1083 did not prevent the subsequent binding of GSA-II to gastric and duodenal epithelial cells (Figures 8c and 8d).
Effect of Pretreatment on Staining
Oxidation and interposed reduction had no significant influence on the affinity for MAb HIK1083 and GSA-II shown by the gastric fundic mucosa. Without pretreatment, ConA labeled most mucous neck cells and some surface mucous cells weakly. Pretreatment involving PA oxidation for 10 min or 60 min, followed by NaBH4 reduction, increased the staining intensity of gastric glandular mucous cells with ConA (Class III ConA reactivity) but abolished ConA reactivity at other sites in the gastric mucosa, as reported previously (Katsuyama and Spicer 1978).
Pretreatment involving PA oxidation for 10 min or 60 min induced ConA reactivity in hepatocytes. This induced reactivity persisted after interposed NaBH4 reduction, as reported previously (Katsuyama and Spicer 1978). GSA-II also showed an affinity for hepatocytes. The reactivity of hepatocytes for GSA-II persisted after pretreatment involving PA oxidation for 10 min or PA oxidation for 10 min followed by NaBH4 reduction, but this reactivity was abolished by PA oxidation for 60 min followed by NaBH4 reduction. Pretreatment by amylase digestion completely abolished the PA oxidation-induced cytoplasmic reaction shown by hepatocytes on PCS or GSA-II-HRP staining, but did not influence HIK1083 reactivity. Hepatocytes were negative for MAb HIK1083 irrespective of the type of pretreatment used.
Discussion
In this study we confirmed that MAb HIK1083 showed an affinity for Class III ConA-reactive mucin not only in normal tissues but also in metaplastic and carcinoma tissues.
Although the biochemical and physiological processes affecting gastric mucins under various conditions have been extensively studied (Kawano et al. 1990; Komuro et al. 1992), little attention has been paid to the fact that there are two types of mucous cell (i.e., surface mucous cells and glandular mucous cells) in the gastric mucosa. In fact, the two types of mucins secreted by these two types of mucous cells exhibit considerably different histochemical properties, suggesting fundamental differences in their sugar moieties (Katsuyama and Spicer 1978; Suganuma et al. 1981; Katsuyama et al. 1985; Ota et al. 1991). As demonstrated earlier (Ota and Katsuyama 1992), these cells collaborate to form the surface mucous gel layer of the stomach, which is one of the most important agents protecting the gastric mucosa. Use of MAb HIK1083 would facilitate the study of these two types of mucin.
PCS requires pretreatment, consisting of PA oxidation and borohydride reduction. A slight change in the conditions used during such pretreatment might have a considerable influence on the results obtained. Immunostaining with HIK1083, on the other hand, is a simple method that allows both detection of Class III ConA-reactive mucin and characterization of the gastric phenotype expressed in a given metaplastic or carcinomatous lesion. Furthermore, this MAb could be easily applied to ultrastructural histochemistry.
(a-d) Normal rat colon, prepared from serial sections and stained as in Figures 1a-1d, respectively. Basal secretory cells of rat colon are positive with PCS
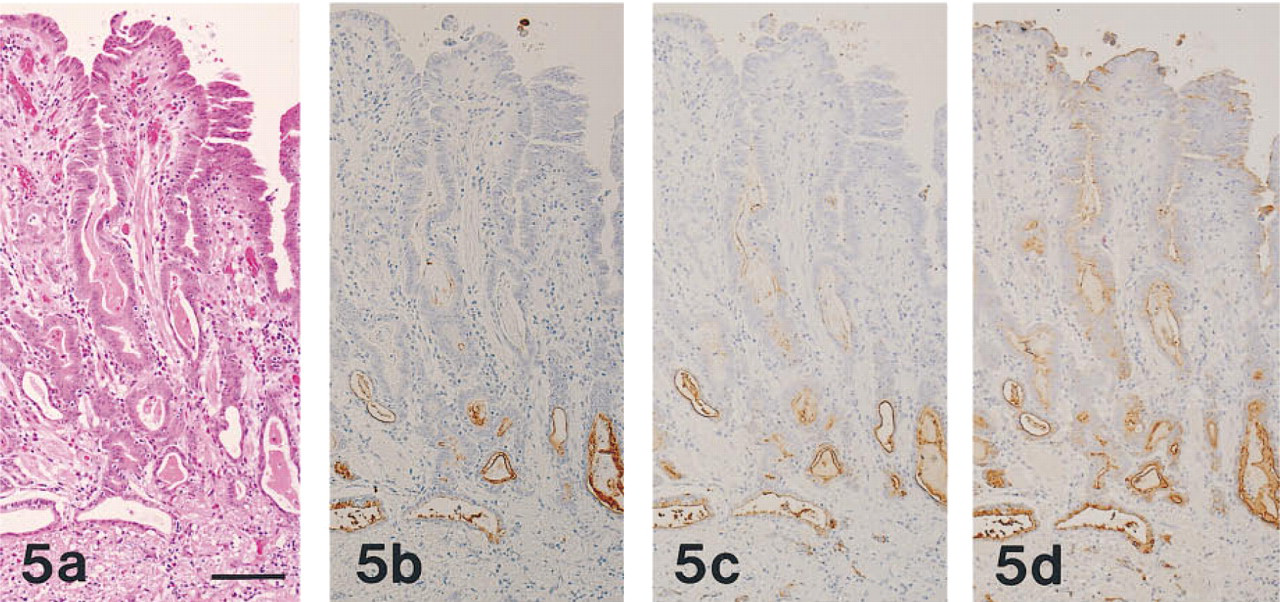
(a-d) Adenocarcinoma of the human stomach, prepared from serial sections stained as in Figures 1a-1d, respectively. The carcinoma tissue has the appearance of moderately differentiated adenocarcinoma
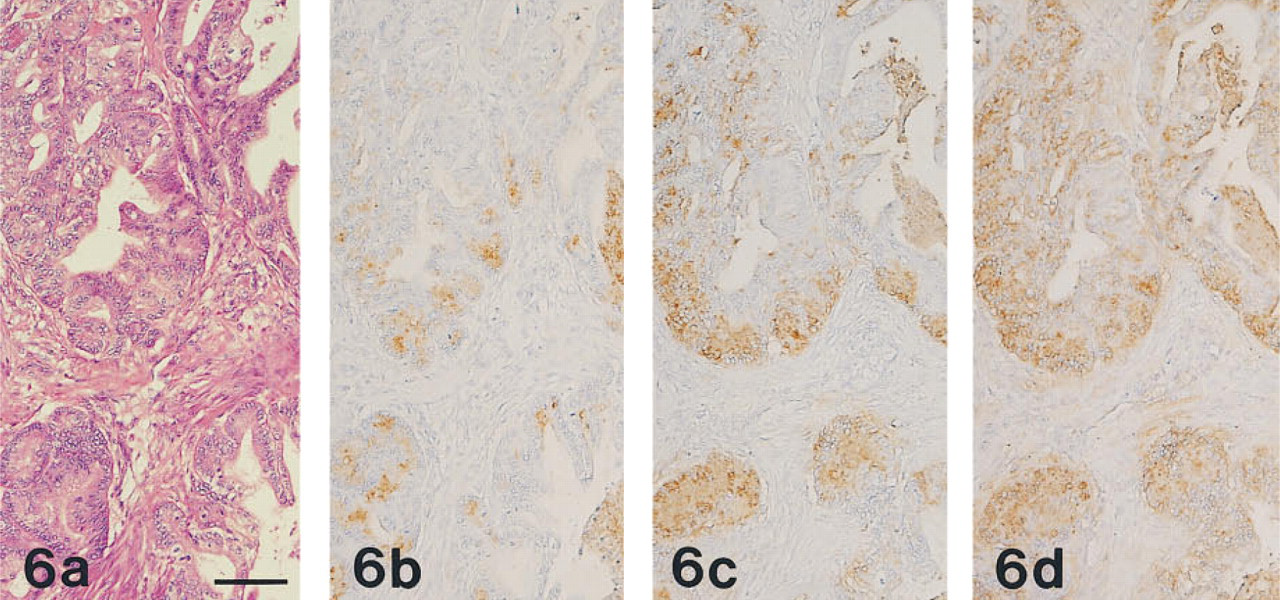
(a-d) Adenocarcinoma of the pancreas, prepared from serial sections stained as in Figures 1a-1d, respectively. The carcinoma tissue had the appearance of well-differentiated adenocarcinoma
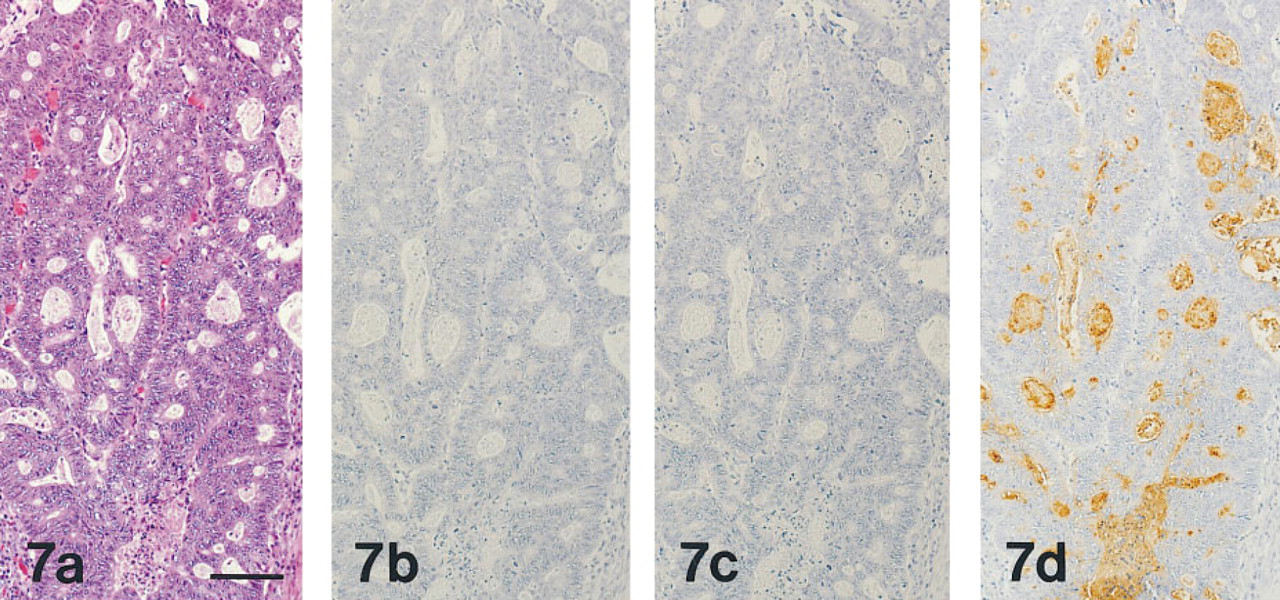
(a-d) Adenocarcinoma of the colon, prepared from serial sections stained as in Figures 1a-1d, respectively. The carcinoma tissues have the appearance of well-differentiated adenocarcinoma
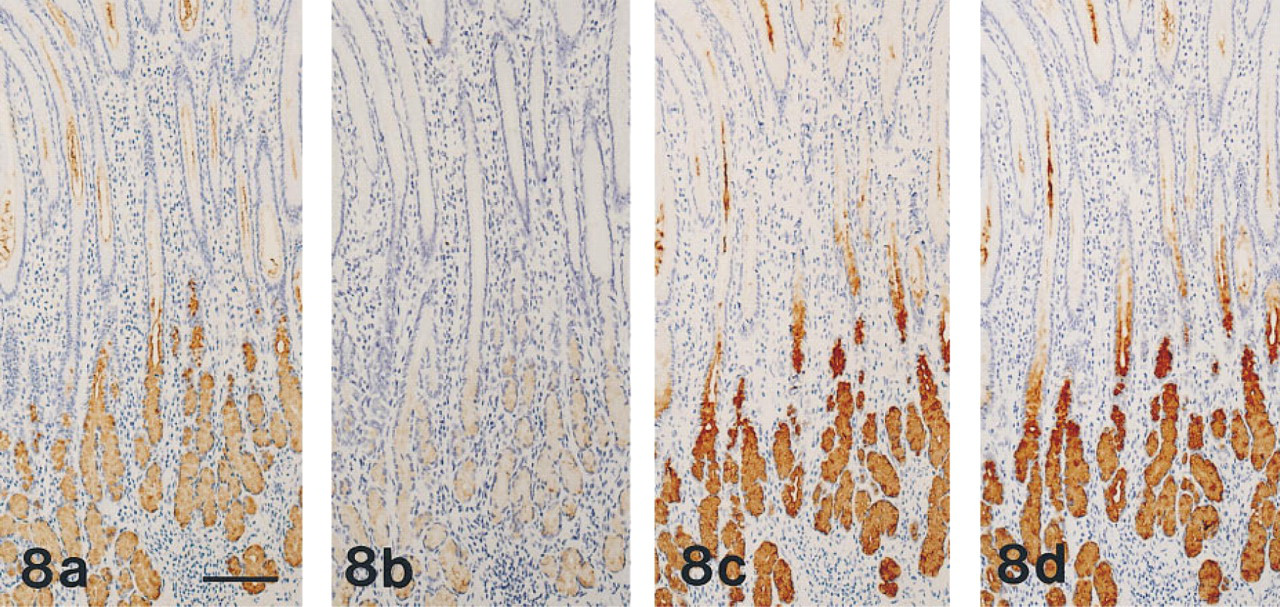
(a-d) Normal human pyloric mucosa, prepared from serial sections. Pyloric gland cells are positive with MAb HIK1083
Biochemical analysis suggests that the most likely candidate for the sugar residue responsible for Class III ConA reactivity is GlcNAc (Hotta et al. 1982). Those authors extracted mucin-type glycoproteins from human gastric mucins using a ConA-Sepharose column after oxidation and reduction of the type used in the PCS sequence. They suggested that GlcNAc residues at the nonreducing terminal of the oligosaccharides were responsible for the Class III ConA reactivity. This idea was based on the fact that, of the three specific sugar residues of ConA, mannose and glucose are absent from epithelial mucins. In line with this idea, Ishihara et al. (1996) had already established that the epitope recognized by MAb HIK1083 consisted of oligosaccharides containing peripheral α-GlcNAc residues. The present study provides further confirmation that the reactivities of mucins to MAb HIK1083 and GSA-II are stable despite the use of PA oxidation followed by NaBH4 reduction, as was Class III ConA-reactive mucin.
Two MAbs with specificities similar to that of MAb HIK1083 have been reported. Hanisch et al. (1993) introduced MAb 2B5, which reacts with terminal β-GlcNAc residues. MAb 2B5 was found to label glandular mucous cells of the human stomach, Brunner's gland cells, metaplastic cells of pancreas and gallbladder, and gastric differentiated carcinomas of the stomach, pancreas, and gallbladder. However, histochemical comparison between the specificity of MAb 2B5 and that of PCS has not been made. In addition, Hughes et al. (1994) and Hughes and Bhathal (1997) introduced MAb 5HL-5D11-D10 (D10). This showed the same reactivity as PCS in human normal and carcinoma tissues, although the reactivity of this MAb in rat tissues has not been investigated nor has its epitope structure been elucidated. The similar specificities shown by these two antibodies may indicate that Class III mucin contains more than one specific oligosaccharide.
In the present study, GSA-II prevented the subsequent binding of HIK1083 to mucous neck cells and to pyloric and Brunner's glands, but MAb HIK1083 did not prevent GSA-II binding. This result is consistent with biochemical reports showing that GSA-II binds to both α- and β-GlcNAc residues (Ishihara et al. 1996) and that the epitope recognized by MAb HIK1083 includes oligosaccharides containing peripheral α-GlcNAc residues (Ishihara et al. 1996).
Colon cancers and some gastric and pancreatic cancers showed GSA-II reactivity but not PCS or MAb HIK 1083 reactivity. In fact, GSA-II binding glycoproteins in human colon carcinoma have been reported to carry GlcNAc at their nonreducing terminals (Nakayama et al. 1985) and, because MAb HIK 1083 recognizes only α-GlcNAc residues and ConA preferentially binds to α-GlcNAc, our results suggest that colon carcinomas may carry β-GlcNAc residues.
GSA-II has been employed to label glandular mucous cell-type mucins of the stomach (Ihida et al. 1988). However, in the present study GSA-II also showed affinities for mucins in gastric surface mucous cells, mucins in the gastric surface mucous cell type of metaplastic cell of the pancreatic duct, colon adenocarcinoma cells, and the supranuclear region of various epithelial cells. To judge from these observations, GSA-II is not suitable for histochemical analysis of Class III ConA-reactive mucin and might be replaced by MAb HIK1083 for identification of glandular mucous celltype mucins.
MAb HIK1083 may have several potentially useful applications. First, histological detection of Class III mucin by this antibody should be useful both for pathological diagnosis and for investigating the histogenesis of adenocarcinomas expressing gastric phenotypes. Second, its application to the biochemical analysis and detection of Class III mucin may be useful for the analysis of gastric mucins. Finally, estimation of Class III mucin in serum or pancreatic juice could serve for the detection of precancerous or carcinomatous lesions.
Footnotes
Acknowledgements
We are indebted to the staff of Kanto Chemical Company for providing MAb HIK1083 and to Masanobu Momose (Central Clinical Laboratory, Shinshu University Hospital) for excellent technical assistance. We also gratefully acknowledge the encouragement given to us by Prof Kiyosawa (The Second Department of Internal Medicine, Shinshu University School of Medicine).