Abstract
Serotonin (5-hydroxytryptamine, 5-HT) mediates many functions of the central and peripheral nervous systems by its interaction with specific neuronal and glial receptors. Fourteen serotonin receptors belonging to seven families have been identified through physiological, pharmacological, and molecular cloning studies. Monoclonal antibodies (MAbs) specific for each of these receptor subtypes are needed to characterize their expression, distribution, and function in embryonic, adult, and pathological tissues. In this article we report the development and characterization of MAbs specific to the serotonin 5-HT2A receptor. To generate MAbs against 5-HT2AR, mice were immunized with the N-terminal domain of the receptor. The antigens were produced as glutathionine S-transferase (GST) fusion proteins in insect cells using a Baculovirus expression system. The hybridomas were initially screened by ELISA against the GST-5-HT2AR recombinant proteins and subsequently against GST control proteins to eliminate clones with unwanted reactivity. They were further tested by Western blotting against recombinant GST-5-HT2AR, rat and human brain lysate, and lysate from cell lines transfected with 5-HT2AR cDNA. One of the MAbs G186-1117, which recognizes a portion of the 5-HT2AR N-terminus, was selected for further characterization. G186-1117 reacted with a band of molecular size 55 kD corresponding to the predicted size of 5-HT2AR in lysates from rat brain and a 5-HT2AR-transfected cell line. Its specificity was further confirmed by adsorption of immunoreactivity with recombinant 5-HT2AR but not with recombinant 5-HT2BR and 5-HT2CR. Rat brain sections and Schwann cell cultures were immunohistochemically labeled with this MAb. G186-1117 showed differential staining in various regions of the rat brain, varying from regions with no staining to regions of intense reactivity. In particular, staining of cell bodies and dendrites of the pyramidal neurons in the cortex was observed, which is in agreement with observations of electrophysiological studies.
S
The 5-HT2 receptors (5-HT2Rs) are implicated in various normal physiological responses, e.g., smooth muscle contraction (Cohen and Wittenauer 1987), aldosterone production (Matsouka et al. 1985), and platelet aggregation (McBride et al. 1987). The 5-HT2 receptors are also involved in various mental disorders (Kalkman and Fozard 1991; Saxena 1995), including migraine headaches, anxiety, depression, schizophrenia, and obsessive-compulsive disorder. Accordingly, 5-HT2 receptors are expressed in diverse tissues such as brain (frontal cortex, nucleus accumbens, and tuberculum olfactorium), vascular smooth muscle, and platelets. These functions of 5-HT2Rs are mediated by the second messenger inositol trisphosphate in a G-protein-coupled pathway. Activation of 5-HT2 receptor also regulates the growth of vascular smooth muscle cells (Nemecek et al. 1978). This activation can be inhibited by ketanserin, a specific 5-HT2A receptor antagonist. Recent studies have shown that activation of the mitogen-activated protein kinase cascade (ERK1/ERK2) of the Ras pathway through the 5-HT2B receptor promotes cell growth, differentiation, and transformation (Launay et al. 1997). Similarly, it was also shown that activation of the Jak/STAT (signal transducers and activators of transcription) pathway through the 5-HT2A receptor upregulates expression of genes involved in myogenic differentiation (Guillet-Deniau et al. 1997).
Localization of these receptors is generally done using radioligand binding followed by autoradiography (Pazos et al. 1985; Mengod et al. 1990; Burnet et al. 1995). This approach is limited by the varying degrees of specificity of the ligands used (Peroutka 1988) and, more importantly, it lacks anatomic resolution. As mentioned above, at least three 5-HT2 receptor subtypes have been identified, designated as 5-HT2AR, 5-HT2BR, and 5-HT2CR. There is a significant amino acid sequence homology among these 5-HT receptors. The overlapping ligand binding profile of 5-HT to different 5-HTR subtypes makes it difficult to precisely locate a specific receptor. Previously, anti-peptide and anti-idiotypic polyclonal antibodies have been raised for localization studies of specific 5-HT2 receptor types (Garlow et al. 1993; Morilak et al. 1993, Backstrom et al. 1995; Choi and Maroteaux 1996). However, the polyclonal antibodies are limited in availability. This problem can be alleviated by the generation of MAbs that are highly specific for each particular receptor subtype. In this article we report the development and characterization of an MAb that is specific to the 5-HT2A receptor. This MAb will aid in the study of the expression and distribution of the 5-HT2A receptor in embryonic and normal adult tissues as well as in pathological tissues.
Materials and Methods
Generation and Purification of Recombinant GST Fusion Proteins as Immunogens
For the generation of immunogens, the cDNA fragments encoding the entire first extracellular domain in the N-terminus (amino acid residues 1-76 of 5-HT2AR, 1-56 of 5-HT2BR, and 1-55 of 5-HT2CR) were amplified from a human retina and human brain cDNA library by the polymerase chain reaction (PCR) using the THERMALASE Tbr Kit (Amresco; Solon, OH) and Biometra Trio-Thermoblock (Biometra; Tampa, FL). The forward primers (5-HT2AR, 5′-ccgggatccatggatattctttgtgaagaaaatact-3′; 5-HT2BR, 5′-cgcggatccaaaatggctctctcttacagagtgtctg-3′; and 5-HT2CR, 5′-cgcggatccaaaatggtgaacctgaggaatgcggtgc-3′) and reverse primers (5-HT2AR, 5′-cagcccgggtcaccagtttttttcctggagatgaag-3′; 5-HT2BR, 5′-ccggaattctgcccagtgcagtttatttcc-3′; and 5-HT2CR, 5′-ccggaattcccagttttgtaccccgtctgg-3′) were chosen from published human 5-HT2 receptor cDNA sequences (Julius et al. 1990; Saltzman et al. 1991; Schmuck et al. 1994). All primers were designed to clone the cDNA fragments in the reading frame of GST-tag of pAcG2T vector (PharMingen; San Diego, CA) using BamHI and SmaI/EcoRI cloning sites. For each cDNA, the condition for PCR thermocycling was 94C for 1 min, 58C for 1 min, 72C for 1 min, for 35 cycles. The amplified PCR fragment was purified from an agarose gel and cut with BamHI and SmaI (for 5-HT2AR) or BamHI and EcoRI (for 5-HT2BR and 5-HT2CR) and then ligated to the Baculovirus transfer vector pAcG2T with T4 DNA ligase (Gibco BRL; Gaithersburg, MD). Minipreps of plasmid DNA were purified by using a QIAwell 8 kit (QIAGEN; Chatsworth, CA). Recombinant Baculovirus transfer plasmids with the cloned 5-HT2 receptor fragments were co-transfected with linearized BaculoGold DNA (PharMingen) into Sf9 insect cells. For recombinant protein production (5-HT2AR, 5-HT2BR, and 5-HT2CR), insect cells were infected with recombinant Baculovirus particles at a multiplicity of infection (MOI) of 10, and the infected insect cells were harvested 3 days later. The cells were lysed in buffer containing Triton X-100 and the protein expression was analyzed by SDS-PAGE gels.
The purification of GST fusion proteins was carried out according to published protocols (Smith and Johnson 1988; Petralia et al. 1997). Briefly, the infected Sf9 cells were lysed with a buffer containing 10 mM Tris, pH 7.5, 130 mM NaCl, 1% Triton X-100, 10 mM NaF, 10 mM NaPi, pH 7.5, 10 mM NaPPi, pH 7.5, and protease inhibitor cocktail (800 μg/ml benzamidine-HCl, 500 μg/ml aprotinin, 500 μg/ml leupeptin, 500 μg/ml pepstatin A, 50 mM PMSF) on ice for 30 min. After centrifuging at 18,800 × g for 20 min, the supernatants were mixed with 0.1 volumes of a 50% slurry of glutathione Sepharose 4B (Pharmacia Biotech; Piscataway, NJ) and incubated for 30 min at 4C. After incubation, the Sepharose beads were spun down for 1 min at 1000 × g. The beads were washed five times with PBS and the fusion proteins were eluted using 10 mM of reduced glutathione in PBS. The purified GST fusion protein was dialyzed against PBS for 4 hr at 4C and analyzed on a SDS-PAGE gel to determine its purity.
Development of Monoclonal Antibodies to 5-HT2AR
Balb/c mice were immunized four times with 50 μg of GST-5-HT2AR fusion protein at 1-week intervals. Four days after the last immunization, the mice were sacrificed and the splenocytes were fused with HGPRT(-) F0 myeloma cells (Patton et al. 1982; Petralia et al. 1997) using polyethylene glycol according to the standard method (Galfre et al. 1977). The hybridomas were selected for by growing in hypoxanthin aminoptherin thymidine (HAT) medium. Supernatants from wells containing hybridomas were screened after 10 days by ELISA using the recombinant GST-5-HT2AR fusion protein. The specificity of positive clones was further confirmed by western blotting on either a GST-5-HT2AR fusion protein blot or a cell lysate from 5-HT2AR-transfected cells, as detailed below. The positive clones were subcloned twice by limiting dilution.
Western Blot Analysis
Rat brain synaptic membrane was a kind gift of Dr. Doug Vetter (Salk Institute; La Jolla, CA). Human brain lysate (a brain medley from a 68-year-old white male who had died of cardiac arrest) was purchased from Clontech Laboratories (Palo Alto, CA). Lysate from NIH-3T3 cell stably transfected with rat 5-HT2AR (Wang et al. 1993) was prepared by harvesting confluent layer of cells in Petri dishes, washing three times in PBS, and lysing in the lysis buffer as described above. Protein concentration was determined by the Lowry assay (Lowry et al. 1951). Equal amounts (about 20 μg/lane) of various protein samples were resolved by SDS-PAGE and electroblotted using a Bio-Rad minigel transfer system (Bio-Rad Laboratories; Cambridge, MA) onto Immobilon P membranes (Millipore; Bedford, MA), using a current of 0.5-0.75 A for 1 hr. The blots were blocked for 1 hr with 5% Carnation nonfat dry milk in TBST (25 mM Tris-Cl, pH 8.0; 125 mM NaCl; 0.1% Tween-20) and then incubated with primary antibodies in 5% milk/TBST overnight at 4C. The primary antibody was either used as a purified MAb at a standard concentration of 2 μg/ml or as a hybridoma tissue culture supernatant diluted 1:4 with 5% Carnation nonfat dry milk in TBST. An IgG1 isotype-matched negative control antibody (PharMingen) was also used. After incubation with the primary antibody, the blots were washed five times in TBST and then incubated with a secondary antibody conjugated to horseradish peroxidase (HRP, 1:3000 dilution; PharMingen) for 45 min at room temperature (RT). After five washes with TBST, the blots were developed for 2 min using a Chemiluminescence kit (SuperSignal CL-HRP Substrate System; Pierce, Rockford, IL). Hyperfilm-ECL films (Amersham Life Sciences; Arlington Heights, IL) were exposed to the blots to visualize the chemiluminescence signal corresponding to specific antibody/antigen reaction.
Absorption of Antibodies with Recombinant 5-HT2R Proteins
To determine the specificity of the MAbs, the antibodies were preadsorbed with the recombinant 5-HT2R proteins. Briefly, 200 μg of either GST-5-HT2AR fusion protein or negative control GST-5-HT2BR or GST-5-HT2CR fusion protein was incubated with 100 μg of MAb and 200 μl of glutathione-agarose beads for 2 hr at 4C. The beads were pelleted at 1000 × g for 3 min. The supernatant containing adsorbed or unadsorbed antibodies was tested by Western blotting and immunocytochemical analysis.
Immunostaining of Cultured Schwann Cells
Schwann cells were isolated from neonatal Sprague-Dawley rat sciatic nerves and purified by depletion of fibroblasts using complement protein, according to a modified protocol (Brockes et al. 1979). Identity of cultured cells was verified by routine immunoblotting for S-100b, a Schwann cell marker, as previously described (Mirsky and Jessen 1990; Gould et al. 1992; Yoder et al. 1996). Subconfluent monolayers of cells were washed with warm Hank's balanced salt solution (HBSS) and fixed with 4% paraformaldehyde (PFA) in PBS for 20 min on ice. The excess fixative was removed by washing with PBS, plasma membranes were permeabilized with 0.05% of Triton X-100, and the nonspecific aldehyde sites were blocked by washing with PBS containing 0.05 M glycine. Cells were then placed in a blocking buffer (3% normal goat serum and 1% bovine serum albumin fraction V in PBS-glycine) for 30 min to block nonspecific protein binding sites. The blocking buffer was diluted 1:3 in PBS-glycine to prepare a working strength buffer. Cells were washed with working strength buffer and then incubated either with one of the anti-5-HT2AR hybridoma culture supernatants or with a negative control supernatant (from unfused myeloma culture), each diluted 1:5 in working strength buffer. The cells were incubated either for 2 hr at RT or overnight at 4C. After several washes, the cells were incubated with FITC-conjugated goat anti-mouse IgG (Jackson ImmunoResearch Labs; West Grove, PA) at a dilution of 1:75 at RT for 1 hr. After another series of washes, the slides were mounted using gelvatol anti-fade medium and the immunolabeling was examined with a Bio-Rad MRC 1000 confocal microscope. As a negative control, the primary antibody incubation step was omitted for some samples. The rest of the Schwann cells were plated and assayed in parallel for calcium imaging experiments. Eighty to 90% of these cells responded to 1 μM 5-HT application during calcium imaging experiments withFluo3-AM (Molecular Probes; Eugene, OR). These responses have been previously characterized as being mediated by 5-HT2A receptors on the basis of transductional and pharmacological data (Yoder et al. 1996). In addition, 5-HT2A receptor message in these cells was confirmed by Northern blot analysis and RT-PCR (Gaietta et al., unpublished observation).
Immunohistochemical Staining of Rat Brain Sections
Adult Sprague-Dawley rats were anesthetized with sodium pentobarbital and perfused transcardially with 50 ml PBS and 250 ml 2% PFA plus 0.2% parabenzoquinone in 0.075 M phosphate buffer. The rat brain tissues were removed, postfixed for 2 hr in the same fixation buffer, and then cryoprotected in 30% sucrose in 0.1 M phosphate buffer overnight. Forty-μm sections were cut with a sliding microtome and stored in PBS until required for immunohistochemistry. Free-floating sections were washed for 10 min in Tris-buffered saline (TBS), incubated for 20 min in TBS containing 0.25% Triton X-100, and blocked for 60 min in TBS + 2% BSA. Staining was performed by incubating sections in 2 μg/ml anti-5-HT2A receptor antibody, G186-1117, in TBS + 0.25% Triton X-100 + 2% BSA for 40 hr at 4C. An IgG1 isotype-matched negative control antibody (PharMingen) was also used. After washing unbound antibodies with TBS + 0.25% Triton X-100, the sections were incubated with 1.5 μg/ml biotinylated donkey anti-mouse antibodies (Jackson ImmunoResearch Labs) in TBS + 0.25% Triton X-100 × 2% BSA for 2 hr at RT. The sections were then washed and incubated with an avidin-biotin-peroxidase reagent (1:200 dilution; DAKO, Carpinteria, CA) for 60 min at RT. After washing, the sections were incubated with freshly prepared diaminobenzidine (DAB) solution (DAB kit; Vector Labs, Burlingame, CA) for 5 min. DAB-stained sections were rinsed three times in PBS and mounted on microscope slides, dehydrated in four changes of 100% alcohol, cleared with four changes of xylene, and mounted with a coverslip.
Immunofluorescence Staining for Confocal Microscopy of Rat Brain Sections
Adult rat brain sections were perfused and cut as described above. G186-1117, an antibody that was directly conjugated with FITC (Molecular Probes) was used for the staining. After several washes and blocking with 2% BSA in TBS + 0.25% Triton X-100, the sections were incubated with 2 μg/ml of the FITC-G186-1117 antibodies for 40 hr at 4C. The sections were then washed extensively in TBS + 0.25% Triton X-100 and mounted in a glycerol/PBS mounting medium. A confocal laser scanning microscope was used to examine the staining, utilizing an immersion lens (X40) and a numerical aperture. For each tissue, focal series of 24 sections each, 0.5 mm apart, were scanned by the confocal laser microscope. The images were reconstructed and modified in Adobe Photoshop software (Adobe Systems; San Jose, CA) and printed on Fujifilm Pictro paper.
Results
Cloning and Expression of Recombinant Proteins
The cDNA fragments corresponding to the N-terminal extracellular domain of 5-HT2A, 5-HT2B, and 5-HT2C receptors were amplified by PCR from a human retina (5-HT2AR) or brain cDNA library (5-HT2BR and 5-HT2CR) and cloned into pAcG2T Baculovirus expression vector. The authenticity of the PCR products was confirmed by nucleotide sequencing (using Sequenase Version 2.0 DNA Sequencing Kit; USB, Cleveland, OH). They were also confirmed to be in frame with the GST coding sequence. The fusion proteins from the insect cell lysate were purified using glutathione-Sepharose beads. The purified proteins were analyzed on SDS-PAGE followed by Coomassie Blue staining to confirm the correct molecular size. These proteins were used to immunize mice, to screen the hybridomas, and to test the specificity of anti-5-HT2AR antibodies by Western blot analysis.
Generation of Hybridomas and Selection of Monoclonal Antibodies
One thousand hybridoma culture supernatants were tested for antibodies to recombinant 5-HT2AR fusion protein (GST-5-HT2AR) by ELISA. Twenty of 200 ELISA-positive hybridomas were further tested by Western blotting using recombinant GST-5-HT2AR protein. Seven of these hybridoma wells gave strong staining with the immunogen. These hybridomas were cloned by limiting dilution and subsequently subcloned to select stable antibody-secreting clones. Four clones that reacted with the N-terminal extracellular portion of the 5-HT2A receptor were selected for further characterization. On the basis of the specificity and intensity of reaction by immunohistochemistry and Western blotting, only one of these four clones, G186-1117, was selected for further characterization.
Characterization of the Specificity of MAb G186-1117 by Western Blot Analyses
We have tested the crossreactivity of the G186-1117 antibody against recombinant 5-HT2B and 5-HT2C receptor proteins. The 5-HT2AR shares approximately 27% sequence homology with the N-terminal extracellular domain of the 5-HT2B and 5-HT2C receptors. To test the crossreactivity of G186-1117 with 5-HT2BR and 5-HT2CR, equal amounts of recombinant GST-5-HT2AR, GST-5-HT2BR, and GST-5-HT2CR were separated on SDS-PAGE gels and analyzed by Western blot. As shown in Figure 1, G186-1117 reacted only with recombinant 5-HT2AR and not with 5-HT2BR and 5-HT2CR.
Specificity of G186-1117 to the full-length 5-HT2A receptor was also tested by Western blot analysis using protein preparations from rat brain, human brain, and a 5-HT2AR cDNA-transfected cell line. G186-1117 detected an immunoreactive band of about 55 kD, the predicted size, in rat brain lysate (Figure 2A).
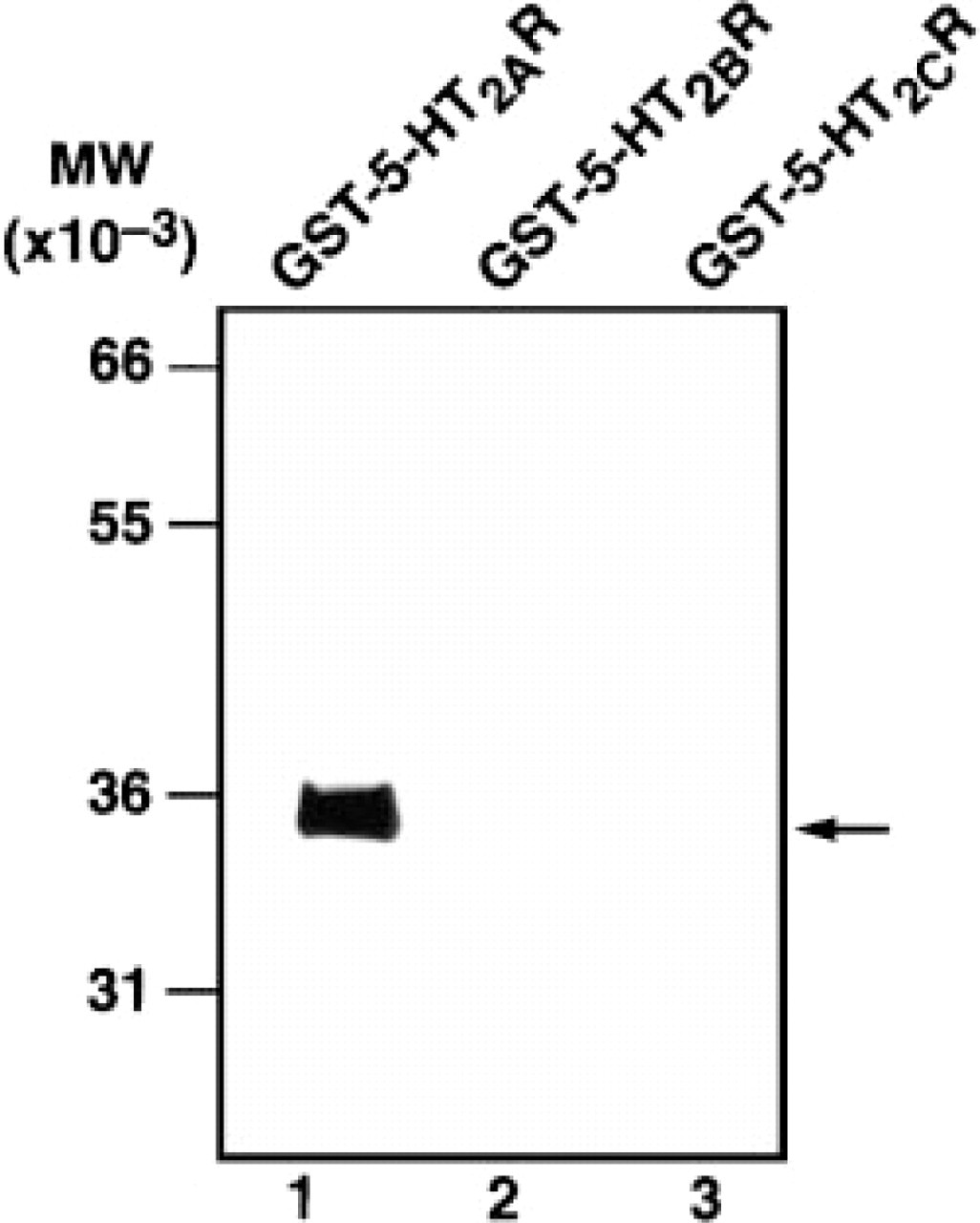
Western blot analysis of G186-1117 against recombinant 5-HT2 receptor proteins. Approximately 50 ng of the N-terminal extracellular domain of 5-HT2AR (Lane 1), 5-HT2BR (Lane 2), and 5-HT2CR (Lane 3) GST fusion proteins were subjected to SDS-PAGE and transferred to an Immobilon membrane. The filters were first incubated with primary antibody and immunoreactivity of G186-1117 was detected with horseradish peroxidase-labeled rabbit antimouse immunoglobulins and enhanced chemiluminiscence. G186-1117 reacted only with GST-5-HT2AR protein.
A smear of immunoreactive proteins of sizes ranging from 40 to 55 kD was observed in human brain lysate (Figure 2B). G186-1117 detected an immunoreactive band of about 55 kD in lysates from NIH-3T3 cells that were transfected with 5-HT2AR cDNA (Figure 2C). GST-5-HT2AR recombinant protein entirely depleted the immunoreactivity of G186-1117 against the positive bands in the purified rat brain synaptic membrane protein preparation, in the human brain lysate as well as in the 5-HT2AR-transfected cell lysates, whereas recombinant GST-5-HT2BR and GST-5-HT2CR failed to adsorb anti-5-HT2AR activity (Figures 2A-2C).
Immunocytochemical Staining of Cultured Schwann Cells
Schwann cells that had been isolated from neonatal Sprague-Dawley rat sciatic nerves (Yoder et al. 1996) were used to test the ability of MAb G186-1117 to stain endogenously expressed 5-HT2AR. As described in Materials and Methods, 5-HT responsiveness was assessed using calcium imaging. G186-1117 immunolabeling was observed in the plasma membrane and cytoplasm but not in the nucleus (Figure 3). No staining was seen when cells were stained without the G186-1117 (data not shown).
Immunohistochemical Staining of Adult Rat Brain Sections with MAb G186-1117
Immunostaining observed with this antibody appeared to be specific (Figure 4B) and could not be reproduced when similar sections were processed with an isotype control (Figure 4A) or with G186-1117 preadsorbed with recombinant protein GST-5-HT2AR (data not shown). The immunoreactivity of G186-1117 could not be blocked by the GST-5-HT2BR and GST-5-HT2CR recombinant proteins (data not shown). An immunohistochemical evaluation was carried out on brain sections from four different animals using G186-1117. Our analysis was not intended to serve as a thorough description of the immunohistochemical labeling seen with this antibody throughout the entire CNS but was aimed at comparing our data with the previously published data of in situ hybridization (Pomperiano et al. 1994), immunohistochemistry using a polyclonal antibody (Morilak et al. 1993), radioligand binding (Pazos et al. 1985), and electrophysiological observations (Andrade and Nicoll 1987; Araneda and Andrade 1991).
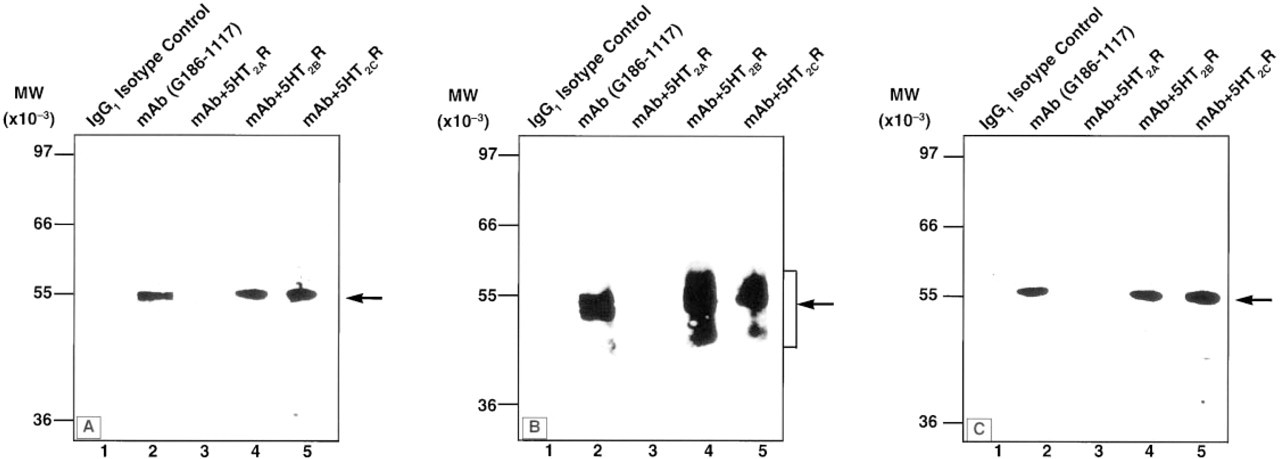
Western blot analysis of 5-HT2AR in rat and human brain and of a transfected cell line. Approximately 20 μg of rat brain synaptic plasma membrane
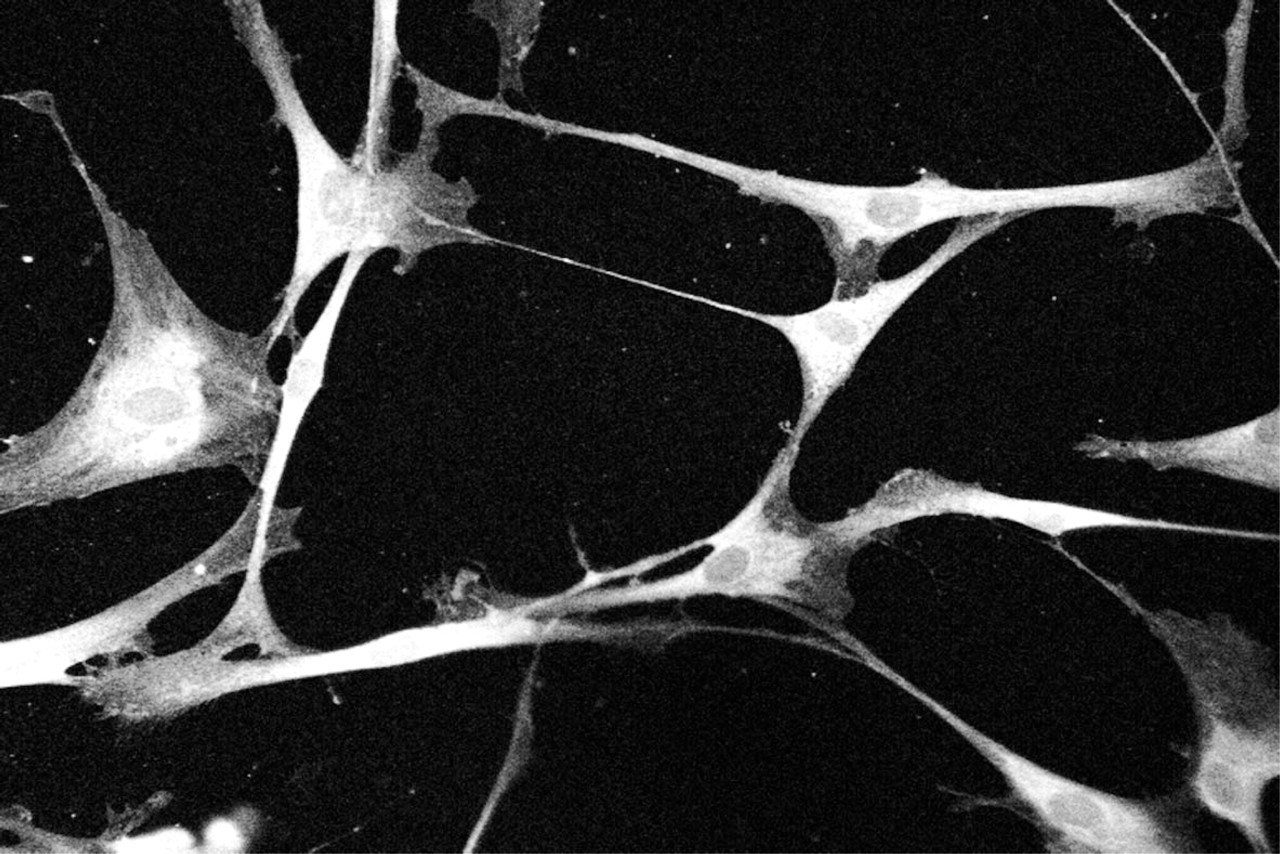
Immunofluorescence staining of rat Schwann cells with anti-5-HT2AR MAb. The monolayer of rat Schwann cells was stained with G186-1117 MAb as described in Materials and Methods and was examined with a Bio-Rad MRC 1000 confocal microscope. Membrane and cytoplasmic staining can be observed. The cytoplasmic staining may be due to the synthesis and/or internalization of the receptor protein in the cytoplasm.
In the main olfactory bulb, intense 5-HT2AR immunoreactivity was observed in cells in the mitral cell layer and in fibers in the external plexiform layer and glomerular layer (Figure 4D). Moderate to strong staining was also seen in most cortical regions. Cortical labeling appeared to be primarily associated with neurons in Layer V and with their proximal dendrites that ramified in cortical Layers II and III (Figure 4C). Staining was almost absent from cortical Layer I and was much less intense in Layers IV and VI. Robust 5-HT2AR immunoreactivity was noted in the piriform cortex (Figure 4E), olfactory tubercle, and ventral pallidum (Figure 4F), but was absent from the claustrum and endopiriform nucleus (Figure 4E). In agreement with previous in situ hybridization studies, staining for the 5-HT2AR was also undetectable throughout the septum (data not shown). Within the hippocampal formation, moderate 5-HT2AR immunostaining was seen in CA1 pyramidal cells and their dendrites in stratum raditum (Figure 4H) and in CA3 pyramidal cells and their dendrites in stratum lucidum (Figure 4G). Some staining was also noted in the dentate gyrus, just above the granule cell layer, although it was not possible to specifically determine the cell structure that stained.
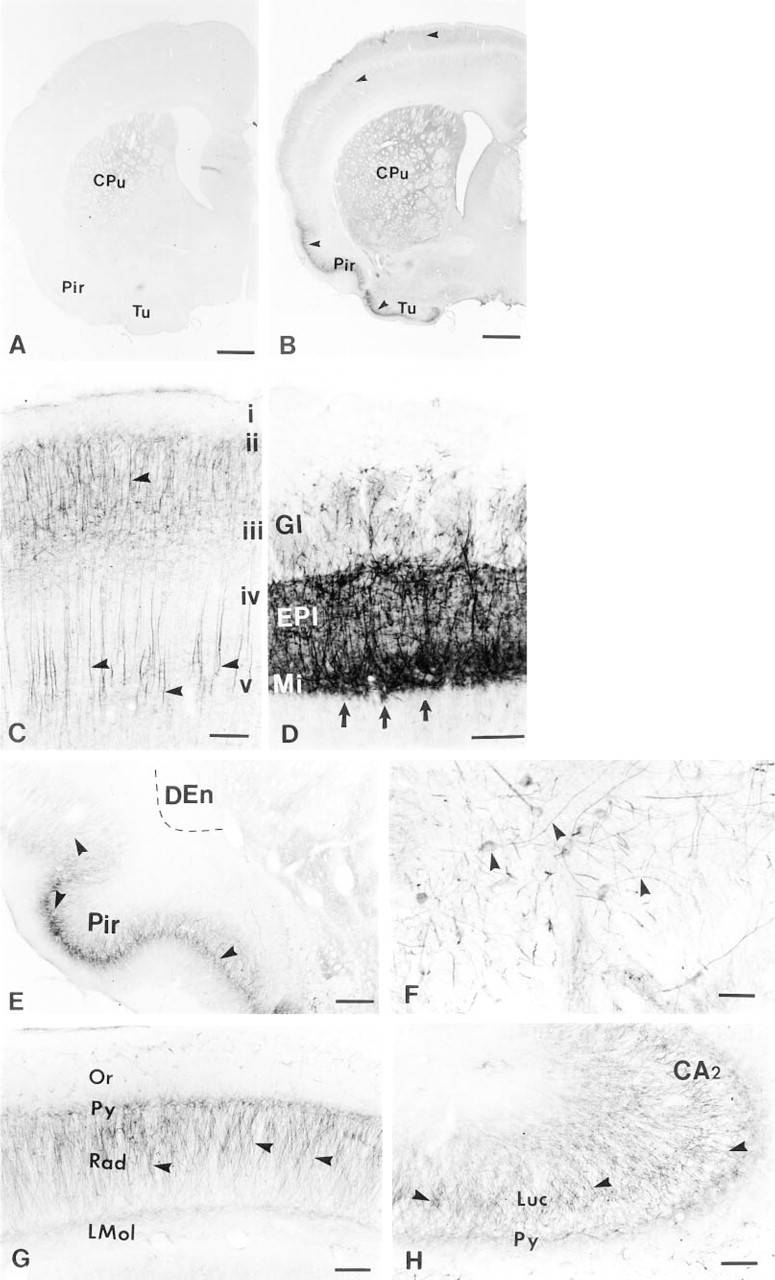
Immunohistochemical staining of adult rat brain sections with G186-1117 antibody. Coronal section of frontal rat brain incubated with an isotype control antibody
In the medial mammillary nucleus, where intense mRNA for the 5-HT2AR has been reported, very little immunostaining was observed (Figure 5A). Similarly, in the pontine nuclei, where high levels of mRNA for the 5-HT2AR have been found, 5-HT2AR immunoreactivity was absent (Figure 5B). We cannot explain the discrepancy between the 5-HT2AR mRNA and the protein level in the mammilary nucleus. However, the absence of 5-HT2AR immunoreactivity in the pontine nuclei is in agreement with radioligand binding data (Pazos et al. 1985), even when there is a high level of mRNA (Pomperiano et al. 1994). Moderate immunostaining was observed in many cells of the red nucleus (Figure 5C), and in the facial nucleus robust 5-HT2AR labeling was observed in almost all cells (Figures 5E and 5F). Intense 5-HT2AR immunoreactivity was also observed in the Purkinje cells and their dendrites throughout the cerebellum (Figure 5D).
Immunofluorescence Staining of 5-HT2A Receptors on the Dendrites of Pyramidal Cells with MAb G186-1117
A typical subcellular localization of 5-HT2A receptor with G186-1117 was seen in the dendrites of pyramidal cells in the frontal cortex of rat brain (Figures 6A-6C). A series of optical sections through the apical dendrites of several neurons showed that most of the immunofluorescence staining was associated with the plasma membrane but some with cytoplasm. Optical sections through the dendrites showed that the staining of the dendrites was on the surface (Figure 6D). Therefore, staining appeared as a hollow tube (Figures 6E and 6F and Figures 6G-6I, arrows). This type of plasma membrane staining was also shown in fusing myoblasts by Guillet-Deniau et al. (1997).
Discussion
A large number of studies have been published on the distribution and localization of the 5-HT receptor by using radioligand binding followed by autoradiography, in situ hybridization, or electrophysiology (Pazos et al. 1985; Peroutka 1988; Mengod et al. 1990; Burnet et al. 1995). However, previously reported antibodies against 5-HT receptors are limited in numbers. These antibodies include anti-peptide rabbit polyclonal antibodies recognizing 5-HT2AR (Garlow et al. 1993; Morilak et al. 1993,1994), 5-HT2BR (Choi and Maroteaux 1996), 5-HT2CR (Abramowski et al. 1995; Backstrom et al. 1995), 5-HT1AR (el Mestikawy et al. 1990; Azmitia et al. 1992; Miquel et al. 1992; Matthiessen et al. 1993; Raymond et al. 1993; Gerard et al. 1994; Hillion et al. 1994; Sundaram and Strange 1994; Verdot et al. 1994,1995; Kia et al. 1996a,b), 5-HT1BR (Grimaldi et al. 1995), 5-HT1DR (Langlois et al. 1995), 5-HT3R (Kia et al. 1995; Morales et al. 1996; Mukerji et al. 1996), and 5-HT6R (Gerard et al. 1997) as well as an anti-idiotypic antibody (TH8, generated by immunizing rabbits with affinity-purified antibodies to serotonin) that recognizes 5-HT1B, 5-HT2A, and 5-HT2C receptor subtypes (Tamir et al. 1991; Ridet et al. 1994). In the present article we have described the development and characterization of an MAb that reacts specifically with the 5-HT2A receptor. A highly specific MAb G186-1117, was obtained from a hybridoma fusion of mouse immunized with a GST fusion protein containing N-terminal extracellular domain (amino acid residues 1-76) of the human 5-HT2A receptor.
We chose to use the N-terminal domain of the 5-HT2A receptor as immunogen because at this region, 5-HT2A, 5-HT2B, and 5-HT2C receptors have low homology (less than 30%) at the amino acid sequence level. In addition, there is 75% homology among human, rat, and mouse 5-HT2A receptors at this same region. This approach had a greater chance of generating 5-HT2AR-specific MAbs that would crossreact with human, rat, and mouse 5-HT2A receptors.
The specificity of G186-1117 to 5-HT2AR was examined by Western blot analysis. G186-1117 reacted with GST-5-HT2AR but not with GST-5-HT2BR or 5-HT2CR (Figure 1). The size of the band on Western blots of rat brain lysate (Figure 2A) and lysates from the NIH-3T3 cells transfected with 5-HT2AR cDNA (Figure 2C) was about 55 kD, which is similar to the value of 56 kD that was detected by the anti-idiotypic polyclonal antibody (Tamir et al. 1991). The Western blot data using GST fusion proteins and membrane preparation from the rat brain plus the cell lysate suggest that G186-1117 reacts specifically with 5-HT2AR protein. Lysates from human brain gave a smear of molecular size ranging from 40 to 55 kD (Figure 2B). Because there is very high homology between the rat and the human 5-HT2AR gene, the 55-kD band may represent the full-length 5-HT2AR protein, as we observed. We suspect that the large smear seen in the human brain lysate may be due to protein degradation because such tissues are often obtained several hours post mortem. Garlow et al. (1993) also obtained a smear reflecting similar degradation. However, their antibody reacted with a band whose size was only 42 kD. The discrepancy in size may be due to different degrees of tissue degradation. The immunoreactivity of G186-1117 can be competitively inhibited by preincubating the MAb with GST-5-HT2AR fusion protein but not with 5-HT2BR and 5-HT2CR fusion protein (Figures 2A-2C), lending further support to the specificity of the MAb.
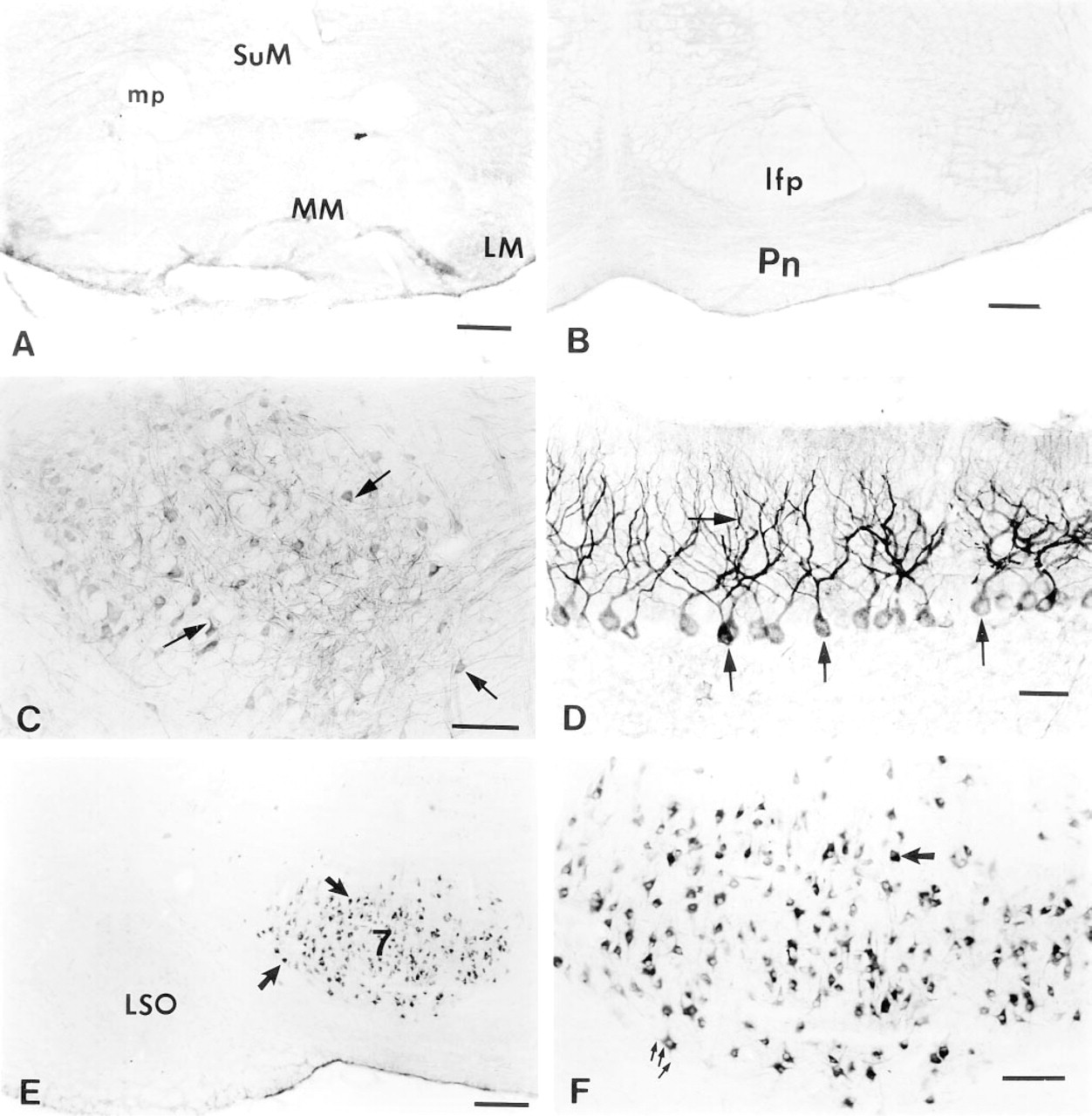
Immunohistochemical staining of adult rat brain sections with G186-1117 antibody (continued). Immunostaining with G186-1117 failed to reveal specific labeling in some brain regions where high levels of 5-HT2AR mRNA have been reported including medial mammillary nuclei
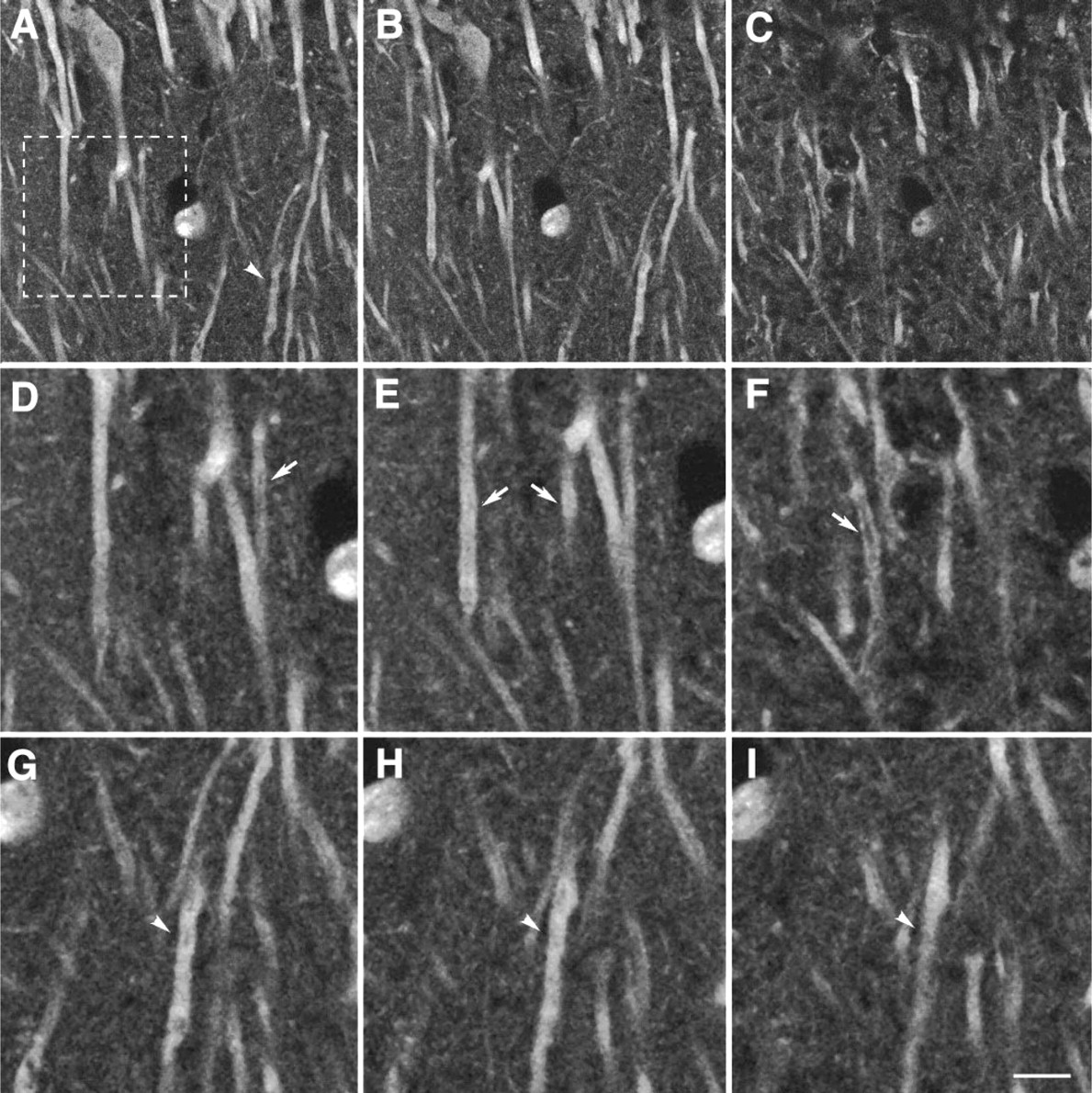
Immunofluorescence staining of 5-HT2A receptors on the dendrites of pyramidal cells by confocal laser scanning microscopy. Coronal section of frontal rat brain was stained with FITC-conjugated MAb G186-1117 and the tissue scanned by confocal microscopy. (
The specificity of G186-1117 was further examined by immunocytochemistry on cells expressing transfected and endogenous 5-HT2A receptors. Cultured rat Schwann cells that naturally express functional 5-HT2A receptors were also labeled with G186-1117 (Figure 3). These cells had previously been shown to label with an anti-idiotypic antibody (TH8) that recognizes 5-HT1B, 5-HT2A, and 5-HT2C receptors (Yoder et al. 1996). The staining pattern observed with G186-1117 was very similar to that observed with TH8. This is a particularly good model for demonstrating MAb G186-1117 reactivity with 5-HT2AR because these cells have previously been shown to express 5-HT2A receptors (Yoder et al. 1996,1997a,b).
G186-1117 was found to stain brain tissues that were processed by different preservation protocols including frozen tissue, paraffin-embedded tissue, and perfused tissue (data not shown). For the perfused tissue, we have also used two different fixation buffers, including 2% paraformaldehyde plus 0.2% parabenzoquinone in 0.075 M phosphate buffer and 4% paraformaldehyde alone in 0.075 M phosphate buffer. These fixation protocols gave different staining patterns with different tissue preservation. In the present study, fixation with 2% paraformaldehyde plus 0.2% parabenzoquinone in 0.075 M phosphate buffer by perfusion gave the best results, which are shown in Figures 4 and 5. Although the distribution of 5-HT2AR protein that we observed was largely in agreement with the distribution of mRNA reported in previous in situ hybridization studies (Pomperiano et al. 1994; Wright et al., 1995), several unique differences were noted. The immunohistochemical staining using G186-1117 clearly revealed prominent 5-HT2AR staining in many brain regions where high levels of mRNA have been found. These include the mitral cell layer of the olfactory bulb, piriform cortex, red nucleus, and the facial nucleus. The staining pattern in the olfactory bulb and in the piriform cortex is consistent with the observations of Morilak et al. (1993). However, the anti-5-HT2AR polyclonal antibody used in their study failed to label the facial nucleus, whereas this was labeled with G186-1117. The other differences include the intense staining of Purkinje cells of the cerebellum by G186-1117. There are conflicting results about expression of the 5-HT2AR in cerebellum. Localization of the 5-HT2AR in cerebellum had not been observed with the polyclonal antiserum or by radioligand binding and in situ hybridization. However, Maura et al. (1988) have shown that endogenous release of glutamate in the cerebellum was inhibited by the 5-HT2 receptor agonist DOI, which was reversed by the selective 5-HT2 receptor antagonist ketanserin. Through the use of 5-HT and various agonists and antagonists, the expression of 5-HT2AR has also been demonstrated in Purkinje cells (Sugihara et al. 1995), which is in agreement with the staining results with our MAb. We do not presently know the reason for this discrepancy. We have also observed moderate staining of cell bodies and dendrites of the pyramidal neurons in the hippocampus, which is in agreement with observations of electrophysiological (Andrade and Nicoll 1987; Araneda and Andrade 1991) and in situ studies (Burnet et al. 1995). However, this staining was not observed with the polyclonal antiserum (Morilak et al. 1993). Recently, using the MAb G186-1117 together with two other anti-5-HT2AR polyclonal antibodies, Willins et al. (1997) were able to show the expression of 5-HT2AR in the pyramidal cells and interneurons in the rat cortex. Therefore, it is likely that the polyclonal antiserum used by Morilak et al. (1993) may not have been sensitive enough. Some regions of the brain in which high levels of 5-HT2AR mRNA have been identified did not stain with our MAb. The reason for this result is unclear. It is possible that in this region there is no translation of the message, the protein is rapidly degraded, or the protein is not stored in sufficient amounts to permit its detection by immunohistochemical staining techniques (such as the medial mammillary nuclei and pontine nuclei). The absence of 5-HT2AR immunoreactivity in the pontine nuclei, where a high level of mRNA for the 5-HT2AR was found (Pomperiano et al. 1994), is in agreement with radioligand binding data (Pazos et al. 1985). It is important to point out that the present investigation was not intended to serve as a comprehensive description of 5-HT2A receptor staining in the rat CNS. Additional and more detailed studies will be required to elucidate the distribution of 5-HT2A receptor protein throughout the rat CNS. However, the immunoreactivity seen with G186-1117 is somewhat consistent with earlier studies using in situ hybridization, radioligand binding, and immunostaining with polyclonal antisera.
The subcellular localization of 5-HT2AR with G186-1117 suggests that most of the labeling is associated with the plasma membrane (Figure 6). In the present study, rat brain sections were stained for G186-1117 and images were analyzed by confocal laser scanning microscopy. These staining results are consistent with observations made by Willins et al. (1997) and Guillet-Deniau et al. (1997) using our MAb. As mentioned previously, Willins et al. have compared three antibodies raised against 5-HT2AR to show the localization of this receptor in pyramidal cells and interneurons in the rat cortex. Two of these were rabbit antisera raised against epitopes, amino acids 22–41 (Roth et al. 1995) and amino acids 330-345 (Backstrom and Sanders-Bush, unpublished data). The staining pattern of these two polyclonal antibodies was identical with the staining pattern obtained with MAb G186-1117. Immunoreactivity in the pyramidal cell dendrites was associated with the plasma membrane. However, a significant portion of 5-HT2AR staining was also observed in the cytoplasmic vesicles in the cell body. Plasma membrane staining of 5-HT2AR using G186-1117 has also been reported by Guillet-Deniau et al. (1997), showing that 5-HT2AR was localized on the plasma membrane of fusing myoblasts. We believe that the cytoplasmic staining for 5-HT2AR seen in the Schwann cells (Figure 3) and in the pyramidal cells (Figure 6) may be due to a higher level of intracellular synthesis of the receptor protein in the rough ER, to accumulation in Golgi for secondary processing, or to internalization of the receptor in the cytoplasm. Previous studies using differential centrifugation followed by radioligand binding of different subcellular fractions have suggested the localization of 5-HT2A receptors in cytoplasmic and mitochondrial extracts (Laubeya et al. 1986). This cytoplasmic pattern of staining is also observed for various other neurotransmitter receptors, including GluR1, AMPA glutamate and mGluR1 receptors (Molnár et al. 1993; Kharazia et al. 1996; Petralia et al. 1997).
In conclusion, we have developed and extensively characterized monoclonal antibodies to the 5-HT2A receptor. We have selected one MAb, G186-1117, that is specific for the 5-HT2A receptor, as defined by its immunoreactivity pattern in various tissues, its reactivity with a protein of expected molecular size, and by selective adsorption using recombinant 5-HT2AR but not 5-HT2BR and 5-HT2CR proteins. We anticipate that G186-1117 will be a useful tool for studying the anatomic distribution, regulation, and function of the 5-HT2A receptor.
Footnotes
Acknowledgements
Supported in part by an SBIR grant from NIMH (Grant #1R43MH54437-1 to SKS) and grants from the NIH: NS14718, NS24635, RR04050 (to MHE), and an Overseas Research Student Award from the CVCP, UK (#ORS/96027038 to CW). SK is supported by the Wellcome Trust, UK.
We thank Dr Chun-Ming Huang for his support of this project. We are grateful to Dr Maryann E. Martone for valuble assistance with the confocal laser microscopy and for her critical reading of the manuscript. We thank Dr L. Descarries and Virginia Cornea-Hebert for their helpful discussions. We would also like to thank Ling-Pu Dong for his help in rat brain section preparation.
