Abstract
Gastric gland mucous cells produce class III mucin, which is also found in Brunner's glands and mucous glands along the pancreaticobiliary tract, and in metaplasia and adenocarcinomas differentiating towards gastric mucosa. Recently, we showed that class III mucin possesses GlcNAcα1→4Galβ→R, formed by α1,4-N-acetylglucosaminyltransferase (α4GnT). Examining the tissue-specific expression of mucin epitopes is useful to clarify cell-lineage differentiation and to identify the site of origin of metastatic carcinomas in histological specimens. Formalin-fixed, paraffin-embedded tissue sections from esophagus, stomach, colon, liver, pancreas, lung, kidney, prostate, breast, and salivary gland resected for carcinoma, as well as salivary gland adenoma, colon adenoma, and metastatic adenocarcinoma of lymph nodes from stomach, pancreas, colon, and breast, were immunostained for MUC6, α4GnT, and GlcNAcα1→4Galβ→R. These were all expressed in normal, metaplastic, and adenocarcinoma tissues of stomach, pancreas, and bile duct, and in pulmonary mucinous bronchioloalveolar carcinomas. Cells expressing α4GnT uniformly expressed GlcNAcα1→4Galβ→R. Only MUC6 was expressed in normal salivary glands, pancreas, seminal vesicles, renal tubules, and colon adenomas, and in normal tissue and adenocarcinomas of prostate and breast. No tissues showed immunoreactivity for α4GnT alone. Immunohistochemistry (IHC) profiles were similar for metastatic carcinomas and primary carcinoma tissues. The IHC profiles for MUC6, α4GnT, and GlcNAcα1→4Galβ→R may be diagnostically relevant.
I
Several years ago, Ishihara et al. (1996) raised a specific monoclonal antibody (MAb) against gastric mucin, designated HIK1083, which recognizes N-acetyl-glucosaminα1→4galactoseβ→R (GlcNAcα1→4Galβ→R). We found that the distribution of GlcNAcα1→4Galβ→R is consistent with the distribution of class III mucin in normal vertebrate tissues and in metaplastic and neoplastic human tissues (Nakamura et al. 1998; Ota et al. 1998,2001). More recently, we showed that class III mucin possesses this particular glycan, GlcNAcα1→4Galβ→R, which is formed by α1,4-N-acetylglucosaminyltransferase (α4GnT) (Nakayama et al. 1999), and that two distinct mucin core proteins, MUC5AC and MUC6, present in gastric mucin carry GlcNAcα1→4Galβ→R (Zhang et al. 2001).
Immunohistochemical expressions of MUC6, a4GnT, and GlcNAc α1→4Galβ→R in non-neoplastic tissues a
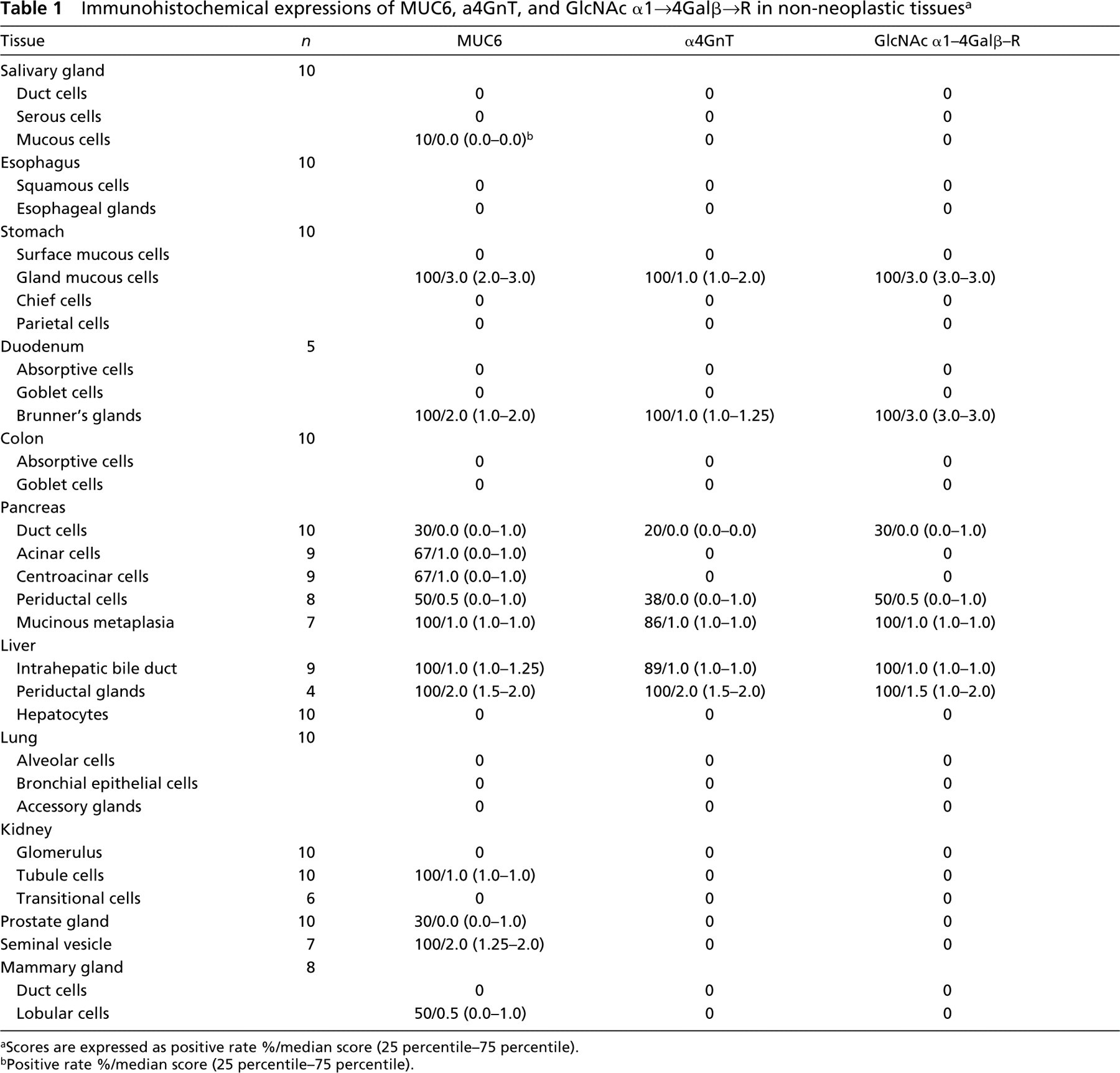
aScores are expressed as positive rate %/median score (25 percentile–75 percentile).
bPositive rate %/median score (25 percentile–75 percentile).
Examining tissue-specific expression of mucin epitopes in human tissues is a useful way to clarify the cell-lineage differentiation of carcinoma cells and to demonstrate the site of origin of metastatic carcinomas in histological specimens.
GlcNAcα1→4Galβ→R is preferentially attached to core2-branched O-glycan (Ishihara et al. 1996). The sialyl Lewis X found in O-glycan are also attached to the terminal end of core2-branched structures (Fukuda 1996). Therefore, it appears that the expressions of GlcNAcα1→4Galβ→R and sialyl Lewis X may be-reciprocally regulated, because these carbohydrates compete for the common precursor oligosaccharide, core2-branched O-glycan. It is well known that sialyl Lewis X serve as preferential ligands for the cell-adhesion molecules E- and P-selectin (Fukushima et al. 1984; Itzkowitz et al. 1988; Rosen and Bertozzi 1994). Recently we demonstrated that, in colorectal and pulmonary cancers, the sialyl Lewis X expressed on core2-branched O-glycans showed a positive correlation with tumor progression (Shimodaira et al. 1997; Machida et al. 2001).
We designed the present study to explore the IHC expressions of MUC6, α4GnT, and GlcNAcα1→4Galβ→R in human normal, metaplastic, and adenocarcinoma tissues. We also examined the IHC expressions of GlcNAcα1→4Galβ→R and sialyl Lewis X in gastric carcinomas and in metastatic gastric carcinomas of lymph nodes.
Immunohistochemical expression of MUC6, α4GnT, GlcNAc α1–4Gal β-R, and sialyl Lewis X in neoplastic tissues at primary and metastatic lesions a
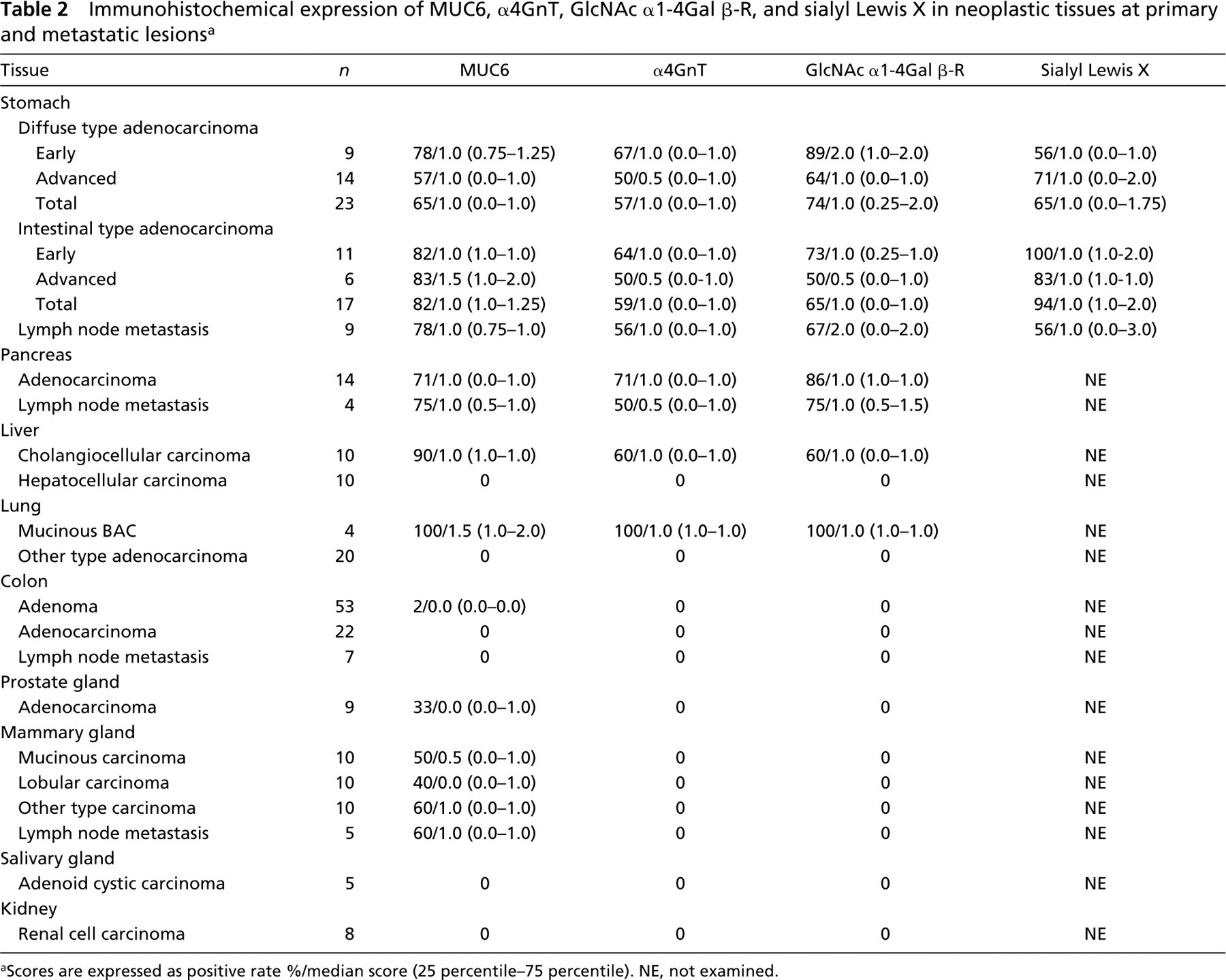
aScores are expressed as positive rate %/median score (25 percentile–75 percentile). NE, not examined.
Materials and Methods
Preparation of Tissues
Formalin-fixed, paraffin-embedded sections of human tissues were obtained from the surgical pathology files of the Department of Clinical Laboratory, Shinshu University Hospital, Matsumoto, Japan. Specimens consisted of histologically normal portions of tissues resected for adenoma or carcinoma and various tumor tissues, as well as metastatic adenocarcinomas of lymph nodes from stomach, colon, pancreas, and breast (Tables 1 and 2). This study was performed after written informed consent had been obtained from the patients.
Histochemistry
Serial paraffin sections of 3-μm thickness were stained with H&E for histological examination or immunostained with anti-MUC6 (Novocastra; Newcastle-upon-Tyne, UK), anti-α4GnT (Zhang et al. 2001), or GlcNAcα1→4Galβ→R (Mab HIK1083; Kanto Chemical, Tokyo, Japan). To facilitate comparison of the distributions of antigens, mirror paraffin sections of 3-μm thickness from gastric adenocarcinomas and metastatic adenocarcinomas of lymph nodes from gastric adenocarcinomas expressing both GlcNAcα1→4Galβ→R and sialyl Lewis X in primary sites were stained with HIK1083 or NCC-ST439 MAb (Kumamoto et al. 1998) (Nippon Kayaku; Tokyo, Japan) specific for the sialyl Lewis X attached to O-glycan.
(
(
(
IHC staining for MUC6 in normal prostate gland, prepared from serial sections. Some prostate gland cells show diffuse cytoplasmic expression of MUC6. H&E staining (
IHC staining for MUC6 in normal mammary gland, prepared from serial sections. Some cells of the terminal duct lobular unit of mammary gland show diffuse cytoplasmic expression of MUC6. H&E staining (
IHC staining was performed using the Envision+ method (DAKO; Carpinteria, CA). Briefly, sections were dewaxed and rehydrated and endogenous peroxidase activity was blocked with 0.3% H2O2 in methanol (30 min). Before immunostaining, antigen retrieval was carried out using a microwave (600 W) for 25 min in 0.01 mol/liter citrate buffer (pH 6.0) for both GlcNAcα1→4Galβ→R and MUC6. For immunostaining with anti-α4GnT and sialyl Lewis X, antigen retrieval was not carried out. The tissue sections were blocked with 5% normal bovine serum albumin in Tris-buffered saline (TBS; 140 mmol/liter NaCl, 50 mmol/liter Tris-HCl, pH 7.6) and incubated with primary antibodies. After washing in TBS, slides were incubated with peroxidase and second antibody-labeled polymer (DAKO) for 60 min. The reaction was developed with 3,3′-diaminobenzidine (Sigma Chemical; Poole, UK) containig 0.02% H2O2. For immunostaining of seminal vesicles, the Envision+ method for immunoalkaline phosphatase (DAKO) was used. Sections were lightly counterstained with hematoxylin, dehydrated, cleared in xylene, and mounted in synthetic medium.
Negative controls were obtained by omitting the primary antibody. The gastric gland mucous cells and Brunner's gland cells in the specimens were used as internal positive controls for MUC6, anti-α4GnT, and GlcNAcα1→4Galβ→R. Colon adenocarcinoma tissues were used as positive controls for sialyl Lewis X.
Evaluation of Immunostaining
The degree of staining in tissues examined with specific antibodies was scored semiquantitatively as 0 (negative), 1 (less than one third of the tissue), 2 (more than one third but less than two thirds), or 3 (more than two thirds). Grading of immunoreactivity was carried out by a single observer (KN). To validate the grading method, all specimens were graded twice, on two separate occasions. There was no significant intraobserver variation.
Statistics
The Mann–Whitney U-test was used to compare the scores given for immunoreactivities. Spearman's correlation coefficient by rank was used to analyze the correlations among the immunoreactivity scores given for MUC6, α4GnT, GlcNAcα1→4Galβ→R, and sialyl Lewis X. Staining scores are nonparametric and are presented as median rather than mean values.
Results
Demonstration of MUC6, α4GnT, and GlcNAcα1→4Galβ→R in Non-neoplastic Tissues
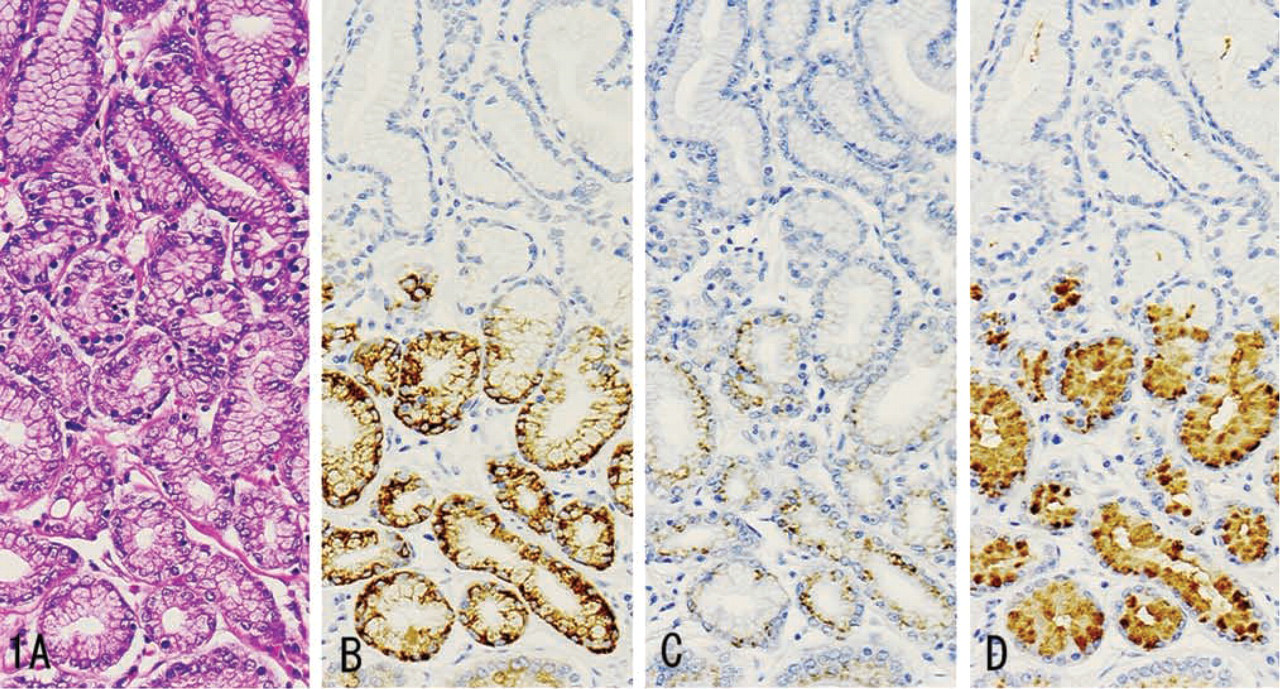
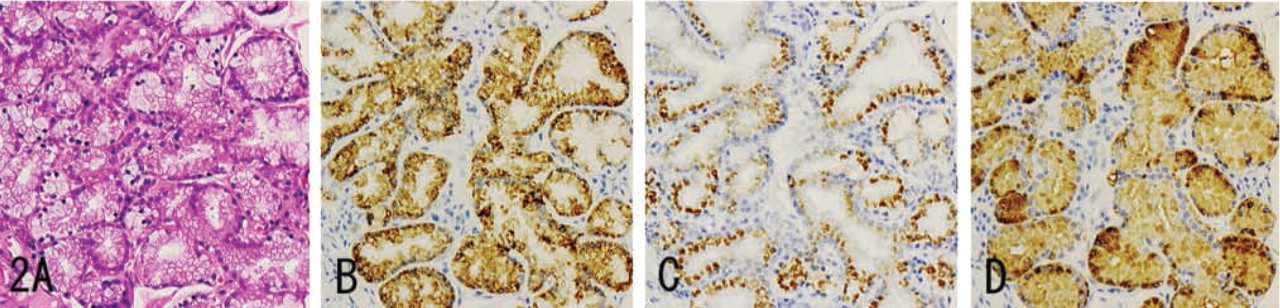
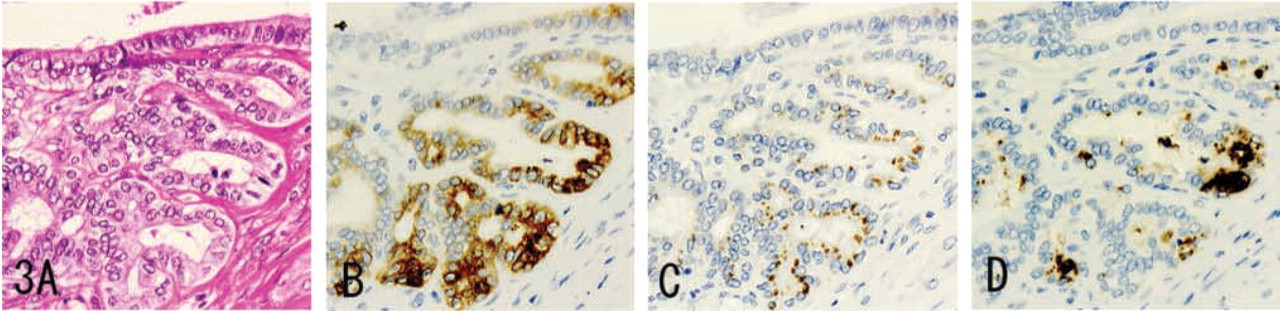
MUC6 was diffusely expressed in the cytoplasm (Figures 1B, 2B, 3B, 4B, and 5B), while α4GnT was expressed in the Golgi area (Figures 1C, 2C, and 3C). GlcNAcα1→4Galβ→R was mainly localized in cytoplasmic mucus granules (Figures 1D, 2D, and 3D), although in some cells it was localized on the apical cytoplasmic membrane.
MUC6, α4GnT, and GlcNAcα1→4Galβ→R were present in discrete cell types to various degrees: gastric gland mucous cells (Figure 1) (cardiac gland cells, mucous neck cells, and pyloric gland cells), Brunner's gland cells (Figure 2), mucous cells of the periductal glands of the pancreaticobiliary tracts (Figure 3), biliary tract epithelial cells, and mucous cells of mucinous metaplasia of the pancreatic ducts. In these tissues, α4GnT-positive cells always exhibited immunoreactivity for GlcNAcα1→4Galβ→R, as described previously (Zhang et al. 2001).
(
(
(
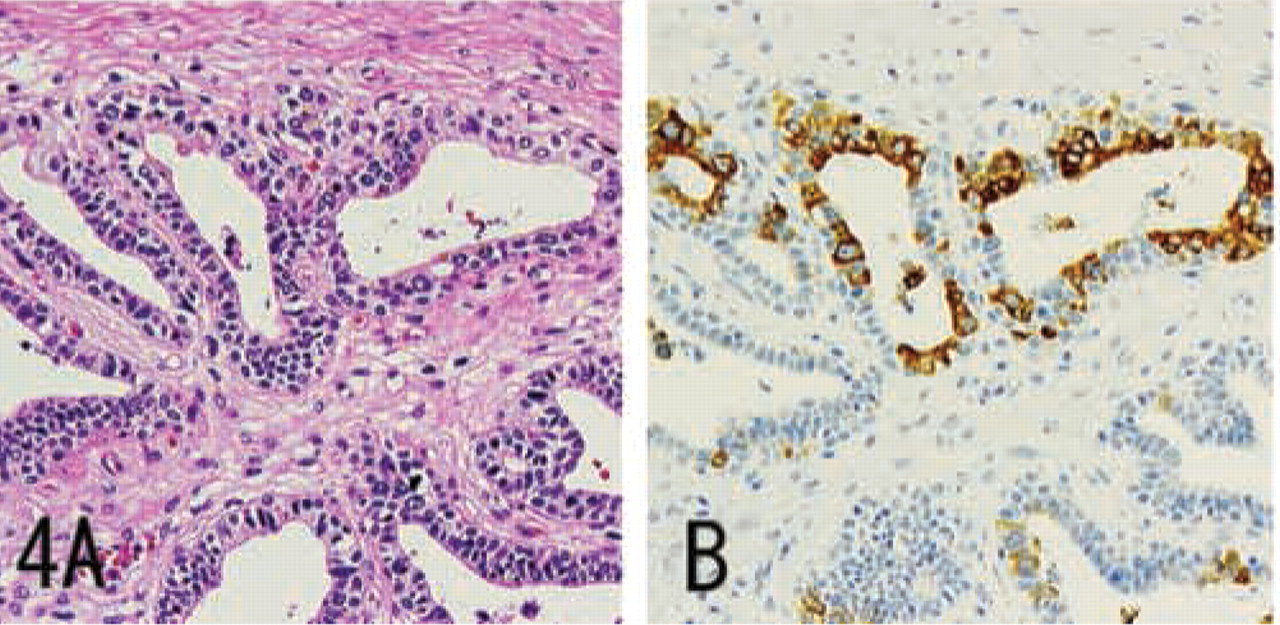
IHC staining for MUC6 in prostatic adenocarcinoma, prepared from serial sections. Some carcinoma cells show diffuse cytoplasmic expression of MUC6. H&E staining (
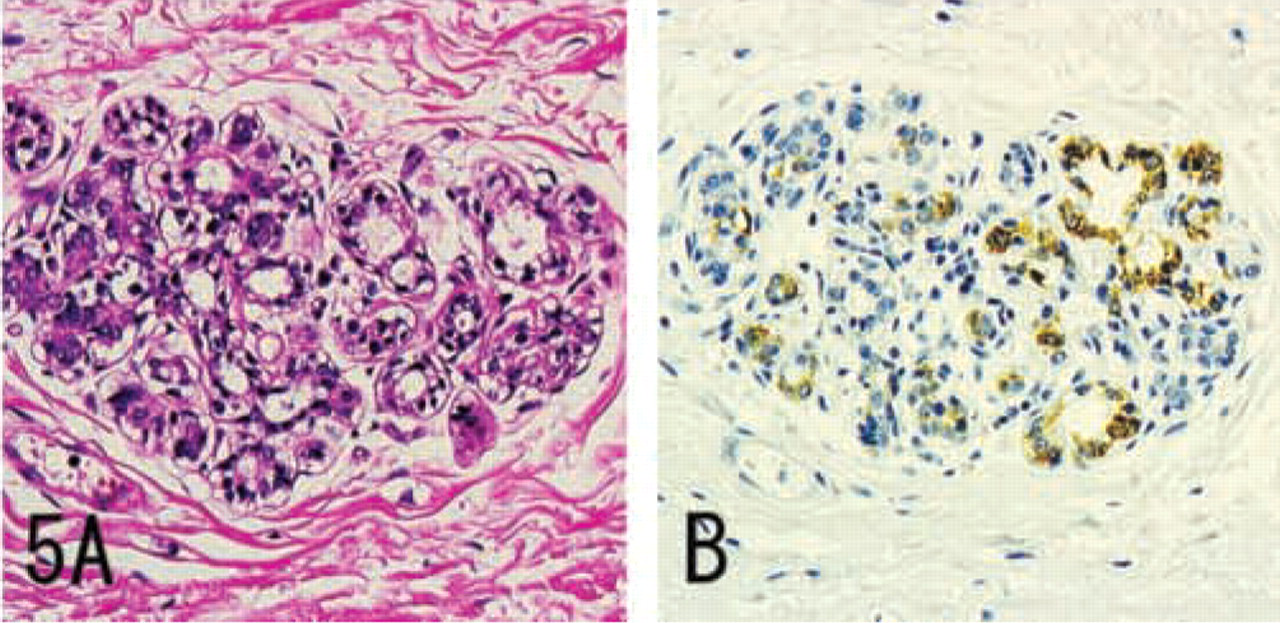
IHC staining for MUC6 in breast carcinoma, prepared from serial sections. Some carcinoma cells show diffuse cytoplasmic expression of MUC6. H&E staining (
Except in the case of biliary tract epithelial cells, GlcNAcα1→4Galβ→R was found in cytoplasmic mucus granules. The biliary tract epithelial cells exhibited cytoplasmic and apical cytoplasmic membrane staining for GlcNAcα1→4Galβ→R.
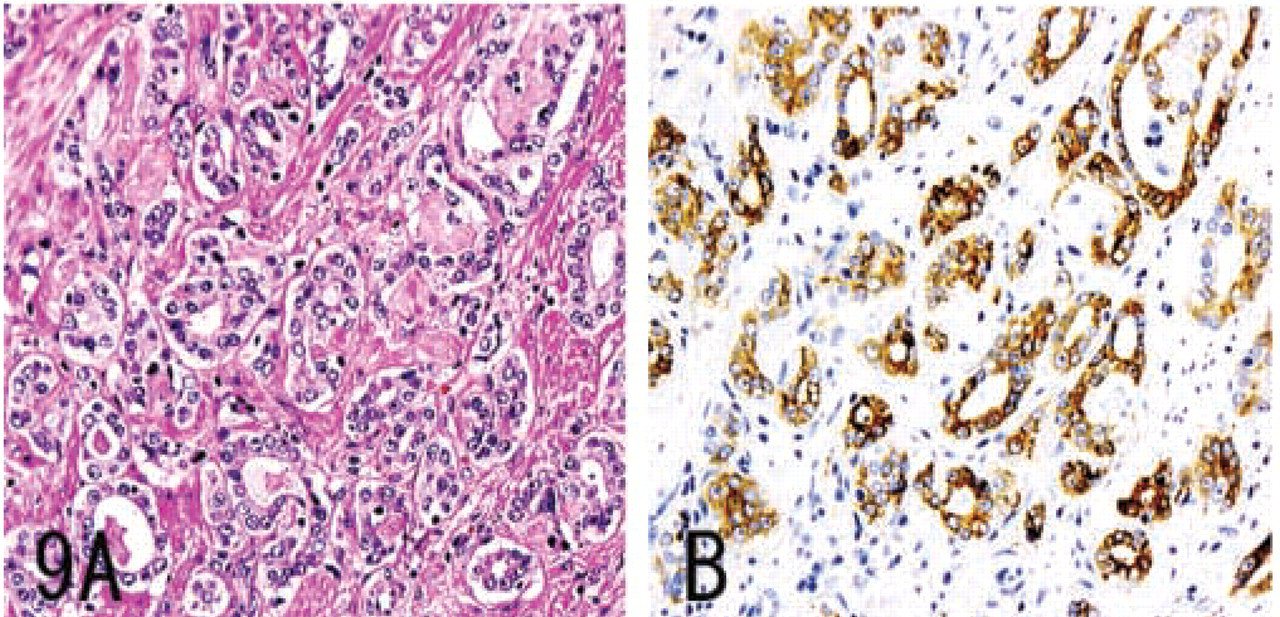
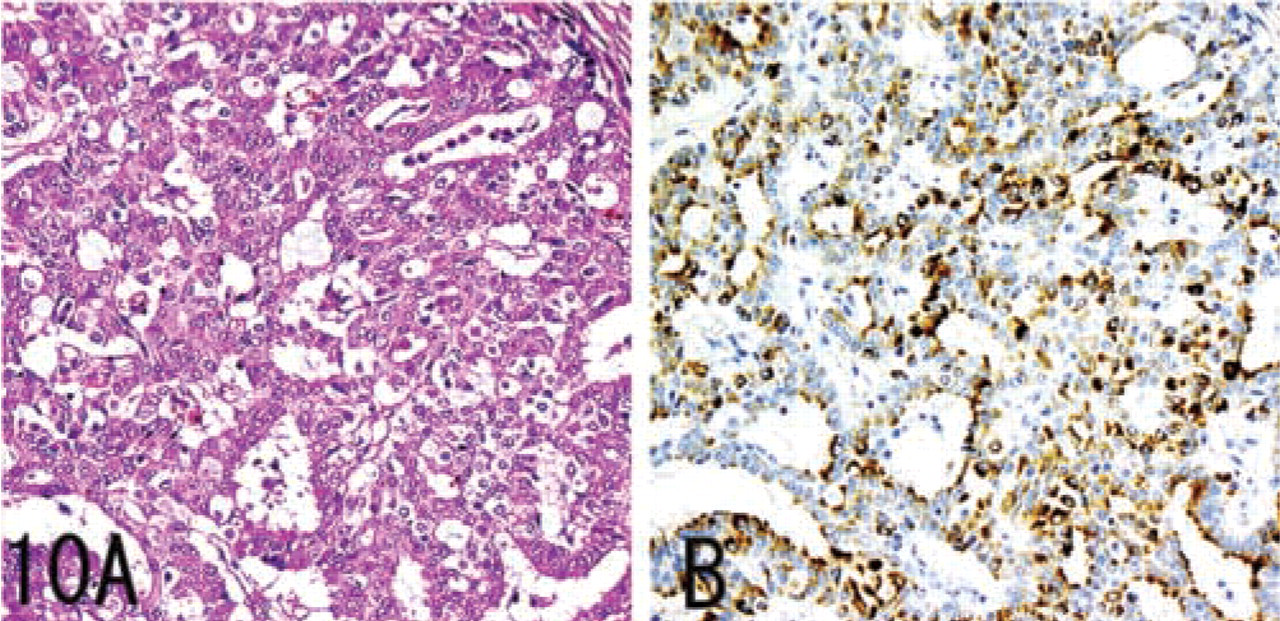
MUC6 without α4GnT and GlcNAcα1→4Galβ→R was expressed in a minority of submandibular gland mucous cells, pancreatic centroacinar cells, renal tubules, prostate glands (Figure 4), and terminal duct lobular units of mammary glands (Figure 5), and was also strongly expressed in seminal vesicle epithelial cells.
MUC6, α4GnT, and GlcNAcα1→4Galβ→R were not detected in normal esophagus, colon, hepatocytes, or lungs. The IHC data presented here are summarized in Table 1.
Demonstration of MUC6, α4GnT, and GlcNAcα1→4Galβ→R in Neoplastic Tissues in Primary Tissues
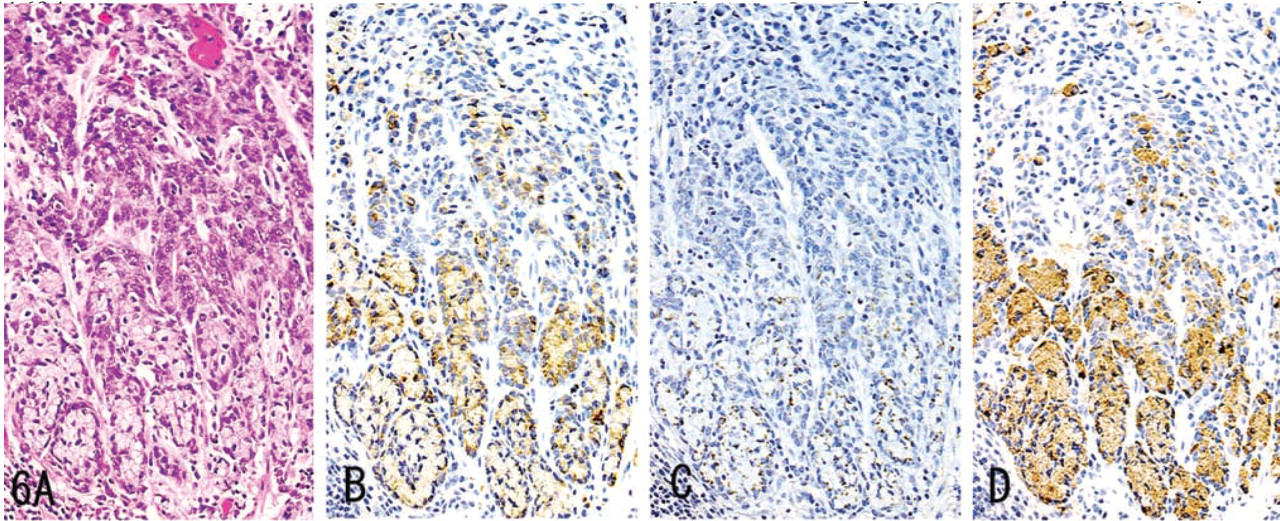
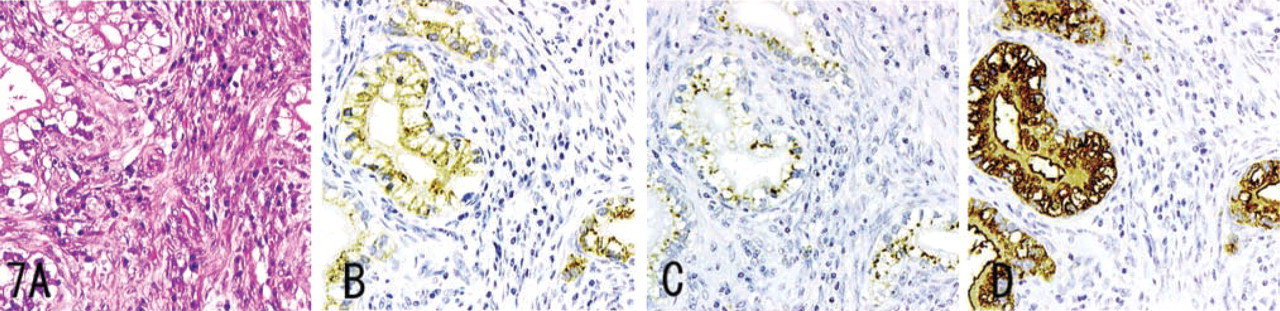
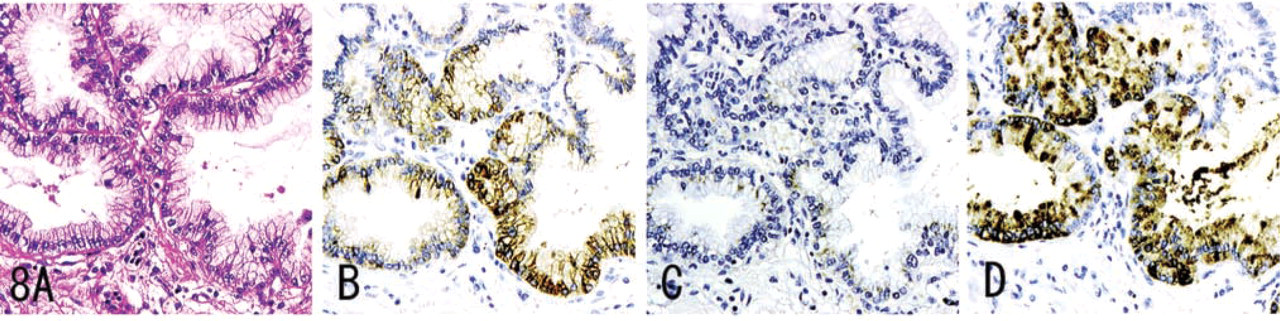
MUC6, α4GnT, and GlcNAcα1→4Galβ→R were detected in carcinoma of the stomach (Figure 6), pancreas (Figure 7), and intrahepatic bile duct, and in mucinous bronchioloalveolar cell carcinoma (Figure 8). Staining differed quantitatively from case to case (Table 2).
In these tumor tissues, MUC6 exhibited a heterogeneous cytoplasmic expression (Figures 6B, 7B, 8B, 9B, and 10B), whereas α4GnT exhibited Golgi staining (Figures 6C, 7C, and 8C). GlcNAcα1→4Galβ→R was expressed on the luminal surface of anaplastic glands and in secreted mucins, as well as in the cytoplasm of carcinoma cells (Figures 6D, 7D, and 8D). In these tissues, α4GnT-positive cells uniformly exhibited GlcNAcα1→4Galβ→R.
In gastric carcinomas, the staining scores given for GlcNAcα1→4Galβ→R in diffuse-type carcinomas were greater in early carcinomas than in advanced carcinomas (p<0.05). The staining scores for α4GnT and MUC6 showed no relation to the depth of invasion of the carcinoma. No significant difference was found among the scores given for MUC6, α4GnT, and GlcNAcα1→4Galβ→R, irrespective of histological type or depth of wall penetration. The scores given for GlcNAcα1→4Galβ→R showed a significant correlation with those given for α4GnT (r = 0.73 in diffuse-type gastric carcinomas; r = 0.86 in intestinal-type carcinomas). There was no correlation between the scores for α4GnT and MUC6 or between those for MUC6 and GlcNAcα1→4Galβ→R.
In pancreatic carcinomas, there was no significant difference among the scores given for MUC6, α4GnT, and GlcNAcα1→4Galβ→R expression. The degree of GlcNAcα1→4Galβ→R expression showed a significant correlation with the degree of expression for α4GnT and for MUC6 (r = 0.65 and r = 0.65, respectively).
In cholangiocarcinomas, there was no significant difference among the staining scores for MUC6, α4GnT, and GlcNAcα1→4Galβ→R. The degree of GlcNAcα1→4Galβ→R expression showed a significant correlation with that of α4GnT expression (r = 0.91). There was no correlation between MUC6 expression and α4GnT expression or between GlcNAcα1→4Galβ→R expression and MUC6 expression.
In mucinous bronchioloalveolar carcinomas, the scores given for GlcNAcα1→4Galβ→R were significantly higher than those for α4GnT expression (p<0.05). There were no correlations among the scores for MUC6, α4GnT, and GlcNAcα1→4Galβ→R.
In a minority of colon adenomas, prostate carcinomas (Figure 9), and breast carcinomas (Figure 10), MUC6 was detected, whereas α4GnT and GlcNAcα1→4Galβ→R were not. These IHC data are summarized in Table 2.
Demonstration of MUC6, α4GnT, and GlcNAcα1→4Galβ→R in Metastatic Adenocarcinomas of Lymph Nodes
Gastric carcinomas and pancreatic carcinomas showed immunoreactivity for MUC6, α4GnT, and GlcNAcα1→4Galβ→R. In gastric carcinomas, the degree of GlcNAcα1→4Galβ→R expression showed a significant correlation with the degree of α4GnT expression in gastric carcinomas (r = 0.79). In both gastric carcinomas and pancreatic carcinomas, α4GnT-positive cells uniformly co-expressed GlcNAcα1→ 4Galβ→R. Breast carcinomas showed immunoreactivity only for MUC6. Colon carcinomas showed immunoreactivity for none of these antigens.
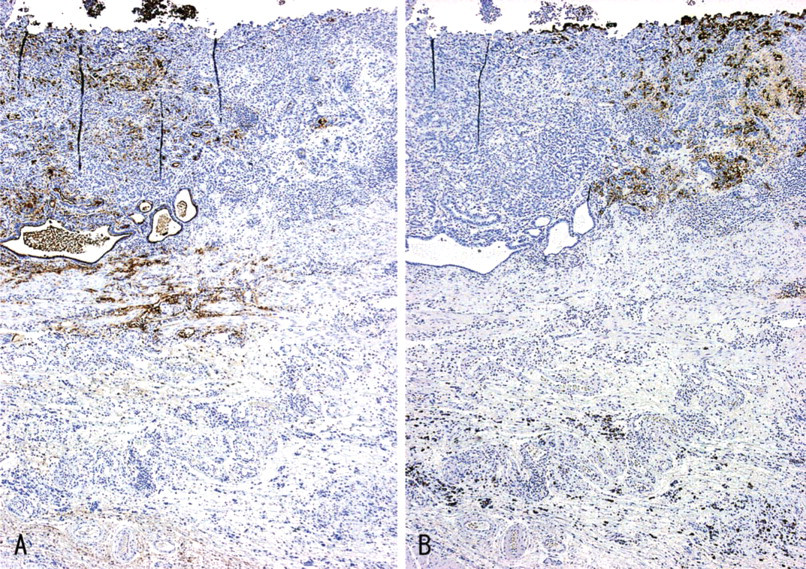
IHC staining for GlcNAcα1 →4Galβ→R and sialyl Lewis X in gastric carcinoma, prepared from mirror sections. The distribution of the carcinoma cells reactive for GlcNAcα1→4Galβ→R was completely different from that of the carcinoma cells reactive for sialyl Lewis X. Immunostaining for GlcNAcα1→4Galβ→R (
These IHC data are summarized in Table 2.
Demonstration of GlcNAcα1→4Galβ→R and Sialyl Lewis X in Gastric Carcinomas and Metastatic Gastric Carcinomas of Lymph Nodes
The distribution of carcinoma cells reactive for GlcNAcα1→4Galβ→R was completely different from that of carcinoma cells reactive for sialyl Lewis X both in primary (Figure 11) and in metastatic sites, although we found no negative correlation between GlcNAcα1→4Galβ→R and sialyl Lewis X expression. Carcinoma cells expressing both GlcNAcα1→4Galβ→R and sialyl Lewis X were not found.
These IHC data are summarized in Table 2.
Discussion
In this study we evaluated the IHC expressions of MUC6, α4GnT, and GlcNAcα1→4Galβ→R in normal, metaplastic, and neoplastic human tissues. The results showed (a) that α4GnT and GlcNAcα1→ 4Galβ→R were co-located in discrete cell-types (a finding consistent with involvement of α4GnT in the formation of GlcNAcα1→4Galβ→R in normal, metaplastic, and neoplastic cells) and (b) that both primary carcinoma tissues and metastatic tissues showed similar IHC profiles with regard to the expressions of MUC6, α4GnT, and GlcNAcα1→ 4Galβ→R.
In this study we have confirmed and extended our knowledge regarding the specific distribution of GlcNAcα1→4Galβ→R in human tissues. GlcNAcα1→4Galβ→R has been reported to be present in gastric gland mucous cells (cardiac gland cells, mucous neck cells, and pyloric gland cells), Brunner's gland cells, mucous cells of the periductal glands of the pancreaticobiliary tract, mucinous metaplasia of gallbladder and pancreas, and neoplastic cells expressing gastric mucins [including mucinous tumors of the ovary, adenocarcinomas of stomach, pancreas, gallbladder, lung (mucinous bronchioloalveolar cell carcinoma), and uterine cervix (adenoma malignum) (Nakamura et al. 1998; Ota et al. 1998,2001; Zhang et al. 2001)], but not in normal esophagus, colon, pancreas, ovary, lung, uterine cervix, normal and neoplastic salivary glands, or adenocarcinomas of colon, breast, kidney, thyroid gland, endometrium, liver, or prostate (Ishii et al. 1998; Nakamura et al. 1998; Ota et al. 2001). In addition, we found that GlcNAcα1→4Galβ→R was present in the normal epithelium of the intrahepatic bile ducts and cholangiocarcinoma but not in seminal vesicle epithelium.
In this study, α4GnT-positive cells uniformly co-expressed GlcNAcα1→4Galβ→R, irrespective of whether the sections were from normal, metaplastic, or neoplastic tissues. The degree of GlcNAcα1→4Galβ→R expression showed a significant correlation with the degree of α4GnT expression in various adenocarcinoma tissues, as well as in normal and metaplastic cells.
The expressions of α4GnT and GlcNAcα1→4Galβ→R were more restricted than that of MUC6. MUC6 has been demonstrated to be present in human tissues in which both α4GnT and GlcNAcα1→4Galβ→R were positive, such as normal stomach (Ho et al. 1995a,b; Buisine et al. 2000b; Machado et al. 2000; Reis et al. 2000), duodenum (Bartman et al. 1998; Buisine et al. 2000a), bile duct (Bartman et al. 1998; Buisine et al. 2000a), gallbladder (Bartman et al. 1998; Buisine et al. 2000a), and in adenocarcinomas of the stomach (Ho et al. 1995b; Machado et al. 2000; Reis et al. 2000), bile duct (Bartman et al. 1998), gallbladder (Bartman et al. 1998; Sasaki et al. 1999), and pancreas (Bartman et al. 1998; Terada et al. 1996). As previously reported and confirmed in this study, MUC6 was distributed in a wide variety of human tissues in which both α4GnT and GlcNAcα1→ 4Galβ→R were negative. Thus, MUC6 has previously been demonstrated in normal endometrium (Bartman et al. 1998) and seminal vesicles (Bartman et al. 1998), adenoma of the colon (Bartman et al. 1999), and normal and adenocarcinoma of the breast (Bartman et al. 1998; Pereira et al. 2001).
The distribution of carcinoma cells reactive for GlcNAcα1→4Galβ→R was completely different from that of carcinoma cells reactive for sialyl Lewis X, both in primary (Figure 11) and metastatic sites, although we found no negative correlation between the GlcNAcα1→4Galβ→R and sialyl Lewis X expressions. GlcNAcα1→4Galβ→R is preferentially attached to core2-branched O-glycan (Ishihara et al. 1996), and the sialyl Lewis X found in O-glycan is also attached to the terminal end of core2-branched structures (Fukuda 1996). Therefore, the expressions of GlcNAcα1→4Galβ→R and sialyl Lewis X may be reciprocally regulated because these carbohydrates compete for the common precursor oligosaccharide, core2-branched O-glycan. It is well known that sialyl Lewis X serve as preferential ligands for the cell-adhesion molecules E- and P-selectin (Fukushima et al. 1984; Itzkowitz et al. 1988; Rosen and Bertozzi 1994). Recently, we demonstrated that the sialyl Lewis X expressed on core2-branched O-glycans were positively correlated with tumor progression in both colorectal and pulmonary cancers (Shimodaira et al. 1997; Machida et al. 2001). Overall, these results suggest that the expression of GlcNAcα1→4Galβ→R in gastric cancer cells may be a favorable predictor of the patient's outcome. Further study will be needed to test this hypothesis.
In summary, primary carcinoma tissues and metastatic tissues showed similar IHC profiles with regard to the expressions of MUC6, α4GnT, and GlcNAcα1→ 4Galβ→R. Determination of the site of origin of metastatic carcinomas using examination of histological slides continues to present a diagnostic challenge for the pathologist. It is possible that immunostaining for GlcNAcα1→4Galβ→R (with MAb HIK1083), or α4GnT, in conjunction with immunostaining for MUC6, could be diagnostically relevant because of their specific distributions in human tissues.
Footnotes
Acknowledgements
Supported by Grants-in-Aid for Scientific Research C-15590482 (to HO) and Priority Area 14082201 (to JN) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
We thank Professor Tsutomu Katsuyama and Katsunori Sasaki at Shinshu University School of Medicine, and Professor David Y. Graham at Baylor College of Medicine (Houston, TX) for their helpful comments and encouragement.
