Abstract
We describe an improved immunohistochemical procedure for detecting regions of hypoxia in normal organs and tumors in mice. The method employs a primary fluorescein-conjugated mouse monoclonal antibody directed against pimonidazole protein adducts that are created in hypoxic tissues and a secondary mouse anti-fluorescein antibody that is conjugated to horseradish peroxidase. Using these reagents, we clearly visualized the regions of relative hypoxia in implanted tumors in mice as well as in normal organs such as liver and kidney. Significantly, the resulting tissue sections were remarkably free of the background staining that is characteristically observed when rodent antibodies are used to detect antigens in rodent tissues.
I
Raleigh et al. (1985) demonstrated that hypoxia could be detected by immunohistochemistry using antibodies that recognize hypoxia marker adducts produced by exogenously administered pimonidazole. Subsequent studies have confirmed that immunochemical detection of pimonidazole protein adducts can be used to detect hypoxia gradients in normal tissues and in a variety of pathological conditions (Arteel et al. 1995).
Most of the pimonidazole-based hypoxia studies, including human-to-animal xenotransplants, are performed in mouse or rat models. Because the primary monoclonal antibody (MAb) that is used to detect pimonidazole adducts in proteins of hypoxic tissues is of mouse origin, detection of hypoxia in murine tissues can be problematic because of the inherent presence of normal mouse immunoglobulins. Therefore, even when special blocking procedures are employed, immunostaining of mouse tissues with primary mouse antibodies produces extensive background staining that can obscure the specific binding pattern of the primary antibody (Samoszuk and Corwin 2003).
To obviate this problem and to facilitate accurate and quantitative detection of hypoxia gradients in animal models, we developed a novel set of reagents for IHC detection of adducts of pimonidazole in mouse tissues without the need to resort to complex blocking procedures. A monoclonal anti-pimonidazole antibody, MAb1, clone 4.3.11.3 (Kennedy et al. 1997), directly conjugated with fluorescein isothiocyanate (FITC), was used as a primary antibody reagent. MAb1 was purified from tissue culture supernatants using protein A affinity chromatography to greater than 95% purity and then conjugated with FITC at elevated temperature (29–32C) and pH 9.5 according to the method of The and Feltkamp (1970). The FITC-to-protein molar ratio in the final preparation was 6.5. The conjugate was concentrated to 1 mg/ml using ultrafiltration and was stored at 4C until use. A standard preparation of protein A-purified monoclonal anti-FITC antibody (clone 5D6.2; Chemicon International, Temecula, CA) conjugated with horseradish peroxidase was used at the initial concentration of 1 mg/ml as a secondary antibody reagent.
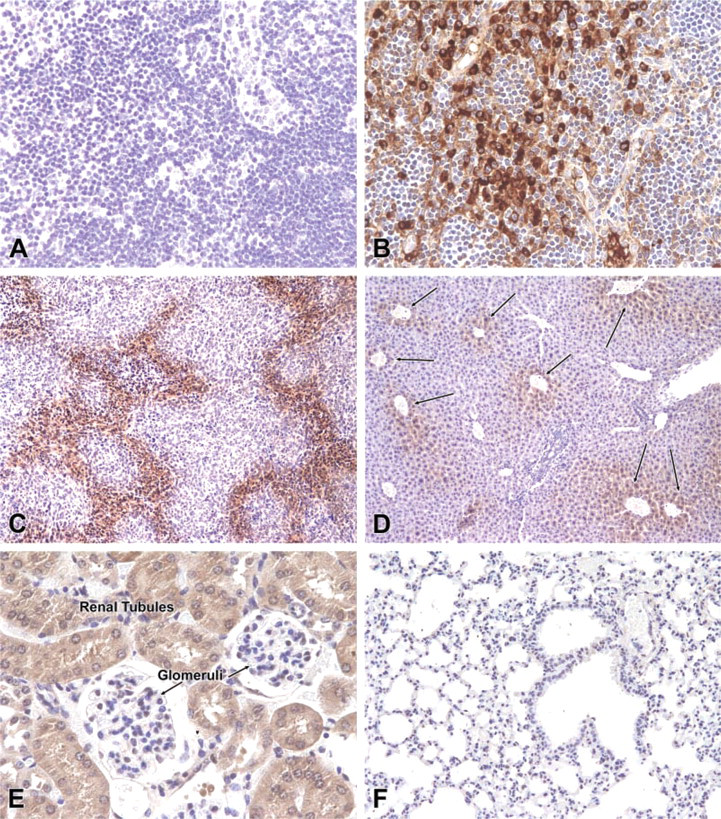
Immunostaining of normal tissues and 4T1 mammary adenocarcinoma tumor for hypoxia using new reagents and procedures described in this report. There was no staining of the stroma or plasma cells in lymph nodes (
The immunostaining was performed on tumors and normal organs obtained from 10 mice that have been previously described in detail (Samoszuk and Corwin 2003). Institutional guidelines for the use of animals were followed. In brief, tissue sections of 4T1 mammary adenocarcinoma tumors, livers, lungs, and kidneys were first deparaffinized and subjected to quenching of endogenous peroxidase and antigen retrieval using microwaving in citrate buffer. The primary conjugate was then applied to the tissues at a predetermined dilution of 1:100 and incubated for 60 min. After a wash with PBS, the slides were then incubated at room temperature for 60 min with a 1:100 dilution of the peroxidase-conjugated, anti-fluorescein antibody. Bound antibody was then visualized with DAB chromogen, followed by counterstaining with hematoxylin. For comparative purposes, we stained duplicate tissue sections with the standard procedure (HypoxyprobeTM-1 Kit; Chemicon) that employed unconjugated primary antibody to pimonidazole. For negative control purposes, we studied similar tumors and normal tissues from mice that had not been treated with pimonidazole.
Using the new reagents, we observed that normal lymph nodes were completely devoid of staining (Figure 1A). In contrast, lymph nodes stained by the conventional procedure using unconjugated primary antibody followed by anti-mouse IgG conjugate with horseradish peroxidase demonstrated extensive staining of the cytoplasm of plasma cells and stroma of lymph nodes (Figure 1B). In the new procedure, sections of tumors produced a mottled, geographic gradient of staining that outlined nodules of growing tumor cells (Figure 1C). Notably, the hypoxic centrilobular regions of livers from the mice treated with pimonidazole had a gradient of staining that extended radially from the central veins (Figure 1D). A similar gradient of staining intensity was noted in the tubules of the renal medulla, whereas the well-vascularized and presumably well-oxygenated glomeruli were devoid of staining (Figure 1E). As expected, the lungs of pimonidazole-treated mice had no evidence of staining for hypoxia (Figure 1F), and the tissues and tumors from the mice that had not received pimonidazole were completely free of staining (not shown).
On the basis of these IHC results, we conclude that the novel reagents we have described are suitable for detecting gradients of hypoxia in mouse tissues. Our method has the advantage that it does not require any complex blocking steps. In addition, the reagents yield tissue sections that are free of the strong nonspecific background staining that is typically seen in rodent tissues reacted with rodent antibodies. Hence, this procedure provides an improved method for detecting hypoxia gradients in mouse tissues and should be a valuable new tool in the study of hypoxia in mouse and rat models of disease, including cancer.
