Abstract
The small leucine-rich proteoglycans (PGs) biglycan and decorin, and their mRNAs, have been localized during neonatal development and aging (3 weeks to 2 years) of collateral and cruciate ligaments and of menisci of the rabbit knee joint. In the collateral ligaments, biglycan and decorin are found between the bundles of collagen fibers at all ages. In cruciate ligaments the PGs are primarily around the cells. In neonatal ligaments all the cells express the mRNAs for biglycan and decorin, but in the collateral ligaments the number expressing the mRNAs is reduced at 8 months. In 3–week menisci the PGs are uniformly distributed in the matrix, but by 8 months biglycan is present primarily in the central fibrocartilaginous regions, whereas decorin is found peripherally. In neonates, all the cells express the mRNAs but the number is reduced in 8-month menisci. The results illustrate the precise localizations of biglycan and decorin in healthy rabbit ligaments and menisci which, after injury, must be reproduced in the repair tissue for normal strength to be regained.
Keywords
T
Ligaments and tendons are composed of bundles of collagen fibrils, of which the major component is Type I, but Types III, V, and VI collagen are also present (Amiel et al. 1984; Bray et al. 1993; Bland and Ashhurst 1996a, 1997). The organization of the fibrillar collagens changes during fetal and postnatal development in both the collateral and cruciate ligaments (Bland and Ashhurst 1996a). Most studies of the proteoglycans (PGs) of ligaments relate to their glycosaminoglycan (GAG) content and are biochemical. In bovine collateral ligaments there are two populations of PGs. The larger population consists of small chondroitin sulfate/dermatan sulfate PGs, most of which are decorin, whereas the smaller population (about 20%) consists of large chondroitin sulfate/keratan sulfate PGs (Hey et al. 1990). One large chondroitin sulfate PG in bovine collateral ligament was identified as versican-like (Campbell et al. 1996). Chondroitin and dermatan sulfates and hyaluronan are present in collateral and cruciate ligaments of human and several other animals (Gässler et al. 1994; Watanabe et al. 1994).
Most mammalian menisci are fibrocartilaginous. The major collagen of menisci is Type I, but Types II, III, V, and VI collagen may also be present (Eyre and Wu 1983; Cheung 1987; McDevitt and Webber 1990; Bland and Ashhurst 1996b). In rabbit fetal menisci, Types I, III, and V collagen are present, but at 3 weeks postnatal Type II collagen appears in the central region (Bland and Ashhurst 1996b). There is little overlap of the regions containing Types I and II collagen in adult menisci, whereas Types III and V collagen are found throughout the tissue.
Binding of antibodies to biglycan and decorin in the matrix and expression of their mRNAs by cells of the collateral and cruciate ligaments and menisci a
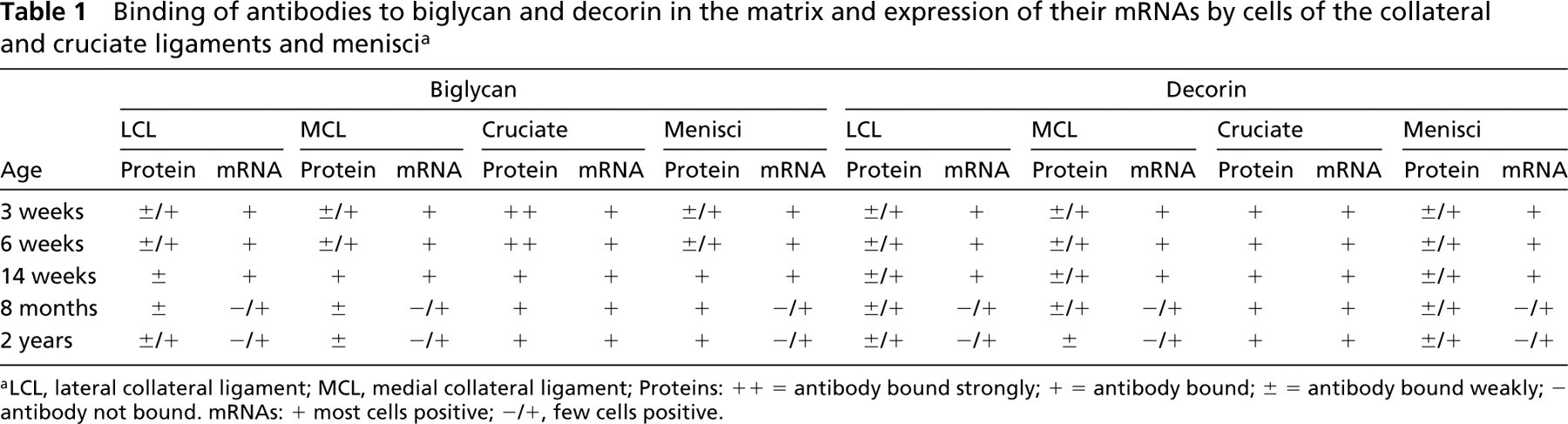
aLCL, lateral collateral ligament; MCL, medial collateral ligament; Proteins: + + = antibody bound strongly; + = antibody bound; ± = antibody bound weakly; – antibody not bound. mRNAs: + most cells positive; −/+, few cells positive.
In adult pig and rabbit menisci the major GAG is chondroitin-6-sulfate, but there is some chondroitin-4-sulfate and dermatan sulfate (Webber et al. 1984; Scott et al. 1997). In the 20-week porcine meniscus biglycan is more abundant than decorin (Nakano et al. 1997; Scott et al. 1997). Aggrecan is the major PG in canine menisci, and its gene expression is reduced by immobilization of the joint (Djurasovic et al. 1998).
The distributions of biglycan and decorin during development and aging of both ligaments and menisci are unknown. There is little immunohistochemically detectable biglycan and decorin in fetal rabbit ligaments and menisci (unpublished observations). Biglycan and decorin and their mRNAs were localized therefore in the collateral and cruciate ligaments and in menisci of the rabbit from 3 weeks postnatal to 2 years. Their distribution between the fiber bundles in the ligaments persists throughout life. In menisci the initial uniform localization changes as fibrocartilage develops. Biglycan is preferentially located in that area. Some cells in these tissues express the mRNAs throughout life.
Materials and Methods
All reagents and labeled antibodies were purchased from Sigma (Poole, UK) and all restriction enzymes, polymerases and yeast tRNA from Roche (Lewes, UK), unless otherwise stated.
Preparation of Tissue for Microscopy
The medial and lateral collateral ligaments, anterior and posterior cruciate ligaments, and lateral and medial menisci were dissected from 3-, 6-, and 12–14-week, 8-month, and 2-year-old rabbit knee joints. Two or more rabbits were used at each stage. The tissues were fixed in 4% paraformaldehyde in 0.05 M Tris-HCl buffer, pH 7.3, for 18 hr, then washed extensively in buffer. After washing, the tissue was dehydrated in graded ethanols, cleared in methyl salicylate, and embedded in paraffin. Serial sagittal longitudinal sections were cut at 7 μm. For histological observations, sections were stained with hematoxylin and eosin.
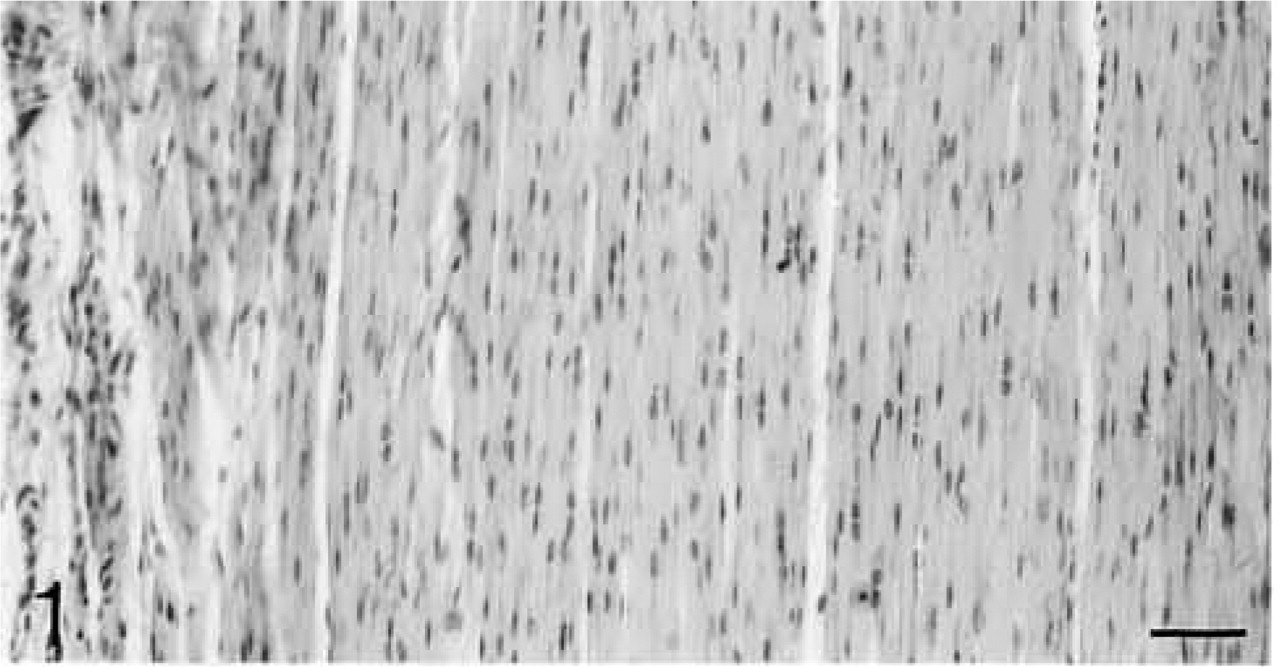
Bundles of collagen fibers with spindle-shaped cells in rows between them. H&E staining. Bar = 50 μm.
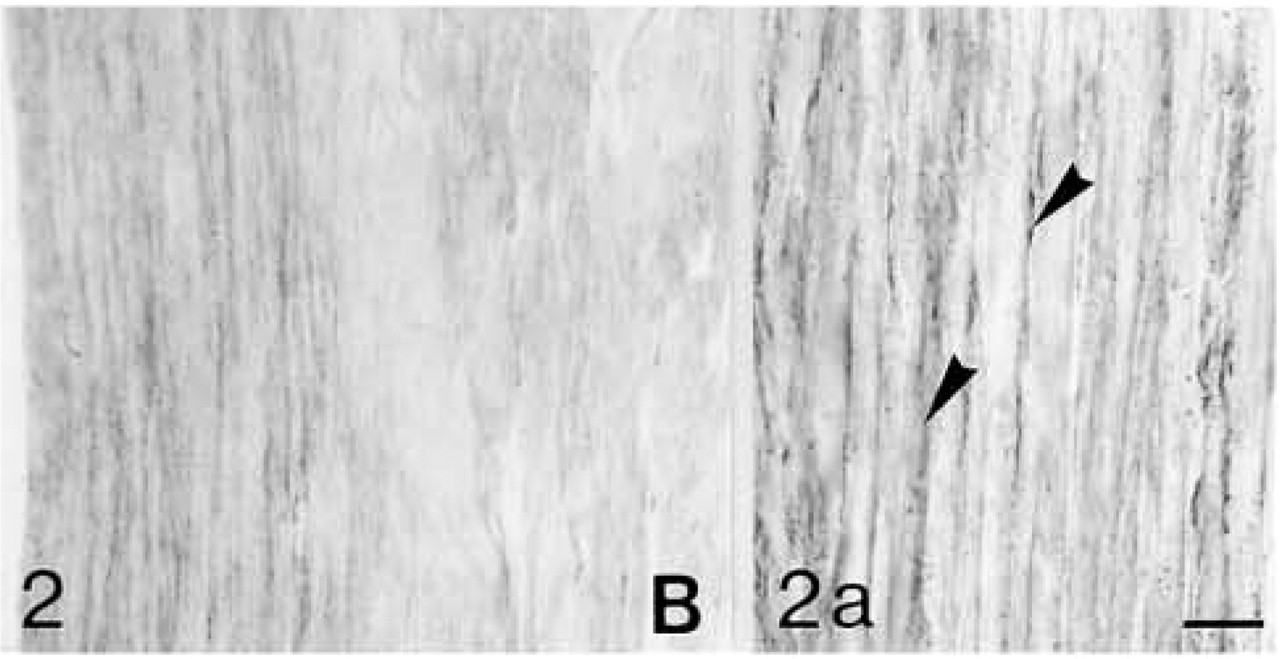
Biglycan (B) antibody binding; biglycan is located primarily between the fiber bundles (arrowheads in
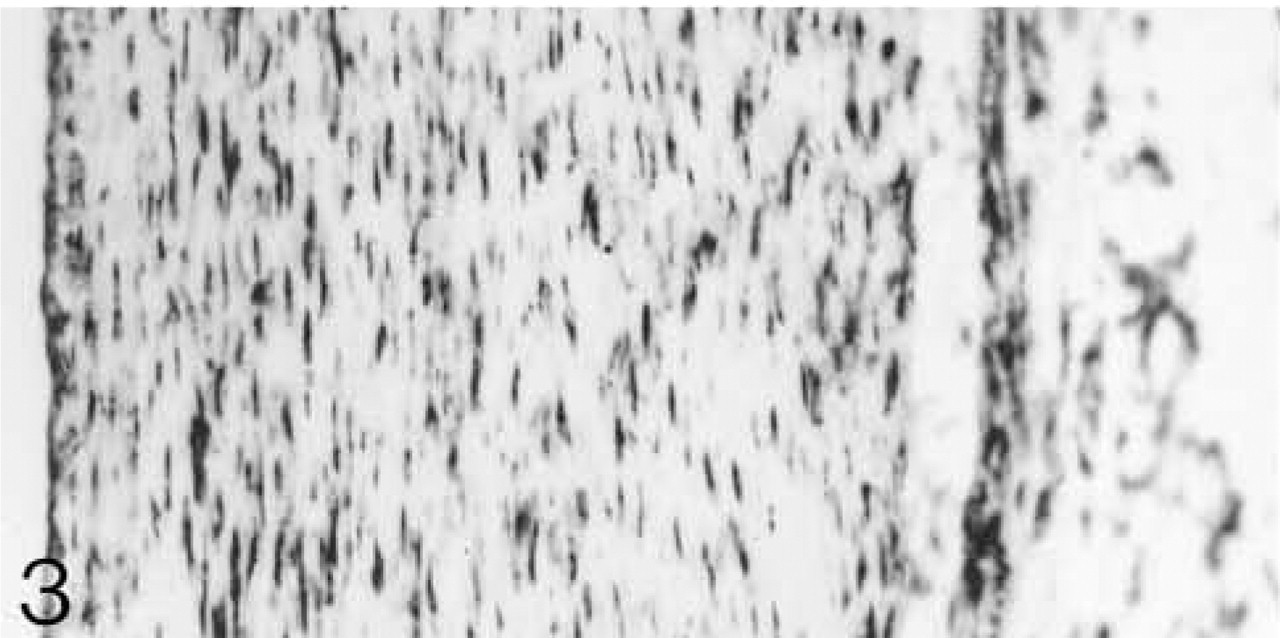
ISH for biglycan mRNA. All cells are expressing the mRNA.
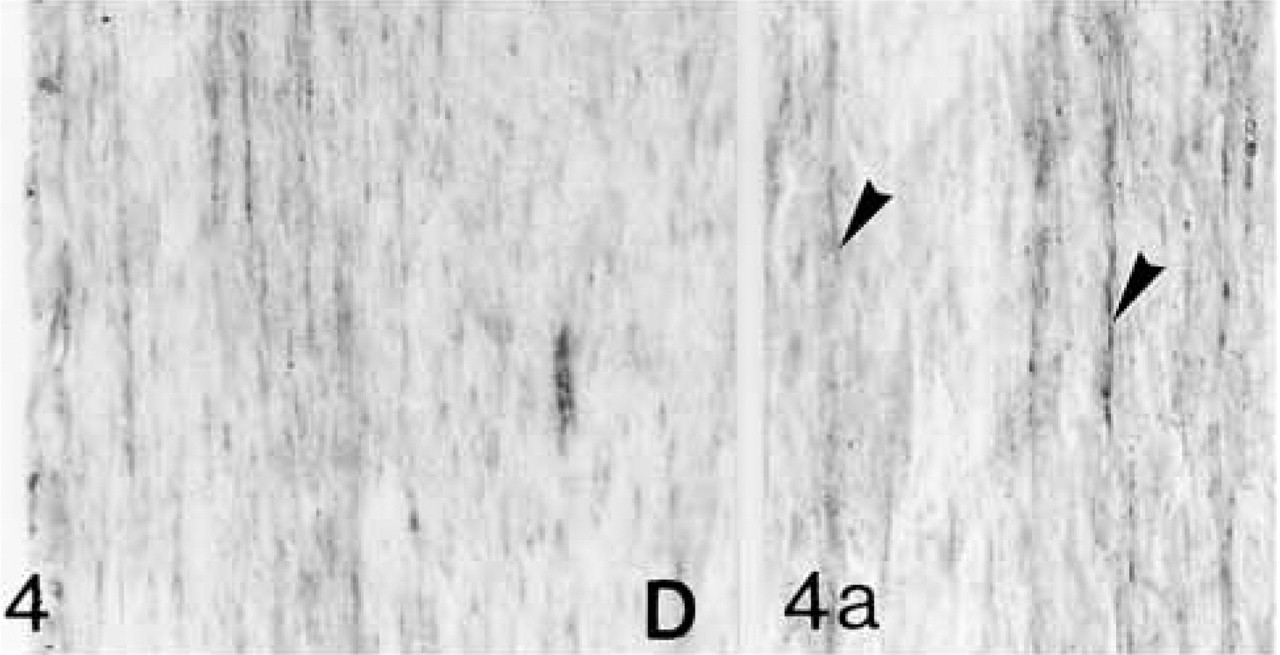
Decorin (D) antibody binding; decorin is located between the fiber bundles (arrowheads in
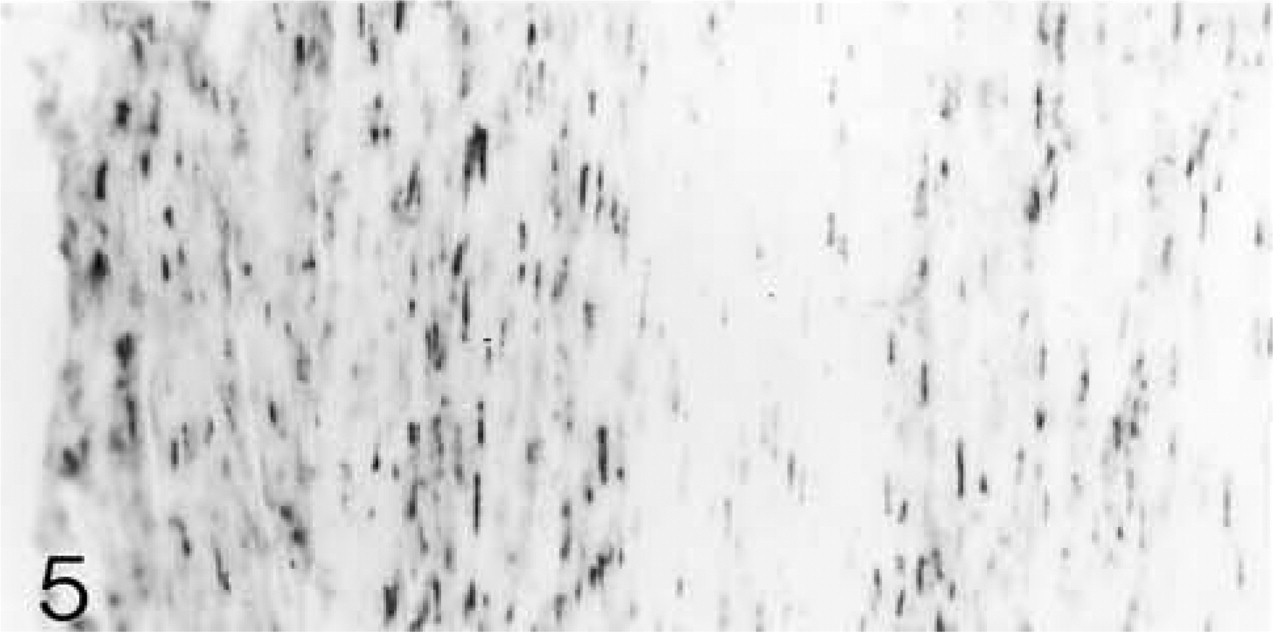
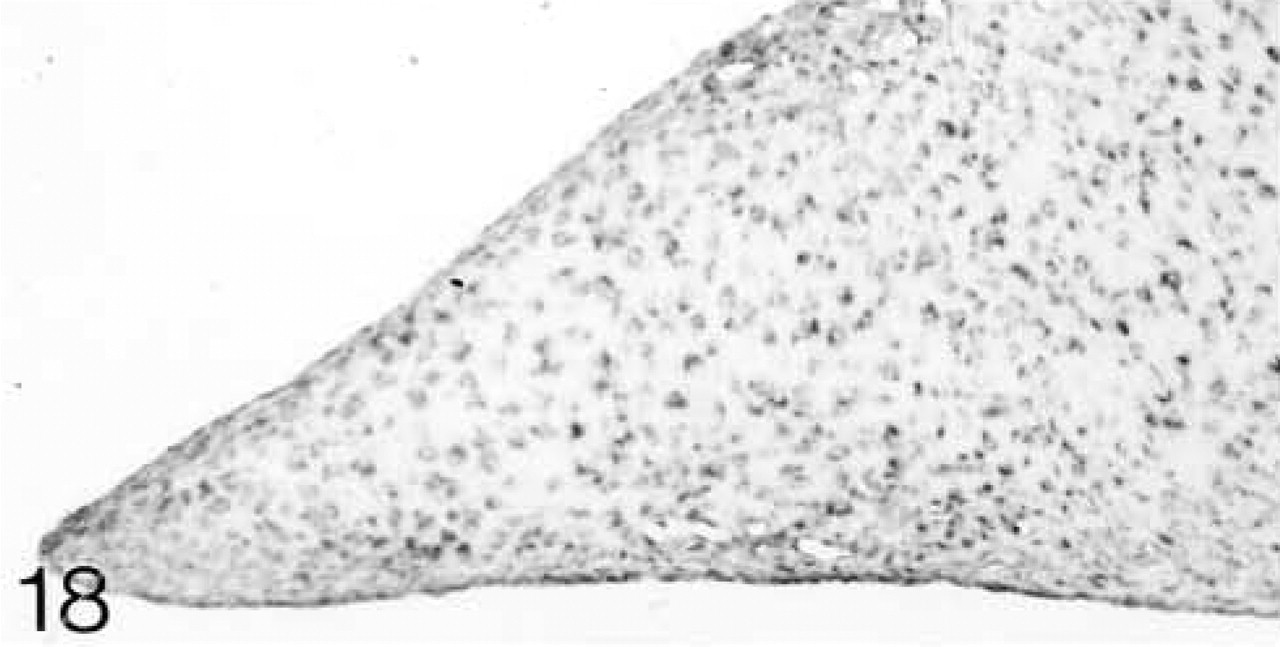
ISH for decorin mRNA. All cells are expressing the mRNA.

Thick bundles of collagen fibrils are interspersed by spindle-shaped cells. H&E staining. Bar = 50 μm.

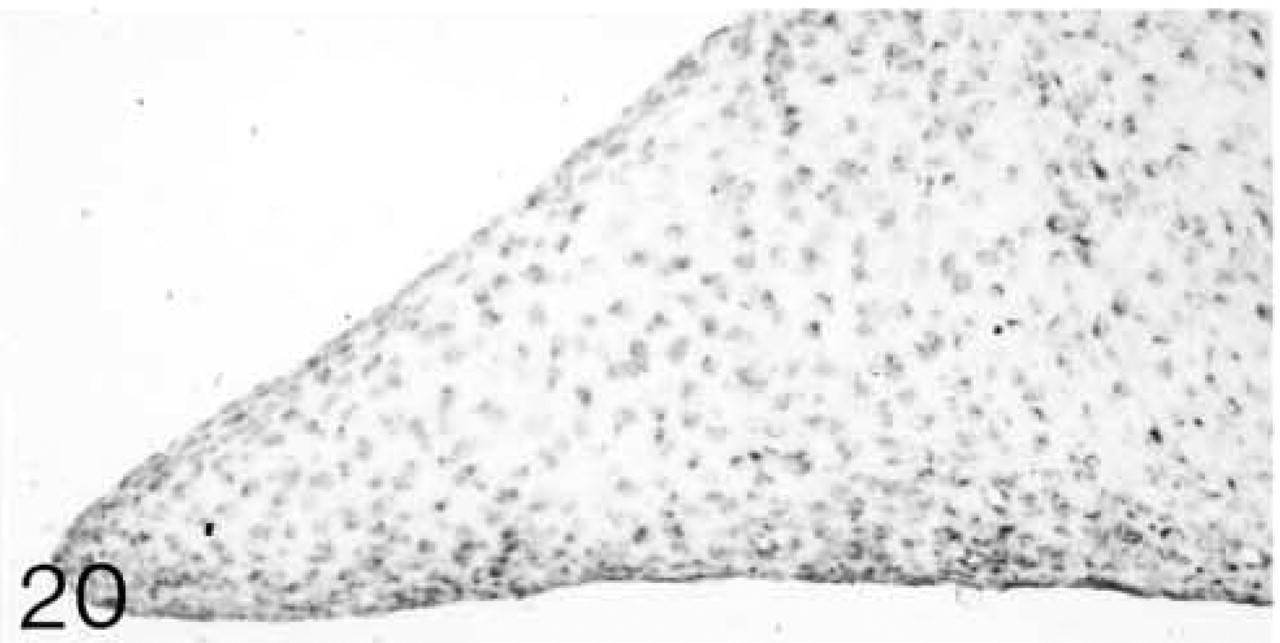
Biglycan (B) antibody binding; biglycan is located between the fiber bundles.

ISH for biglycan mRNA; only a few scattered cells are expressing the mRNA.

Decorin (D) antibody binding; decorin is located between the fiber bundles.
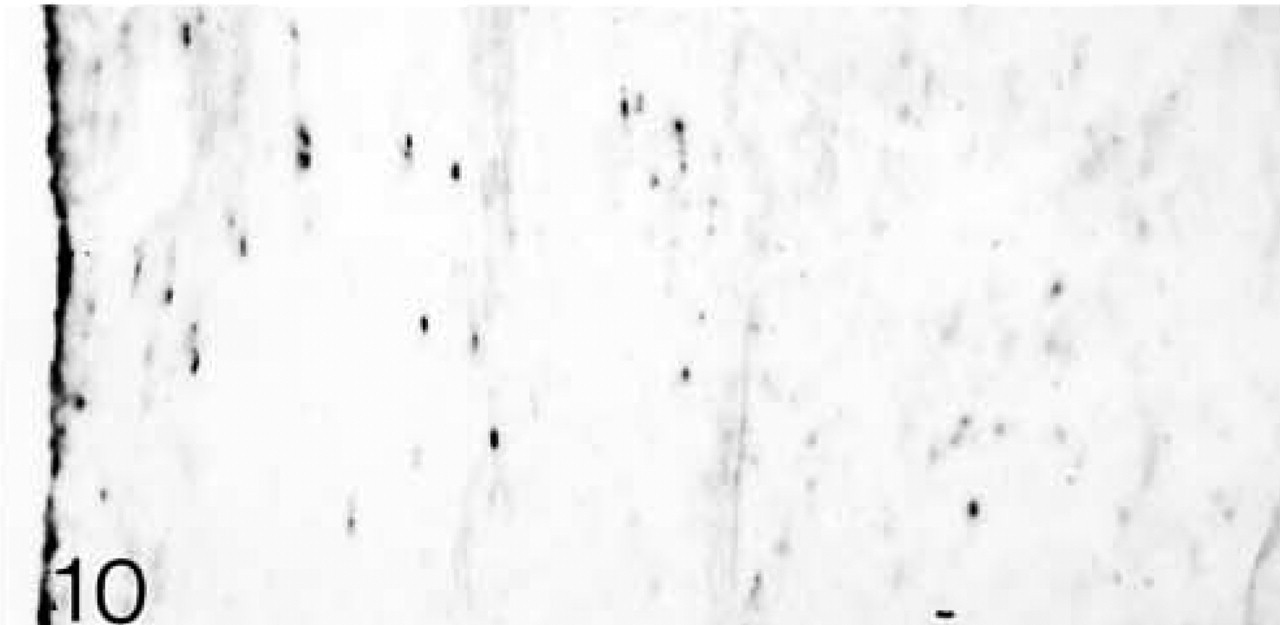
ISH for decorin mRNA; only a few cells are expressing the mRNA.
Immunohistochemistry
Procedures for Antibodies to Biglycan and Decorin. Chicken polyclonal antibodies to purified bovine biglycan and to purified bovine decorin were a gift from Prof. D. Heinegård (Lund University, Sweden). The total IgG was purified as described by Heinegård et al. (1985).
Sections were dewaxed and rehydrated before the following pretreatments that are usual for paraffin-embedded tissues: (a) 0.1% trypsin in 0.05 M Tris–saline, pH 7.8, with 0.1% CaCl2 at 37C for 1 hr to aid tissue permeability; (b) 2% hyaluronidase in PBS, pH 7.3, at 37C for 1 hr to remove GAGs and unmask epitopes; (c) 2%
For controls the primary antibody was replaced by normal chicken serum. At no time was there any binding in the controls.
In Situ Hybridization
cDNA probes to human biglycan and decorin were a gift from Dr. L. Fisher (NIH; Bethesda, MD) (Fisher et al. 1989); they recognize rabbit biglycan and decorin mRNAs in Northern blots (Demoor–Fossard et al. 1998). Antisense and sense probes were generated by linearizing with Kpn I and T3 polymerase and Xba I and T7 polymerase, respectively, for biglycan and with Bam HI and T7 polymerase and Kpn I and T3 polymerase, respectively, for decorin. Both probes were labeled with digoxigenin by in vitro transcription using the DIG-RNA labeling (SP6/T7) kit (Roche). The riboprobes are 1685 bp (biglycan) and 1600 bp (decorin) long. The transcripts were hydrolyzed at 60C for 40 min in 100 mM bicarbonate buffer to yield fragments of ~200 bp and were stored at –20C until use.
The sections were dewaxed and rehydrated. The following pretreatments were performed: (a) 0.2 N HCl for 20 min; (b) 6% H2O2 for 30 min; (c) proteinase K (20 μg/ml) for 10 min at 37C; (d) 4% paraformaldehyde in PBS for 20 min at 4C; (e) 0.1 M glycine twice for 10 min; (f) 0.25% acetic anhydride in triethanolamine (pH 8.0) for 10 min. Between each treatment the sections were washed in PBS. They were then dehydrated through graded ethanols and allowed to dry. Fifteen μl of hybridization solution was applied to each section. The hybridization solution (low stringency) contained 50% formamide, 10 mM Tris-HCl (pH 7.4), 1 mM EDTA, 1 × Denhardt's, 0.5% SDS, 600 mM NaCl, 10% dextran sulfate, 0.5 μg/ml yeast tRNA, and labeled antisense or sense probe. Hybridization was for 18 hr at 50C for biglycan mRNA and 45C for decorin mRNA. After hybridization, the coverslips were removed in 2 × SSC before rinsing in Tris–EDTA buffer. The sections were then treated with 20 μg/ml RNase in Tris–EDTA–NaCl buffer, pH 8.0. The sections were washed twice in 2 × SSC for 15 min, then in 1 × SSC for 10 min at 55C. A final wash in 1 × SSC was at room temperature. The digoxigenin label was detected using the Roche kit, except that 0.3% Triton X-100 was added to the antibody solution. This blocks the nonspecific binding in the cartilage matrices that arises after treatment with proteolytic enzymes (Bland et al. 1991). The sections were mounted in glycerin jelly.
At no time was there any reaction with the sense, i.e., control, probes.
Results
The results are summarized in Table 1.
Collateral Ligaments
Both lateral and medial collateral ligaments consist of bundles of collagen fibrils with spindle-shaped cells irregularly arranged between the fiber bundles. The number of cells appears to decrease with age (Figures 1 and 6). Antibodies to biglycan and decorin are preferentially bound between the fiber bundles. They tend to be bound more strongly in young ligaments (Figures 2, 4, 7, and 9). Biglycan and decorin mRNAs are expressed by most cells up to 14 weeks, but by 8 months the number of cells expressing the mRNAs has decreased (Figures 3, 5, 8, and 10).
Cruciate Ligaments
The anterior and posterior ligaments are composed of parallel bundles of collagen fibrils separated by rows of large round cells (Figure 11). The binding of antibodies to biglycan and decorin is similar at all ages and is closely associated with the rows of cells (Figures 12 and 14). Among the fibers, the binding is weaker and more variable. Both biglycan and decorin mRNAs are expressed by the cells throughout life (Figures 13 and 15).
Menisci
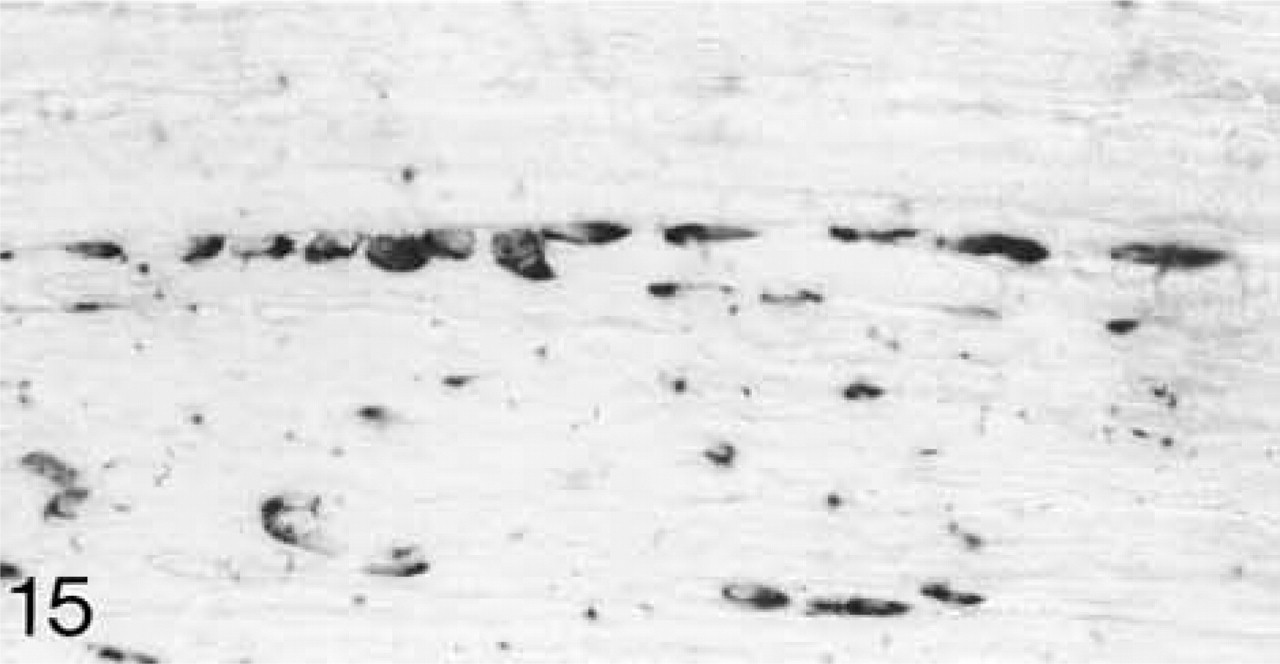
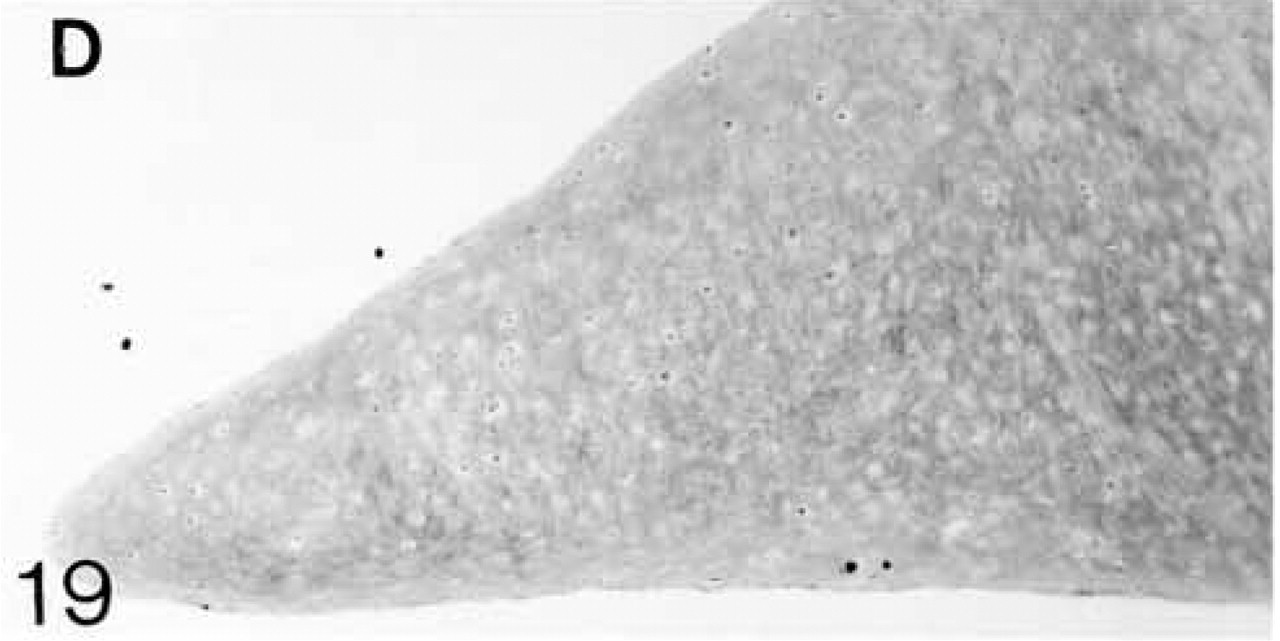
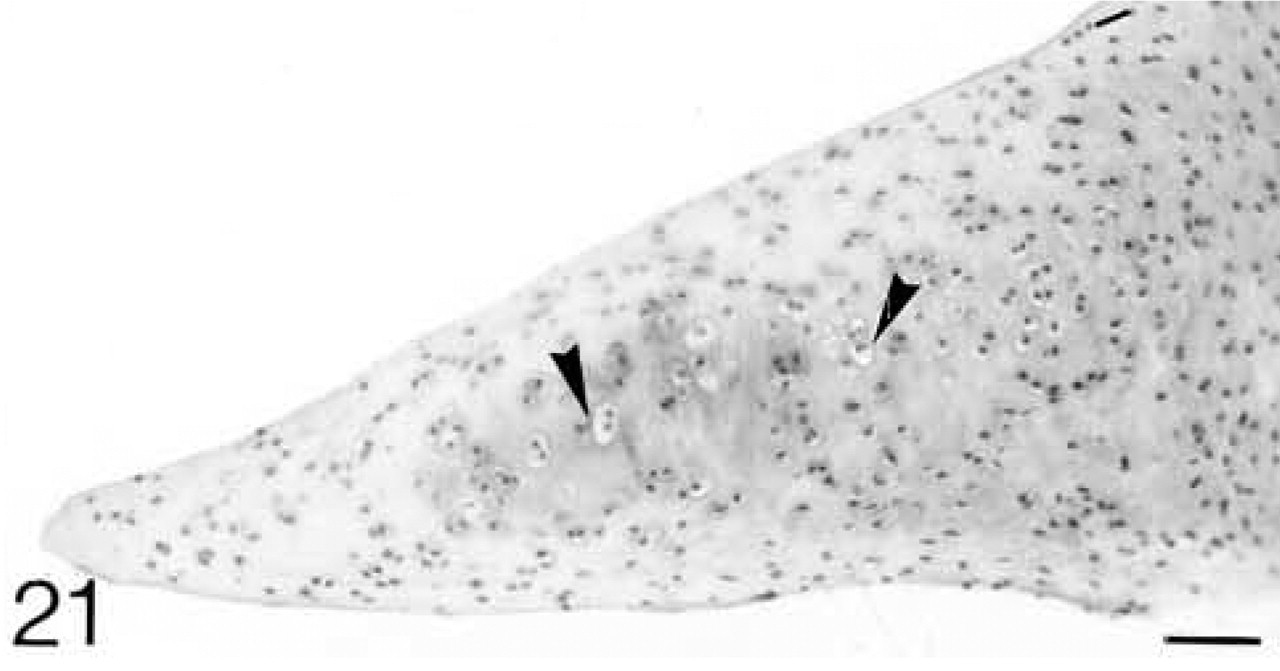
At 3 weeks postnatal, a fibrocartilaginous region starts to develop in the center of both the lateral and medial menisci (Figure 16) and it is much larger at 8 months (Figure 21). It is surrounded by a dense fibrous layer. At 3 weeks, both antibodies are bound throughout the matrix (Figures 17 and 19). Thereafter, as the fibrocartilaginous regions develop, their PG content changes so that at 8 months these regions strongly bind antibodies to biglycan (Figure 22), whereas antibodies to decorin are bound only in the periphery of the fibrocartilage (Figure 24). Their distributions appear to be mutually exclusive. In non-cartilaginous regions, biglycan antibodies are bound around the cells. Biglycan, but not decorin, antibodies are bound in the surface fibrous tissue. The mRNAs for both biglycan and decorin are expressed by most of the cells at 3 and 6 weeks postnatal (Figures 18 and 20), but with increasing maturity the numbers of cells expressing the mRNAs decreases (Figures 23 and 25).
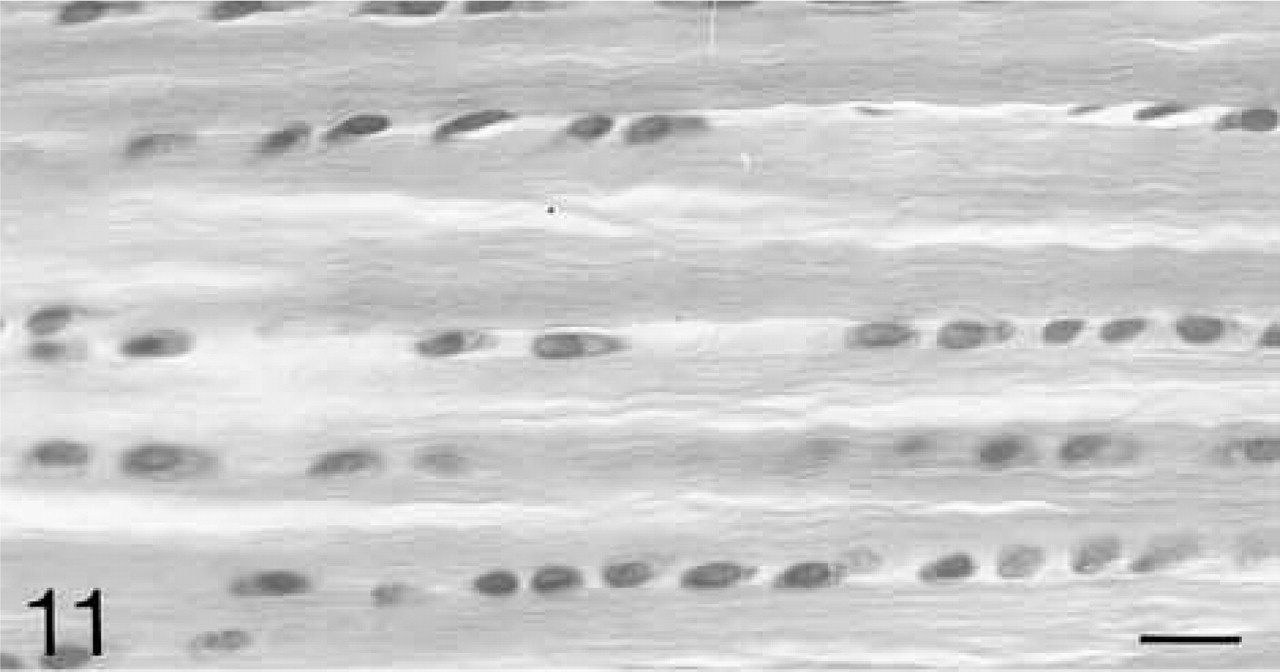
The bundles of collagen fibers are separated by rows of rounded cells. H&E staining. Bar = 20 μm.

Biglycan (B) antibody binding; biglycan is located primarily around the cells (arrowheads).
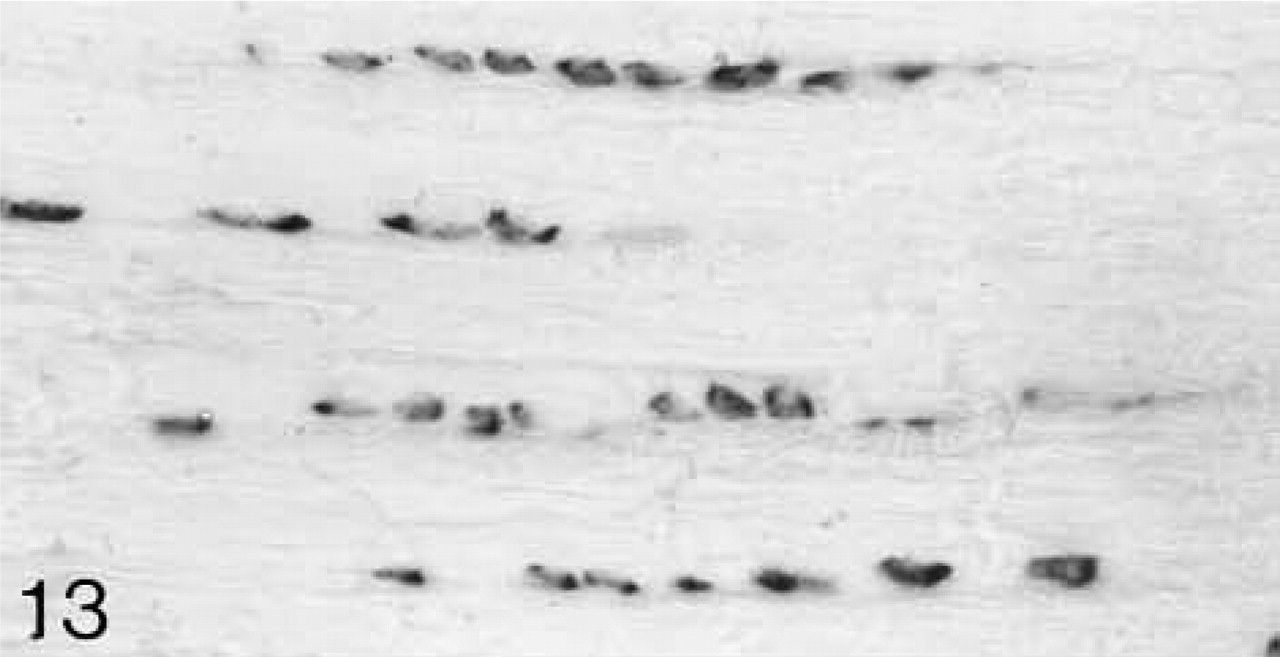
ISH for biglycan mRNA; the majority of the cells are expressing the mRNA.

Decorin (D) antibody binding; decorin is located within the fiber bundles and around the cells (arrowheads).
ISH for decorin mRNA; most cells are expressing the mRNA.
Discussion
Ligaments
The lateral and medial collateral ligaments are similar structurally and in their collagen content (Bland and Ashhurst 1996a). Antibodies to biglycan and decorin localize these small PGs between the fiber bundles. Biochemical data suggest that 80% of the PG in mature rabbit medial collateral ligament is decorin, while the remaining 20% is biglycan and a large unidentified PG (Plaas et al. 2000). The immunohistochemical data do not indicate a predominance of decorin over biglycan in mature ligaments.
The cruciate ligaments differ in appearance from the collateral ligaments principally because the cells are round and in prominent rows. Both biglycan and decorin are primarily associated with the cells. The stronger binding of the biglycan antibody compared to that of the decorin antibody may indicate a greater concentration of this PG, but immunohistochemistry cannot be regarded as strictly quantitative.
Over the period from 3 weeks to the mature adult, the distribution of biglycan and decorin in rabbit knee ligaments does not change. Previous studies of PGs of ligaments have been restricted to the localization of their GAGs. Using electron microscopic histochemistry, Bray et al. (1990, 1993) found chondroitin sulfate around the cells and in seams radiating between the bundles of collagen fibrils in human and rabbit medial collateral ligament. This agrees with the distributions of biglycan and decorin reported here.
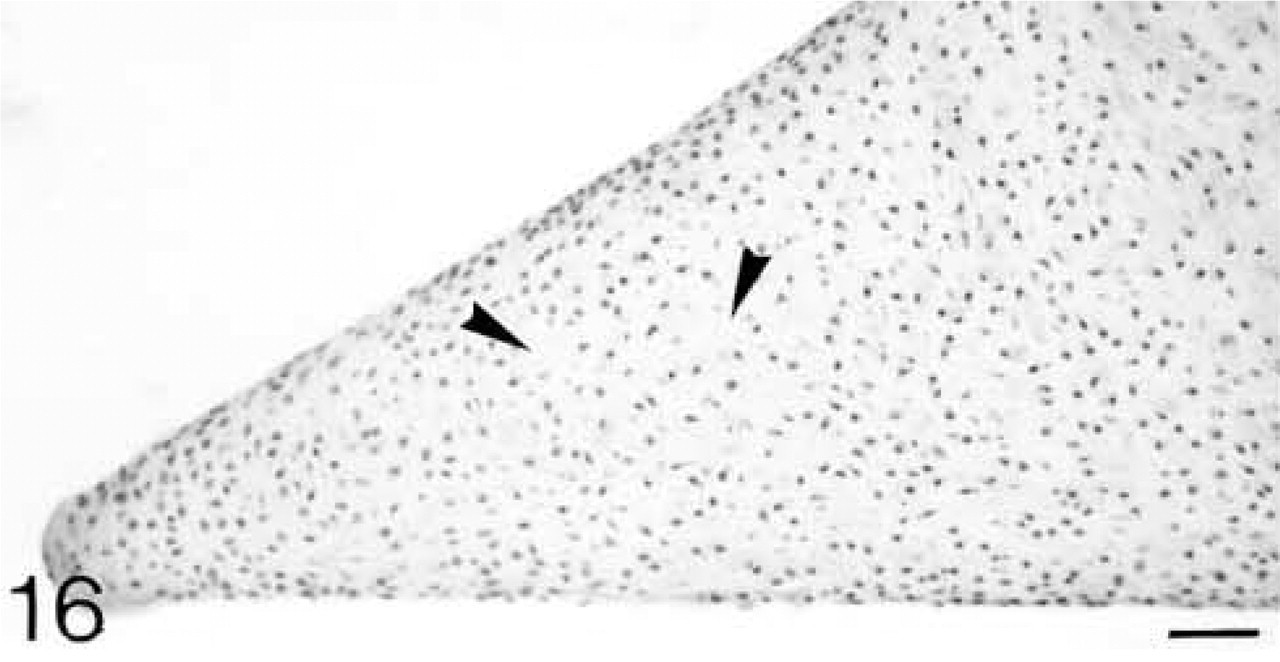
Random distribution of the cells, but in some internal areas in which fibrocartilage is developing (arrowheads) they are farther apart. H&E staining. Bar = 50 μm.
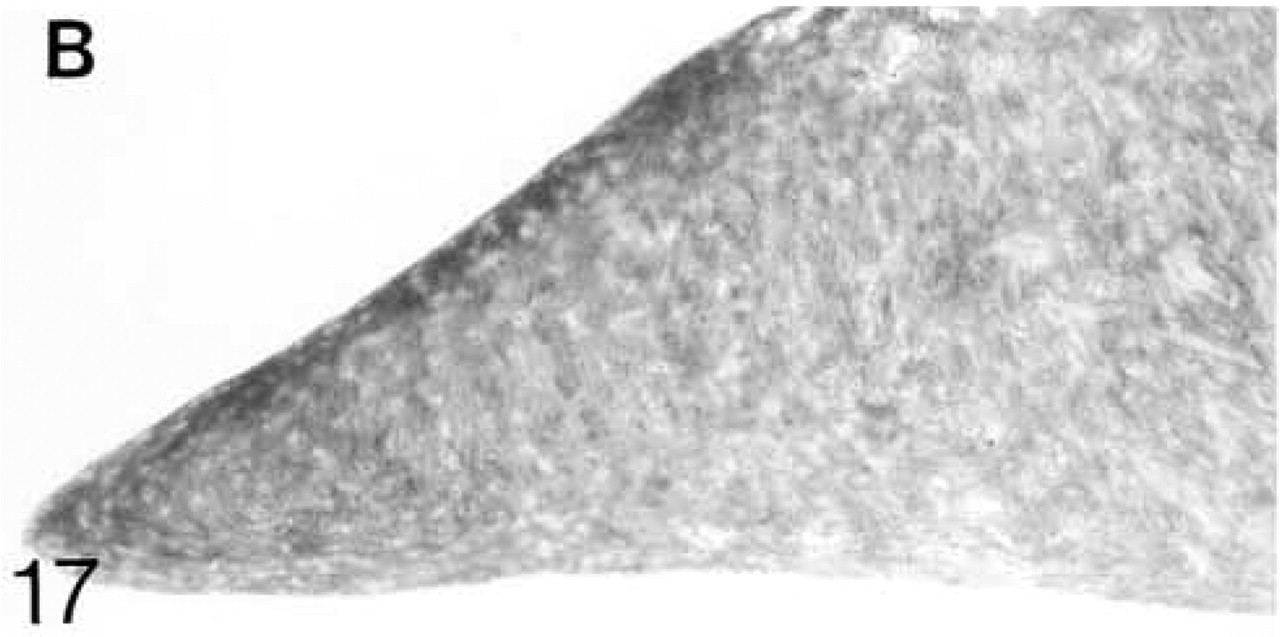
Biglycan (B) antibody binding; biglycan is present throughout the matrix, but antibody binding is stronger in some surface regions.
ISH for biglycan mRNA; all cells are expressing the mRNA.
Decorin (D) antibody binding; decorin is located throughout the matrix.
ISH for decorin mRNA; all cells are expressing the mRNA.
The central fibrocartilaginous region with chondrocyte-like cells (arrowheads) and the peripheral fibrous layer. H&E staining. Bar = 50 μm.
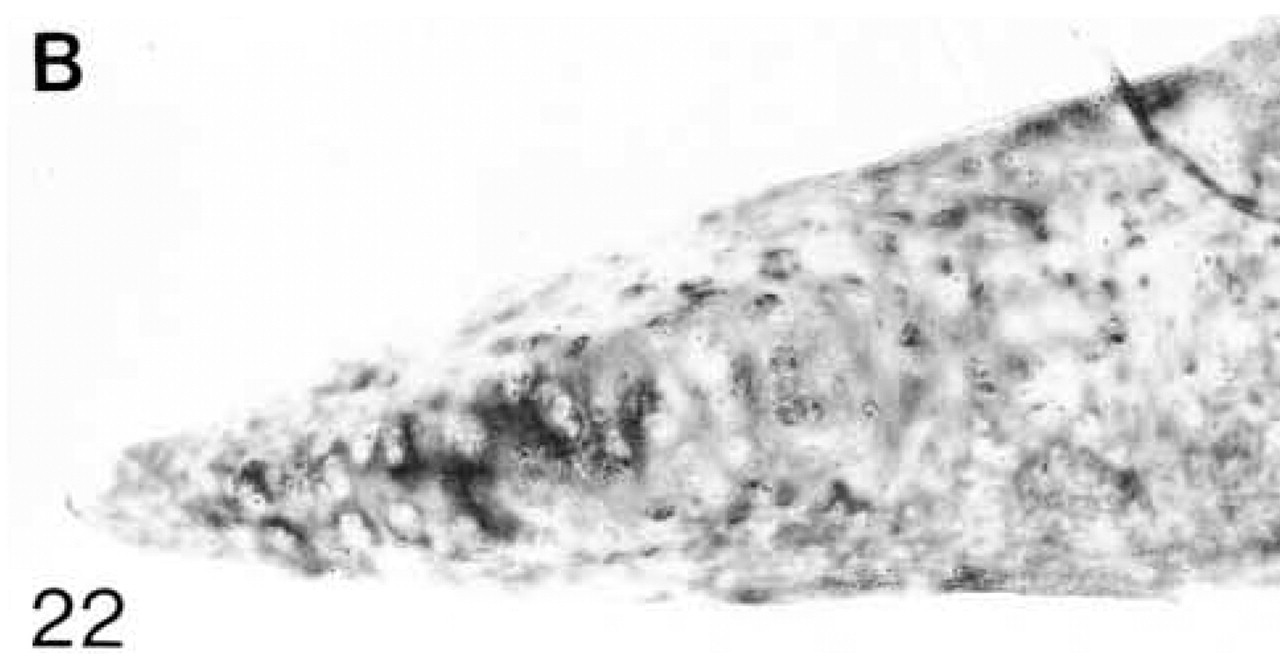
Biglycan (B) antibody binding; biglycan is present throughout the matrix, but antibody binding is much stronger in the central cartilaginous region.

ISH for biglycan mRNA; only a few cells (arrowheads) are expressing the mRNA.
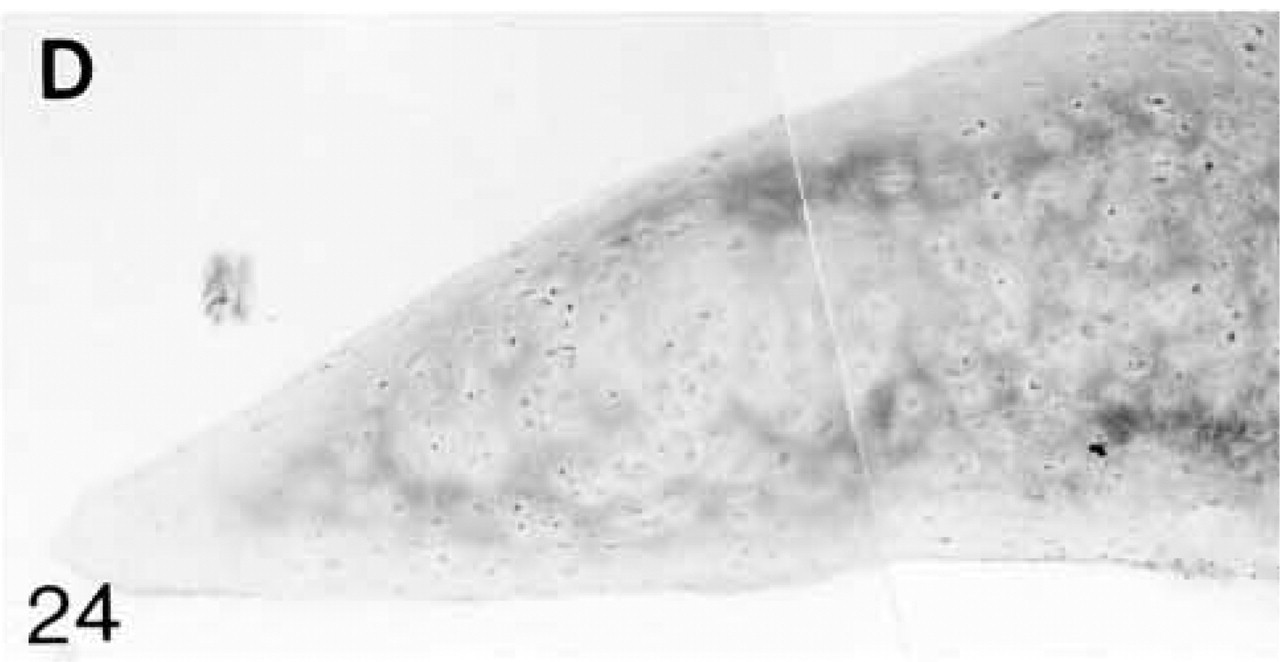
Decorin (D) antibody binding; decorin is located primarily in a region surrounding the cartilaginous area. Antibody binding is very weak both in the surface layers and in the cartilaginous region.
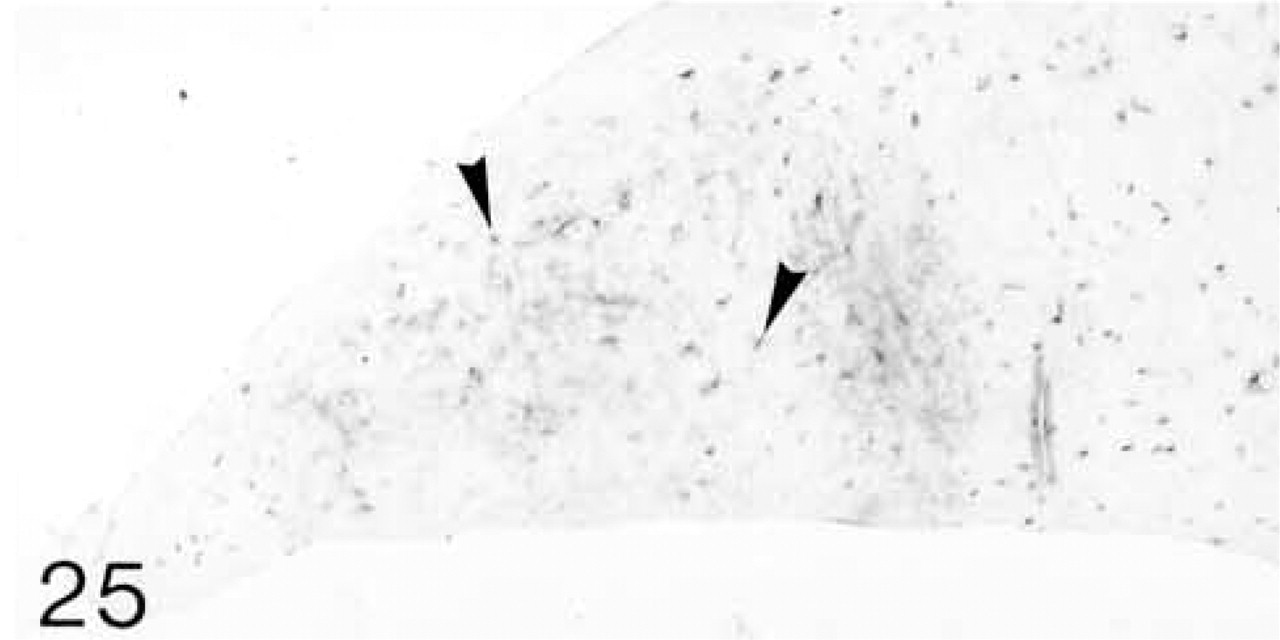
ISH for decorin mRNA; few cells (arrowheads) are expressing the mRNA.
Menisci
At 3 weeks postnatal, a Type II collagen-containing region develops in the inner part of the rabbit meniscus and persists as fibrocartilage throughout life (Bland and Ashhurst 1996b). At 3 weeks biglycan and decorin are present throughout the fibrocartilage, but by 8 months their distributions are mutually exclusive. Biglycan is present in the center and decorin around the periphery of the fibrocartilage. There are no biochemical data on PGs in rabbit menisci, but data on 20-week porcine menisci indicates that biglycan comprises around 50% and decorin 30% of the small PGs present (Nakano et al. 1997; Scott et al. 1997). This may also be true in rabbits because the area occupied by biglycan is greater than that of decorin. An accumulation of biglycan with increasing age also occurs in rabbit articular cartilage, but here decorin is colocalized with biglycan (Kavanagh and Ashhurst 1999).
Functions of Biglycan and Decorin and Their Association with Collagens
Biglycan is said to be involved in matrix organization, and, together with decorin, in cell–matrix interactions and binding of growth factors (Hildebrand et al. 1994; Buckwalter and Mankin 1997). Decorin binds to both Types I and II collagen and may be involved in the control of fibrillogenesis (Hedbom and Heinegård 1993; Keene et al. 2000; Neame et al. 2000). Developing ligaments are formed primarily of Types I and V collagen; Type III appears in the collateral ligaments at 12 weeks and in the cruciate ligaments at 8 months (Bland and Ashhurst 1996a). In neither the collateral nor the cruciate ligament can the acquisition of Type III collagen be correlated with changes in the biglycan or decorin content. In the 3-week meniscus Types I, III, and V collagen are present throughout the tissue, whereas Type II is just beginning to appear in the central regions (Bland and Ashhurst 1996b). As this central fibrocartilaginous region develops, Type I collagen disappears. The findings presented here show that at 3 weeks both biglycan and decorin are co-localized in the meniscus, but that at 8 months only biglycan is found in the central fibrocartilage. Thus, biglycan colocalizes with Type II and decorin with Type I collagen in the adult rabbit meniscus (compare Figures 22 and 24 with Figures 25 and 26 in Bland and Ashhurst 1996b). The role of decorin in promoting fibrillogenesis must be fulfilled before its loss from the fibrocartilage. The function of biglycan in menisci has been related to the greater compressive forces on the inner region of the meniscus where it predominates (Nakano et al. 1997; Scott et al. 1997).
Expression of Biglycan and Decorin mRNAs
In both the ligaments and menisci, all cells express the mRNAs for biglycan and decorin during growth, i.e., up to 14 weeks, but, except in the cruciate ligaments, only a few cells express the mRNAs by 8 months when the rabbit is skeletally mature. The continuing expression of the mRNAs for biglycan and decorin by a few cells in the adult ligaments and menisci corresponds to our previous observations of rabbit articular cartilage (Kavanagh and Ashhurst 1999). PGs are therefore continually turned over throughout life in contrast to the collagens, whose mRNAs are not expressed by any cells after 8 months (Bland and Ashhurst 1996a,b).
Estimations of mRNA levels in human and rabbit ligaments indicate that expression of biglycan and decorin mRNAs continues throughout life (Boykiw et al. 1998; Lo et al. 1998). Similarly, quantification of biglycan and decorin mRNAs in rabbit knee menisci confirms their continued expression after skeletal maturity (Hellio Le Graverand et al. 1999). After injury, the level of biglycan, but not decorin, mRNA is elevated in both species (Boykiw et al. 1998; Lo et al. 1998). This agrees with the observation that in repairing rabbit medial collateral ligament the amount of biglycan increases, but decorin is barely detectable (Plaas et al. 2000).
Conclusion
Immunohistochemical studies show that matrix components, i.e, collagens, proteoglycans, and other molecules, are distributed in a specific pattern within each tissue. The strength and proper functioning of each ligament and meniscus are dependent on the unique organization of these molecules. During development they are laid down over a protracted period in a precise and sequential manner. The problem for tissue repair is not how to stimulate the cells to secrete the appropriate molecules but how to achieve their correct organization in the regenerated tissues.
Footnotes
Acknowledgements
Supported by the Arthritis and Rheumatism Campaign UK (grant no. A052226).
We thank Prof D. Heinegård for the antibodies, Dr L. Fisher for the cDNA probes, and Ms Y.S. Bland for expert technical assistance.
