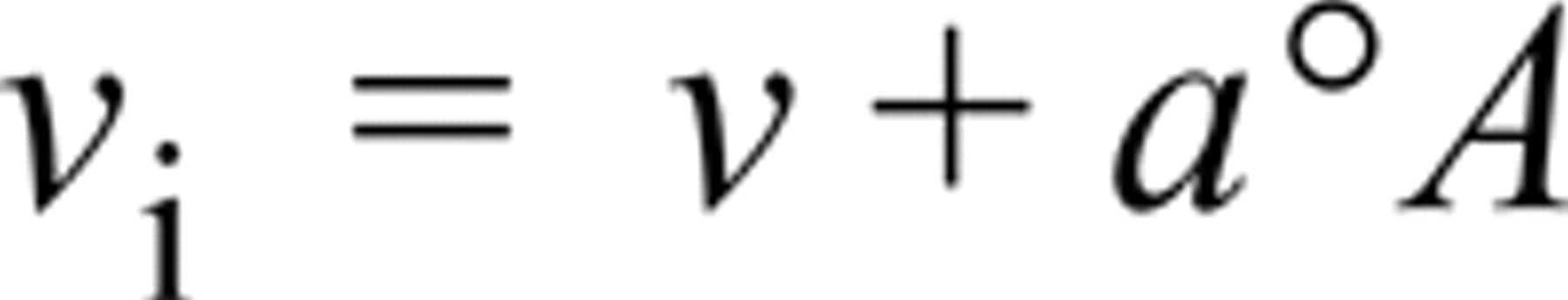Abstract
We determined the Michaelis constant (K
m) and maximal velocity (V
max) of lactate dehydrogenase (LDH) in periportal hepatocytes and skeletal muscle fibers by three different histochemical assay methods. Unfixed sections of mouse liver and gastrocnemius were incubated at 37C either on substrate (
Keywords
I
Furthermore, the Michaelis constants (K m) of enzymes determined histochemically with assay media containing high concentrations of PVA are up to 10 times higher than those determined biochemically (Butcher 1970; Robertson et al. 1982; Sinowatz et al. 1983; Van Noorden and Butcher 1986; Van Noorden and Jonges 1987; Jonges and Van Noorden 1989; Lawrence et al. 1990; Frederiks et al. 1994). However, it is difficult to conclude that this discrepancy is due entirely to interactions of enzymes with intracellular components, for at least two reasons. First, addition of 18% PVA to aqueous assay media reduces the activities (initial velocities) of LDH in tissue cells and polyacrylamide gel sections by more than half (Nakae and Stoward 1997). Second, no evidence has been published to confirm the validity of K m values determined in situ with PVA-containing media. Previously, we found that the K m values of LDH determined in various types of cell with the agarose gel film method are on the same order of magnitude as those determined biochemically (Nakae and Stoward 1993b, 1994b).
Hanes plots of mean s/v
i on
Hanes plots of mean s/v
i on
Hanes plots of mean s/v
i on
The purpose of the work reported here was to find out whether measurements of the kinetic parameters of “soluble” enzymes in situ, such as LDH, in different cell types was significantly affected by the presence of tissue protectants in assay media. The kinetic parameters of pure LDH incorporated into polyacrylamide gel sections and of LDH in periportal hepatocytes and skeletal muscle fibers in sections of mouse unfixed tissues determined with PVA-containing media were compared with those obtained using PVA-free media and agarose gel media.
Materials and Methods
The tissues and experimental procedures described in the previous article (Nakae and Stoward 1997) were used.
Kinetic Analyses
The initial velocities (v i) of the linear LDH and “nothing dehydrogenase” reactions observed for purified LDH incorporated into polyacrylamide gel sections incubated on substrate gel films and in PVA media were determined from the gradients of the linear regression lines of absorbance on time for incubation times between 0 and 3 min (Nakae and Stoward 1997).
The v i of the nonlinear LDH and “nothing dehydrogenase” reactions for purified LDH in model polyacrylamide sections incubated in aqueous media and for LDH in periportal hepatocytes and skeletal muscle fibers in unfixed sections incubated with three assay media were calculated as described previously (Nakae and Stoward 1993a, b, 1994a, b, 1997) from the equation
where v and °A are, respectively, the gradient (steady-state velocity) and intercept on the absorbance axis of the linear regression line of absorbance on time for incubation times between 1 and 3 min. The parameter a is a “diffusion” constant characteristic for LDH in each cell type in tissue sections and pure LDH in the model gel sections (see Table 1 and also Nakae and Stoward, 1997, for details). The v i of the intrinsic LDH reaction was obtained by subtracting the v i of the “nothing dehydrogenase” reaction from that of the uncorrected LDH reaction.
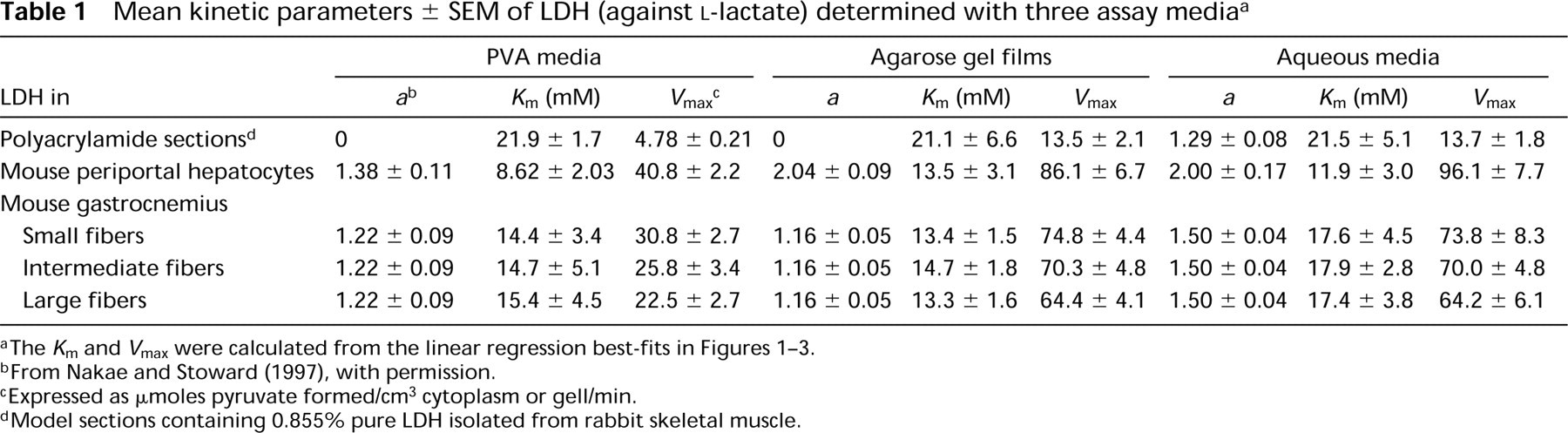
The Michaelis constant (K m) and the maximal velocity (V max) for the intrinsic LDH reactions in each cell type and in polyacrylamide gel sections were determined from Hanes plots of s/v i against s, where s is the substrate concentration. The V max was expressed as μoles pyruvate formed/cm3 cytoplasm or gel/min, assuming a molar extinction coefficient of 16,000 for Nitro BT formazans (Butcher 1978).
Results
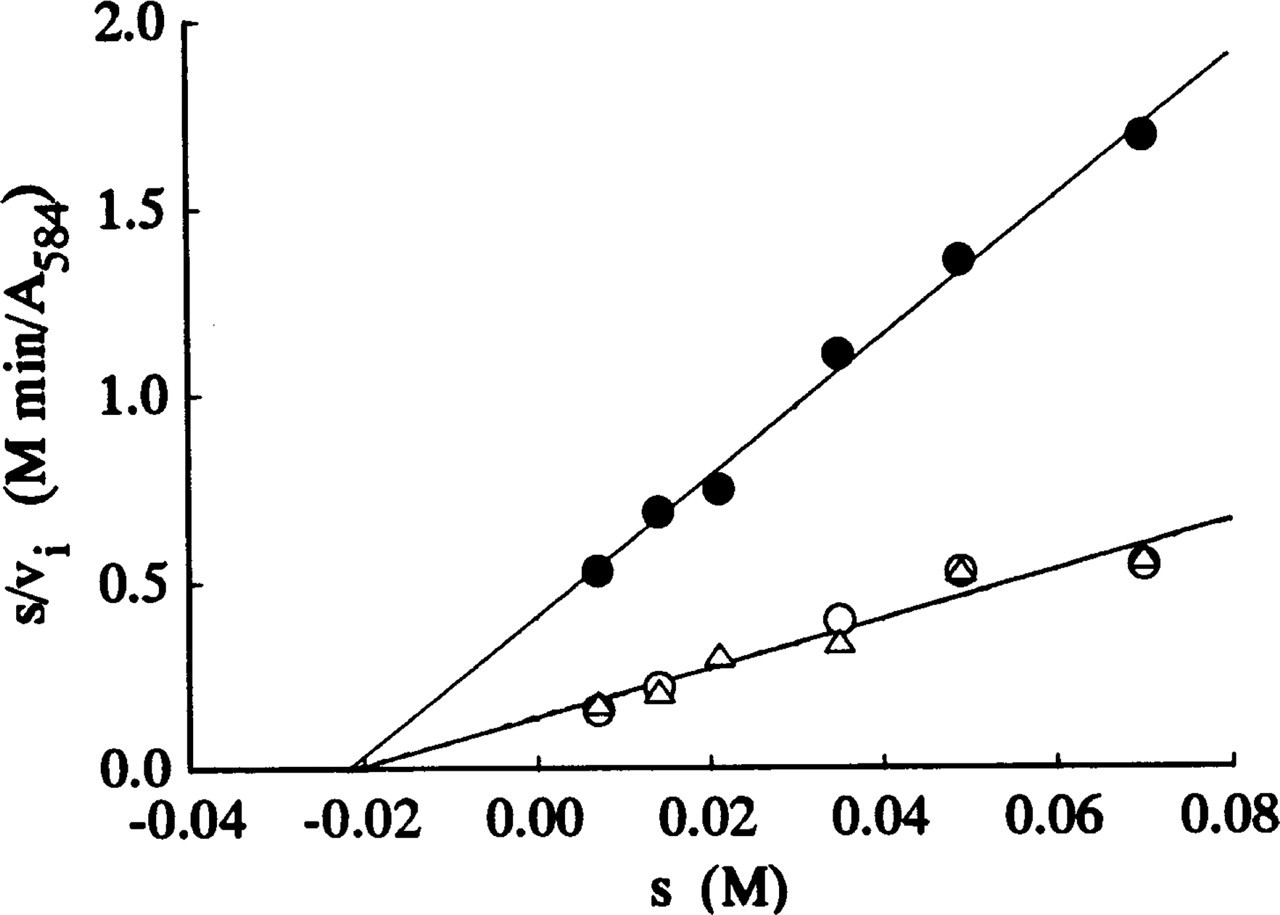
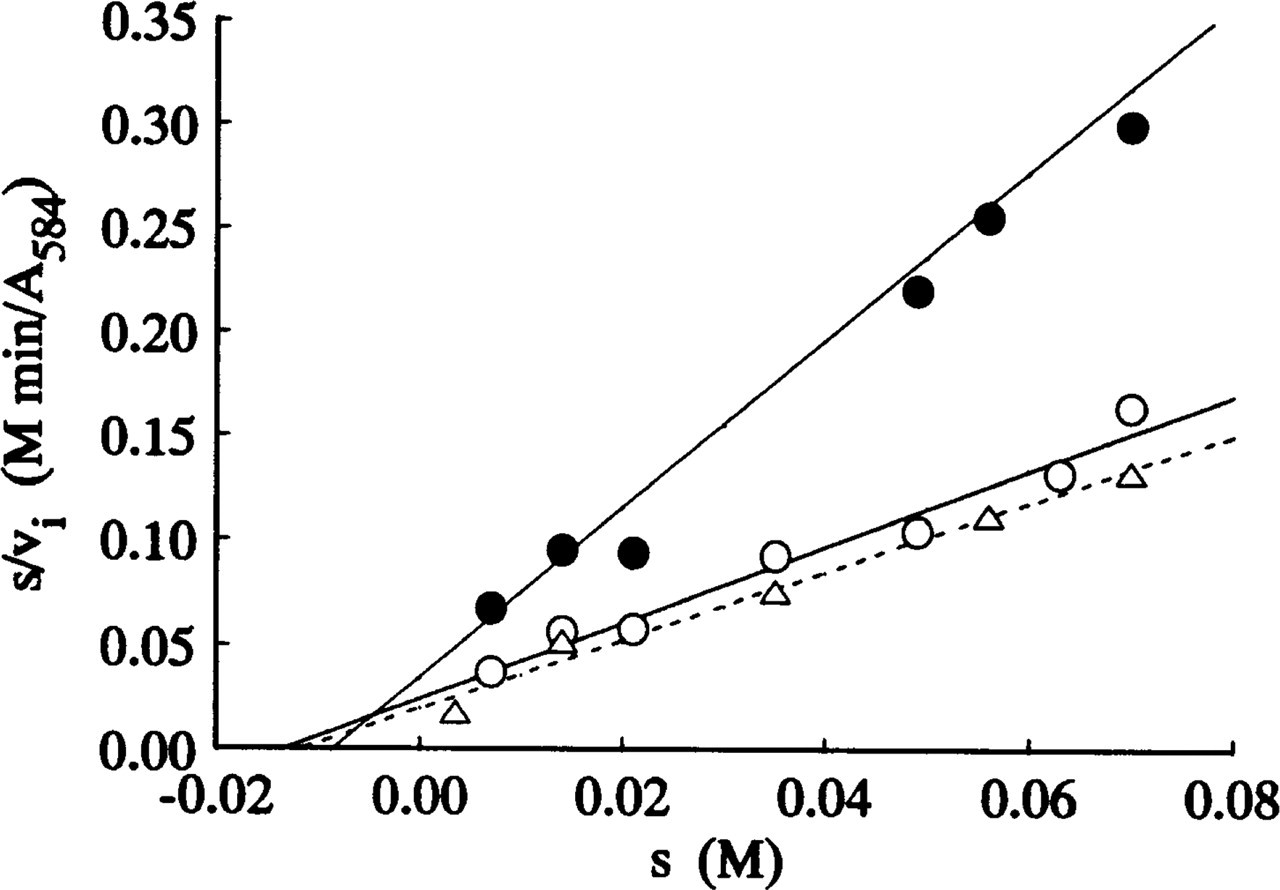
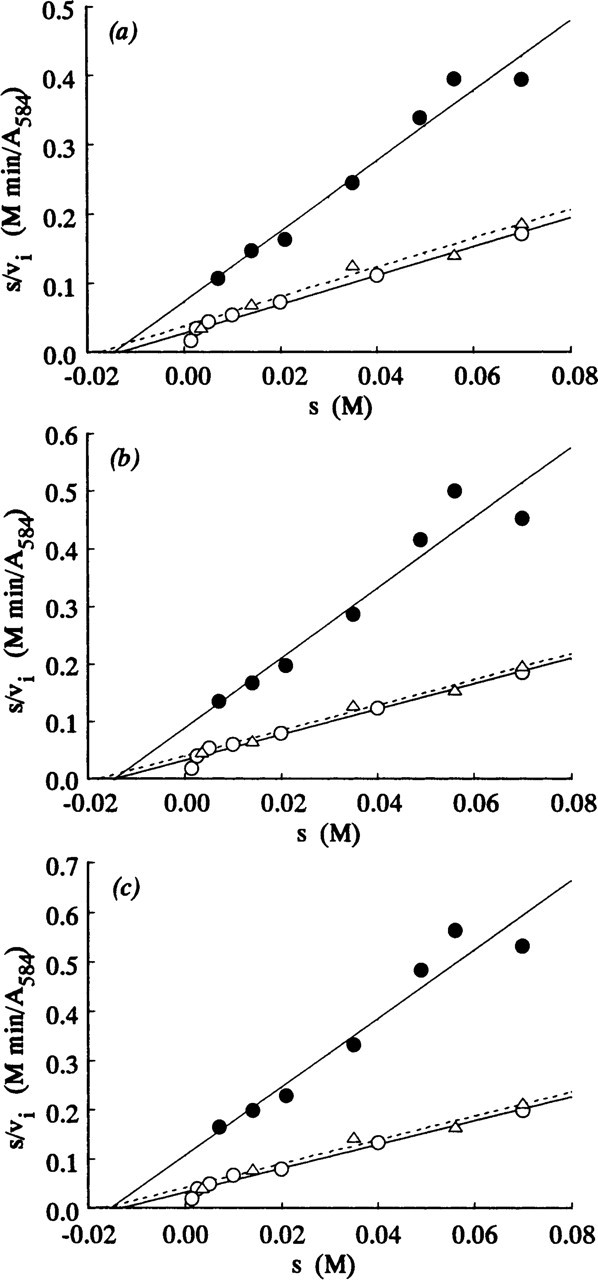
Figures 1–3 show Hanes plots of s/v i on s for pure LDH isolated from rabbit skeletal muscle and for LDH in periportal hepatocytes and skeletal muscle fibers in the sections incubated with three assay media. The regression line best-fits for the LDH in the model and tissue sections incubated on the substrate gel films and in the aqueous media were similar, but differed from those for sections incubated in PVA media. The intercepts on the abscissae and the reciprocals of the gradients of the regression best-fits correspond to -K m and V max, respectively. The K m and V max values obtained are shown in Table 1.
Discussion
The K m values obtained in this investigation (21.1–21.9 mM; Table 1) for pure LDH in model polyacrylamide gel sections incubated on lactate-containing gel films and in aqueous assay media in the presence and absence of 18% PVA are remarkably similar. The coincidence indicates that agarose gel and 18% PVA do not affect the Michaelis constant. The K m values obtained in our study are only slightly lower than the K m of 23.0 mM determined biochemically for LDH-M4 (muscle type) isolated from rabbit skeletal muscle (Stambaugh and Post 1966).
kinetic parameters ± SEM of LDH (against
bFrom Nakae and Stoward (1997), with permission.
cExpressed as μoles pyruvate formed/cm3 cytoplasm or gell/min.
dModel sections containing 0.855% pure LDH isolated from rabbit skeletal muscle.
Liver LDH consists mainly of the M4 isozyme also (e.g., Battellino and Blanco 1970). However, the mean K m of LDH in periportal hepatocytes determined with PVA media (8.62 mM) was lower than those determined on agarose gel films (13.5 mM) and in PVA-free aqueous media (11.9 mM), but the differences are not significant (0.2 p >0.5). The K m value of 13.5 mM for periportal hepatocytes is in good agreement with the values of 14.3–15.6 mM reported previously (Nakae and Stoward 1993b, 1994b). These values in situ were similar to those determined in vitro for the M4 isozyme purified from human liver (14.3–15.5 mM) (Nisselbaum and Bodansky 1963; Nisselbaum et al. 1964), but they are 1.6–2.5 times higher and 1.5–2.3 times lower, respectively, than the in vitro values for the LDH-M4 purified from mouse liver (Battellino and Blanco 1970) and rabbit liver (Battellino et al. 1968).
Three types of skeletal muscle fibers in gastrocnemius are discriminated quantitatively by the highest, intermediate, and lowest V max values, respectively (Table 1). There are, however, no significant differences in the K m between the three fiber types. This confirms a previous report (Nakae and Stoward 1994b). The K m values (17.4–17.9 mM) determined in aqueous assay media for the three fiber types were slightly higher than those determined on agarose gel films (13.3–14.7 mM) and in PVA media (14.4–15.4 mM), but the differences are not significant (0.5 p>0.2 and p>0.5, respectively). The K m values of 13.3–14.7 mM for skeletal muscle fibers determined by the agarose gel film method are similar to the values (10.4–12.5 mM) determined previously (Nakae and Stoward 1994b). As shown in Table 1, these K m values are closer to those for LDH in periportal hepatocytes of the same animal (8.62–13.5 mM) rather than to those for pure LDH isolated from rabbit skeletal muscle (21.1–21.9 mM). Our K m values determined for LDH in situ in skeletal muscle fibers (13.3–17.9 mM) are similar to the K m value determined in vitro for the M4 isozyme purified from bovine skeletal muscle (12.5 mM; Pesce et al. 1964). They are, respectively, 2.7–3.6 times and 3.4–4.6 times higher than the in vitro K m values for the LDH-M4 isolated from pig (Bennett and Gutfreund 1973) and mouse (Hawtrey et al. 1975) muscles, and 1.3–1.7 times lower than the in vitro K m value for the M4 isozyme isolated from rabbit muscle (Stambaugh and Post 1966). Wachsmuth (1980) found that the K m values of LDH in two types of human skeletal muscle fibers, determined with the mixed-aggregation immunocytochemical technique, were 20 and 30 mM for muscle fibers stained respectively for the LDH-M4 and LDH-H4 (heart type), whereas in solution they were 5 and 1 mM.
The intrinsic maximal velocities (V max) of pure LDH and LDH in periportal hepatocytes and skeletal muscle fibers determined on substrate-containing agarose gel films or with aqueous assay media were similar, but were reduced to 35–47% by addition of 18% PVA to the aqueous media (Table 1). The reduction in the V max may be due to the catalytic center activity k cat being retarded by the high viscosity of the PVA media because PVA does not inhibit the LDH reaction itself, as shown by the nonsignificant differences between the K m values determined by the three assay methods (Table 1).
There are a number of reports in the literature that the affinity of LDH for its substrates or co-enzymes is perturbed by interactions of the enzyme with intracellular components. Nitisewojo and Hultin (1976), for example, reported that the K m value of LDH-M4 isozyme against pyruvate is doubled when the isozyme binds to a particulate fraction obtained from a homogenate of skeletal muscle. Poglazov and Livanova (1986) found that the K m of purified LDH against NADH is 2.5 times higher when associated with F-actin. Moreover, cytochemical and immunohistochemical studies have shown that muscle LDH is predominantly localized in the isotropic zones of myofibrils (e.g., Dölken et al. 1975, and references cited therein). Therefore, the discrepancies observed in the present study between the in situ and in vitro K m values of LDH of mouse liver and skeletal muscle may be attributed to the interaction of the enzyme with intracellular structures or with macromolecules such as F-actin, tropomyosin, and troponin (see, e.g., Clarke et al. 1985; Poglazov and Livanova 1986; Yasykova et al. 1990), microtubules, and tubulin (Karkhoff–Schweizer and Knull 1987; Walsh et al. 1989; Marmillot et al. 1994). This interpretation is currently being tested experimentally.